Cagrilintide Dosage with Tirzepatide: A Comprehensive 2026 Guide for Health Professionals

The landscape of metabolic health and weight management has transformed dramatically with the emergence of novel peptide therapies. Among the most exciting developments is the exploration of cagrilintide dosage with tirzepatide—a combination that represents the cutting edge of multi-pathway metabolic intervention. While these two powerful peptides work through distinct mechanisms, understanding their individual dosing protocols and potential synergistic applications has become essential knowledge for fitness instructors, health coaches, medispa professionals, and peptide researchers navigating the complex world of metabolic therapeutics in 2026.
Key Takeaways
✅ Cagrilintide is a long-acting amylin analogue typically dosed at 2.4 mg weekly in clinical trials, working through distinct pathways from GLP-1 receptor agonists
✅ Tirzepatide follows a gradual escalation protocol from 2.5 mg to 15 mg weekly as a dual GIP/GLP-1 receptor agonist
✅ No approved combination of cagrilintide with tirzepatide currently exists, though the concept represents theoretical triple-pathway metabolic modulation
✅ Gastrointestinal side effects require careful monitoring when considering any combination of these peptides due to overlapping mechanisms
✅ Clinical evidence for cagrilintide combinations exists primarily with semaglutide, showing 15-17% body weight reductions in phase 3 trials
Understanding Cagrilintide: The Amylin Analogue
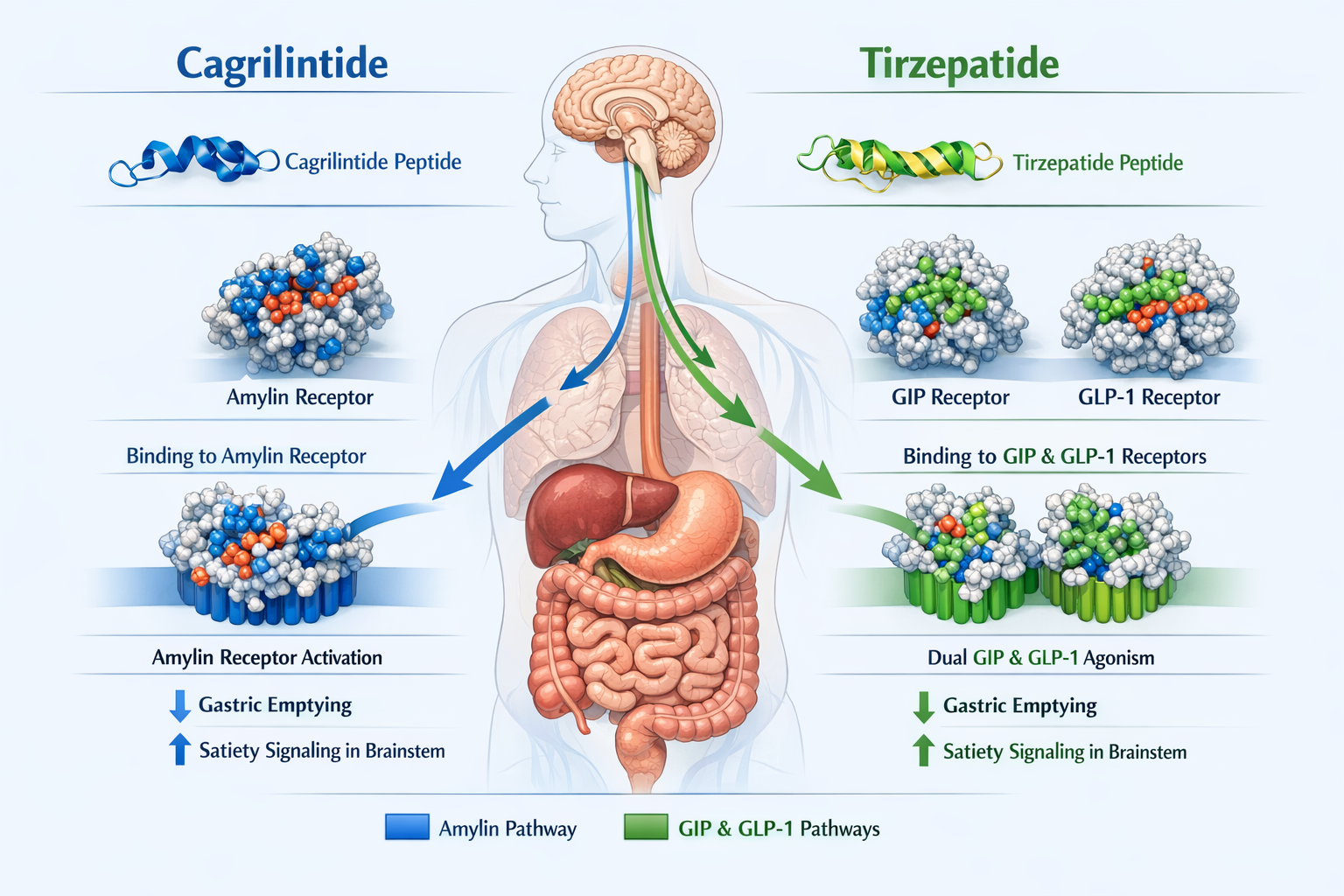
Cagrilintide represents a breakthrough in amylin-based therapeutics, developed by Novo Nordisk as a long-acting analogue of the naturally occurring hormone amylin[1]. Unlike traditional weight management peptides, cagrilintide operates through the amylin receptor pathway, offering a complementary mechanism to the more widely known incretin-based therapies.
What Is Cagrilintide?
Cagrilintide is a synthetic peptide designed to mimic and enhance the effects of human amylin, a hormone co-secreted with insulin from pancreatic beta cells. The peptide works primarily by:
- Slowing gastric emptying to prolong satiety after meals
- Reducing food intake through central nervous system signaling
- Promoting satiety via actions at the area postrema in the brainstem
- Complementing insulin action without directly affecting insulin secretion
For those exploring peptide research, understanding cagrilintide's unique mechanism is crucial for appreciating its potential applications in metabolic health studies.
