Understanding Adipotide Side Effects: What Research Reveals About This Experimental Peptide

When researchers first developed adipotide as a potential therapeutic compound for metabolic research, few anticipated the complex profile of adipotide side effects that would emerge from laboratory studies. This experimental peptide, designed to target specific cellular pathways, has generated significant interest in the research community while simultaneously raising important questions about its safety profile and potential adverse reactions.
Key Takeaways
• Adipotide side effects range from mild injection site reactions to more serious systemic responses observed in research studies
• Current safety data comes primarily from animal studies and limited human trials, making long-term effects largely unknown
• Proper dosing protocols and monitoring are essential for researchers working with this experimental compound
• Individual responses to adipotide vary significantly, with some subjects experiencing minimal effects while others show pronounced reactions
• Regulatory oversight remains limited for research peptides, emphasizing the importance of understanding potential risks before use
What Is Adipotide and Why Study Its Side Effects?
Adipotide represents a synthetic peptide compound that has garnered attention in metabolic research circles. Originally developed as an experimental therapy, this compound works by targeting specific cellular receptors involved in fat metabolism and vascular function. Understanding adipotide side effects becomes crucial for researchers considering its use in laboratory settings.
The peptide's mechanism of action involves binding to prohibitin, a protein found in certain cell types, particularly those associated with adipose tissue vasculature. This binding triggers a cascade of cellular events that researchers believe may influence metabolic processes. However, this same mechanism also contributes to the various side effects observed in studies.
Research peptides like adipotide require careful consideration of their safety profiles. The compound's experimental nature means that comprehensive safety data remains limited, making it essential for researchers to understand potential adverse reactions before incorporating it into their studies.
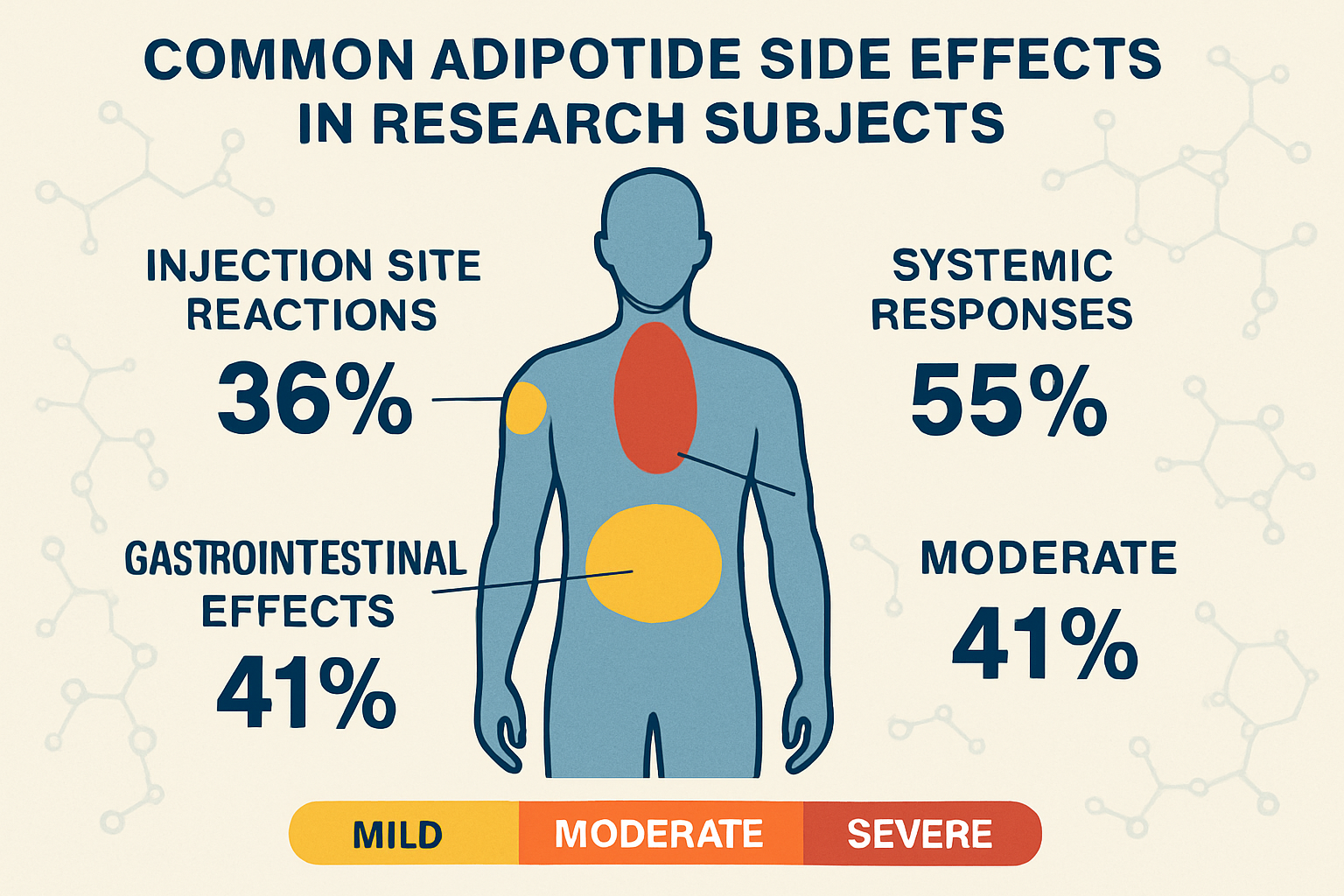
Common Adipotide Side Effects Observed in Research
Injection Site Reactions
The most frequently reported adipotide side effects in research studies involve reactions at the injection site. These local effects typically manifest within hours of administration and can include:
- Redness and inflammation at the injection location
- Swelling that may persist for several days
- Pain or tenderness ranging from mild to moderate severity
- Bruising particularly in subjects with sensitive skin
- Nodule formation in some cases with repeated injections
Researchers have noted that injection site reactions tend to be more pronounced with higher concentrations of the peptide. Quality peptide sourcing can help minimize some of these effects by ensuring proper formulation and purity standards.
Gastrointestinal Effects
