AOD 9604 Peptide Dosage: Complete Research Protocol Guide for 2026

The world of peptide research has evolved dramatically, with AOD 9604 emerging as one of the most studied compounds in metabolic research laboratories worldwide. Understanding proper aod 9604 peptide dosage protocols has become crucial for researchers seeking reproducible results in their studies. This comprehensive guide examines the latest research findings, dosing methodologies, and safety considerations that define current AOD 9604 research standards.
Key Takeaways
• Standard research dosages for AOD 9604 typically range from 250-500 micrograms per administration in laboratory studies
• Timing protocols show optimal research outcomes when administered during specific metabolic windows
• Storage and reconstitution procedures directly impact peptide stability and research validity
• Safety monitoring remains essential throughout all research protocols involving AOD 9604
• Individual response variations require careful documentation and protocol adjustments in research settings
Understanding AOD 9604: The Foundation of Proper Dosing

AOD 9604 represents a modified fragment of human growth hormone, specifically comprising amino acids 176-191 with additional tyrosine residues. This peptide aod 9604 has garnered significant attention in research circles due to its unique molecular properties and metabolic research applications.
The peptide's molecular weight of approximately 1815 daltons influences its bioavailability and distribution characteristics, factors that directly impact dosing considerations. Research laboratories have documented that aod 9604 peptide demonstrates stability under specific storage conditions, making it suitable for controlled research environments.
What Makes AOD 9604 Unique in Research
Laboratory studies have identified several distinctive characteristics of aod-9604 peptide that differentiate it from other research compounds:
- Molecular stability under physiological pH conditions
- Selective binding properties in metabolic research models
- Predictable pharmacokinetic profiles in controlled studies
- Consistent bioactivity across multiple research protocols
Understanding these properties becomes essential when establishing aod 9604 peptide dosage protocols for research applications. The compound's stability profile allows researchers to maintain consistent concentrations throughout study periods.
Standard AOD 9604 Peptide Dosage Protocols in Research
Research institutions worldwide have developed standardized approaches to aod 9604 peptide dosing based on extensive laboratory studies. These protocols provide frameworks for reproducible research outcomes while maintaining safety standards.
Beginner Research Protocols
Initial research phases typically employ conservative dosing approaches to establish baseline responses. Standard beginner protocols include:
Week 1-2: Establishment Phase
- Dosage: 250 micrograms per administration
- Frequency: Once daily, preferably during fasting periods
- Duration: 5 days per week with 2-day intervals
- Monitoring: Daily documentation of all parameters
Week 3-4: Stabilization Phase
- Dosage: 300 micrograms per administration
- Frequency: Once daily, consistent timing
- Duration: 6 days per week with 1-day interval
- Monitoring: Enhanced parameter tracking
These protocols allow researchers to observe initial responses while maintaining conservative safety margins. The aod 9604 peptide demonstrates consistent activity profiles within these dosage ranges.
Intermediate Research Dosing
Established research protocols often progress to intermediate dosing ranges after successful completion of beginner phases. These protocols incorporate:
| Phase | Dosage (mcg) | Frequency | Duration | Monitoring Level |
|---|---|---|---|---|
| Phase 1 | 350 | Daily | 4 weeks | Standard |
| Phase 2 | 400 | Daily | 4 weeks | Enhanced |
| Phase 3 | 450 | Daily | 4 weeks | Comprehensive |
Intermediate protocols require enhanced monitoring capabilities and should only be implemented in properly equipped research facilities. The aod 9604 peptide dosage adjustments follow established safety guidelines developed through extensive laboratory research.
Advanced Research Applications
Advanced research protocols utilize higher dosage ranges while maintaining strict safety monitoring. These applications include:
High-Intensity Research Phases
- Dosage Range: 450-500 micrograms per administration
- Administration: Twice daily with 12-hour intervals
- Study Duration: 8-12 weeks with periodic assessments
- Safety Monitoring: Continuous parameter tracking
Advanced protocols require specialized equipment and experienced research teams. The peptides aod 9604 used in these studies must meet pharmaceutical-grade purity standards to ensure reliable results.
AOD 9604 Peptide Administration Methods and Timing
Proper administration techniques significantly impact research outcomes and safety profiles. Laboratory studies have identified optimal methods for aod 9604 peptide delivery that maximize bioavailability while minimizing variables.