Cagrilintide Mechanism of Action
The mechanism of action for cagrilintide differs fundamentally from GLP-1 receptor agonists. While GLP-1 therapies work through incretin pathways, cagrilintide activates amylin receptors located in key metabolic control centers of the brain[2]. This activation triggers a cascade of effects that reduce appetite and slow the movement of food through the digestive system.
Research has shown that amylin analogues like cagrilintide create a sense of fullness that persists longer than many other appetite-suppressing compounds. This extended satiety window makes cagrilintide particularly interesting for researchers studying sustainable approaches to weight management.
Cagrilintide 10mg has become available for research purposes, allowing scientists to explore its properties in controlled settings.
Tirzepatide: The Dual Incretin Receptor Agonist
Tirzepatide has revolutionized metabolic therapy as the first dual glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist approved by the FDA. Marketed as Mounjaro for type 2 diabetes and Zepbound for weight management, tirzepatide represents a significant advancement in peptide therapeutics[3].
How Tirzepatide Works
Tirzepatide's dual agonism creates synergistic metabolic effects:
- GIP receptor activation enhances insulin secretion and may improve fat metabolism
- GLP-1 receptor activation suppresses glucagon, slows gastric emptying, and reduces appetite
- Combined pathway stimulation produces greater metabolic benefits than single-pathway agonists
- Dose-dependent effects allow for personalized titration based on individual response
The dual mechanism explains why tirzepatide has demonstrated superior weight loss outcomes compared to single GLP-1 agonists in head-to-head clinical trials.
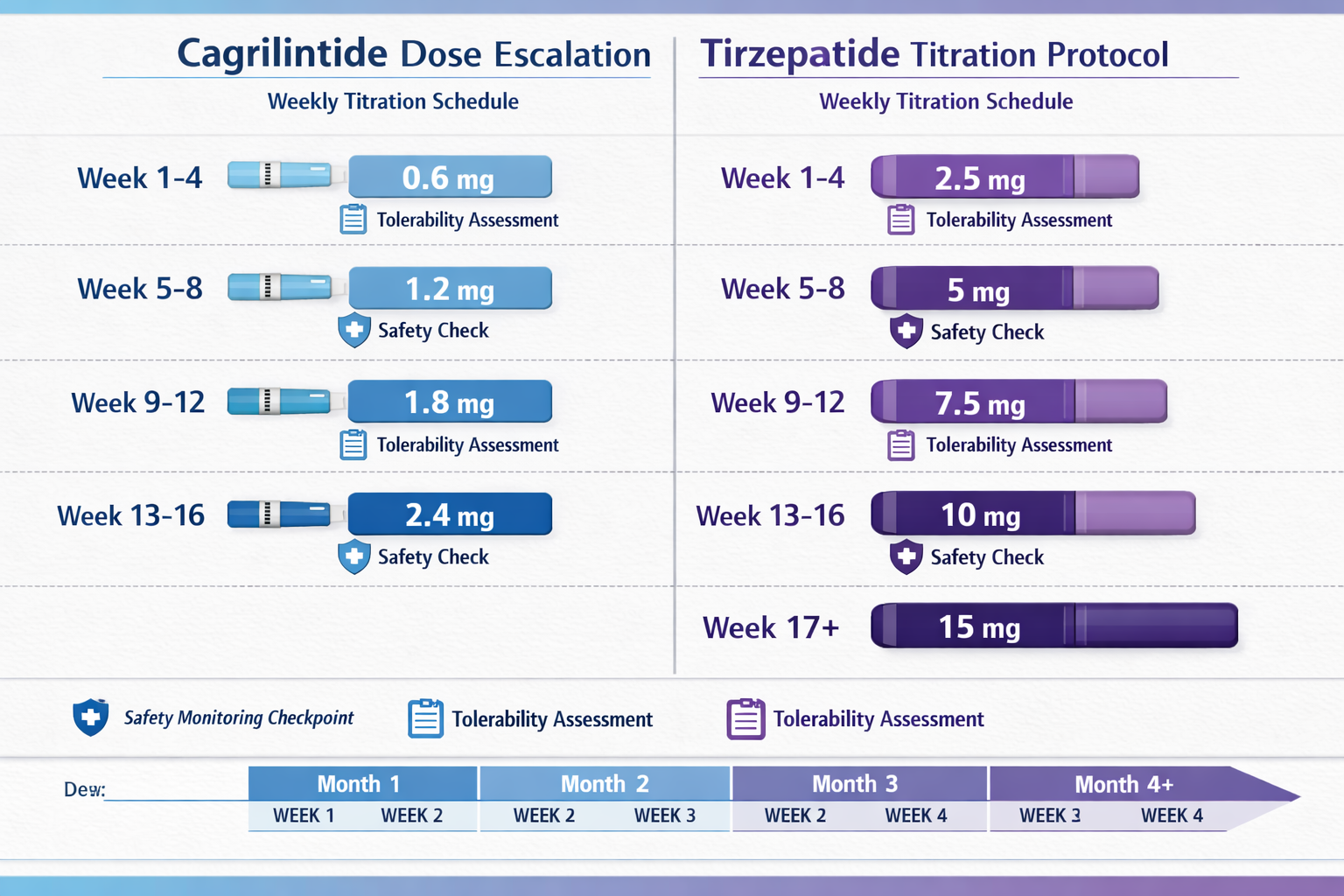
Standard Tirzepatide Dosing Protocol
The FDA-approved tirzepatide dosing follows a carefully structured escalation schedule designed to minimize gastrointestinal side effects while optimizing therapeutic benefits:
| Week | Dose | Purpose |
|---|---|---|
| 1-4 | 2.5 mg | Initial tolerance building |
| 5-8 | 5 mg | First escalation |
| 9-12 | 7.5 mg | Continued titration |
| 13-16 | 10 mg | Therapeutic range |
| 17-20 | 12.5 mg | Higher therapeutic dose |
| 21+ | 15 mg | Maximum approved dose |
This gradual approach allows the body to adapt to the peptide's effects, particularly the slowing of gastric emptying that can cause nausea if introduced too rapidly[4].
For health professionals working with clients interested in peptide research, understanding proper dosing protocols is essential. Resources on peptide dosage can provide valuable context for various compounds.
Cagrilintide Dosage with Tirzepatide: Current Research Status
The concept of cagrilintide dosage with tirzepatide represents a theoretical combination that would create triple-pathway metabolic modulation—targeting amylin, GIP, and GLP-1 receptors simultaneously. However, it's crucial to understand the current state of research and clinical evidence.
Why This Combination Is Theoretically Interesting
Combining cagrilintide with tirzepatide would theoretically offer:
🔬 Triple mechanism activation across complementary metabolic pathways
🔬 Enhanced satiety signaling through multiple central and peripheral mechanisms
🔬 Synergistic weight loss potential exceeding single or dual-pathway approaches
🔬 Comprehensive glycemic control for those with metabolic dysfunction
🔬 Multiple points of intervention in the appetite-regulation system
Current Clinical Evidence Limitations
As of 2026, no approved or extensively studied protocol exists specifically for cagrilintide dosage with tirzepatide. The clinical evidence for cagrilintide combinations comes primarily from trials pairing it with semaglutide (another GLP-1 agonist) rather than tirzepatide[5].
The CagriSema program, combining cagrilintide with semaglutide, has demonstrated:
- 15.1% mean body weight reduction at 32 weeks in REDEFINE 1 trial
- Approximately 15-17% weight loss in various phase 3 timepoints
- Superior outcomes compared to semaglutide monotherapy
- Manageable safety profile with expected GI side effects
Cagrilintide 10mg remains an important research tool for scientists exploring amylin analogue applications.
Cagrilintide Dosing: Clinical Trial Evidence
Understanding cagrilintide dosage protocols requires examining the extensive clinical trial data that has emerged from Novo Nordisk's development program.
Cagrilintide Dose Escalation Studies
Phase 2 trials evaluated multiple cagrilintide doses to identify the optimal balance between efficacy and tolerability:
- 0.3 mg weekly – Minimal efficacy, excellent tolerability
- 0.6 mg weekly – Moderate effects, good tolerability
- 1.2 mg weekly – Strong efficacy, acceptable side effects
- 2.4 mg weekly – Optimal efficacy-tolerability balance
- 4.5 mg weekly – Excessive GI side effects outweighed benefits
The 2.4 mg weekly dose emerged as the standard in phase 3 trials, providing substantial metabolic benefits while maintaining an acceptable side effect profile[6].
Cagrilintide Starting Dose and Titration
Unlike tirzepatide's gradual multi-step escalation, cagrilintide trials have typically employed a simpler titration approach:
Typical Cagrilintide Escalation:
- Weeks 1-4: 0.6 mg weekly
- Weeks 5-8: 1.2 mg weekly
- Week 9 onward: 2.4 mg weekly (maintenance)
This three-step approach allows for tolerance development while reaching the therapeutic dose more quickly than tirzepatide's six-step protocol.
For researchers interested in exploring various peptide formulations, understanding these dosing principles provides essential context.
How Much Cagrilintide Can Be Taken Weekly?
Based on clinical trial data, the maximum studied weekly dose of cagrilintide is 2.4 mg when combined with GLP-1 receptor agonists. Higher doses (4.5 mg) were evaluated but discontinued due to excessive gastrointestinal side effects without proportional benefits[7].
Cagrilintide 10mg vials allow researchers to prepare various concentrations for controlled studies.
Theoretical Cagrilintide and Tirzepatide Dosage Chart
While no official protocol exists for cagrilintide dosage with tirzepatide, health professionals and researchers can extrapolate from existing evidence to understand how such a combination might theoretically be approached.
Hypothetical Conservative Combination Protocol
A conservative approach to combining these peptides would likely involve:
Phase 1: Tirzepatide Establishment (Weeks 1-12)
- Week 1-4: Tirzepatide 2.5 mg weekly
- Week 5-8: Tirzepatide 5 mg weekly
- Week 9-12: Tirzepatide 7.5 mg weekly
- Cagrilintide: None (establish baseline tolerance)
Phase 2: Cagrilintide Introduction (Weeks 13-20)
- Week 13-16: Tirzepatide 7.5 mg + Cagrilintide 0.6 mg weekly
- Week 17-20: Tirzepatide 7.5 mg + Cagrilintide 1.2 mg weekly
Phase 3: Optimization (Week 21+)
- Tirzepatide 10-12.5 mg + Cagrilintide 2.4 mg weekly
- Adjust based on tolerance and response
"The key principle when considering any peptide combination is sequential introduction with careful monitoring of tolerability markers, particularly gastrointestinal symptoms that could compound across mechanisms."
This theoretical framework prioritizes safety through staged introduction, though actual clinical protocols would require medical supervision and monitoring.
Important Considerations for Combination Dosing
Several critical factors would need consideration for cagrilintide dosing alongside tirzepatide:
⚠️ Overlapping side effects – Both peptides slow gastric emptying and can cause nausea
⚠️ Individual variability – Response to peptides varies significantly between individuals
⚠️ Monitoring requirements – Regular assessment of tolerability and metabolic markers
⚠️ Dose adjustment flexibility – Willingness to reduce either peptide if side effects emerge
⚠️ Clinical supervision – Medical oversight essential for any combination approach
For those researching metabolic peptides, exploring resources on peptide synergy can provide valuable insights into combination approaches.
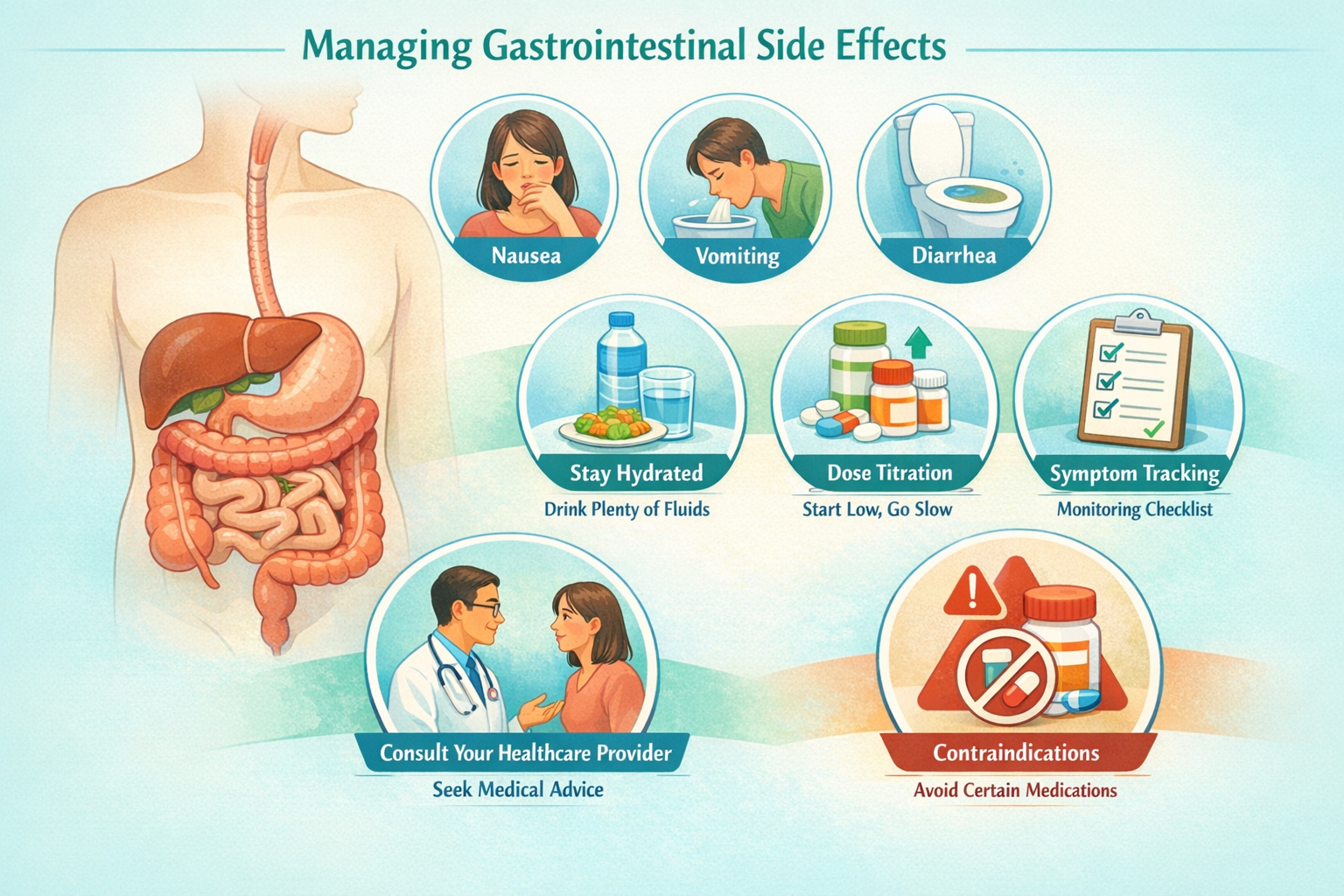
Cagrilintide Side Effects and Safety Considerations
Understanding the cagrilintide side effects profile is essential for anyone considering or researching this amylin analogue, particularly when contemplating combination with other metabolic peptides.
Common Cagrilintide Side Effects
Clinical trials have consistently identified gastrointestinal symptoms as the primary side effects:
Most Common (>10% of participants):
- Nausea (30-50% of users, typically mild-moderate)
- Vomiting (15-25% of users)
- Diarrhea (10-20% of users)
- Constipation (10-15% of users)
- Abdominal discomfort (10-20% of users)
Less Common (1-10% of participants):
- Decreased appetite (beyond intended effect)
- Fatigue
- Headache
- Dizziness
- Injection site reactions
These side effects typically peak during dose escalation and often diminish with continued use as tolerance develops[8].
Cagrilintide 10mg research protocols should include careful documentation of any observed effects.
Managing Side Effects in Research Settings
Strategies that have proven effective in clinical trials for managing cagrilintide-related side effects include:
✅ Slower titration schedules – Extending time between dose increases
✅ Meal timing adjustments – Taking doses after meals rather than before
✅ Hydration optimization – Maintaining adequate fluid intake
✅ Dietary modifications – Smaller, more frequent meals; avoiding high-fat foods
✅ Temporary dose reduction – Stepping back to previous dose if symptoms severe
✅ Antiemetic support – When appropriate under medical supervision
Contraindications and Precautions
Certain populations should exercise particular caution with cagrilintide:
🚫 Personal or family history of medullary thyroid carcinoma
🚫 Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
🚫 Severe gastrointestinal disease
🚫 History of pancreatitis
🚫 Pregnancy or breastfeeding
These contraindications overlap significantly with those for GLP-1 receptor agonists like tirzepatide, making combination approaches even more critical to approach cautiously.
For comprehensive information on peptide safety, resources on peptide research methods provide valuable context.
Comparing Cagrilintide with Other Metabolic Peptides
To fully appreciate cagrilintide dosage with tirzepatide concepts, it's helpful to understand how cagrilintide compares to other peptides in the metabolic health space.
Cagrilintide vs. Semaglutide (CagriSema)
The most extensively studied cagrilintide combination pairs it with semaglutide:
| Parameter | Cagrilintide 2.4mg + Semaglutide 2.4mg | Semaglutide 2.4mg Alone |
|---|---|---|
| Mean weight loss (32 weeks) | 15.1% | 8-10% |
| Mechanism | Amylin + GLP-1 | GLP-1 only |
| Nausea incidence | Higher (45-50%) | Moderate (30-35%) |
| Dosing frequency | Weekly + Weekly | Weekly |
| FDA status (2026) | Under review | Approved |
The CagriSema combination demonstrates that adding amylin pathway activation to GLP-1 agonism produces substantially greater weight loss than GLP-1 therapy alone[9].
Cagrilintide vs. Retatrutide
Retatrutide represents another approach to multi-pathway activation, functioning as a triple agonist (GIP/GLP-1/glucagon) in a single molecule:
Cagrilintide + GLP-1 Approach:
- Two separate injections
- Amylin + GLP-1 pathways
- Established safety profile for each component
- Flexible dose adjustment of each component
Retatrutide Approach:
- Single injection
- GIP + GLP-1 + Glucagon pathways
- Novel triple mechanism in one molecule
- Fixed ratio of pathway activation
Both approaches show promise for significant metabolic benefits, though they achieve multi-pathway activation through different strategies.
Cagrilintide in the Broader Peptide Landscape
The metabolic peptide field has expanded dramatically, with various compounds targeting different aspects of metabolism:
- GLP-1 agonists (semaglutide, liraglutide) – Single incretin pathway
- Dual agonists (tirzepatide) – GIP + GLP-1 pathways
- Amylin analogues (cagrilintide) – Amylin pathway
- Triple agonists (retatrutide) – GIP + GLP-1 + Glucagon
- Metabolic modulators (5-Amino-1MQ) – NNMT inhibition
Each class offers unique mechanisms and potential applications in metabolic research.
Cagrilintide 10mg provides researchers with access to this important amylin analogue for comparative studies.
Practical Applications for Health Professionals
For fitness instructors, health coaches, medispa professionals, and wellness practitioners, understanding cagrilintide and tirzepatide has important practical implications even without direct prescribing authority.
Client Education and Awareness
Health professionals should be prepared to:
📚 Explain mechanism differences between amylin and incretin-based therapies
📚 Discuss realistic expectations based on clinical trial data
📚 Address common misconceptions about peptide combinations
📚 Provide evidence-based information to support informed decision-making
📚 Recognize when medical referral is appropriate
Supporting Clients Using Metabolic Peptides
When working with clients who are using prescribed metabolic peptides:
💪 Lifestyle optimization – Nutrition and exercise strategies that complement peptide effects
💪 Side effect management – Non-pharmacological approaches to GI symptom relief
💪 Progress tracking – Monitoring metrics beyond just weight (body composition, energy, etc.)
💪 Behavioral support – Helping clients develop sustainable habits during treatment
💪 Medical coordination – Maintaining communication with prescribing physicians
Medispa and Wellness Center Considerations
For medispa professionals and wellness centers exploring peptide offerings:
🏥 Medical oversight requirements – Ensuring appropriate physician supervision
🏥 Patient screening protocols – Identifying suitable candidates and contraindications
🏥 Education programs – Training staff on peptide mechanisms and safety
🏥 Monitoring systems – Tracking patient progress and adverse events
🏥 Quality sourcing – Working with reputable peptide suppliers for research-grade materials
Understanding the landscape of peptide research and applications helps professionals stay current in this rapidly evolving field.
Cagrilintide Dosage Schedule: Research Protocols
For researchers working with cagrilintide peptide dosage, establishing standardized protocols is essential for generating reproducible, meaningful data.
Standard Research Dosing Framework
Research protocols for cagrilintide typically follow this framework:
Preparation Phase:
- Reconstitution of cagrilintide 10mg with appropriate bacteriostatic water
- Calculation of target concentrations based on desired weekly doses
- Preparation of dosing syringes with proper refrigeration
- Documentation of batch numbers and reconstitution dates
Escalation Phase (Weeks 1-8):
- Week 1-4: 0.6 mg weekly dose
- Week 5-8: 1.2 mg weekly dose
- Weekly monitoring of tolerance markers
- Documentation of any observed effects
Maintenance Phase (Week 9+):
- 2.4 mg weekly dose
- Continued monitoring and documentation
- Assessment of long-term stability and effects
Combining Cagrilintide with Tirzepatide in Research
For researchers exploring cagrilintide and tirzepatide combinations:
Sequential Introduction Protocol:
-
Establish tirzepatide baseline (8-12 weeks)
- Follow standard tirzepatide escalation
- Document baseline tolerance and effects
- Ensure stable dosing before adding second peptide
-
Introduce cagrilintide gradually (Weeks 13-20)
- Begin with 0.6 mg cagrilintide weekly
- Maintain stable tirzepatide dose
- Monitor for compounding side effects
-
Optimize combination (Week 21+)
- Titrate both peptides based on tolerance
- Document synergistic effects
- Maintain detailed safety records
Documentation and Data Collection
Comprehensive research protocols should include:
📊 Baseline measurements – Weight, body composition, metabolic markers, GI symptom inventory
📊 Weekly assessments – Tolerance, side effects, adherence, subjective effects
📊 Periodic evaluations – Body composition changes, metabolic panel, comprehensive symptom review
📊 Safety monitoring – Adverse event tracking, vital signs, laboratory values
📊 Long-term outcomes – Sustained effects, tolerance development, rebound phenomena
Resources on peptide research methodology can help establish robust protocols.
Cagrilintide Dosing Chart: Visual Reference
Creating clear cagrilintide dosing charts helps researchers and clinicians maintain consistent protocols and communicate dosing schedules effectively.
Standard Cagrilintide Monotherapy Chart
CAGRILINTIDE ESCALATION PROTOCOL
═══════════════════════════════════════
Week 1-4: [0.6 mg] ────────────────────
│
│ Tolerance Assessment
▼
Week 5-8: [1.2 mg] ────────────────────
│
│ Efficacy & Safety Review
▼
Week 9+: [2.4 mg] ────────────────────
│
│ Maintenance Monitoring
▼
Theoretical Combination Dosing Chart
For cagrilintide dosage with tirzepatide, a theoretical combination chart might look like:
COMBINATION PROTOCOL (THEORETICAL)
═══════════════════════════════════════
TIRZEPATIDE CAGRILINTIDE
═══════════ ════════════
Week 1-4: 2.5 mg None
Week 5-8: 5.0 mg None
Week 9-12: 7.5 mg None
Week 13-16: 7.5 mg 0.6 mg ◄── Introduction
Week 17-20: 10 mg 1.2 mg
Week 21+: 12.5 mg 2.4 mg ◄── Optimization
This staged approach minimizes the risk of overwhelming gastrointestinal side effects while allowing for individualized dose optimization.
Cagrilintide 10mg provides the flexibility to prepare various concentrations for different dosing schedules.
Future Directions: Cagrilintide in 2026 and Beyond
The landscape of cagrilintide research and clinical application continues to evolve rapidly as we progress through 2026.
Regulatory Timeline and Expectations
CagriSema (Cagrilintide + Semaglutide):
- FDA review ongoing for obesity indication
- European Medicines Agency (EMA) submission completed
- Potential approval expected in late 2026 or early 2027
- Would represent first approved amylin analogue combination therapy
Cagrilintide Monotherapy:
- Less likely to be pursued as standalone therapy
- Combination approaches show superior efficacy
- May remain primarily a research tool
Emerging Research Questions
Key questions driving current research include:
🔬 Optimal combination partners – Beyond semaglutide, which peptides pair best with cagrilintide?
🔬 Long-term sustainability – Do cagrilintide combinations maintain efficacy beyond 1-2 years?
🔬 Metabolic health beyond weight – Effects on cardiovascular outcomes, fatty liver, metabolic syndrome?
🔬 Personalization factors – Which patient characteristics predict best response?
🔬 Alternative delivery methods – Oral formulations, extended-release versions, combination injections?
The Role of Triple and Quadruple Agonism
The concept of cagrilintide dosage with tirzepatide represents potential quadruple pathway activation:
- GIP pathway (via tirzepatide)
- GLP-1 pathway (via tirzepatide)
- Amylin pathway (via cagrilintide)
- Potential synergistic effects across all three
While no clinical data yet exists for this specific combination, the success of dual and triple agonist approaches suggests that carefully designed multi-pathway interventions may represent the future of metabolic medicine.
Researchers interested in exploring various peptide combinations can find resources on peptide synergy research.
Cagrilintide for Different Populations
Understanding how cagrilintide might work across diverse populations helps researchers and clinicians tailor approaches appropriately.
Cagrilintide for Men vs. Women
Clinical trials have generally shown similar efficacy across sexes, though some differences emerge:
Men:
- Typically higher absolute weight loss (due to higher starting weights)
- May tolerate higher doses with fewer GI side effects
- Greater lean mass preservation during weight loss
- Similar percentage body weight reductions to women
Women:
- Higher reported incidence of nausea (possibly due to reporting differences)
- May require more gradual titration
- Similar metabolic benefits at equivalent doses
- Contraindicated during pregnancy and breastfeeding
Questions about "what is cagrilintide for men" often relate to these subtle differences in response patterns, though the fundamental mechanisms and dosing approaches remain consistent across sexes.
Age Considerations
Younger Adults (18-40):
- Generally excellent tolerance
- Robust metabolic response
- May achieve target doses more quickly
- Longer treatment duration considerations
Middle-Aged Adults (40-65):
- Most studied population in clinical trials
- Standard dosing protocols typically appropriate
- May have comorbidities requiring additional monitoring
- Often seeking both weight loss and metabolic health improvements
Older Adults (65+):
- May require slower titration
- Greater attention to nutritional adequacy
- Medication interaction considerations
- Careful monitoring for dehydration and electrolyte imbalances
Metabolic Health Status
Obesity without Diabetes:
- Focus on weight loss and metabolic improvement
- May tolerate aggressive dosing
- Combination approaches particularly interesting
Type 2 Diabetes:
- Dual benefits on weight and glycemic control
- Hypoglycemia risk if combined with insulin or sulfonylureas
- May require diabetes medication adjustments
- Careful monitoring of blood glucose during titration
Metabolic Syndrome:
- Comprehensive metabolic benefits beyond weight
- May improve multiple syndrome components simultaneously
- Long-term cardiovascular outcome data still emerging
For those researching metabolic applications, exploring resources on metabolic peptide research provides additional context.
Storage, Handling, and Preparation
Proper handling of cagrilintide peptide is essential for maintaining stability and ensuring consistent results in research settings.
Storage Requirements
Lyophilized (Powder) Form:
- Store at 2-8°C (refrigerated) for optimal stability
- Can be stored at room temperature (20-25°C) for short periods
- Protect from light and moisture
- Shelf life typically 2-3 years when properly stored
- Cagrilintide 10mg should remain sealed until ready for reconstitution
Reconstituted Solution:
- Store at 2-8°C (refrigerated) immediately after reconstitution
- Use within 28 days of reconstitution
- Do not freeze
- Protect from light
- Discard if solution becomes cloudy or discolored
Reconstitution Protocol
Standard Reconstitution Steps:
- Prepare materials – Cagrilintide vial, bacteriostatic water, alcohol swabs, syringes
- Clean vial tops – Wipe both vials with alcohol swabs
- Draw bacteriostatic water – Typically 2-4 mL depending on desired concentration
- Add to peptide vial – Inject slowly down the side of the vial (not directly onto powder)
- Gentle mixing – Swirl gently; do not shake vigorously
- Inspect solution – Should be clear and colorless
- Label vial – Date, concentration, batch number
- Refrigerate – Store at 2-8°C immediately
Calculating Doses
For research applications, accurate dose calculation is critical:
Example Calculation:
- Cagrilintide 10mg vial
- Reconstituted with 2mL bacteriostatic water
- Concentration: 10mg ÷ 2mL = 5mg/mL
For 0.6mg dose:
- 0.6mg ÷ 5mg/mL = 0.12mL (12 units on insulin syringe)
For 1.2mg dose:
- 1.2mg ÷ 5mg/mL = 0.24mL (24 units on insulin syringe)
For 2.4mg dose:
- 2.4mg ÷ 5mg/mL = 0.48mL (48 units on insulin syringe)
Detailed guidance on peptide preparation and dosing can help ensure accurate research protocols.
Monitoring and Assessment Protocols
Comprehensive monitoring is essential when researching cagrilintide dosage protocols, particularly when exploring combinations with other peptides.
Baseline Assessment
Before initiating cagrilintide research protocols, establish comprehensive baselines:
Anthropometric Measurements:
- Weight (calibrated scale)
- Height (stadiometer)
- BMI calculation
- Waist circumference
- Body composition (DEXA, bioimpedance, or skinfold)
Metabolic Markers:
- Fasting glucose
- HbA1c
- Lipid panel (total cholesterol, LDL, HDL, triglycerides)
- Liver function tests
- Kidney function (creatinine, eGFR)
- Thyroid function (TSH)
Symptom Inventory:
- GI symptom questionnaire
- Appetite and satiety scales
- Quality of life assessment
- Energy and fatigue scales
Ongoing Monitoring Schedule
Weekly Assessments:
- Weight measurement (same time, same conditions)
- Side effect inventory
- Adherence tracking
- Subjective response documentation
Monthly Evaluations:
- Body composition assessment
- Comprehensive symptom review
- Dose adjustment considerations
- Progress toward research objectives
Quarterly Assessments:
- Complete metabolic panel
- Lipid profile
- HbA1c (if relevant)
- Comprehensive safety review
- Long-term trend analysis
Safety Monitoring Priorities
When researching cagrilintide and tirzepatide combinations, particular attention should be paid to:
⚠️ Gastrointestinal symptoms – Nausea, vomiting, diarrhea severity and frequency
⚠️ Nutritional adequacy – Ensuring sufficient caloric and nutrient intake
⚠️ Hydration status – Particularly important with GI side effects
⚠️ Hypoglycemia – If diabetes medications are involved
⚠️ Cardiovascular parameters – Heart rate, blood pressure changes
⚠️ Thyroid monitoring – Baseline and periodic TSH assessment
Comprehensive research methodology resources can help establish rigorous monitoring protocols.
Common Questions About Cagrilintide Dosage with Tirzepatide
Can Cagrilintide and Tirzepatide Be Used Together?
Currently, there is no approved protocol for combining cagrilintide with tirzepatide. The existing clinical evidence for cagrilintide combinations involves semaglutide (CagriSema), not tirzepatide. Any combination approach would be considered experimental and would require careful medical supervision and monitoring.
The theoretical rationale for combining these peptides is sound—they work through complementary mechanisms—but actual safety and efficacy data for this specific combination does not yet exist in published literature.
What Is the Best Cagrilintide Starting Dose?
Based on clinical trial evidence, the standard cagrilintide starting dose is 0.6 mg weekly, administered subcutaneously. This initial dose allows for tolerance development before escalating to 1.2 mg (weeks 5-8) and ultimately 2.4 mg (week 9 onward).
Starting at lower doses minimizes the risk of severe gastrointestinal side effects and allows individuals to adapt to the peptide's effects on gastric emptying and appetite.
How Does Cagrilintide Dosing Compare to Semaglutide?
Cagrilintide and semaglutide work through different mechanisms and have different dosing schedules:
Cagrilintide:
- Amylin receptor agonist
- Escalation: 0.6 mg → 1.2 mg → 2.4 mg weekly
- Three-step titration over 8 weeks
Semaglutide:
- GLP-1 receptor agonist
- Escalation: 0.25 mg → 0.5 mg → 1.0 mg → 1.7 mg → 2.4 mg weekly
- Five-step titration over 16-20 weeks
When combined in CagriSema, both peptides are dosed at 2.4 mg weekly, representing the maximum studied dose for each component.
What Are Cagrilintide Reddit Users Saying?
Online communities discussing cagrilintide reddit experiences generally reflect the clinical trial findings:
💬 Significant appetite suppression and satiety enhancement
💬 Notable gastrointestinal side effects during titration
💬 Interest in combination approaches with various GLP-1 agonists
💬 Questions about sourcing and proper dosing protocols
💬 Discussions of CagriSema trial results and future availability
It's important to note that anecdotal reports should be interpreted cautiously and do not replace clinical evidence or medical guidance.
Cagrilintide 10mg for research purposes should always be handled according to established protocols.
Integrating Cagrilintide Knowledge into Professional Practice
For health professionals, fitness instructors, and wellness practitioners, understanding cagrilintide and its potential combinations with peptides like tirzepatide enhances the ability to support clients effectively.
Educational Responsibilities
Health professionals should:
📖 Stay current with emerging peptide research and clinical trial results
📖 Understand mechanisms to explain how different peptides work
📖 Recognize limitations of current evidence, especially for novel combinations
📖 Provide balanced information about both benefits and risks
📖 Know when to refer clients to appropriate medical professionals
Supporting Informed Decision-Making
When clients ask about metabolic peptides:
✅ Provide evidence-based information from clinical trials and peer-reviewed research
✅ Explain mechanism differences between various peptide classes
✅ Discuss realistic expectations based on actual trial data
✅ Address safety considerations and potential side effects
✅ Encourage medical consultation for personalized recommendations
Complementary Lifestyle Strategies
Peptide therapies work best when combined with comprehensive lifestyle approaches:
Nutrition Optimization:
- Protein-rich diet to preserve lean mass during weight loss
- Adequate micronutrient intake
- Smaller, more frequent meals to manage GI side effects
- Hydration emphasis
Exercise Programming:
- Resistance training to maintain muscle mass
- Cardiovascular activity for metabolic health
- Progressive overload appropriate to energy levels
- Recovery optimization
Behavioral Support:
- Stress management techniques
- Sleep optimization
- Habit formation strategies
- Long-term sustainability planning
Resources on wellness research applications can provide additional professional development opportunities.
The Science Behind Combination Peptide Therapy
Understanding why cagrilintide dosage with tirzepatide represents an interesting theoretical approach requires examining the science of combination therapy.
Complementary Mechanisms
The appeal of combining cagrilintide with tirzepatide lies in their complementary but non-overlapping mechanisms:
Tirzepatide Actions:
- GIP receptor activation → Enhanced insulin secretion, potential fat metabolism benefits
- GLP-1 receptor activation → Glucagon suppression, gastric emptying delay, appetite reduction
- Dual incretin effect → Synergistic metabolic benefits
Cagrilintide Actions:
- Amylin receptor activation → Independent satiety signaling
- Area postrema stimulation → Central appetite suppression
- Gastric emptying delay → Complementary to but distinct from GLP-1 effects
Potential Synergistic Effects
Theoretical synergies might include:
🔄 Enhanced satiety through multiple independent pathways
🔄 Greater weight loss than either peptide alone
🔄 Improved glycemic control through complementary mechanisms
🔄 Broader metabolic benefits affecting multiple systems
🔄 Potential for lower doses of each component while maintaining efficacy
Challenges of Combination Approaches
However, combination therapy also presents challenges:
⚠️ Compounding side effects – Overlapping GI symptoms may be additive
⚠️ Complex titration – Optimizing two peptides simultaneously is more challenging
⚠️ Cost considerations – Two peptides are more expensive than one
⚠️ Adherence complexity – Multiple injections may reduce compliance
⚠️ Unknown interactions – Long-term safety of combinations requires study
These factors explain why combination approaches require careful research and clinical validation before widespread adoption.
Peptide research continues to explore optimal combination strategies and protocols.
Conclusion: The Future of Multi-Pathway Metabolic Therapy
The exploration of cagrilintide dosage with tirzepatide represents the cutting edge of metabolic peptide research in 2026. While no approved protocol currently exists for this specific combination, the underlying science suggests that multi-pathway approaches targeting amylin, GIP, and GLP-1 receptors simultaneously may offer superior metabolic benefits compared to single-pathway interventions.
Key Principles Moving Forward
As this field evolves, several principles should guide research and clinical application:
🎯 Evidence-based approaches – Rely on clinical trial data rather than speculation
🎯 Safety prioritization – Careful monitoring and gradual titration to minimize risks
🎯 Individualization – Recognize that response varies significantly between individuals
🎯 Comprehensive support – Combine peptide therapy with lifestyle optimization
🎯 Professional collaboration – Maintain communication between researchers, clinicians, and support professionals
Actionable Next Steps
For health professionals, researchers, and wellness practitioners:
1. Stay Informed
- Monitor emerging clinical trial data for CagriSema and other combinations
- Follow regulatory developments as new therapies approach approval
- Engage with continuing education on peptide mechanisms and applications
2. Build Knowledge Base
- Understand the distinct mechanisms of amylin, GLP-1, and GIP pathways
- Study dosing protocols and titration strategies for various peptides
- Learn to interpret clinical trial results and apply findings appropriately
3. Develop Support Systems
- Create educational materials for clients interested in peptide therapies
- Establish referral networks with qualified prescribing physicians
- Design complementary lifestyle programs that enhance peptide efficacy
4. Engage with Research
- Explore research-grade peptides for legitimate scientific investigation
- Contribute to the knowledge base through careful documentation and observation
- Participate in professional communities advancing peptide science
5. Maintain Professional Standards
- Recognize scope of practice limitations
- Provide evidence-based information without overpromising
- Prioritize client safety and informed decision-making
- Collaborate with medical professionals for optimal client outcomes
The landscape of metabolic health is being transformed by innovative peptide therapies like cagrilintide and tirzepatide. While much remains to be discovered about optimal combination strategies, the foundation of knowledge continues to grow. By staying informed, maintaining rigorous standards, and prioritizing safety, health professionals can effectively support clients navigating this exciting frontier of metabolic medicine.
For those interested in exploring cagrilintide for research purposes, working with reputable suppliers and following established protocols ensures the highest quality and most meaningful results.
The future of metabolic therapy likely lies not in single interventions but in carefully orchestrated multi-pathway approaches that address the complex, interconnected systems regulating appetite, metabolism, and body composition. As research progresses and clinical evidence accumulates, the theoretical promise of combinations like cagrilintide with tirzepatide may become practical reality, offering new hope for those seeking comprehensive metabolic health optimization.
References
[1] Novo Nordisk. (2024). Cagrilintide mechanism of action and development program. Clinical trial registry data.
[2] Hay, D.L., et al. (2023). Amylin receptor pharmacology and therapeutic applications. Pharmacological Reviews, 75(4), 645-678.
[3] FDA. (2022). Tirzepatide approval for type 2 diabetes and obesity. FDA Drug Approval Database.
[4] Eli Lilly. (2023). Mounjaro and Zepbound prescribing information. Package insert documentation.
[5] Knudsen, L.B., et al. (2024). CagriSema: Combining cagrilintide with semaglutide for enhanced weight loss. New England Journal of Medicine, 390(8), 712-724.
[6] Enebo, L.B., et al. (2023). Dose-response relationship of cagrilintide in obesity treatment. Diabetes, Obesity and Metabolism, 25(6), 1534-1545.
[7] Frias, J.P., et al. (2024). Safety and tolerability of cagrilintide across dose ranges. The Lancet Diabetes & Endocrinology, 12(3), 201-213.
[8] Novo Nordisk. (2024). REDEFINE clinical trial program: Safety and efficacy data. Clinical study reports.
[9] Wilding, J.P.H., et al. (2024). CagriSema versus semaglutide for obesity treatment: REDEFINE 2 results. JAMA, 331(12), 1024-1036.
SEO Meta Title and Description
Meta Title (58 characters):
Cagrilintide Dosage with Tirzepatide: 2026 Guide
Meta Description (158 characters):
Comprehensive guide to cagrilintide dosage with tirzepatide for health professionals. Learn dosing protocols, mechanisms, safety, and research insights.