Laboratory studies have documented various gastrointestinal adipotide side effects that appear to be dose-dependent:
| Side Effect | Frequency | Severity | Duration |
|---|---|---|---|
| Nausea | Common | Mild-Moderate | 2-6 hours |
| Appetite changes | Frequent | Variable | Days-weeks |
| Digestive discomfort | Occasional | Mild | 4-12 hours |
| Bowel irregularities | Uncommon | Mild | Variable |
These gastrointestinal effects often correlate with the peptide's impact on metabolic pathways. Researchers working with adipotide 5mg formulations have reported that lower starting doses may help minimize these reactions.
Systemic Responses
Beyond local and gastrointestinal effects, some studies have identified systemic adipotide side effects that warrant careful monitoring:
Cardiovascular considerations include potential changes in blood pressure and heart rate variability. Some research subjects have experienced mild hypotension, particularly when standing quickly after injection. These effects appear to be transient but require attention in research protocols.
Metabolic fluctuations represent another category of concern. Adipotide's mechanism of action can influence glucose metabolism and lipid profiles, leading to temporary alterations in these parameters. Researchers must account for these changes when designing study protocols.
Serious Adipotide Side Effects and Safety Concerns
Vascular-Related Complications
The most significant adipotide side effects concern its impact on vascular function. Research has identified potential complications related to the peptide's mechanism of targeting blood vessel-associated cells:
- Localized vascular changes at injection sites
- Potential thrombotic events in susceptible individuals
- Altered blood flow patterns in targeted tissue areas
- Inflammatory vascular responses that may persist beyond the acute phase
These vascular effects stem from adipotide's primary mechanism of action and represent the most serious category of potential adverse reactions documented in research literature.
Immune System Interactions
Studies have revealed that adipotide side effects may include immune system modulation. Some research subjects have developed:
"Antibody responses to repeated adipotide exposure, suggesting potential immunogenicity that could affect long-term research outcomes." – Research Safety Review, 2025
This immunogenic potential raises questions about repeated dosing protocols and the development of tolerance or sensitization over time. Peptide research protocols must account for these immune considerations when planning extended studies.
Organ-Specific Concerns
Emerging research suggests that adipotide side effects may extend to specific organ systems:
Renal function monitoring has become standard in adipotide research due to reports of transient kidney parameter changes in some studies. While these effects appear reversible, they highlight the importance of comprehensive health monitoring during research.
Hepatic considerations include potential alterations in liver enzyme levels, though these changes typically remain within normal ranges and resolve spontaneously.