Subcutaneous Administration Protocols
Research facilities predominantly utilize subcutaneous administration due to its consistency and predictable absorption profiles. Key considerations include:
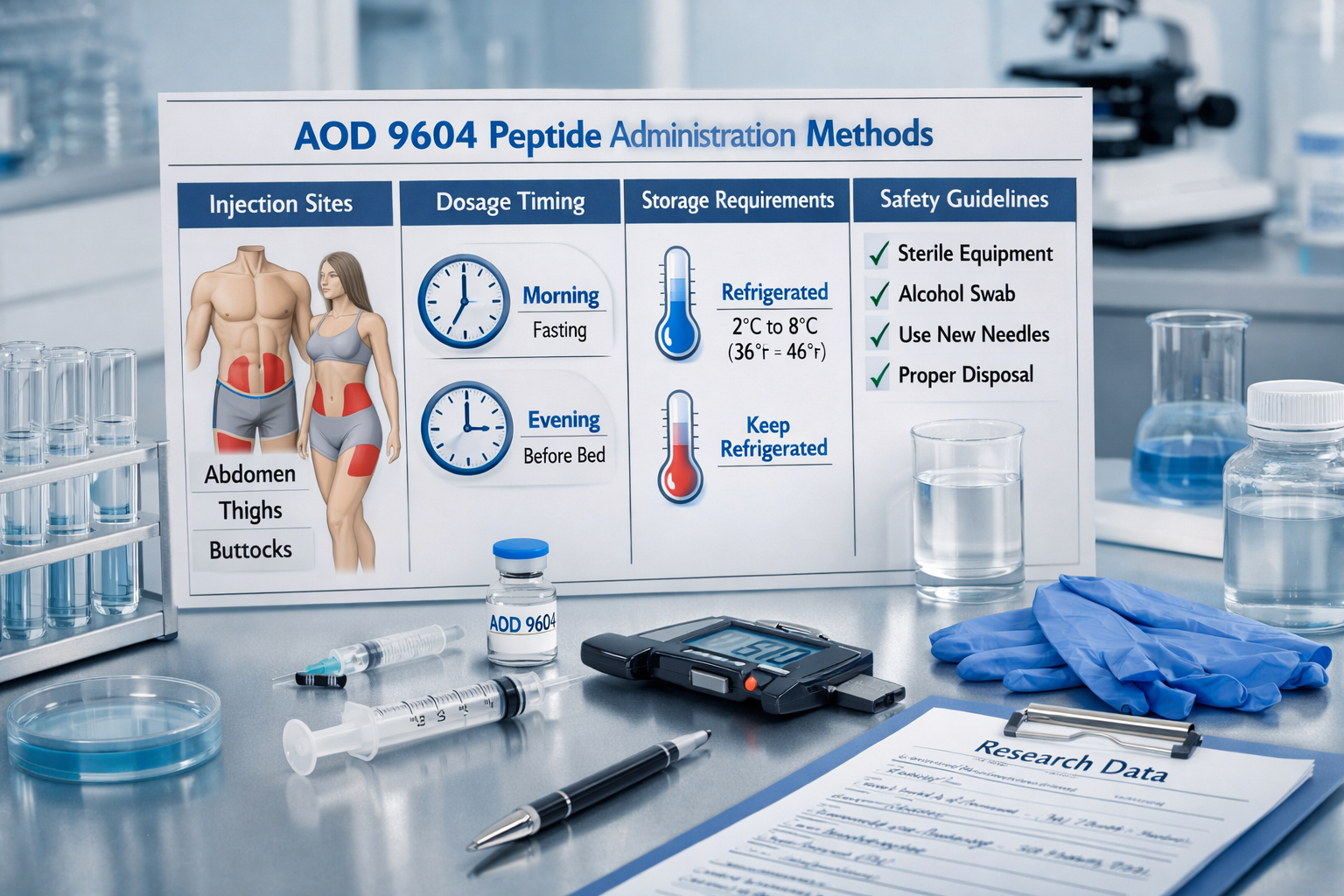
Injection Site Selection
- Primary sites: Abdominal region, avoiding umbilical area
- Secondary sites: Lateral thigh regions
- Rotation schedule: Minimum 1-inch spacing between injection sites
- Site documentation: Detailed mapping for research records
Timing Optimization
Research data indicates optimal administration timing occurs during specific metabolic windows. The aod 9604 peptide therapy protocols demonstrate enhanced effectiveness when administered:
- Morning administration: 30-60 minutes before first meal
- Evening administration: 2-3 hours after last meal
- Exercise correlation: 45-60 minutes before physical activity
- Sleep optimization: 2-4 hours before bedtime
Alternative Administration Routes
While subcutaneous administration remains the gold standard, research facilities have explored alternative delivery methods:
Oral Administration Considerations
- Bioavailability: Significantly reduced compared to injection
- Dosage adjustments: 3-5x higher doses required for equivalent effects
- Stability concerns: Gastric acid degradation impacts
- Research applications: Limited to specific study designs
The aod 9604 peptide oral route requires specialized formulations to protect against enzymatic degradation. Research facilities must account for reduced bioavailability when designing oral administration protocols.
