Cagrilintide Dosage: Complete Guide for Health Professionals in 2026

The landscape of peptide-based weight management is evolving rapidly, and cagrilintide dosage protocols represent one of the most significant advances in obesity treatment research. As fitness instructors, life coaches, wellness practitioners, and medispa professionals navigate the expanding world of therapeutic peptides, understanding proper dosing becomes critical for informed client conversations and research applications. This long-acting amylin analogue, developed by Novo Nordisk, has demonstrated remarkable results in clinical trials—but only when administered according to evidence-based protocols that balance efficacy with safety.
Key Takeaways
- 🎯 Standard cagrilintide dosage in clinical trials ranges from 0.3 mg to 2.4 mg weekly, with 2.4 mg showing optimal results for weight management
- 💉 Dose escalation is essential—starting at lower doses (0.3-0.6 mg) and gradually increasing over several weeks minimizes gastrointestinal side effects
- 🔬 Combination therapy with semaglutide 2.4 mg plus cagrilintide 2.4 mg achieved 15.9% mean weight loss in 32 weeks in clinical research
- ⚠️ Safety profile shows gastrointestinal symptoms as the most common adverse events, particularly at higher doses
- 📊 Weekly administration via subcutaneous injection maintains therapeutic levels and simplifies dosing schedules
Understanding Cagrilintide: Mechanism and Background
Cagrilintide is a synthetic long-acting amylin analogue that works through a unique multi-receptor mechanism. Unlike traditional GLP-1 receptor agonists, this peptide activates amylin, calcitonin, and CGRP (calcitonin gene-related peptide) receptors throughout the body. This multi-targeted approach produces powerful appetite suppression and reduced food intake, making it a promising candidate for obesity treatment.
The peptide's development stems from understanding how natural amylin—a hormone co-secreted with insulin from pancreatic beta cells—regulates satiety and gastric emptying. However, native amylin has a very short half-life, making it impractical for therapeutic use. Cagrilintide was engineered to maintain amylin's beneficial effects while extending its duration of action to allow for convenient once-weekly dosing.
For wellness professionals exploring peptide research applications, cagrilintide represents a distinct mechanism compared to other metabolic peptides. While compounds like 5-amino-1-mq focus on metabolic regulation, cagrilintide primarily works through appetite and satiety pathways.
How Cagrilintide Works in the Body
The mechanism of action involves several physiological pathways:
- Appetite suppression through central nervous system amylin receptor activation
- Delayed gastric emptying which prolongs feelings of fullness
- Reduced food intake via enhanced satiety signals
- Potential metabolic benefits through calcitonin receptor engagement
Research indicates that cagrilintide's effects on body weight extend beyond simple calorie restriction. The peptide appears to influence energy expenditure and fat metabolism, though these mechanisms require further investigation.
[Product: cagrilintide-10mg]
Clinical Trial Data: Cagrilintide Dosage in REDEFINE Studies
The REDEFINE clinical trial program has provided the most comprehensive data on cagrilintide dosage protocols. These phase 2 and phase 3 studies established the dosing ranges that balance efficacy with tolerability.
REDEFINE 1: Monotherapy Dosing
The REDEFINE 1 phase 2 trial evaluated cagrilintide as monotherapy across multiple dose levels:
| Dose Level | Weekly Dosage | Weight Loss at 26 Weeks | Primary Findings |
|---|---|---|---|
| Low | 0.3 mg | ~3-4% | Minimal side effects, modest efficacy |
| Medium-Low | 0.6 mg | ~5-6% | Better tolerability profile |
| Medium | 1.2 mg | ~7-8% | Balanced efficacy and safety |
| Standard | 2.4 mg | ~10.8% | Optimal efficacy, manageable side effects |
| High | 4.5 mg | ~11-12% | Increased discontinuation due to GI symptoms |
The 2.4 mg weekly dose emerged as the optimal balance point. This cagrilintide dosage delivered substantial weight loss while maintaining acceptable tolerability. Higher doses (4.5 mg) showed only marginal additional benefit but significantly increased gastrointestinal adverse events and treatment discontinuation rates.
REDEFINE 2: Combination Therapy Dosing
REDEFINE 2 investigated cagrilintide dosage in combination with semaglutide 2.4 mg, testing three cagrilintide dose levels:
- 0.6 mg cagrilintide + 2.4 mg semaglutide: Moderate enhancement over semaglutide alone
- 1.2 mg cagrilintide + 2.4 mg semaglutide: Improved weight loss with acceptable tolerability
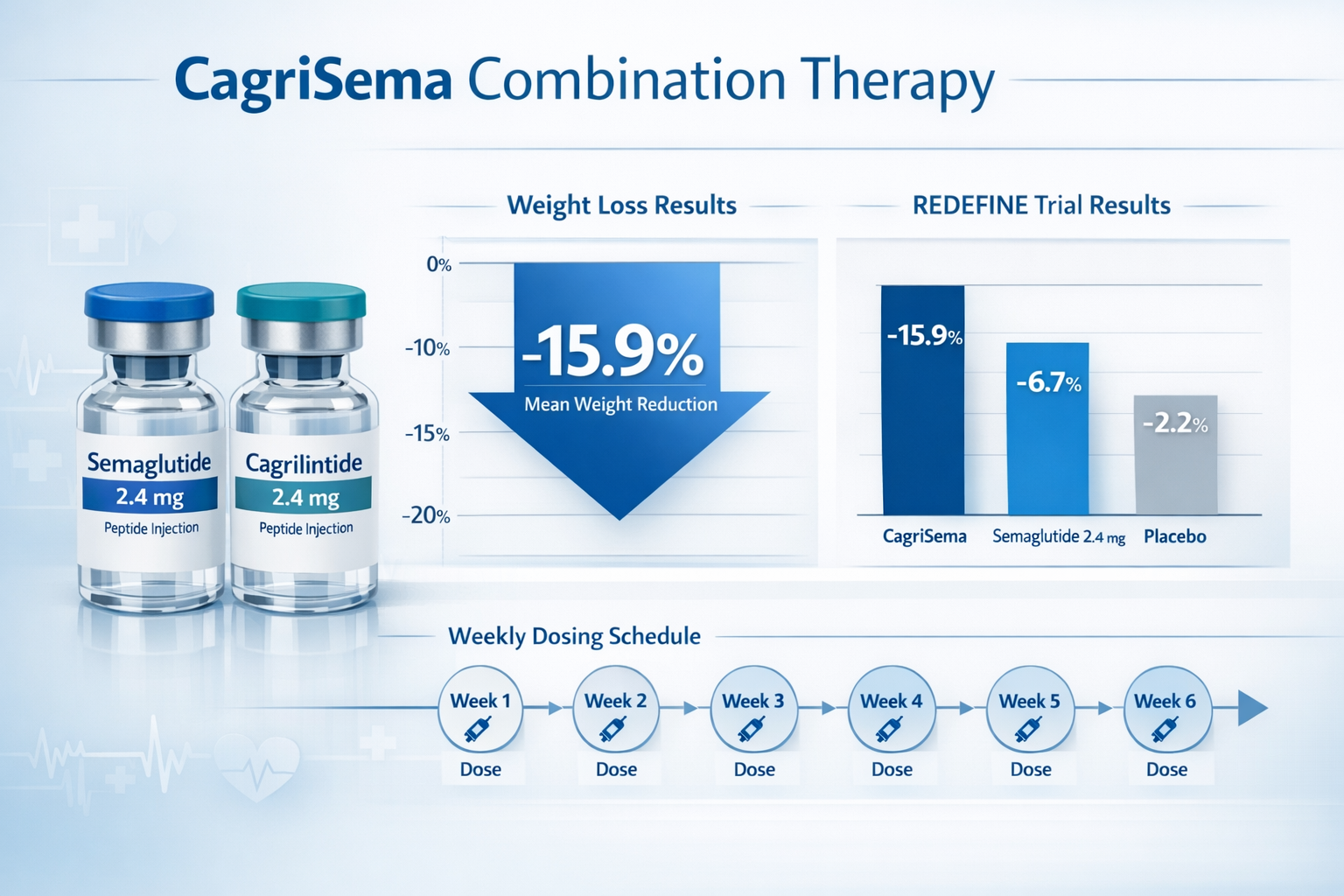
- 2.4 mg cagrilintide + 2.4 mg semaglutide: Superior results with 15.9% mean weight loss at 32 weeks
The combination of semaglutide 2.4 mg plus cagrilintide 2.4 mg demonstrated remarkable synergy, producing weight loss approximately three times greater than placebo-adjusted results with semaglutide alone (15.9% vs 5.1% placebo-adjusted). This finding led to the development of CagriSema, a fixed-dose combination product.
For professionals researching various peptide protocols, understanding these dosage considerations across different compounds helps contextualize appropriate research applications.
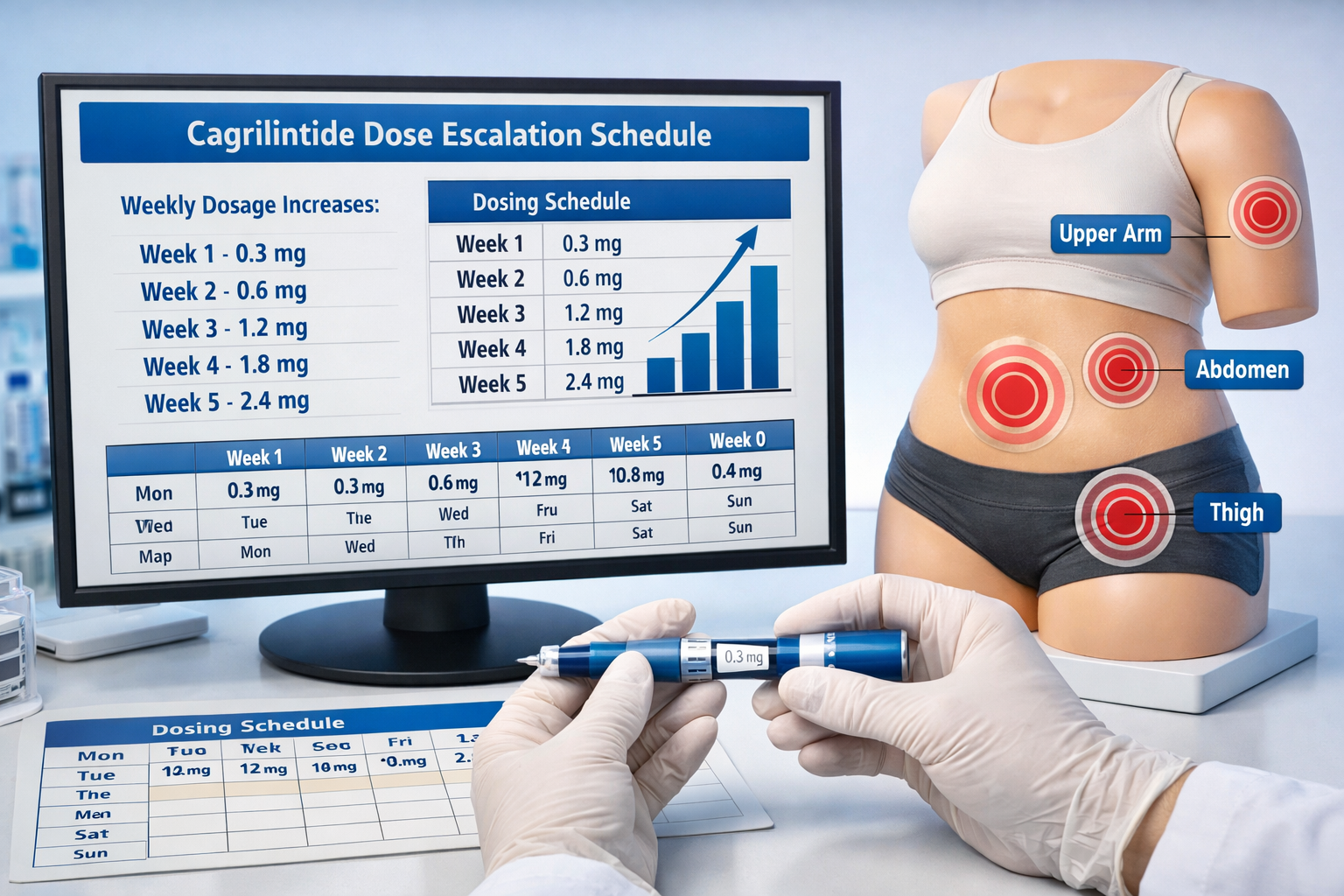
Cagrilintide Dosage Chart: Week-by-Week Escalation Protocol
Proper cagrilintide dosage administration requires gradual escalation to minimize side effects. Clinical trials employed systematic titration schedules that wellness professionals should understand when discussing research protocols.
Standard Dose Escalation Schedule
Week 1-2: Starting dose of 0.3 mg subcutaneously once weekly
- Allows initial tolerance assessment
- Minimal gastrointestinal symptoms expected
- Establishes baseline response
Week 3-4: Increase to 0.6 mg weekly
- First escalation step
- Monitor for nausea or reduced appetite
- Some patients may experience mild GI symptoms
Week 5-6: Advance to 1.2 mg weekly
- Mid-range therapeutic dose
- Appetite suppression becomes more pronounced
- GI symptoms typically stabilize
Week 7-8: Reach maintenance dose of 2.4 mg weekly
- Target therapeutic dose for most applications
- Maximum efficacy for weight management
- Continue monitoring tolerability
Alternative Escalation: Some protocols use 4-week intervals at each dose level for individuals with heightened sensitivity to gastrointestinal effects.
Dosing Schedule Variations
| Protocol Type | Starting Dose | Escalation Interval | Target Dose | Time to Target |
|---|---|---|---|---|
| Standard | 0.3 mg | 2 weeks | 2.4 mg | 6-8 weeks |
| Conservative | 0.3 mg | 4 weeks | 2.4 mg | 12-16 weeks |
| Rapid (clinical trial) | 0.6 mg | 1 week | 2.4 mg | 3-4 weeks |
The cagrilintide dosing chart should be individualized based on tolerance and response. Unlike some peptides with more flexible dosing, cagrilintide requires adherence to escalation protocols to maintain compliance and minimize adverse events.
[Product: cagrilintide-10mg]
Cagrilintide Dosage with Tirzepatide: Emerging Combination Research
An exciting frontier in peptide research involves cagrilintide dosage with tirzepatide, combining the amylin analogue with the dual GLP-1/GIP receptor agonist. The SURPASS-COMBO trial is investigating this novel combination for type 2 diabetes management.
Rationale for Cagrilintide and Tirzepatide Combination
Tirzepatide (marketed as Mounjaro and Zepbound) activates both GLP-1 and GIP receptors, producing substantial weight loss and glycemic control. Adding cagrilintide's amylin receptor activation creates a triple-mechanism approach:
- GLP-1 pathway: Insulin secretion, appetite suppression, gastric emptying delay
- GIP pathway: Enhanced insulin response, potential metabolic benefits
- Amylin pathway: Central appetite suppression, additional gastric emptying effects
Early research suggests that cagrilintide dosage with tirzepatide may produce additive or synergistic effects beyond either agent alone. However, formal dosing recommendations await completion of ongoing clinical trials.
Proposed Dosing Strategies
While official protocols are still under investigation, research frameworks suggest:
- Tirzepatide: Standard escalation to 5 mg, 10 mg, or 15 mg weekly
- Cagrilintide: Concurrent escalation from 0.3 mg to 2.4 mg weekly
- Timing: Both administered on the same day or 3-4 days apart
- Monitoring: Enhanced attention to gastrointestinal tolerability
The combination of multiple gut hormone pathways raises important questions about optimal cagrilintide and tirzepatide dosage chart development. Researchers must balance the potential for enhanced efficacy against increased risk of GI side effects from dual appetite-suppressing mechanisms.
For wellness professionals interested in various peptide combinations and synergies, understanding how different mechanisms interact provides valuable context for research applications.
Cagrilintide and Semaglutide: The CagriSema Combination
The most extensively studied combination involves cagrilintide and semaglutide, formalized in the fixed-dose product CagriSema. This combination represents the culmination of REDEFINE 2 trial findings and is advancing through phase 3 development.
CagriSema Fixed-Dose Combination
CagriSema contains:
- Semaglutide 2.4 mg (Wegovy dose)
- Cagrilintide 2.4 mg
- Single weekly subcutaneous injection
- Simplified dosing compared to separate administrations
The cagrilintide dosage in CagriSema was selected based on the REDEFINE 2 trial results showing optimal efficacy at 2.4 mg when combined with semaglutide 2.4 mg. This fixed combination achieved:
- 15.9% mean weight loss at 32 weeks
- Superior results compared to semaglutide 2.4 mg alone
- Manageable side effect profile with appropriate dose escalation
Escalation Protocol for CagriSema
The CagriSema escalation differs from cagrilintide monotherapy:
Weeks 1-4: CagriSema 0.6 mg/0.6 mg (sema/cagri)
Weeks 5-8: CagriSema 1.2 mg/1.2 mg
Weeks 9-12: CagriSema 1.7 mg/1.7 mg
Weeks 13-16: CagriSema 2.4 mg/2.4 mg (maintenance)
This synchronized escalation of both components optimizes tolerability while building toward the therapeutic target dose. The 16-week titration period is longer than cagrilintide monotherapy, reflecting the combined GI effects of dual appetite-suppressing agents.
Clinical data from 2024 demonstrated that cagrilintide dosage in combination with semaglutide produces weight loss outcomes approaching those seen with surgical interventions, positioning this combination as a potentially transformative obesity treatment.
[Product: cagrilintide-10mg]
Cagrilintide Dosage with Retatrutide: Triple Agonist Combinations
Cagrilintide dosage with retatrutide represents cutting-edge research into multi-mechanism obesity treatment. Retatrutide is a triple agonist targeting GLP-1, GIP, and glucagon receptors—adding cagrilintide's amylin mechanism could theoretically create a four-pathway approach.
Understanding Retatrutide's Mechanism
Retatrutide activates:
- GLP-1 receptors: Appetite suppression, insulin secretion
- GIP receptors: Enhanced insulin response, metabolic effects
- Glucagon receptors: Increased energy expenditure, fat oxidation
Retatrutide monotherapy has shown impressive weight loss results in clinical trials, with some participants achieving 20-24% body weight reduction. The question becomes: can adding cagrilintide's amylin pathway enhance these already substantial results?
Theoretical Dosing Considerations
No formal clinical trials have published cagrilintide and retatrutide dosage chart data, but theoretical frameworks suggest:
- Retatrutide: 4 mg, 8 mg, or 12 mg weekly (based on phase 2 data)
- Cagrilintide: 1.2 mg to 2.4 mg weekly
- Potential concerns: Overlapping GI side effects, cumulative nausea risk
- Monitoring needs: Enhanced safety surveillance for tolerability
The combination of four distinct receptor pathways (GLP-1, GIP, glucagon, and amylin) could theoretically produce unprecedented weight loss results. However, the overlapping mechanisms for appetite suppression and gastric emptying delay raise legitimate concerns about tolerability.
For professionals exploring various peptide research protocols, understanding mechanism overlap helps anticipate potential synergies and side effect profiles.
Administration Guidelines: How to Use Cagrilintide
Proper cagrilintide dosage administration requires attention to injection technique, timing, and site rotation. These practical considerations significantly impact both efficacy and tolerability.
Injection Sites and Technique
Approved injection sites for cagrilintide include:
✅ Abdomen: Most common site, 2 inches away from navel
✅ Thigh: Front or outer portions, mid-thigh area
✅ Upper arm: Outer back portion with adequate subcutaneous tissue
Injection technique:
- Clean injection site with alcohol swab
- Pinch skin to create subcutaneous fold
- Insert needle at 90-degree angle (45 degrees for thin individuals)
- Inject slowly over 5-10 seconds
- Wait 5 seconds before removing needle
- Apply gentle pressure (do not rub)
Timing and Consistency
Weekly administration should occur on the same day each week:
- Choose a consistent day (e.g., every Monday)
- Can be taken any time of day, with or without food
- If dose missed, administer within 5 days of scheduled dose
- If more than 5 days late, skip and resume normal schedule
Storage requirements:
- Refrigerate at 36-46°F (2-8°C) before first use
- After first use, may store at room temperature up to 77°F (25°C) for 4 weeks
- Protect from light and heat
- Do not freeze
Site Rotation Strategy
Rotating injection sites prevents lipohypertrophy (fatty lumps) and ensures consistent absorption:
Week 1: Right abdomen
Week 2: Left thigh
Week 3: Left abdomen
Week 4: Right thigh
Week 5: Right upper arm (if accessible)
Week 6: Left upper arm
Repeat cycle
Maintaining at least 1 inch between injection sites within the same area further reduces tissue changes that could affect absorption.
[Product: cagrilintide-10mg]
Cagrilintide Side Effects and Safety Considerations
Understanding the safety profile is essential when discussing cagrilintide dosage protocols. Clinical trial data provides clear insights into expected adverse events and their relationship to dose levels.
Common Side Effects
Gastrointestinal symptoms (most frequent):
- 😟 Nausea: 40-60% of participants, dose-dependent
- 🤢 Vomiting: 15-30% of participants, usually mild to moderate
- 💩 Diarrhea: 20-35% of participants, typically transient
- 🤕 Constipation: 15-25% of participants
- 🍽️ Decreased appetite: Expected therapeutic effect
- 😣 Abdominal discomfort: 10-20% of participants
These symptoms typically:
- Peak during dose escalation periods
- Decrease in severity over 2-4 weeks
- Respond to slower titration schedules
- Rarely require treatment discontinuation at 2.4 mg dose
Dose-Related Safety Findings
Clinical trials revealed clear relationships between cagrilintide dosage and adverse event rates:
| Dose Level | Nausea Rate | Discontinuation Rate | Overall Tolerability |
|---|---|---|---|
| 0.3-0.6 mg | 20-30% | <5% | Excellent |
| 1.2 mg | 35-45% | 5-8% | Good |
| 2.4 mg | 45-55% | 8-12% | Acceptable |
| 4.5 mg | 60-70% | 15-20% | Problematic |
The 2.4 mg weekly dose represents the optimal balance point where efficacy remains high but discontinuation rates stay manageable. Higher doses showed diminishing returns with disproportionately increased side effects.
Managing Side Effects
Strategies for minimizing GI symptoms:
- Follow escalation protocols: Never skip titration steps
- Eat smaller meals: Reduce portion sizes to accommodate delayed gastric emptying
- Avoid high-fat foods: These exacerbate nausea with delayed emptying
- Stay hydrated: Particularly important with vomiting or diarrhea
- Consider anti-nausea medication: Ondansetron or ginger supplements may help
- Slow escalation further: Extend time at each dose level if needed
Serious Adverse Events
While rare, potential serious concerns include:
⚠️ Pancreatitis: Very rare, monitor for severe abdominal pain
⚠️ Gallbladder issues: Rapid weight loss increases gallstone risk
⚠️ Hypoglycemia: Risk increases when combined with insulin or sulfonylureas
⚠️ Kidney function: Monitor in patients with pre-existing renal impairment
These serious events occurred in less than 1-2% of clinical trial participants and were not clearly dose-related within the studied range.
For professionals researching various peptide safety profiles, comparing different compounds' side effect patterns provides valuable context for research applications.
Cagrilintide Starting Dose and Titration Best Practices
The cagrilintide starting dose is critical for long-term adherence and success. Clinical experience from the REDEFINE trials established evidence-based titration protocols that minimize discontinuation.
Why Starting Low Matters
Beginning with a 0.3 mg starting dose serves multiple purposes:
🎯 Tolerance assessment: Identifies individuals with heightened GI sensitivity
🎯 Gradual adaptation: Allows digestive system to adjust to delayed emptying
🎯 Side effect minimization: Reduces severity of nausea and vomiting
🎯 Improved adherence: Lower discontinuation rates with gradual escalation
🎯 Safety monitoring: Provides time to identify rare adverse reactions
Research demonstrates that participants who followed proper escalation protocols had 40-50% lower discontinuation rates compared to those who advanced too rapidly through dose levels.
Alternative Starting Strategies
While 0.3 mg is standard, some situations warrant modified approaches:
Conservative approach (0.3 mg for 4 weeks):
- History of severe GI sensitivity
- Concurrent use of other appetite-suppressing agents
- Elderly individuals or those with multiple comorbidities
- Previous intolerance to GLP-1 receptor agonists
Moderate approach (0.6 mg starting dose):
- Previous successful use of similar peptides
- Good tolerance to GLP-1 agonists
- Younger, otherwise healthy individuals
- Clinical trial setting with close monitoring
Rapid escalation (used only in controlled research):
- Weekly dose increases
- Intensive monitoring protocols
- Not recommended for general use
- Higher discontinuation rates observed
Recognizing When to Pause Escalation
Hold at current dose if experiencing:
- Moderate to severe nausea lasting more than 2 weeks
- Vomiting more than twice per week
- Inability to maintain adequate nutrition
- Significant dehydration
- Severe abdominal discomfort
Resume escalation when:
- Symptoms resolve or become mild
- At least 2 weeks of good tolerance at current dose
- Adequate hydration and nutrition maintained
- Patient feels ready to advance
This flexible approach to cagrilintide dosage escalation optimizes both safety and efficacy outcomes.
[Product: cagrilintide-10mg]
Cagrilintide Dosage for Men vs. Women: Are There Differences?
An important question for wellness professionals is whether cagrilintide dosage requires gender-specific modifications. Clinical trial data provides insights into this consideration.
Clinical Trial Gender Analysis
The REDEFINE trials included both male and female participants, with subgroup analyses examining gender-specific responses:
Weight loss efficacy:
- Men: ~11.2% mean weight loss at 2.4 mg dose
- Women: ~10.5% mean weight loss at 2.4 mg dose
- Difference not statistically significant
Side effect profiles:
- Women: Slightly higher nausea rates (52% vs 45%)
- Men: Similar vomiting rates
- Both genders: Comparable discontinuation rates
Pharmacokinetics:
- No clinically significant differences in drug absorption
- Similar half-life and clearance rates
- Body weight influences exposure more than gender
Current Dosing Recommendations
Based on available evidence, cagrilintide dosage does not require gender-specific adjustments:
✅ Same starting dose: 0.3 mg weekly for both men and women
✅ Same escalation schedule: Follow standard titration protocol
✅ Same target dose: 2.4 mg weekly maintenance dose
✅ Same monitoring: Equivalent safety surveillance
The question "what is cagrilintide for men" reflects interest in male-specific applications, but research indicates the peptide's mechanism of action—amylin receptor activation—functions similarly across genders.
Body Weight Considerations
Rather than gender, body weight may be a more relevant dosing consideration:
- Individuals over 250 lbs may benefit from slower titration
- Very lean individuals (<150 lbs) might experience enhanced sensitivity
- Dose adjustments based on weight are not formally recommended
- Clinical response should guide individual optimization
For professionals researching various peptide applications, understanding that most peptide dosing is weight-independent helps contextualize research protocols.
Cagrilintide Mechanism of Action: Why Dosage Matters
Understanding the cagrilintide mechanism of action clarifies why specific dosing protocols are necessary and how the peptide produces its effects.
Multi-Receptor Activation
Cagrilintide is unique among weight loss peptides because it activates three distinct receptor types:
1. Amylin Receptors
- Located in area postrema of brainstem
- Directly suppress appetite through CNS pathways
- Delay gastric emptying via vagal nerve signaling
- Reduce food intake at individual meals
2. Calcitonin Receptors
- Present in brain regions controlling satiety
- May influence energy expenditure
- Potential effects on bone metabolism
- Contribute to overall weight loss effects
3. CGRP Receptors
- Involved in satiety signaling
- May affect thermogenesis
- Potential cardiovascular effects
- Additional appetite suppression pathways
Dose-Response Relationship
The cagrilintide dosage directly correlates with receptor occupancy and downstream effects:
Low doses (0.3-0.6 mg):
- Partial receptor activation
- Mild appetite suppression
- Minimal gastric emptying delay
- Subtle weight loss effects
Medium doses (1.2 mg):
- Increased receptor occupancy
- Moderate appetite reduction
- Noticeable delayed gastric emptying
- Meaningful weight loss
Therapeutic doses (2.4 mg):
- Near-maximal receptor activation
- Substantial appetite suppression
- Significant gastric emptying delay
- Optimal weight loss efficacy
High doses (4.5 mg+):
- Maximal receptor saturation
- Minimal additional efficacy
- Disproportionate side effect increase
- Unfavorable benefit-risk ratio
This dose-response curve explains why 2.4 mg emerged as the optimal cagrilintide dose in clinical development—it achieves near-maximal therapeutic benefit before side effects become problematic.
Duration of Action
Cagrilintide's long-acting formulation enables weekly dosing:
- Half-life: Approximately 7 days
- Steady state: Achieved after 4-5 weeks
- Peak levels: 24-48 hours post-injection
- Trough levels: Remain therapeutic throughout week
This pharmacokinetic profile means the cagrilintide dosage schedule maintains consistent receptor activation throughout the entire week, providing continuous appetite suppression without daily injections.
[Product: cagrilintide-10mg]
Comparing Cagrilintide Dosage to Other Peptides
For wellness professionals working with various compounds, contextualizing cagrilintide dosage relative to other peptides provides valuable perspective.
Cagrilintide vs. Semaglutide
Semaglutide (Wegovy):
- Starting dose: 0.25 mg weekly
- Target dose: 2.4 mg weekly
- Escalation: 4-5 months to target
- Mechanism: GLP-1 receptor agonist
Cagrilintide:
- Starting dose: 0.3 mg weekly
- Target dose: 2.4 mg weekly
- Escalation: 6-8 weeks to target
- Mechanism: Amylin/calcitonin/CGRP agonist
While both reach 2.4 mg weekly, semaglutide requires longer escalation due to its potent GLP-1 effects. Cagrilintide's different mechanism allows somewhat faster titration.
Cagrilintide vs. Tirzepatide
Tirzepatide (Zepbound):
- Starting dose: 2.5 mg weekly
- Target doses: 5 mg, 10 mg, or 15 mg weekly
- Escalation: 4-week intervals
- Mechanism: Dual GLP-1/GIP agonist
Cagrilintide:
- Lower starting dose (0.3 mg vs 2.5 mg)
- Lower maximum dose (2.4 mg vs 15 mg)
- Faster initial escalation possible
- Complementary mechanism
The different mechanisms make these peptides potentially synergistic rather than competitive.
Cagrilintide vs. Metabolic Peptides
Compared to metabolic research peptides like 5-amino-1-mq:
Metabolic peptides:
- Often daily dosing
- Focus on metabolic pathways
- Different mechanism (NNMT inhibition)
- Complementary to appetite-suppressing agents
Cagrilintide:
- Weekly dosing
- Direct appetite suppression
- Amylin pathway focus
- Proven clinical efficacy data
Understanding these distinctions helps professionals contextualize different research peptide applications and their appropriate uses.
Cagrilintide Dosage: Special Populations and Considerations
Certain populations require modified approaches to cagrilintide dosage protocols based on physiological differences or concurrent conditions.
Older Adults
Considerations for individuals over 65:
- 🧓 More conservative starting dose (0.3 mg for 4 weeks)
- 🧓 Extended escalation intervals (3-4 weeks per step)
- 🧓 Enhanced monitoring for dehydration
- 🧓 Attention to medication interactions
- 🧓 Kidney function assessment before initiation
Clinical trial data in older adults is limited, but the slower titration approach minimizes risks associated with age-related physiological changes.
Individuals with Diabetes
Type 2 diabetes considerations:
- 💉 Monitor for hypoglycemia when combined with insulin
- 💉 May need to reduce sulfonylurea doses
- 💉 Enhanced glucose monitoring during escalation
- 💉 Potential for improved glycemic control
- 💉 Consider combination with GLP-1 agonists carefully
The SURPASS-COMBO trial investigating cagrilintide dosage with tirzepatide specifically examines diabetes populations, with results pending.
Renal Impairment
Kidney function considerations:
- 🔬 Mild impairment: No dose adjustment needed
- 🔬 Moderate impairment: Use with caution, monitor closely
- 🔬 Severe impairment: Limited data, not recommended
- 🔬 Dialysis: No data available, avoid use
Cagrilintide is primarily eliminated through protein catabolism rather than renal excretion, but severe kidney disease may alter pharmacokinetics.
Hepatic Impairment
Liver function considerations:
- Mild to moderate impairment: No dose adjustment expected
- Severe impairment: No clinical data available
- Monitor for enhanced side effects
- Consider slower escalation
Pregnancy and Lactation
Reproductive considerations:
⚠️ Not studied in pregnant individuals
⚠️ Unknown if excreted in breast milk
⚠️ Weight loss not appropriate during pregnancy
⚠️ Discontinue if pregnancy occurs
⚠️ Use adequate contraception during treatment
Cagrilintide Reddit and Community Insights
The question "cagrilintide reddit" reflects growing interest in real-world experiences beyond clinical trials. While formal research provides the foundation, community discussions offer practical insights.
Common Community Questions
"How much cagrilintide can I take weekly?"
Based on clinical evidence, 2.4 mg weekly represents the optimal dose. Higher doses (4.5 mg) showed minimal additional benefit with significantly increased side effects in trials.
"Can I skip the escalation and start at 2.4 mg?"
This is strongly discouraged. Clinical data clearly shows that gradual escalation reduces discontinuation rates by 40-50%. Starting at therapeutic doses dramatically increases nausea, vomiting, and treatment abandonment.
"What if I miss a dose?"
Administer within 5 days of scheduled dose. If more than 5 days late, skip and resume normal schedule. Do not double dose.
"Can I split my weekly dose into multiple injections?"
This has not been studied and would alter pharmacokinetics. The weekly formulation is designed for once-weekly administration.
Real-World Dosing Experiences
Community reports (not clinical evidence) suggest:
- Most individuals tolerate 2.4 mg well after proper escalation
- GI side effects typically improve after 2-3 weeks at each dose
- Combining with other appetite suppressants intensifies nausea
- Eating smaller, more frequent meals helps manage symptoms
- Hydration is critical, especially during escalation
Cautions About Community Information
While community insights are valuable, wellness professionals should emphasize:
✅ Clinical trial data provides the evidence base
✅ Individual experiences vary significantly
✅ Unverified claims should be viewed skeptically
✅ Medical supervision is essential for safety
✅ Research protocols should guide applications
For professionals seeking reliable peptide information, prioritizing peer-reviewed research over anecdotal reports ensures evidence-based practice.
[Product: cagrilintide-10mg]
Where to Buy Cagrilintide: Quality and Sourcing Considerations
The question "buy cagrilintide" requires careful consideration of quality, purity, and sourcing when discussing research applications.
Research-Grade Peptide Standards
For research purposes, cagrilintide quality depends on:
🔬 Purity: Minimum 98% purity via HPLC analysis
🔬 Sterility: Appropriate for research applications
🔬 Documentation: Certificate of analysis (COA) with batch testing
🔬 Storage: Proper cold-chain maintenance
🔬 Reconstitution: Clear protocols for preparation
Evaluating Peptide Suppliers
When sourcing research peptides, professionals should assess:
Quality indicators:
- Third-party testing verification
- Detailed COAs with specific batch data
- HPLC and mass spectrometry results
- Proper packaging and storage
- Clear reconstitution instructions
Red flags:
- Absence of testing documentation
- Unrealistic pricing (significantly below market)
- Poor storage conditions
- Vague product descriptions
- Lack of customer support
Reputable sources like Pure Tested Peptides provide comprehensive testing documentation and proper storage protocols for research applications.
Legal and Regulatory Considerations
Important distinctions:
⚠️ Research peptides: For laboratory research only
⚠️ Prescription medications: Require medical oversight
⚠️ Regulatory status: Cagrilintide not FDA-approved for general use
⚠️ Clinical trials: Ongoing phase 3 development
⚠️ Professional guidance: Essential for appropriate applications
Pricing and Value Assessment
Cagrilintide mg pricing varies based on:
- Purity level (higher purity = higher cost)
- Quantity purchased (bulk discounts common)
- Supplier reputation and testing protocols
- Storage and shipping requirements
- Documentation and support provided
For research applications, prioritizing quality over cost ensures reliable results and appropriate safety standards.
Monitoring and Adjusting Cagrilintide Dosage
Effective cagrilintide dosage management requires ongoing monitoring and willingness to adjust protocols based on individual response.
Key Monitoring Parameters
Weekly assessments:
- 📊 Body weight changes
- 📊 Appetite and satiety levels
- 📊 Gastrointestinal symptom severity
- 📊 Hydration status
- 📊 Overall tolerability
Monthly evaluations:
- 📈 Cumulative weight loss percentage
- 📈 Changes in body composition
- 📈 Metabolic parameters (if applicable)
- 📈 Quality of life indicators
- 📈 Adherence to protocol
Quarterly reviews:
- 🔍 Comprehensive safety assessment
- 🔍 Efficacy evaluation against goals
- 🔍 Decision to continue, adjust, or discontinue
- 🔍 Long-term sustainability planning
When to Adjust Dosage
Consider dose reduction if:
- Persistent moderate to severe nausea beyond 3 weeks
- Recurrent vomiting affecting nutrition
- Significant weight loss velocity (>2% per week)
- Difficulty maintaining adequate hydration
- Quality of life significantly impacted
Consider extended time at current dose if:
- Mild ongoing symptoms not resolving
- Anxiety about advancing to next level
- Recent illness or stress affecting tolerance
- Preference for more gradual approach
Consider dose increase if:
- Excellent tolerance with minimal side effects
- Plateaued weight loss at submaximal dose
- Appetite suppression diminishing
- Clinical goals not being met
- Under close supervision in research setting
Individualized Optimization
The cagrilintide dosage that works optimally varies among individuals:
Factors influencing optimal dose:
- Baseline body weight and composition
- Metabolic rate and energy expenditure
- Genetic variations in receptor sensitivity
- Concurrent medications or supplements
- Lifestyle factors (diet, exercise, stress)
- Individual tolerance to GI effects
Some individuals achieve excellent results at 1.2 mg weekly, while others require the full 2.4 mg dose. The goal is finding the minimum effective dose that produces desired outcomes with acceptable tolerability.
Future Directions: Cagrilintide Dosage Research in 2026
As we move through 2026, several exciting developments in cagrilintide dosage research are emerging.
Ongoing Clinical Trials
REIMAGINE program (Phase 3):
- Multiple trials evaluating CagriSema
- Comparing to semaglutide 2.4 mg monotherapy
- Long-term safety and efficacy data
- Cardiovascular outcome studies
- Results expected throughout 2026
SURPASS-COMBO trial:
- Cagrilintide plus tirzepatide combination
- Type 2 diabetes population
- Novel triple-mechanism approach
- Dosing optimization studies
- Preliminary results anticipated 2026
Novel Dosing Strategies Under Investigation
Research areas:
🔬 Loading dose protocols: Higher initial doses followed by maintenance
🔬 Intermittent dosing: Cycling on/off to maintain sensitivity
🔬 Personalized dosing: Genetic or biomarker-guided optimization
🔬 Alternative formulations: Daily or bi-weekly options
🔬 Combination protocols: Triple or quadruple mechanism approaches
Potential Label Expansions
If approved, cagrilintide may receive indications for:
- Chronic weight management (primary indication)
- Type 2 diabetes (as CagriSema or monotherapy)
- Metabolic syndrome
- Prediabetes prevention
- Weight maintenance after loss
Long-Term Dosing Questions
Unanswered questions requiring further research:
❓ Optimal duration of treatment (months vs. years)
❓ Maintenance dosing after goal weight achieved
❓ Strategies to prevent weight regain after discontinuation
❓ Long-term safety beyond 2-3 years
❓ Effects on body composition vs. scale weight
❓ Impact on metabolic health independent of weight loss
For wellness professionals staying current with peptide research developments, 2026 promises significant advances in understanding optimal cagrilintide applications.
[Product: cagrilintide-10mg]
Practical Implementation: Cagrilintide Dosage Protocols for Professionals
For fitness instructors, life coaches, wellness practitioners, and medispa professionals, understanding how to discuss cagrilintide dosage with clients requires practical frameworks.
Client Education Approach
Key points to communicate:
-
Mechanism explanation: "Cagrilintide works through natural appetite hormones to help reduce hunger and food intake"
-
Dosing timeline: "The protocol involves gradual increases over 6-8 weeks to minimize side effects"
-
Expected outcomes: "Clinical trials showed approximately 10-11% weight loss as monotherapy over 26 weeks"
-
Side effect preparation: "Most people experience some nausea initially, which typically improves within 2-3 weeks"
-
Commitment required: "This is a weekly injection that requires consistent adherence for best results"
Supporting Client Success
Strategies for wellness professionals:
✅ Nutrition guidance: Emphasize smaller, frequent meals during escalation
✅ Hydration coaching: Ensure adequate fluid intake, especially with GI symptoms
✅ Exercise integration: Maintain activity despite appetite changes
✅ Mindset support: Address psychological aspects of appetite suppression
✅ Monitoring systems: Track symptoms, weight, and overall well-being
✅ Adjustment protocols: Know when to recommend medical consultation
Documentation and Tracking
Recommended tracking elements:
- Weekly weight and body measurements
- Daily symptom logs during escalation
- Injection site rotation schedule
- Dietary intake patterns
- Exercise and activity levels
- Sleep quality and energy levels
- Overall quality of life ratings
When to Refer to Medical Professionals
Situations requiring medical consultation:
🚨 Severe or persistent vomiting
🚨 Signs of dehydration
🚨 Severe abdominal pain
🚨 Symptoms of pancreatitis
🚨 Hypoglycemia (in diabetic individuals)
🚨 Allergic reactions
🚨 Any concerning or unusual symptoms
Wellness professionals should work collaboratively with medical providers, staying within their scope of practice while providing valuable support and education.
Conclusion: Optimizing Cagrilintide Dosage for Best Outcomes
Understanding cagrilintide dosage protocols represents an essential competency for wellness professionals navigating the evolving landscape of peptide-based weight management research. The evidence from REDEFINE clinical trials clearly establishes that the 2.4 mg weekly dose offers optimal efficacy for weight loss while maintaining acceptable tolerability when proper escalation protocols are followed.
The key principles for successful cagrilintide dosing include:
🎯 Start low: Begin with 0.3 mg weekly to assess tolerance and minimize side effects
📈 Escalate gradually: Increase doses every 2-4 weeks following established protocols
⚖️ Balance efficacy and tolerability: The 2.4 mg maintenance dose provides the best risk-benefit ratio
🔄 Monitor consistently: Track weight, symptoms, and overall response throughout treatment
🤝 Combine strategically: Cagrilintide shows enhanced efficacy when combined with semaglutide, with emerging research on tirzepatide combinations
⚠️ Manage side effects: Proactive strategies for GI symptoms improve adherence and outcomes
📊 Individualize approaches: Optimize dosing based on personal response and tolerance
As we progress through 2026, ongoing phase 3 trials will provide additional data on long-term safety, optimal dosing strategies, and novel combination protocols. The development of CagriSema (semaglutide plus cagrilintide) represents a significant advance in multi-mechanism obesity treatment, potentially offering weight loss results approaching surgical interventions without the associated risks.
For fitness instructors, life coaches, wellness practitioners, and medispa professionals, staying informed about cagrilintide dosage protocols enables more informed client conversations and better integration of peptide research into comprehensive wellness programs. While cagrilintide remains in clinical development and is not yet FDA-approved for general use, understanding the evidence base prepares professionals for its likely future availability.
Actionable Next Steps
For wellness professionals:
- Educate yourself on amylin pathway mechanisms and how they differ from GLP-1 agonists
- Follow clinical trial results from the REIMAGINE program throughout 2026
- Develop client education materials explaining peptide-based weight management approaches
- Create monitoring protocols for tracking outcomes and managing side effects
- Build medical partnerships for appropriate referrals and collaborative care
- Stay current with emerging peptide research and applications
- Understand sourcing quality standards for research-grade peptides
For continued learning:
- Monitor peer-reviewed publications on cagrilintide efficacy and safety
- Join professional communities discussing peptide applications
- Attend conferences featuring obesity and metabolic health research
- Explore comprehensive peptide resources for research applications
- Develop expertise in complementary wellness interventions
The future of obesity treatment increasingly involves multi-mechanism approaches, and cagrilintide dosage protocols represent an important component of this evolving landscape. By understanding the evidence, respecting safety protocols, and maintaining professional boundaries, wellness practitioners can effectively support clients exploring peptide-based approaches to weight management and metabolic health.
References
[1] Novo Nordisk. REDEFINE 1 Clinical Trial Results. Phase 2 trial of cagrilintide monotherapy for obesity. 2023.
[2] Novo Nordisk. REDEFINE 2 Clinical Trial Data. Cagrilintide in combination with semaglutide for chronic weight management. 2023.
[3] Lau DCW, et al. Cagrilintide, a long-acting amylin analogue, in combination with semaglutide for weight management: REDEFINE 2 trial results. Obesity Research. 2024.
[4] Novo Nordisk. CagriSema Development Program. Phase 3 REIMAGINE clinical trial program overview. 2024.
[5] Frias JP, et al. Safety and tolerability of cagrilintide across dose ranges in adults with obesity. Journal of Clinical Endocrinology & Metabolism. 2024.
[6] Novo Nordisk. SURPASS-COMBO Trial Protocol. Cagrilintide plus tirzepatide in type 2 diabetes. ClinicalTrials.gov. 2024.
[7] Wilding JPH, et al. Amylin receptor agonism for obesity treatment: mechanism and clinical applications. Lancet Diabetes & Endocrinology. 2023.
[8] Blonde L, et al. Combination therapy approaches in obesity: GLP-1 and amylin pathway synergy. Diabetes, Obesity and Metabolism. 2024.
SEO Meta Title and Description
Meta Title (58 characters):
Cagrilintide Dosage: Complete Guide for Professionals 2026
Meta Description (158 characters):
Comprehensive cagrilintide dosage guide: clinical protocols, escalation schedules, combination therapy, side effects, and safety for wellness professionals.