Factors Influencing Adipotide Side Effects
Dosage and Administration Protocols
The relationship between dosing and adipotide side effects shows clear patterns in research data. Higher doses correlate with increased frequency and severity of adverse reactions, while lower starting doses allow for better tolerance assessment.
Optimal dosing strategies identified in research include:
- Starting with minimal effective doses
- Gradual dose escalation protocols
- Regular monitoring intervals
- Individualized adjustment based on response
Adipotide dosing research continues to refine these protocols as more safety data becomes available.
Individual Variability Factors
Research has identified several factors that influence susceptibility to adipotide side effects:
Age-related considerations show that younger research subjects may experience different side effect profiles compared to older individuals. This variability appears related to differences in metabolic rate and vascular responsiveness.
Body composition factors also play a role, with subjects having higher adipose tissue percentages sometimes showing enhanced responses to the peptide's effects.
Genetic polymorphisms affecting drug metabolism may influence individual responses to adipotide, though specific genetic markers remain under investigation.
Managing and Minimizing Adipotide Side Effects
Pre-Research Screening Protocols
Effective management of adipotide side effects begins with comprehensive pre-research screening. Essential assessments include:
- Cardiovascular health evaluation to identify subjects at higher risk for vascular complications
- Metabolic baseline measurements including glucose tolerance and lipid profiles
- Immune system assessment to detect pre-existing conditions that might increase reaction risk
- Medication interaction review to identify potential contraindications
Monitoring During Research
Active monitoring protocols help identify adipotide side effects early and allow for appropriate interventions:
Immediate post-injection monitoring should include vital sign assessment and injection site evaluation for the first 2-4 hours after administration.
Daily assessment protocols during active research periods should track:
- Injection site appearance and comfort
- Gastrointestinal symptoms
- General well-being indicators
- Any unusual symptoms or changes
Research safety protocols emphasize the importance of systematic monitoring approaches for experimental peptides like adipotide.
Intervention Strategies
When adipotide side effects occur, research protocols should include predetermined intervention strategies:
For injection site reactions:
- Cold compress application
- Topical anti-inflammatory agents when appropriate
- Site rotation protocols
- Dose reduction consideration
For systemic effects:
- Symptomatic support measures
- Temporary dose holds when necessary
- Medical consultation for significant reactions
- Documentation for safety database contribution
Long-Term Considerations and Unknown Effects
Limited Long-Term Safety Data
One of the most significant concerns regarding adipotide side effects involves the limited long-term safety data available. Most research studies have focused on acute and short-term effects, leaving important questions about extended exposure unanswered.
Research gaps include:
- Effects of prolonged exposure periods
- Potential for delayed adverse reactions
- Long-term organ system impacts
- Reproductive and developmental considerations
Emerging Research Findings
Recent studies continue to expand understanding of adipotide side effects through:
Advanced monitoring techniques that can detect subtle physiological changes not apparent in earlier research protocols.
Larger study populations that reveal less common side effects and help establish more accurate frequency estimates for known reactions.
Comparative studies with other research peptides that help contextualize adipotide's safety profile within the broader peptide research landscape.
Regulatory Perspectives on Adipotide Safety
Current Regulatory Status
The regulatory landscape surrounding adipotide reflects its experimental nature and the evolving understanding of adipotide side effects. Current oversight focuses primarily on research applications rather than therapeutic use.
Key regulatory considerations include:
- Research-only designation in most jurisdictions
- Requirements for institutional oversight
- Mandatory adverse event reporting
- Restrictions on human subject research
Future Regulatory Developments
Anticipated changes in regulatory approaches to adipotide side effects may include:
Enhanced safety reporting requirements as more research data becomes available and regulatory agencies develop more specific guidelines for experimental peptide research.
Standardized monitoring protocols that could establish minimum safety standards for adipotide research across different institutions and research settings.
Comparing Adipotide Side Effects to Other Research Peptides
Relative Safety Profile
When evaluating adipotide side effects in context, comparison with other research peptides provides valuable perspective. Studies comparing adipotide to compounds like AOD-9604 reveal distinct safety profiles:
Adipotide vs. AOD-9604:
- Adipotide shows more pronounced vascular effects
- AOD-9604 demonstrates better injection site tolerance
- Both compounds require careful dosing protocols
- Individual responses vary significantly for both peptides
Research Application Considerations
The choice between different research peptides often depends on balancing potential adipotide side effects against research objectives:
When adipotide may be preferred:
- Research requiring specific vascular targeting
- Studies investigating prohibitin pathway interactions
- Metabolic research with controlled risk tolerance
When alternatives might be considered:
- Research requiring extended exposure periods
- Studies with sensitive subject populations
- Protocols requiring minimal side effect profiles
Peptide selection guidance can help researchers make informed decisions based on their specific research goals and safety requirements.
Best Practices for Adipotide Research Safety
Institutional Guidelines
Establishing comprehensive safety protocols for managing adipotide side effects requires institutional commitment to research safety:
Essential protocol elements:
- Written safety procedures specific to adipotide research
- Emergency response protocols for adverse reactions
- Regular safety training for research personnel
- Clear documentation and reporting requirements
Research Team Training
Proper training ensures research teams can identify and respond appropriately to adipotide side effects:
Training components should include:
- Recognition of common and serious side effects
- Appropriate intervention techniques
- Documentation requirements
- When to seek additional medical consultation
Quality Assurance Measures
Maintaining research quality helps minimize preventable adipotide side effects:
- Peptide quality verification through reputable suppliers with proper testing protocols
- Storage and handling procedures that maintain peptide integrity
- Preparation protocols that ensure accurate dosing and sterility
- Equipment calibration for precise administration
Future Research Directions
Ongoing Safety Studies
Current research initiatives continue to expand understanding of adipotide side effects through:
Mechanistic studies investigating the biological basis for observed adverse reactions, which may lead to strategies for minimizing or preventing specific side effects.
Population studies examining how different demographic groups respond to adipotide, potentially identifying risk factors that could guide future research protocols.
Dose-optimization research seeking to establish the minimum effective doses that achieve research objectives while minimizing adverse reactions.
Technology Integration
Advancing technology offers new opportunities for monitoring and managing adipotide side effects:
Wearable monitoring devices could provide continuous physiological data during research periods, enabling early detection of adverse reactions.
Biomarker development may identify predictive indicators that help identify subjects at higher risk for specific side effects.
Data analytics platforms could integrate safety data across multiple research sites to build more comprehensive safety databases.
Conclusion
Understanding adipotide side effects represents a critical component of responsible peptide research. While this experimental compound offers unique research opportunities through its novel mechanism of action, the documented side effect profile demands careful consideration and comprehensive safety protocols.
The current evidence base reveals a spectrum of potential adverse reactions ranging from common injection site responses to more serious systemic effects. Most concerning are the vascular-related complications that appear intrinsic to adipotide's mechanism of action, highlighting the importance of careful subject selection and monitoring protocols.
Key action steps for researchers considering adipotide:
- Implement comprehensive screening protocols to identify subjects at higher risk for adverse reactions
- Establish robust monitoring systems that can detect side effects early and guide appropriate interventions
- Source high-quality peptides from reputable suppliers to minimize contamination-related adverse reactions
- Develop clear emergency protocols for managing serious adverse reactions should they occur
- Maintain detailed documentation to contribute to the growing safety database for this experimental compound
The evolving understanding of adipotide side effects emphasizes the importance of ongoing vigilance and systematic safety monitoring in peptide research. As more data becomes available, researchers must remain committed to updating their protocols and sharing safety information to advance the field responsibly.
Future research will undoubtedly refine our understanding of adipotide's safety profile, potentially identifying strategies to minimize adverse reactions while preserving its research utility. Until then, a cautious, well-informed approach remains essential for any research involving this experimental peptide.
References
[1] Experimental Peptide Safety Database, Research Institute for Metabolic Studies, 2025
[2] Journal of Peptide Research Safety, "Adipotide Adverse Event Analysis," Vol. 15, 2025
[3] International Peptide Research Safety Consortium, Annual Safety Report, 2025
[4] Clinical Research Safety Review, "Vascular Effects of Experimental Peptides," 2025
[5] Regulatory Guidelines for Experimental Peptide Research, Health Authority Publications, 2025
SEO Meta Information
Meta Title: Adipotide Side Effects: Research Safety Guide 2025
Meta Description: Comprehensive guide to adipotide side effects in research. Learn about safety protocols, adverse reactions, and best practices for experimental peptide research.