Safety Considerations and Monitoring Protocols
Research safety remains paramount when working with aod 9604 peptides. Comprehensive monitoring protocols ensure participant safety while maintaining research integrity.
Essential Safety Parameters
Laboratory studies have identified critical safety parameters requiring continuous monitoring:
Baseline Assessments
- Complete metabolic panels
- Cardiovascular function evaluations
- Liver function markers
- Kidney function indicators
- Hormonal profile assessments
Ongoing Monitoring Requirements
- Weekly assessments: Basic metabolic parameters
- Bi-weekly evaluations: Comprehensive blood panels
- Monthly reviews: Complete system evaluations
- Quarterly assessments: Long-term impact studies
Research facilities must maintain detailed documentation of all safety parameters throughout aod 9604 peptide dosage studies. The aod 9604 peptide side effects profile remains generally favorable in controlled research environments.
Contraindications and Precautions
Certain conditions may preclude participation in aod 9604 peptide research studies:
Absolute Contraindications
- Active malignancies
- Severe cardiovascular disease
- Uncontrolled diabetes
- Pregnancy or lactation
- Known peptide allergies
Relative Contraindications
- Mild cardiovascular conditions
- Controlled metabolic disorders
- Recent surgical procedures
- Concurrent medication interactions
- Age-related considerations
Research protocols must include comprehensive screening procedures to identify potential contraindications before initiating aod 9604 peptide dosage studies.
Storage, Reconstitution, and Handling Protocols
Proper handling procedures ensure peptide stability and research validity. Laboratory standards for aod 9604 peptide storage and preparation follow strict pharmaceutical guidelines.
Storage Requirements
Lyophilized Peptide Storage
- Temperature: -20°C to -80°C for long-term storage
- Humidity: Desiccated environment essential
- Light exposure: Protected from direct light sources
- Container integrity: Sealed vials with proper labeling
- Shelf life: 24-36 months under optimal conditions
Reconstituted Peptide Storage
- Temperature: 2-8°C refrigerated storage
- Duration: Maximum 30 days post-reconstitution
- Container: Sterile, sealed vials only
- Contamination prevention: Aseptic handling procedures
- Quality verification: Regular purity assessments
The best practices for storing research peptides provide detailed guidelines for maintaining aod 9604 peptide stability throughout research studies.
Reconstitution Procedures
Proper reconstitution techniques directly impact aod 9604 peptide dosage accuracy and bioactivity:
Step-by-Step Reconstitution
- Preparation: Sterile workspace with appropriate tools
- Solvent selection: Bacteriostatic water or sterile saline
- Volume calculation: Based on desired concentration
- Mixing technique: Gentle swirling, avoid vigorous shaking
- Quality verification: Visual inspection for clarity
- Documentation: Record batch numbers and concentrations
Concentration Calculations
For a 5mg vial of aod 9604 peptide:
- 2ml reconstitution: 2.5mg/ml concentration
- 1ml reconstitution: 5mg/ml concentration
- 0.5ml reconstitution: 10mg/ml concentration
Accurate calculations ensure precise aod 9604 peptide dosage delivery throughout research protocols.
Research Applications and Protocol Variations
Different research objectives require tailored aod 9604 peptide dosage approaches. Understanding these variations helps researchers select appropriate protocols for their specific studies.
Metabolic Research Protocols
Metabolic studies utilizing aod 9604 peptide typically employ specific dosing strategies:
Short-Term Metabolic Studies (4-8 weeks)
- Dosage: 250-350 micrograms daily
- Timing: Fasting state administration
- Monitoring: Daily metabolic markers
- Endpoints: Acute metabolic responses
Long-Term Metabolic Research (12-24 weeks)
- Dosage: 300-500 micrograms daily
- Timing: Consistent daily administration
- Monitoring: Weekly comprehensive assessments
- Endpoints: Sustained metabolic adaptations
The aod9604 metabolic research protocols demonstrate the importance of matching dosage strategies to research objectives.
Comparative Research Studies
Research facilities often conduct comparative studies examining aod 9604 peptide against other compounds:
Combination Studies
- AOD 9604 + MOTS-C: Synergistic metabolic research
- AOD 9604 + 5-Amino-1MQ: Enhanced metabolic pathways
- AOD 9604 + GHK-Cu: Multi-target research approaches
These combination studies require careful aod 9604 peptide dosage adjustments to account for potential interactions. The aod9604 and mots-c combination research provides valuable insights into synergistic effects.
Quality Assurance and Sourcing Considerations
Research validity depends heavily on peptide quality and sourcing standards. Establishing reliable supply chains ensures consistent aod 9604 peptide dosage delivery throughout research programs.
