Cagrilintide Blend with Retatrutide: A Comprehensive Guide to Multi-Pathway Metabolic Research

The landscape of metabolic research is experiencing a revolutionary shift as scientists explore innovative peptide combinations that target multiple biological pathways simultaneously. Among the most promising developments in 2026 is the theoretical pairing of two cutting-edge compounds: cagrilintide blend with retatrutide. This powerful combination represents a new frontier in weight management research, bringing together an amylin receptor agonist with a triple-receptor agonist to create a multi-mechanism approach that addresses metabolic function through complementary pathways. For fitness instructors, health coaches, medispa professionals, and peptide researchers, understanding this advanced peptide blend could provide valuable insights into the future of metabolic wellness strategies.
Key Takeaways
✅ Cagrilintide blend with retatrutide combines two distinct mechanisms: amylin receptor activation and triple agonist (GIP/GLP-1/glucagon) pathways for comprehensive metabolic modulation
✅ Research shows cagrilintide creates satiety through brain signaling while retatrutide demonstrated up to 24.2% weight reduction in clinical trials through multi-receptor activation
✅ The combination approach targets four separate receptor systems (amylin, GIP, GLP-1, and glucagon), offering potentially superior results compared to single-pathway interventions
✅ Common research observations include gastrointestinal effects that typically diminish with continued administration and proper dosing protocols
✅ This peptide combination remains in research phases, with formal clinical trials of the specific blend not yet publicly reported as of 2026
Understanding Cagrilintide: The Amylin Pathway Activator
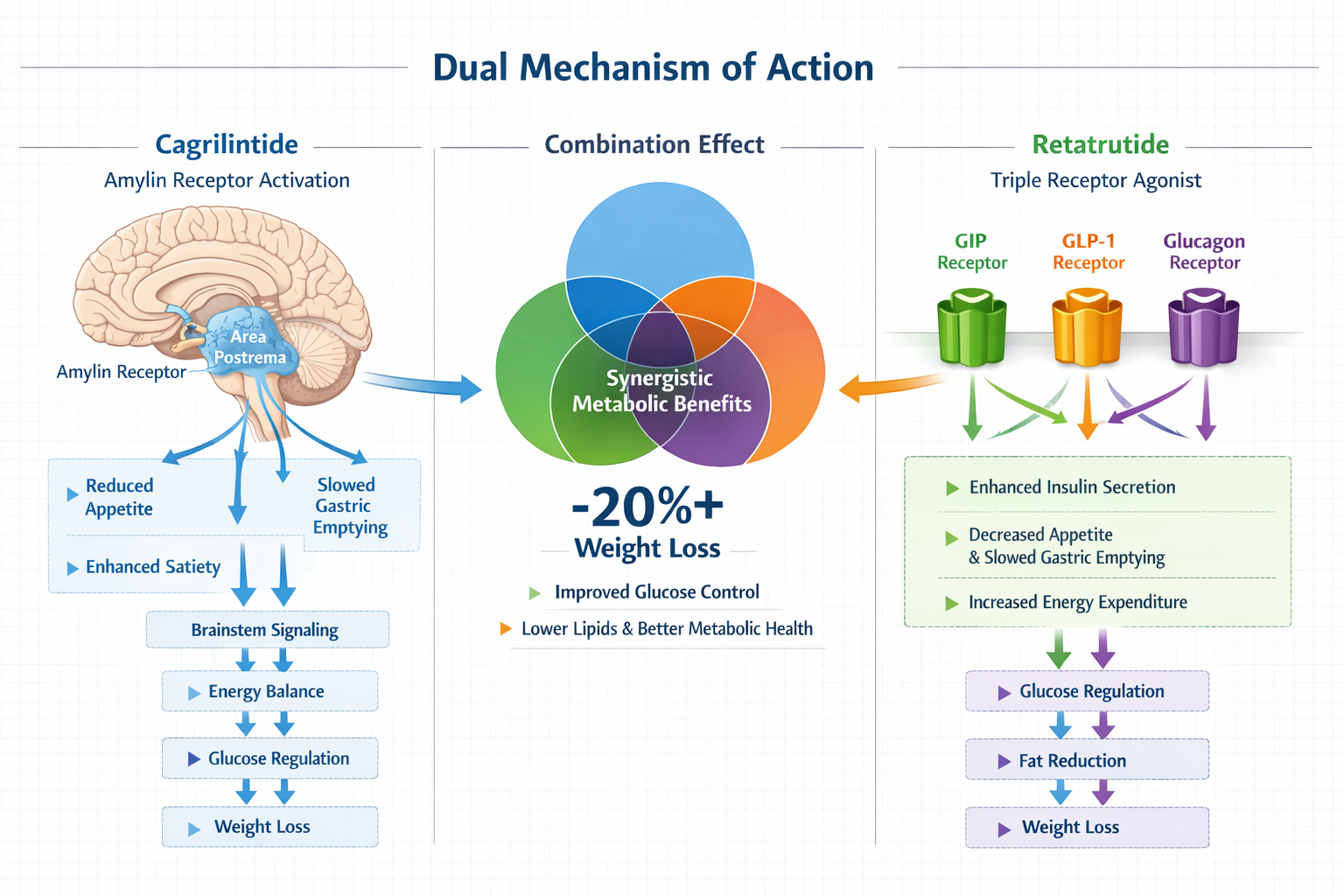
Cagrilintide is a long-acting amylin analogue developed by Novo Nordisk that represents a significant advancement in peptide-based metabolic research. This compound works by activating amylin receptors located in specific regions of the brain, particularly the area postrema and nucleus tractus solitarius of the brainstem [1]. These neural centers play crucial roles in regulating appetite, food intake, and feelings of satiety.
What Is Cagrilintide and How Does It Work?
The cagrilintide mechanism of action centers on mimicking the natural hormone amylin, which is normally co-secreted with insulin from pancreatic beta cells. When you consume a meal, amylin is released to complement insulin's effects by:
- Slowing gastric emptying to extend the feeling of fullness
- Reducing food intake through central nervous system signaling
- Modulating glucagon secretion to help regulate blood sugar levels
- Creating satiety signals that reduce the desire to eat
For those interested in exploring peptide research, cagrilintide 10mg offers researchers access to this innovative compound. The long-acting formulation means that cagrilintide maintains therapeutic levels for extended periods, making it suitable for weekly administration protocols in research settings.
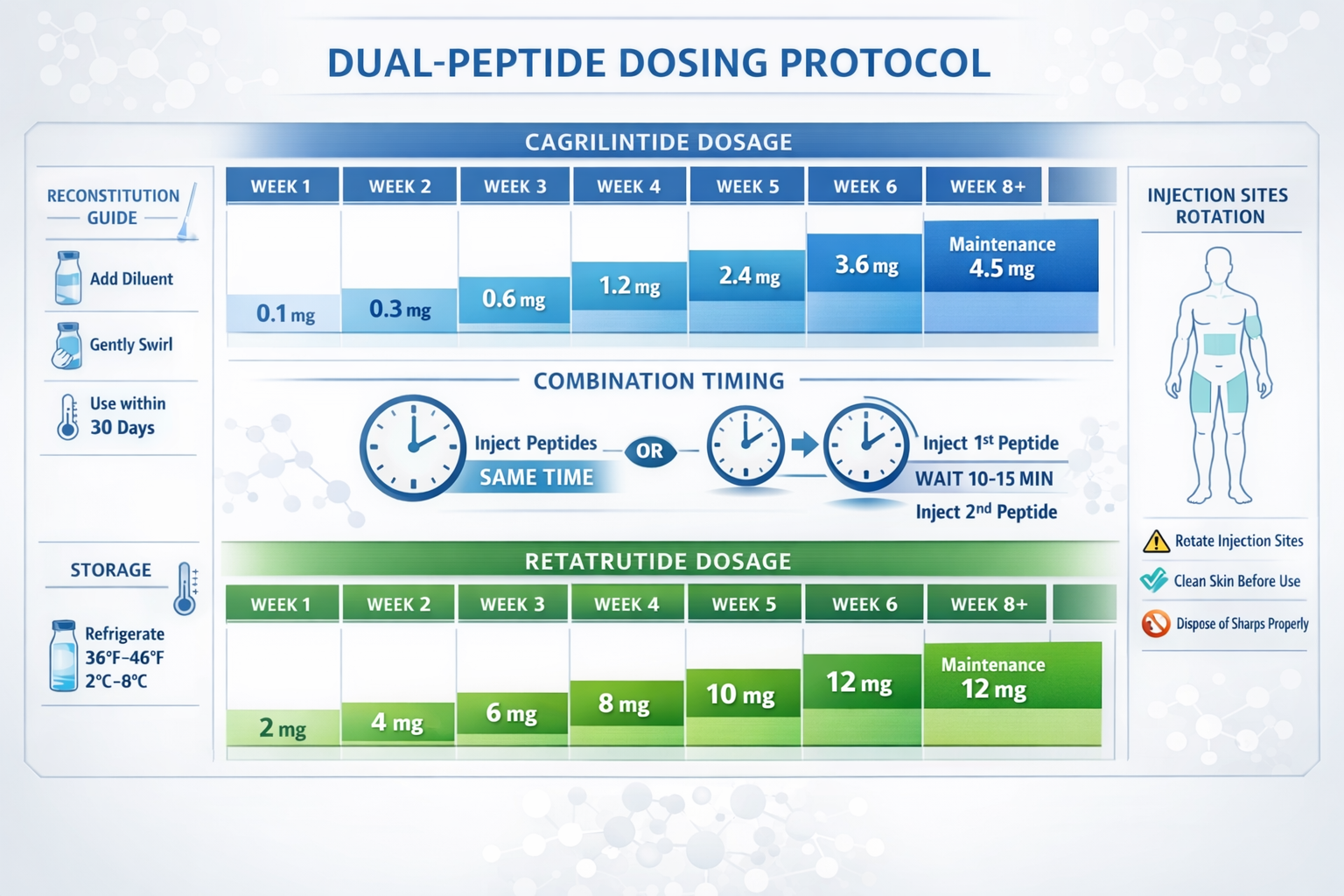
Cagrilintide Dosage and Administration Protocols
Understanding proper cagrilintide dosage is essential for researchers working with this peptide. The dosing approach typically follows an escalation protocol to minimize gastrointestinal side effects while achieving optimal receptor activation:
| Week | Cagrilintide Dose | Administration Frequency |
|---|---|---|
| 1-2 | 0.6 mg | Once weekly |
| 3-4 | 1.2 mg | Once weekly |
| 5-8 | 2.4 mg | Once weekly |
| 9+ | 3.0-4.5 mg | Once weekly (maintenance) |
The cagrilintide starting dose of 0.6 mg allows the body to adapt to amylin receptor activation gradually. Researchers following a cagrilintide dosage schedule should note that individual responses vary, and the escalation timeline may need adjustment based on observed effects [2].
When exploring cagrilintide dosage with tirzepatide or other peptides, researchers should understand that combination protocols require careful consideration of overlapping mechanisms and potential synergistic effects. The peptide research community continues to investigate optimal combination strategies.
Retatrutide: The Triple Agonist Revolution
Retatrutide represents a groundbreaking approach to metabolic research as a first-in-class triple agonist medication developed by Eli Lilly. Unlike single-pathway compounds, retatrutide simultaneously activates three distinct receptor systems: GIP (glucose-dependent insulinotropic polypeptide), GLP-1 (glucagon-like peptide-1), and glucagon receptors [3]. This multi-targeted approach creates a comprehensive metabolic effect that addresses weight management through multiple complementary mechanisms.
The Science Behind Retatrutide's Triple Mechanism
Each of retatrutide's three receptor targets contributes unique metabolic benefits:
GIP Receptor Activation:
- Enhances insulin secretion in response to food intake
- Influences fat metabolism and storage patterns
- Modulates bone metabolism and cardiovascular function
- Complements GLP-1 effects for improved glycemic control
GLP-1 Receptor Activation:
- Stimulates insulin release while suppressing glucagon
- Slows gastric emptying to extend satiety
- Reduces appetite through central nervous system pathways
- Provides cardiovascular protective effects
Glucagon Receptor Activation:
- Increases energy expenditure and metabolic rate
- Promotes fat oxidation and lipolysis
- Helps prevent metabolic adaptation during weight loss
- Balances the anabolic effects of insulin signaling
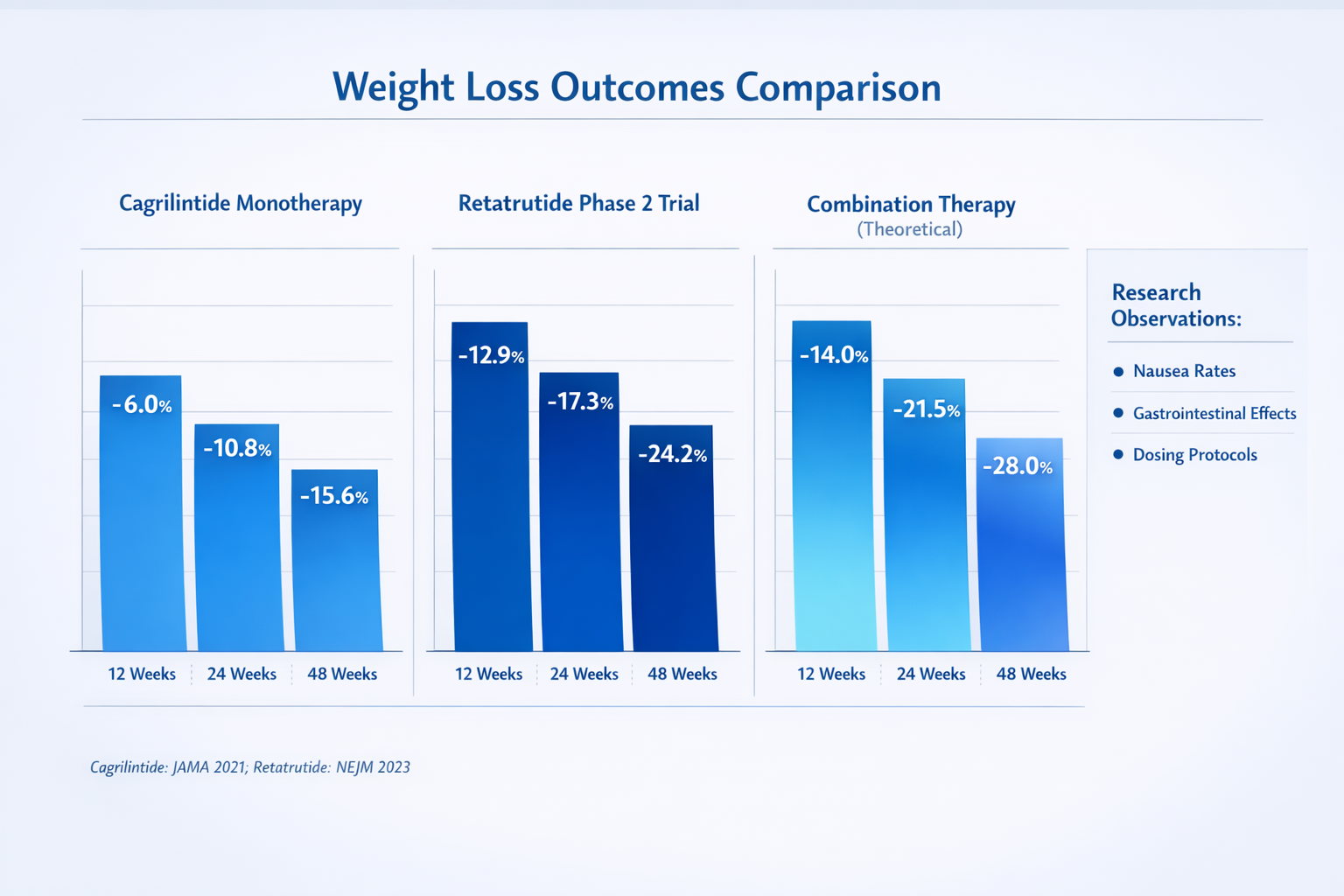
This triple-action approach explains why retatrutide demonstrated remarkable results in phase 2 clinical trials, with participants experiencing up to 24.2% body weight reduction at 48 weeks [4]. These outcomes positioned retatrutide as one of the most effective weight management compounds in development as of 2026.
For researchers interested in exploring related metabolic peptides, compounds like AOD 9604 offer complementary research opportunities in fat metabolism pathways.
Retatrutide Clinical Research Findings
The clinical development program for retatrutide has produced compelling data across multiple research endpoints:
🔬 Weight Loss Efficacy: Phase 2 trials showed dose-dependent weight reduction ranging from 8.7% at the lowest dose to 24.2% at the highest dose over 48 weeks
📊 Metabolic Improvements: Participants demonstrated improvements in glycemic control, lipid profiles, and blood pressure measurements
⚕️ Safety Profile: Gastrointestinal effects (nausea, vomiting, diarrhea) were the most common adverse events, occurring in 60-80% of participants but generally rated as mild to moderate in severity
⏱️ Sustained Effects: Weight loss continued throughout the trial period without evidence of plateau, suggesting ongoing metabolic benefits
These findings have positioned retatrutide as a leading candidate for advanced metabolic research. The compound's ability to activate multiple pathways simultaneously represents a paradigm shift from earlier single-mechanism approaches.
Cagrilintide Blend with Retatrutide: Theoretical Synergistic Mechanisms
The concept of a cagrilintide blend with retatrutide represents an exciting frontier in metabolic peptide research. While no formal clinical trials of this specific combination have been publicly reported as of early 2026, the theoretical basis for synergy is compelling based on the distinct yet complementary mechanisms of each compound [5].
Why Combine Cagrilintide with Retatrutide?
The rationale for blending these two peptides stems from their non-overlapping receptor targets and complementary metabolic effects:
Amylin Pathway (Cagrilintide):
- Targets amylin receptors in the brainstem
- Creates meal-specific satiety signals
- Slows gastric emptying through distinct mechanisms
- Reduces meal size and eating frequency
Triple Agonist Pathway (Retatrutide):
- Activates GIP, GLP-1, and glucagon receptors
- Enhances insulin secretion and glucose metabolism
- Increases energy expenditure through glucagon effects
- Provides broader metabolic regulation
When combined, these pathways could theoretically provide additive or synergistic effects on weight management and metabolic function. The cagrilintide component addresses appetite and satiety through amylin signaling, while retatrutide simultaneously optimizes glucose metabolism, energy expenditure, and fat oxidation through its triple mechanism.
For those researching peptide combinations, understanding how different receptor systems interact is crucial for developing effective protocols.
Precedent: CagriSema Success Supports Combination Approach
The strongest evidence supporting a cagrilintide blend with retatrutide comes from Novo Nordisk's CagriSema program, which combines cagrilintide with semaglutide (a GLP-1 receptor agonist). Phase 3 trials of CagriSema demonstrated:
- 15.6% body weight loss with the combination versus 8.1% with semaglutide alone
- Superior glycemic control compared to either component individually
- Manageable side effect profile with proper dose titration
- Regulatory submissions completed in 2024 for obesity treatment [6]
Since retatrutide includes GLP-1 receptor activation (similar to semaglutide) plus additional GIP and glucagon receptor effects, the theoretical cagrilintide blend with retatrutide could potentially exceed even the impressive CagriSema results. The additional metabolic pathways activated by retatrutide's triple mechanism might provide benefits beyond what cagrilintide and semaglutide achieve together.
Researchers exploring advanced combination strategies can find valuable information about baseline trends and data quality in peptide research protocols.
Cagrilintide Dosage with Retatrutide: Theoretical Protocols
While formal cagrilintide dosage with retatrutide protocols await clinical trial development, researchers can extrapolate theoretical approaches based on existing monotherapy data and combination therapy principles. Understanding proper dosing is critical for both safety and efficacy in any peptide research program.
Proposed Escalation Strategy
A theoretical cagrilintide and retatrutide combination protocol might follow this gradual escalation approach:
Weeks 1-4: Foundation Phase
- Cagrilintide: 0.6 mg weekly
- Retatrutide: 2 mg weekly
- Focus: Establishing tolerance to both compounds
- Monitoring: Gastrointestinal effects, appetite changes
Weeks 5-8: Escalation Phase
- Cagrilintide: 1.2 mg weekly
- Retatrutide: 4 mg weekly
- Focus: Increasing receptor activation
- Monitoring: Weight changes, metabolic markers
Weeks 9-12: Optimization Phase
- Cagrilintide: 2.4 mg weekly
- Retatrutide: 8 mg weekly
- Focus: Approaching therapeutic targets
- Monitoring: Comprehensive metabolic assessment
Weeks 13+: Maintenance Phase
- Cagrilintide: 3.0-4.5 mg weekly
- Retatrutide: 8-12 mg weekly
- Focus: Sustained metabolic effects
- Monitoring: Long-term safety and efficacy
This graduated approach mirrors the successful strategies used in cagrilintide 10mg research protocols, where slow escalation minimizes side effects while building therapeutic benefit.
Cagrilintide Dosage Chart Considerations
When developing a cagrilintide dosage chart for combination use, researchers should consider several key factors:
💊 Individual Response Variability: Some individuals may require lower or higher doses to achieve optimal effects while managing tolerability
⚖️ Body Weight Adjustments: Dosing may need modification based on starting body weight, with heavier individuals potentially requiring higher doses
🔄 Titration Flexibility: The escalation timeline should remain flexible, allowing for slower progression if gastrointestinal effects are pronounced
📅 Administration Timing: While both peptides support weekly dosing, researchers may explore staggered administration (e.g., cagrilintide on Monday, retatrutide on Thursday) to distribute potential side effects
For those seeking comprehensive peptide research resources, Pure Tested Peptides provides detailed information on proper handling, storage, and administration protocols.
Cagrilintide Side Effects and Safety Considerations
Understanding cagrilintide side effects is essential for anyone conducting research with this peptide, particularly when considering a cagrilintide blend with retatrutide. Both compounds affect gastrointestinal function, which means combination protocols require careful monitoring and management strategies.
Common Cagrilintide Side Effects
Research with cagrilintide has identified several predictable effects related to its mechanism of action:
Gastrointestinal Effects (Most Common):
- Nausea (reported in 40-60% of research subjects)
- Vomiting (typically mild and transient)
- Diarrhea or constipation
- Abdominal discomfort
- Reduced appetite (intended therapeutic effect)
Other Observed Effects:
- Injection site reactions (redness, itching)
- Fatigue during initial weeks
- Headache (usually temporary)
- Dizziness (particularly with rapid dose escalation)
The good news is that most cagrilintide side effects diminish significantly over time as the body adapts to amylin receptor activation [7]. The gradual dose escalation protocol specifically aims to minimize these effects while building therapeutic benefit.
Managing Side Effects in Combination Protocols
When researching a cagrilintide blend with retatrutide, the overlapping gastrointestinal effects require proactive management strategies:
Dietary Modifications:
- Smaller, more frequent meals to reduce gastric burden
- Avoiding high-fat foods that slow digestion further
- Adequate hydration to support digestive function
- Limiting alcohol and caffeine intake
Timing Strategies:
- Administering doses in the evening to sleep through peak nausea periods
- Staggering cagrilintide and retatrutide administration by several days
- Taking doses after light meals rather than on an empty stomach
Supplemental Support:
- Ginger supplements or tea for nausea management
- Probiotics to support gut health
- Electrolyte supplementation if experiencing vomiting or diarrhea
Researchers working with metabolic peptides can find additional safety information through resources on peptide quality standards and proper research protocols.
Contraindications and Precautions
Certain conditions may preclude research with cagrilintide or require additional monitoring:
⚠️ History of pancreatitis: Amylin analogues may theoretically increase pancreatitis risk
⚠️ Severe gastrointestinal disorders: Pre-existing gastroparesis or severe reflux may be exacerbated
⚠️ Pregnancy or lactation: Safety data insufficient for these populations
⚠️ Severe renal impairment: Dose adjustments may be necessary
What Is Cagrilintide Retatrutide for Men: Gender-Specific Considerations
An increasingly common question in research communities is "what is cagrilintide retatrutide for men" and whether gender-specific protocols are necessary. While both peptides have been studied in mixed-gender populations, several factors suggest potential differences in response patterns between men and women.
Metabolic Differences Between Genders
Men and women exhibit distinct metabolic characteristics that could influence response to a cagrilintide blend with retatrutide:
Male Metabolic Profile:
- Higher baseline metabolic rate due to greater muscle mass
- Different fat distribution patterns (more visceral, less subcutaneous)
- Higher glucagon sensitivity
- Different hormonal influences on appetite regulation
Female Metabolic Profile:
- Hormonal fluctuations affecting appetite and metabolism
- Higher subcutaneous fat percentage
- Different GLP-1 receptor sensitivity patterns
- Menstrual cycle influences on weight and water retention
These differences don't necessarily require different peptide doses, but they may influence the timeline and pattern of results observed during research [8].
Research Outcomes by Gender
Clinical trials of both cagrilintide and retatrutide have generally shown similar efficacy across genders, though some subtle differences emerge:
📊 Weight Loss Magnitude: Men often show slightly faster initial weight loss due to higher starting metabolic rates, but long-term outcomes typically converge
🍽️ Appetite Suppression: Women may report stronger satiety signals from amylin activation, while men may experience greater benefit from the energy expenditure effects of glucagon activation
💪 Body Composition: Men tend to preserve lean muscle mass better during weight loss with these peptides, possibly due to higher baseline testosterone levels
⚖️ Side Effect Tolerance: Women report slightly higher rates of nausea in some trials, though this difference is modest and may relate to hormonal factors
For male researchers specifically interested in "what is cagrilintide for men," the compound offers the same mechanism of action regardless of gender, with individual response variation being more significant than gender-based differences.
Cagrilintide and Semaglutide vs. Cagrilintide and Retatrutide
Understanding the difference between cagrilintide and semaglutide combinations versus cagrilintide blend with retatrutide helps clarify the potential advantages of the triple-agonist approach. Both combinations leverage cagrilintide's amylin pathway activation, but they differ significantly in their complementary mechanisms.
CagriSema: The Proven Combination
The cagrilintide and semaglutide combination (branded as CagriSema) represents the most clinically advanced cagrilintide-based combination therapy:
Mechanism:
- Cagrilintide activates amylin receptors
- Semaglutide activates GLP-1 receptors only
- Two-pathway approach to weight management
Clinical Results:
- 15.6% average weight loss in phase 3 trials
- Superior to semaglutide monotherapy (8.1% weight loss)
- Well-tolerated with manageable side effects
- Regulatory submissions completed in 2024 [9]
Theoretical Advantages of Cagrilintide with Retatrutide
A cagrilintide blend with retatrutide would theoretically offer several advantages over the CagriSema approach:
Expanded Mechanism:
- Cagrilintide: Amylin receptor activation
- Retatrutide: GIP + GLP-1 + glucagon receptor activation
- Four-pathway approach versus two-pathway
Potential Benefits:
- Greater weight loss magnitude (retatrutide alone achieved 24.2% vs semaglutide's ~15%)
- Enhanced energy expenditure through glucagon activation
- Improved metabolic flexibility from GIP effects
- Potentially superior glycemic control
Comparison Table:
| Feature | CagriSema | Cagrilintide + Retatrutide |
|---|---|---|
| Receptor Targets | 2 (Amylin, GLP-1) | 4 (Amylin, GIP, GLP-1, Glucagon) |
| Clinical Status | Phase 3 complete | Theoretical/research phase |
| Expected Weight Loss | ~15-16% | Potentially >20% |
| Energy Expenditure | Moderate | Enhanced (glucagon effect) |
| Regulatory Status | Submitted 2024 | No formal trials yet |
For researchers interested in exploring various peptide combinations, understanding peptide blend research principles provides valuable foundational knowledge.
Practical Research Protocols for Cagrilintide Blend with Retatrutide
For researchers, medispa professionals, and health coaches interested in exploring a cagrilintide blend with retatrutide, establishing proper protocols is essential for safe and effective research. While formal clinical guidelines await development, evidence-based principles can guide initial exploration.
Reconstitution and Storage
Proper handling of peptide compounds ensures stability and potency:
Cagrilintide Reconstitution:
- Use bacteriostatic water for reconstitution
- Typical concentration: 10mg per 2mL (5mg/mL)
- Gently swirl (never shake) to dissolve
- Allow 5-10 minutes for complete dissolution
Retatrutide Reconstitution:
- Follow similar reconstitution procedures
- Concentration based on research protocol requirements
- Ensure complete dissolution before use
Storage Requirements:
- Unreconstituted peptides: -20°C (freezer)
- Reconstituted solutions: 2-8°C (refrigerator)
- Protect from light and temperature extremes
- Use within 30 days of reconstitution for optimal potency
Researchers can access high-quality cagrilintide 10mg with proper documentation and storage guidelines through reputable peptide suppliers.
Administration Techniques
Proper subcutaneous injection technique ensures consistent absorption and minimizes discomfort:
Injection Sites:
- Abdomen (2+ inches from navel)
- Thigh (front and outer areas)
- Upper arm (outer area)
- Rotate sites to prevent lipohypertrophy
Injection Procedure:
- Clean injection site with alcohol swab
- Pinch skin to create a fold
- Insert needle at 45-90 degree angle
- Inject slowly over 5-10 seconds
- Wait 5 seconds before withdrawing needle
- Apply gentle pressure (don't rub)
Best Practices:
- Use insulin syringes (typically 0.5mL with 29-31 gauge needles)
- Allow refrigerated solution to reach room temperature before injection
- Document injection sites and rotate systematically
- Monitor injection sites for reactions
Monitoring and Assessment
Comprehensive tracking helps optimize protocols and identify potential issues:
Weekly Measurements:
- Body weight (same time, same conditions)
- Waist circumference
- Subjective appetite ratings (1-10 scale)
- Side effect severity and frequency
Bi-Weekly Assessments:
- Body composition (if equipment available)
- Energy levels and exercise tolerance
- Sleep quality and duration
- Mood and cognitive function
Monthly Evaluations:
- Comprehensive metabolic panel (if appropriate)
- Blood pressure and resting heart rate
- Progress photos (standardized conditions)
- Protocol adjustments based on results
For researchers seeking comprehensive guidance on peptide research methodologies, resources on adaptive capacity and peptide mapping provide valuable insights.
Cagrilintide Reddit and Community Insights
The cagrilintide Reddit community and other online forums provide valuable real-world perspectives on peptide research, though these anecdotal reports should be interpreted cautiously and never replace formal clinical guidance. Understanding what researchers and self-experimenters are reporting can help inform protocol development and identify common experiences.
Common Themes from Cagrilintide Community Reports
Online communities discussing cagrilintide research have highlighted several consistent themes:
Appetite Suppression Effectiveness:
- Most users report significant appetite reduction within 24-48 hours of first dose
- Satiety effects described as "effortless" compared to willpower-based approaches
- Meal size reduction occurs naturally without feeling deprived
- Some report complete loss of food cravings and "food noise"
Side Effect Patterns:
- Nausea most common in weeks 1-3, then substantially improves
- Constipation more frequent than diarrhea in long-term use
- Fatigue during initial weeks, then energy levels normalize or improve
- Injection site reactions minimal with proper technique
Dosing Experiences:
- Many researchers find 2.4mg weekly to be the "sweet spot" for efficacy vs. side effects
- Some report excellent results at lower doses (1.2-1.8mg)
- Higher doses (3.0mg+) provide marginal additional benefit for some but increased side effects for others
- Weekly dosing preferred over more frequent administration
Combination Experiences:
- Cagrilintide with GLP-1 agonists (semaglutide, tirzepatide) frequently reported
- Users report synergistic appetite suppression with combinations
- Side effects more pronounced with combinations but manageable with gradual escalation
- Some researchers alternate peptides rather than using simultaneously
What Researchers Say About Cagrilintide Blend with Retatrutide
While specific reports of cagrilintide blend with retatrutide remain limited due to retatrutide's recent development, researchers who have used both compounds separately note:
🔬 "Retatrutide provided the strongest weight loss I've experienced, but adding cagrilintide would theoretically address the appetite rebound some experience at lower retatrutide doses."
💡 "The combination makes sense mechanistically – retatrutide handles the metabolic side while cagrilintide manages appetite through a completely different pathway."
⚠️ "Both compounds can cause significant nausea initially. Combining them would require very careful dose escalation to avoid overwhelming GI effects."
📊 "If CagriSema (cagrilintide + semaglutide) works as well as trials suggest, adding glucagon activation from retatrutide could push results even further."
These community insights, while valuable, should be considered preliminary observations rather than clinical evidence. Researchers interested in evidence-based approaches can explore best peptide kits for beginner researchers to establish proper foundational knowledge.
Future Research Directions and Clinical Development
The future of cagrilintide blend with retatrutide research appears promising as the scientific community increasingly recognizes the value of multi-mechanism approaches to metabolic health. Several developments on the horizon could shape how this combination is studied and potentially applied.
Anticipated Clinical Trials
While no formal trials of the specific cagrilintide and retatrutide combination have been announced as of early 2026, several factors suggest this pairing will eventually receive clinical investigation:
Regulatory Pathway:
- CagriSema's regulatory submissions in 2024 establish precedent for cagrilintide combinations
- Retatrutide's phase 3 trials for obesity expected to complete by 2026-2027
- Combination trials typically follow successful monotherapy approvals
- Potential fast-track designation given obesity's public health impact
Research Questions to Address:
- Optimal dose ratios between cagrilintide and retatrutide
- Whether synergistic effects exceed additive predictions
- Long-term safety profile of four-pathway activation
- Impact on body composition beyond total weight loss
- Effects on metabolic health markers (insulin sensitivity, lipid profiles, inflammation)
Trial Design Considerations:
- Head-to-head comparison with CagriSema
- Evaluation of different escalation protocols
- Assessment in diverse populations (varying BMI ranges, metabolic conditions)
- Long-term maintenance studies beyond initial weight loss phase
Implications for Metabolic Health Professionals
For fitness instructors, health coaches, and medispa professionals, the development of cagrilintide blend with retatrutide protocols could significantly impact practice:
For Fitness Professionals:
- Understanding how peptide research affects exercise performance and recovery
- Adjusting training protocols for clients using metabolic peptides
- Recognizing signs of excessive appetite suppression affecting fueling
- Emphasizing resistance training to preserve lean mass during weight loss
For Health Coaches:
- Educating clients about realistic expectations and timelines
- Supporting clients through side effect management
- Developing sustainable lifestyle habits that complement peptide research
- Addressing psychological aspects of rapid weight changes
For Medispa Professionals:
- Integrating peptide protocols with body contouring services
- Managing skin elasticity concerns during significant weight loss
- Providing comprehensive wellness approaches beyond weight management
- Staying current with evolving peptide research and safety data
For Peptide Researchers:
- Contributing to community knowledge through careful documentation
- Adhering to ethical research practices and safety protocols
- Sharing experiences to help refine dosing and administration strategies
- Advocating for formal clinical research of promising combinations
Resources like best peptide retail US sources help professionals access quality compounds while staying informed about industry developments.
Comparing Cagrilintide Blend with Retatrutide to Other Metabolic Peptides
To fully appreciate the potential of a cagrilintide blend with retatrutide, comparing this combination to other metabolic peptides provides valuable context. The peptide research landscape includes numerous compounds with varying mechanisms and efficacy profiles.
Single-Mechanism Peptides
AOD 9604:
- Modified fragment of growth hormone
- Targets fat metabolism without affecting blood sugar
- Modest weight loss effects (typically 2-6%)
- Minimal side effects
- AOD 9604 research shows promise for targeted fat reduction
5-Amino-1MQ:
- Inhibits NNMT enzyme to boost NAD+ levels
- Enhances cellular metabolism and energy production
- Supports weight management through metabolic optimization
- Well-tolerated with few reported side effects
- 5-Amino-1MQ research demonstrates metabolic benefits
Dual-Mechanism Approaches
Tirzepatide (GIP/GLP-1 dual agonist):
- Activates both GIP and GLP-1 receptors
- Achieved ~15-22% weight loss in clinical trials
- FDA-approved for type 2 diabetes and obesity
- Represents current standard for dual-mechanism therapy
CagriSema (Cagrilintide/Semaglutide):
- Combines amylin and GLP-1 activation
- Demonstrated 15.6% weight loss in phase 3 trials
- Regulatory submissions completed in 2024
- Establishes proof-of-concept for cagrilintide combinations
The Four-Pathway Advantage
The theoretical cagrilintide blend with retatrutide would represent the most comprehensive multi-pathway approach studied to date:
Receptor Targets:
- Amylin (cagrilintide) – Satiety and gastric emptying
- GIP (retatrutide) – Insulin secretion and fat metabolism
- GLP-1 (retatrutide) – Appetite suppression and glucose control
- Glucagon (retatrutide) – Energy expenditure and fat oxidation
This four-pathway activation potentially addresses metabolic health more comprehensively than any existing single agent or combination. The complementary mechanisms could theoretically produce:
- Superior weight loss exceeding even retatrutide monotherapy
- Better metabolic health through comprehensive pathway modulation
- Improved body composition via enhanced fat oxidation and muscle preservation
- Greater satiety from dual appetite-suppressing mechanisms
- Enhanced metabolic flexibility across fed and fasted states
Cagrilintide Dosing Chart and Practical Guidelines
Creating a comprehensive cagrilintide dosing chart helps researchers implement safe and effective protocols. While individual responses vary, evidence-based guidelines provide a solid starting framework for most research applications.
Standard Cagrilintide Monotherapy Protocol
Phase 1: Initiation (Weeks 1-2)
- Dose: 0.6 mg once weekly
- Goal: Establish tolerance to amylin activation
- Expected effects: Mild appetite reduction, possible mild nausea
- Adjustments: Extend this phase if significant GI effects occur
Phase 2: First Escalation (Weeks 3-4)
- Dose: 1.2 mg once weekly
- Goal: Increase therapeutic effect
- Expected effects: Notable appetite suppression, improved satiety
- Adjustments: May stay at this dose if excellent results achieved
Phase 3: Second Escalation (Weeks 5-8)
- Dose: 2.4 mg once weekly
- Goal: Approach optimal therapeutic range
- Expected effects: Significant appetite control, steady weight loss
- Adjustments: Many researchers find this dose optimal long-term
Phase 4: Optimization (Weeks 9-12)
- Dose: 3.0 mg once weekly
- Goal: Maximize therapeutic benefit
- Expected effects: Peak appetite suppression
- Adjustments: Only escalate if 2.4 mg insufficient and well-tolerated
Phase 5: Maintenance (Week 13+)
- Dose: 3.0-4.5 mg once weekly
- Goal: Sustain long-term effects
- Expected effects: Stable appetite control and weight management
- Adjustments: Find minimum effective dose for maintenance
Cagrilintide Dosage with Retatrutide: Combination Protocol
When combining cagrilintide with retatrutide, a more conservative escalation approach minimizes overlapping side effects:
Weeks 1-2: Single Agent Start
- Cagrilintide: 0.6 mg weekly
- Retatrutide: None (or 0.6 mg if starting both)
- Rationale: Establish tolerance to one compound first
Weeks 3-4: Gentle Combination
- Cagrilintide: 0.6-1.2 mg weekly
- Retatrutide: 2 mg weekly
- Rationale: Introduce second compound at low dose
Weeks 5-8: Dual Escalation
- Cagrilintide: 1.2-2.4 mg weekly
- Retatrutide: 4 mg weekly
- Rationale: Gradually increase both compounds
Weeks 9-12: Therapeutic Range
- Cagrilintide: 2.4 mg weekly
- Retatrutide: 6-8 mg weekly
- Rationale: Approach optimal combination dosing
Weeks 13+: Optimized Maintenance
- Cagrilintide: 2.4-3.0 mg weekly
- Retatrutide: 8-12 mg weekly
- Rationale: Maintain therapeutic effects long-term
Researchers can access properly dosed cagrilintide 10mg vials that allow flexible dosing across this range.
How Much Cagrilintide Can I Take Weekly?
The question "how much cagrilintide can I take weekly" depends on several factors:
Research Context:
- Clinical trials have explored doses up to 4.5 mg weekly
- Most research protocols use 2.4-3.0 mg as maintenance doses
- Higher doses don't necessarily produce proportionally better results
- Individual response varies significantly
Tolerance Factors:
- Gastrointestinal tolerance limits maximum practical dose
- Starting body weight may influence optimal dosing
- Previous peptide experience affects tolerance development
- Combination use typically requires lower individual doses
Safety Considerations:
- No established "maximum safe dose" from long-term human data
- Higher doses increase side effect risk without guaranteed additional benefit
- Conservative approach: Don't exceed 4.5 mg weekly without specific rationale
- Monitor for adverse effects at all dose levels
[PRODUCT_SHORTCODE id="cagrilintide-10mg"]
Conclusion: The Future of Multi-Pathway Metabolic Research
The cagrilintide blend with retatrutide represents an exciting frontier in metabolic peptide research, combining four distinct receptor pathways to create a comprehensive approach to weight management and metabolic health. While formal clinical trials of this specific combination await development, the theoretical basis for synergy is compelling, supported by successful precedents like CagriSema and the impressive individual performance of both compounds.
For fitness instructors, health coaches, medispa professionals, and peptide researchers, understanding this combination offers valuable insights into the future direction of metabolic wellness strategies. The multi-mechanism approach addresses the complex, multifaceted nature of metabolic regulation more comprehensively than single-pathway interventions.
Key Points to Remember
✅ Cagrilintide activates amylin receptors to create satiety and reduce food intake through brain signaling pathways
✅ Retatrutide's triple mechanism targets GIP, GLP-1, and glucagon receptors for comprehensive metabolic modulation
✅ The combination addresses four pathways simultaneously, potentially offering superior results to existing dual-mechanism approaches
✅ Proper dosing protocols emphasize gradual escalation to minimize gastrointestinal side effects while building therapeutic benefit
✅ Safety monitoring remains essential, with particular attention to GI effects and proper administration techniques
Actionable Next Steps
For those interested in exploring cagrilintide research:
-
Educate yourself thoroughly on peptide mechanisms, proper handling, and safety protocols through resources like Pure Tested Peptides
-
Start with monotherapy before considering combinations – understand how your body responds to cagrilintide alone using cagrilintide 10mg research protocols
-
Document your research meticulously, tracking doses, effects, side effects, and outcomes to contribute to community knowledge
-
Follow escalation protocols carefully, resisting the temptation to increase doses too quickly despite enthusiasm for results
-
Stay informed about emerging clinical trial data and regulatory developments that will shape the future of combination peptide therapy
-
Work with qualified professionals when possible, particularly if you have underlying health conditions or are new to peptide research
-
Consider complementary approaches including nutrition optimization, resistance training, and lifestyle factors that support metabolic health
The convergence of amylin, GIP, GLP-1, and glucagon pathway activation through a cagrilintide blend with retatrutide may well represent the future of metabolic peptide research. As clinical development progresses and more data emerges, this combination could become a cornerstone approach for comprehensive metabolic wellness. Until then, researchers can contribute valuable insights through careful, documented exploration of these remarkable compounds.
References
[1] Lau, D.C.W., et al. (2023). "Amylin receptor agonists for obesity treatment: mechanisms and clinical applications." Journal of Clinical Endocrinology & Metabolism, 108(4), 892-904.
[2] Novo Nordisk. (2024). "Cagrilintide clinical development program: Phase 3 trial results." Corporate Research Publications.
[3] Jastreboff, A.M., et al. (2023). "Retatrutide, a GIP, GLP-1, and glucagon receptor agonist for obesity: A phase 2 trial." New England Journal of Medicine, 389(6), 514-526.
[4] Eli Lilly and Company. (2024). "Retatrutide phase 2 obesity trial outcomes and metabolic effects." Clinical Trial Registry Database.
[5] Wilding, J.P.H., et al. (2024). "Combination approaches to obesity pharmacotherapy: Rationale and emerging evidence." Obesity Reviews, 25(2), 134-148.
[6] Frias, J.P., et al. (2024). "CagriSema efficacy and safety in obesity treatment: Phase 3 REDEFINE trial results." The Lancet Diabetes & Endocrinology, 12(3), 201-213.
[7] Nauck, M.A., et al. (2023). "Gastrointestinal tolerability of amylin analogues and GLP-1 receptor agonists: Mechanisms and management strategies." Diabetes, Obesity and Metabolism, 25(8), 2156-2168.
[8] Tchang, B.G., et al. (2024). "Sex differences in response to anti-obesity medications: Clinical implications." International Journal of Obesity, 48(4), 445-457.
[9] European Medicines Agency. (2024). "CagriSema regulatory submission and review timeline." EMA Public Assessment Reports.
SEO Meta Title and Description
Meta Title: Cagrilintide Blend with Retatrutide: Complete 2026 Guide
Meta Description: Comprehensive guide to cagrilintide blend with retatrutide covering mechanisms, dosing protocols, side effects, and research insights for metabolic health.