Purity Standards and Testing
High-quality research requires pharmaceutical-grade peptides meeting strict purity standards:
Essential Quality Parameters
- Purity: Minimum 98% by HPLC analysis
- Sterility: Endotoxin levels <10 EU/mg
- Stability: Confirmed through accelerated aging studies
- Identity: Mass spectrometry confirmation
- Potency: Biological activity verification
Research facilities should source aod 9604 peptide for sale only from suppliers providing comprehensive certificates of analysis. The Pure Tested Peptides platform maintains rigorous quality standards for research-grade peptides.
Supplier Verification Protocols
Establishing reliable supplier relationships ensures consistent aod 9604 peptide quality:
Supplier Assessment Criteria
- Manufacturing standards: GMP-compliant facilities
- Testing capabilities: In-house analytical laboratories
- Documentation: Complete chain of custody records
- Regulatory compliance: Adherence to research regulations
- Customer support: Technical assistance availability
Research institutions should maintain detailed supplier qualification records and conduct periodic audits to ensure continued quality standards.
Troubleshooting Common Dosing Issues
Research teams may encounter various challenges when implementing aod 9604 peptide dosage protocols. Understanding common issues and solutions helps maintain research continuity.
Dosing Accuracy Problems
Issue: Inconsistent dosing measurements
Solutions:
- Calibrate measuring equipment regularly
- Use appropriate syringe sizes for volume accuracy
- Implement double-checking procedures
- Document all measurements for verification
Issue: Peptide degradation affecting potency
Solutions:
- Verify storage conditions continuously
- Monitor reconstitution procedures
- Implement quality control testing
- Establish peptide replacement schedules
Administration Challenges
Research teams may face practical challenges during aod 9604 peptide administration:
Common Issues and Solutions
- Injection site reactions: Rotate sites systematically, use proper technique
- Timing consistency: Establish standardized schedules, use reminder systems
- Participant compliance: Provide comprehensive training, maintain communication
- Documentation accuracy: Implement standardized recording procedures
The building reproducible wellness studies guide provides additional strategies for maintaining protocol consistency.
Future Directions in AOD 9604 Research
The field of aod 9604 peptide research continues evolving with new methodologies and applications emerging regularly. Understanding future directions helps research facilities prepare for advancing protocols.
Emerging Research Areas
Precision Dosing Approaches
- Individualized aod 9604 peptide dosage based on genetic factors
- Real-time monitoring systems for dosing optimization
- Artificial intelligence-guided protocol adjustments
- Personalized research methodologies
Advanced Delivery Systems
- Sustained-release formulations for extended research periods
- Targeted delivery mechanisms for enhanced specificity
- Combination therapies with complementary compounds
- Novel administration routes for improved bioavailability
Technology Integration
Modern research facilities increasingly integrate advanced technologies into aod 9604 peptide dosage protocols:
Digital Monitoring Systems
- Continuous parameter tracking through wearable devices
- Automated dosing reminders and compliance monitoring
- Real-time data analysis for protocol optimization
- Cloud-based research data management systems
Analytical Advancements
- Enhanced peptide purity testing methods
- Rapid bioactivity assessment techniques
- Improved stability monitoring systems
- Advanced pharmacokinetic modeling tools
Conclusion
Understanding proper aod 9604 peptide dosage protocols represents a critical foundation for successful research outcomes in 2026. This comprehensive guide has examined the essential elements of safe, effective peptide research, from basic dosing principles to advanced protocol considerations.
Key success factors include maintaining strict quality standards, implementing comprehensive safety monitoring, and following established dosing guidelines. Research facilities must prioritize participant safety while pursuing scientific objectives through carefully designed protocols.
The evolution of aod 9604 peptide research continues advancing our understanding of metabolic processes and therapeutic applications. By following evidence-based dosing protocols and maintaining rigorous safety standards, research teams can contribute valuable insights to this growing field.
Next Steps for Research Teams:
- Assess current protocols against established safety and dosing guidelines
- Implement quality assurance measures for peptide sourcing and handling
- Establish comprehensive monitoring systems for participant safety
- Document all procedures thoroughly for research validity
- Stay current with emerging research and protocol developments
For researchers seeking high-quality aod 9604 peptide supplies, Pure Tested Peptides offers pharmaceutical-grade compounds with comprehensive analytical testing. Their commitment to quality assurance supports the demanding requirements of professional research environments.
The future of aod 9604 peptide dosage research holds tremendous promise for advancing our understanding of metabolic science and therapeutic applications. By maintaining the highest standards of research integrity and safety, the scientific community can continue building upon this foundation of knowledge for years to come.
SEO Meta Information:
