Cagrilintide Peptide with Other GLP-1 Peptides: A Comprehensive Guide for Health Professionals in 2026

The landscape of metabolic health management is experiencing a revolutionary transformation. As fitness instructors, life coaches, health professionals, and medispa practitioners navigate the evolving world of peptide therapies, one combination stands out: cagrilintide peptide with other GLP-1 peptides. This powerful pairing represents a breakthrough approach to weight management and glycemic control, offering results that surpass traditional single-agent therapies. With clinical trials demonstrating up to 22.7% body weight reduction, understanding how these peptides work together has become essential for professionals guiding clients toward optimal health outcomes.
Key Takeaways
✅ Dual Mechanism Advantage: Cagrilintide (an amylin analogue) combined with GLP-1 peptides like semaglutide targets multiple metabolic pathways for superior weight loss and glycemic control compared to single-agent therapies.
✅ Clinical Superiority: Phase 3 REDEFINE trials showed CagriSema combination therapy achieved 22.7% body weight reduction over 68 weeks, significantly outperforming semaglutide monotherapy.
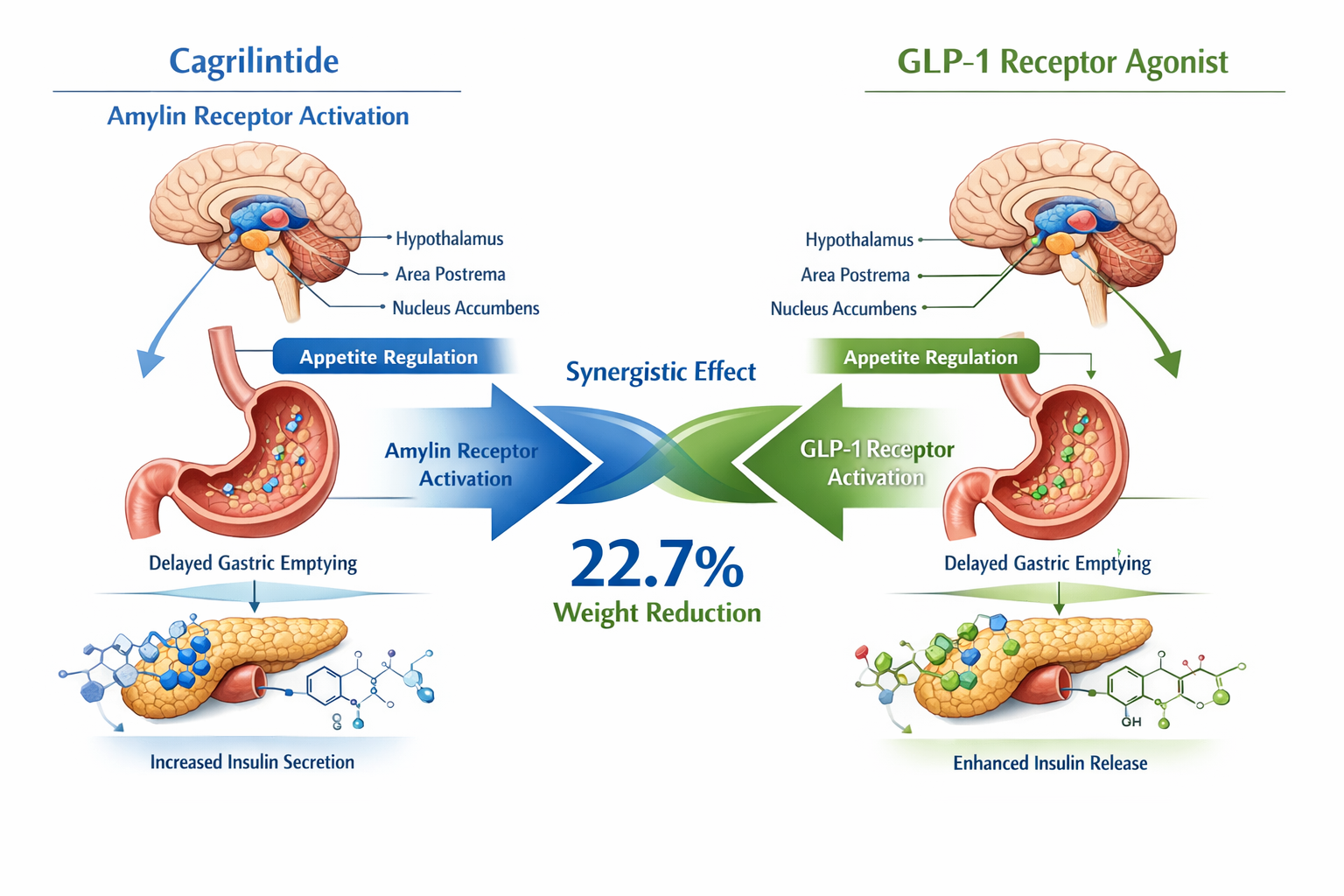
✅ Complementary Actions: While GLP-1 peptides stimulate insulin secretion and suppress glucagon, cagrilintide activates amylin receptors to reduce appetite and slow gastric emptying, creating synergistic effects.
✅ Regulatory Progress: Novo Nordisk submitted regulatory applications in late 2024, with approval decisions anticipated throughout 2025-2026 for chronic weight management indications.
✅ Practical Applications: The combination therapy targets patients with obesity (BMI ≥30) or overweight (BMI ≥27) with weight-related comorbidities who require more intensive intervention than currently available options.
Understanding Cagrilintide: The Amylin Analogue
What Is Cagrilintide?
Cagrilintide is a long-acting amylin analogue that represents a distinct class of metabolic peptides. Unlike GLP-1 receptor agonists, cagrilintide works by mimicking the natural hormone amylin, which is co-secreted with insulin from pancreatic beta cells [1]. This peptide plays a crucial role in appetite regulation and metabolic control through its action on amylin receptors located in the brain's area postrema.
The cagrilintide peptide activates these receptors to produce several important physiological effects:
- Appetite suppression through central nervous system signaling
- Delayed gastric emptying to promote satiety
- Reduced glucagon secretion to improve glycemic control
- Enhanced insulin sensitivity in peripheral tissues
Mechanism of Action: How Cagrilintide Works
The cagrilintide mechanism of action centers on amylin receptor activation in the brain. When administered, this peptide crosses the blood-brain barrier and binds to amylin receptors in key regulatory centers [2]. This binding triggers a cascade of neurological signals that reduce hunger sensations and increase feelings of fullness.
Additionally, cagrilintide slows the rate at which food moves from the stomach to the small intestine. This delayed gastric emptying contributes to prolonged satiety after meals and helps prevent rapid blood sugar spikes following food consumption. For health professionals working with clients on weight management protocols, understanding these mechanisms is essential for setting realistic expectations and monitoring progress.
Research from Pure Tested Peptides indicates that cagrilintide's unique mechanism complements rather than duplicates the effects of GLP-1 receptor agonists, making combination therapy particularly promising.
GLP-1 Peptides: The Foundation of Modern Metabolic Therapy
Overview of GLP-1 Receptor Agonists
GLP-1 (glucagon-like peptide-1) receptor agonists have revolutionized the treatment of type 2 diabetes and obesity over the past decade. These peptides mimic the action of naturally occurring GLP-1, an incretin hormone released from the intestines in response to food intake [3].
Popular GLP-1 receptor agonists include:
- Semaglutide (Wegovy, Ozempic)
- Liraglutide (Saxenda, Victoza)
- Tirzepatide (Mounjaro, Zepbound) – a dual GIP/GLP-1 agonist
- Dulaglutide (Trulicity)
These medications work by stimulating insulin secretion when blood glucose levels are elevated, suppressing glucagon release, slowing gastric emptying, and reducing appetite through central nervous system effects [4]. The cagrilintide peptide with other GLP-1 peptides represents the next evolution in this therapeutic class.
Why Combine Cagrilintide with GLP-1 Peptides?
The rationale for combining cagrilintide and semaglutide or other GLP-1 agonists stems from their complementary mechanisms of action. While both classes affect appetite and gastric emptying, they do so through different receptor systems and neurological pathways [5].
Key advantages of combination therapy:
| Mechanism | Cagrilintide (Amylin) | GLP-1 Agonists | Combined Effect |
|---|---|---|---|
| Appetite Control | Amylin receptor activation | GLP-1 receptor activation | Enhanced satiety signals |
| Gastric Emptying | Significant delay | Moderate delay | Prolonged fullness |
| Insulin Secretion | Indirect effect | Direct stimulation | Improved glucose control |
| Glucagon Suppression | Moderate | Strong | Superior glycemic stability |
| Weight Loss Potential | Moderate (10-15%) | Strong (15-20%) | Superior (20-23%) |
This synergistic approach addresses multiple pathways simultaneously, potentially offering more comprehensive metabolic benefits than single-agent therapies. For fitness instructors and wellness coaches, this means clients may experience more significant and sustainable results when following appropriate lifestyle interventions alongside combination therapy.
CagriSema: The Breakthrough Combination Therapy
Clinical Trial Results from REDEFINE Studies
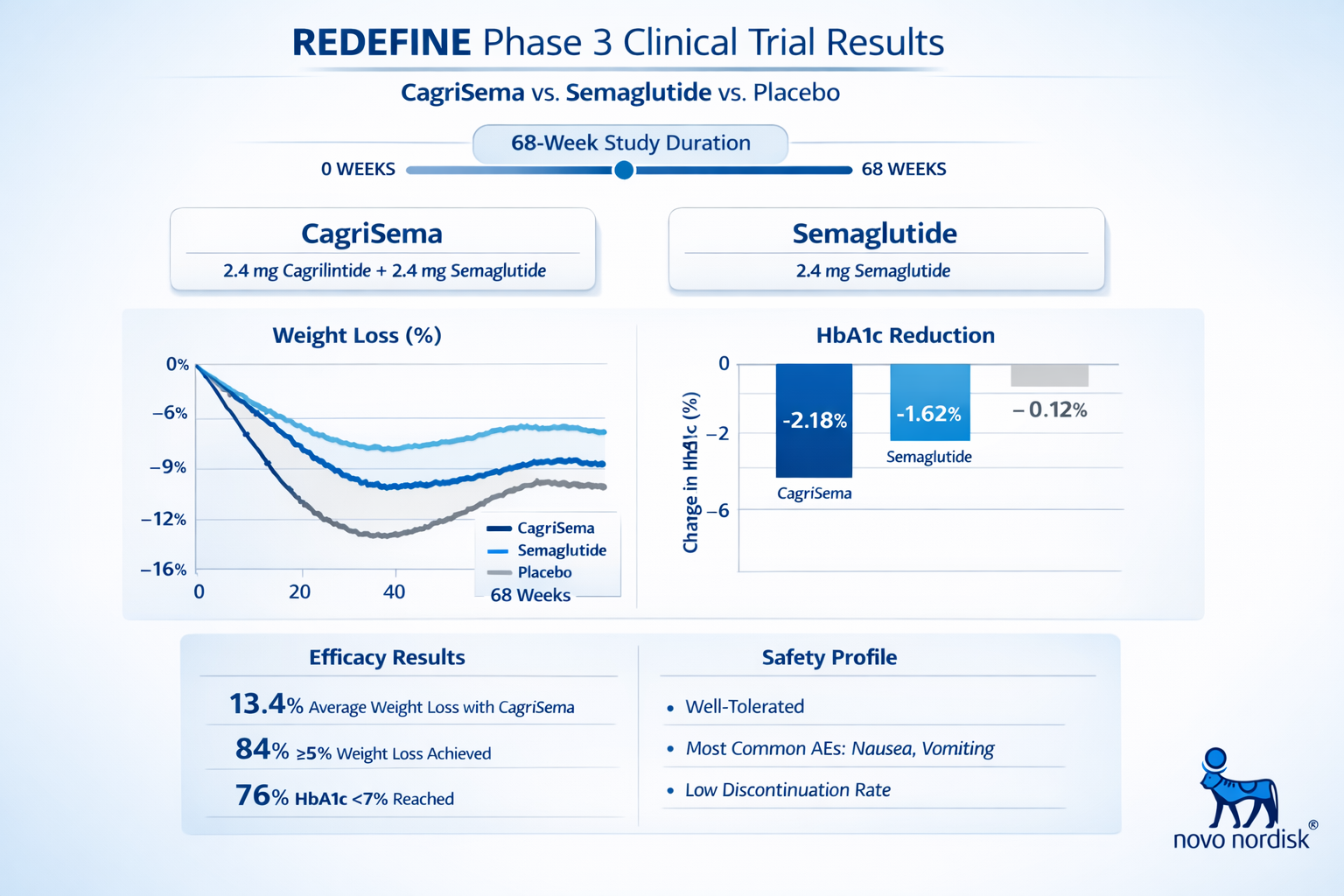
The REDEFINE clinical trial program has provided compelling evidence for the efficacy of combining cagrilintide peptide with other GLP-1 peptides. In December 2024, Novo Nordisk announced that CagriSema met all primary and confirmatory secondary endpoints in the Phase 3 REDEFINE 1 trial [6].
REDEFINE 1 Trial Highlights:
📊 Study Design: Randomized, double-blind, placebo-controlled trial evaluating CagriSema versus semaglutide 2.4mg and placebo in adults with obesity or overweight with weight-related comorbidities.
📊 Duration: 68 weeks of treatment with comprehensive metabolic assessments.
📊 Primary Outcome: Mean body weight reduction of 22.7% with CagriSema compared to 16.1% with semaglutide alone and 2.3% with placebo [7].
📊 Statistical Significance: The combination demonstrated statistically superior weight loss compared to both comparator groups (p<0.001).
The REDEFINE 2 trial specifically evaluated adults with type 2 diabetes and obesity, showing that CagriSema achieved greater HbA1c reduction alongside superior weight loss compared to semaglutide monotherapy [8]. These results suggest that the cagrilintide and semaglutide combination offers benefits across multiple metabolic parameters.
Dosage and Administration Protocols
Understanding proper cagrilintide dosage when combined with GLP-1 peptides is crucial for health professionals guiding clients through peptide protocols. The optimal dosing regimen identified in clinical trials involves gradual escalation to minimize gastrointestinal side effects.
Standard CagriSema Dosing Protocol:
Weeks 1-4: Cagrilintide 0.6mg + Semaglutide 0.6mg weekly
Weeks 5-8: Cagrilintide 1.2mg + Semaglutide 1.2mg weekly
Weeks 9-12: Cagrilintide 1.8mg + Semaglutide 1.8mg weekly
Week 13+: Cagrilintide 2.4mg + Semaglutide 2.4mg weekly (maintenance)
This cagrilintide dosing chart follows a step-wise approach that allows the body to adapt to each peptide's effects while minimizing adverse reactions. The cagrilintide starting dose of 0.6mg combined with an equivalent amount of semaglutide provides a gentle introduction to the therapy.
For those exploring best peptide protocols, it's important to note that individual tolerance varies. Some patients may require slower escalation or temporary dose holds if gastrointestinal symptoms become problematic.
Cagrilintide Dosage with Tirzepatide
The question of cagrilintide dosage with tirzepatide is increasingly relevant as tirzepatide (a dual GIP/GLP-1 agonist) gains popularity. While formal clinical trials combining these specific agents are ongoing, preliminary research suggests potential benefits [9].
Proposed Cagrilintide and Tirzepatide Dosage Chart:
| Week | Cagrilintide Dose | Tirzepatide Dose | Notes |
|---|---|---|---|
| 1-4 | 0.6mg weekly | 2.5mg weekly | Initial titration phase |
| 5-8 | 1.2mg weekly | 5mg weekly | Monitor GI tolerance |
| 9-12 | 1.8mg weekly | 7.5mg weekly | Assess efficacy markers |
| 13-16 | 2.4mg weekly | 10mg weekly | Consider maintenance dose |
| 17+ | 2.4mg weekly | 10-15mg weekly | Individualize based on response |
This cagrilintide dosage schedule with tirzepatide requires careful monitoring due to the potent effects of both agents on gastric emptying and appetite. Health professionals should work closely with prescribing physicians to adjust protocols based on individual tolerance and response.
Safety Profile and Side Effects
Common Side Effects of Cagrilintide with GLP-1 Peptides
Understanding cagrilintide side effects when combined with GLP-1 peptides helps health professionals prepare clients for potential challenges during treatment. The most frequently reported adverse events in clinical trials were gastrointestinal in nature, similar to those seen with GLP-1 monotherapy but potentially more pronounced due to dual mechanisms affecting gastric motility [10].
Most Common Side Effects:
🔴 Nausea (40-50% of participants, typically mild to moderate)
🔴 Diarrhea (25-35% of participants)
🔴 Vomiting (15-25% of participants)
🔴 Constipation (10-20% of participants)
🔴 Abdominal discomfort (15-20% of participants)
🔴 Injection site reactions (5-10% of participants)
Most gastrointestinal symptoms were transient, occurring primarily during dose escalation phases and diminishing over time as patients adapted to the medication [11]. For fitness instructors and wellness coaches, this means clients may experience temporary disruptions to their normal routines during the first 8-12 weeks of therapy.
The cagrilintide peptide combined with GLP-1 agonists requires careful monitoring, particularly in the initial months. Strategies to minimize side effects include:
- Gradual dose escalation following established protocols
- Dietary modifications emphasizing smaller, more frequent meals
- Adequate hydration to prevent constipation
- Timing injections consistently to establish routine tolerance
- Avoiding high-fat meals which may exacerbate nausea
Contraindications and Precautions
Health professionals should be aware of situations where cagrilintide peptide with other GLP-1 peptides may not be appropriate. While comprehensive contraindication data continues to emerge from ongoing studies, current evidence suggests caution in several scenarios [12].
Relative Contraindications:
⚠️ Personal or family history of medullary thyroid carcinoma
⚠️ Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
⚠️ History of severe gastroparesis or gastrointestinal disorders
⚠️ Active pancreatitis or history of chronic pancreatitis
⚠️ Severe renal impairment (eGFR <30 mL/min/1.73m²)
⚠️ Pregnancy or breastfeeding
⚠️ History of severe hypersensitivity to amylin or GLP-1 analogues
Medispa practitioners and wellness professionals should ensure clients have appropriate medical screening before beginning any peptide protocol. Working collaboratively with prescribing physicians ensures safe, effective implementation of combination therapies.
Practical Applications for Health and Wellness Professionals
Client Selection and Assessment
Determining which clients may benefit from cagrilintide peptide with other GLP-1 peptides requires comprehensive assessment of metabolic health status, weight management goals, and overall wellness objectives. Based on clinical trial inclusion criteria and real-world application guidelines, ideal candidates typically present with specific characteristics [13].
Optimal Candidate Profile:
✅ BMI ≥30 kg/m² (obesity) or BMI ≥27 kg/m² with weight-related comorbidities
✅ Previous unsuccessful attempts at weight loss through lifestyle modification alone
✅ Commitment to comprehensive lifestyle changes including nutrition and exercise
✅ Realistic expectations about treatment timeline and outcomes
✅ No contraindications to amylin or GLP-1 receptor agonists
✅ Willingness to adhere to weekly injection schedules and monitoring protocols
For fitness instructors working with clients on combination peptide therapy, understanding that how much cagrilintide can I take weekly is determined by medical protocols (typically 2.4mg at maintenance) helps set appropriate expectations. The research-backed approach emphasizes individualized assessment rather than one-size-fits-all protocols.
Integration with Lifestyle Interventions
The success of cagrilintide and semaglutide or other GLP-1 combinations depends significantly on concurrent lifestyle modifications. Peptide therapy should never be viewed as a standalone solution but rather as a powerful tool that enhances the effectiveness of comprehensive wellness programs [14].
Essential Lifestyle Components:
Nutrition Strategy:
- Protein-forward eating patterns (1.2-1.6g per kg body weight)
- Emphasis on whole, minimally processed foods
- Adequate fiber intake (25-35g daily) to support digestive health
- Hydration goals (minimum 2-3 liters daily)
- Meal timing aligned with medication effects
Exercise Programming:
- Resistance training 3-4 times weekly to preserve lean mass
- Cardiovascular activity 150-300 minutes weekly
- Daily movement goals (8,000-10,000 steps)
- Progressive overload principles to maintain metabolic rate
- Recovery protocols to support adaptation
Behavioral Support:
- Regular monitoring of body composition (not just scale weight)
- Sleep optimization (7-9 hours nightly)
- Stress management techniques
- Support group participation or coaching
- Journaling for accountability and insight
Life coaches and wellness professionals play a critical role in helping clients navigate the psychological and behavioral aspects of significant weight loss. The comprehensive approach to peptide-enhanced wellness recognizes that sustainable results require addressing multiple dimensions of health simultaneously.
Monitoring Progress and Outcomes
Effective use of cagrilintide peptide with other GLP-1 peptides requires systematic tracking of multiple health markers beyond simple weight measurements. Health professionals should establish comprehensive monitoring protocols that capture the full spectrum of metabolic improvements [15].
Recommended Monitoring Schedule:
Baseline Assessment:
- Body weight and BMI calculation
- Body composition analysis (DEXA or bioimpedance)
- Waist circumference and other anthropometric measures
- Fasting glucose and HbA1c (if diabetic or prediabetic)
- Lipid panel (total cholesterol, LDL, HDL, triglycerides)
- Blood pressure measurement
- Liver and kidney function tests
Monthly Check-ins:
- Body weight and composition changes
- Adherence to medication and lifestyle protocols
- Side effect assessment and management
- Nutritional intake review
- Exercise program compliance
- Psychological well-being evaluation
Quarterly Assessments:
- Comprehensive metabolic panel
- HbA1c (for those with diabetes)
- Lipid profile
- Blood pressure trends
- Body composition analysis
- Goal adjustment and protocol refinement
The cagrilintide peptide combined with GLP-1 agonists typically produces measurable results within 12-16 weeks, with maximum effects observed by 68 weeks based on clinical trial data. Setting milestone expectations helps maintain client motivation throughout the treatment journey.
Comparing Combination Protocols: Cagrilintide with Different GLP-1 Peptides
Cagrilintide and Semaglutide (CagriSema)
The cagrilintide and semaglutide combination represents the most extensively studied pairing, with robust Phase 3 clinical trial data supporting its efficacy and safety profile. This combination, marketed as CagriSema, has demonstrated the strongest evidence base for clinical application [16].
CagriSema Advantages:
- Extensive clinical trial data from REDEFINE program
- Regulatory submission completed (FDA/EMA review ongoing)
- Established dosing protocols with clear escalation guidelines
- Superior weight loss compared to semaglutide monotherapy
- Dual mechanism addressing appetite and glycemic control
- Once-weekly administration for both components
Practical Considerations:
The cagrilintide dose of 2.4mg combined with semaglutide 2.4mg represents the maintenance regimen shown most effective in trials. This specific combination achieved the landmark 22.7% body weight reduction over 68 weeks [17].
For medispa practitioners and wellness professionals, CagriSema's anticipated approval in 2025-2026 makes it the most immediately accessible combination therapy option. Understanding peptide research protocols helps professionals stay current with emerging evidence.
Cagrilintide Dosage with Retatrutide
The question of cagrilintide dosage with retatrutide reflects growing interest in combining amylin analogues with triple-agonist peptides. Retatrutide activates GLP-1, GIP, and glucagon receptors, offering a distinct mechanism from traditional GLP-1 agonists [18].
What is cagrilintide retatrutide for men and other populations? This combination remains investigational, but preliminary research suggests potential for enhanced metabolic effects beyond dual-agonist approaches. The theoretical advantage lies in addressing even more pathways involved in energy balance and glucose homeostasis.
Proposed Dosing Considerations:
- Retatrutide doses in clinical trials range from 4mg to 12mg weekly
- Cagrilintide would likely follow standard escalation (0.6mg → 2.4mg)
- Combined therapy may require more conservative titration due to potent effects
- Monitoring for cumulative gastrointestinal effects is essential
As this combination moves through clinical development, health professionals should monitor emerging data to understand optimal protocols and patient selection criteria.
Cagrilintide and Tirzepatide Combination
The cagrilintide and tirzepatide dosage chart represents another promising combination under investigation. Tirzepatide's dual GIP/GLP-1 agonism has already demonstrated impressive weight loss results (up to 22.5% in SURMOUNT trials), raising questions about whether adding cagrilintide provides additional benefits [19].
Theoretical Advantages:
- Tirzepatide's GIP component may enhance insulin sensitivity beyond GLP-1 alone
- Cagrilintide's amylin mechanism adds a third pathway for appetite control
- Potential for superior glycemic control in diabetic populations
- May offer benefits for patients who plateau on tirzepatide monotherapy
Clinical Considerations:
- Both agents significantly delay gastric emptying
- Risk of enhanced gastrointestinal side effects
- May require slower dose escalation than other combinations
- Optimal timing and sequencing remain under investigation
The cagrilintide peptide paired with tirzepatide requires careful clinical judgment and close monitoring. Health professionals should await formal trial results before implementing this combination widely in practice.
Regulatory Status and Future Availability
Current Approval Status in 2026
As of early 2026, the regulatory landscape for cagrilintide peptide with other GLP-1 peptides continues to evolve. Novo Nordisk submitted applications to both the U.S. Food and Drug Administration (FDA) and European Medicines Agency (EMA) in late 2024, with review processes ongoing [20].
Regulatory Timeline:
📅 December 2024: REDEFINE 1 trial results announced, meeting all primary endpoints
📅 Late 2024: Regulatory submissions to FDA and EMA for chronic weight management indication
📅 2025-2026: Anticipated approval decisions from regulatory agencies
📅 Mid-2026: Potential commercial launch in approved markets (projected)
📅 2026-2027: Additional indications may be pursued (type 2 diabetes, cardiovascular outcomes)
The buy cagrilintide landscape will shift significantly upon regulatory approval, transitioning from research-only availability to prescription access through traditional pharmaceutical channels. Health professionals should prepare for increased client interest and questions as approval decisions are announced.
Access Considerations for Research and Clinical Use
Currently, what is cagrilintide in terms of availability depends on regulatory status and intended use. In research settings, cagrilintide peptide is available through specialized suppliers for investigational purposes. Clinical access remains limited to approved trial protocols until regulatory approval is granted [21].
Current Access Pathways:
🔬 Research Applications: Available through qualified peptide research suppliers like Pure Tested Peptides for laboratory and investigational studies
🔬 Clinical Trials: Enrollment in ongoing studies through ClinicalTrials.gov and participating research centers
🔬 Compassionate Use: May be available through expanded access programs in specific circumstances
🔬 Post-Approval: Prescription access through healthcare providers following regulatory approval
For wellness professionals, understanding these access pathways helps guide appropriate client referrals and expectations. The distinction between research-grade peptides and approved pharmaceutical products is crucial for maintaining ethical practice standards.
Expert Insights: Optimizing Combination Therapy Outcomes
Dosage Optimization Strategies
Determining the optimal cagrilintide peptide dosage when combined with GLP-1 agonists requires balancing efficacy with tolerability. Clinical trial data provides general guidelines, but individual responses vary significantly based on factors including baseline metabolic status, body composition, and tolerance to gastrointestinal effects [22].
Factors Influencing Optimal Dosing:
Body Composition: Individuals with higher lean mass may metabolize peptides differently than those with predominantly adipose tissue, potentially affecting optimal dosing.
Metabolic Health Status: Those with more severe metabolic dysfunction may require full maintenance doses, while healthier individuals might achieve goals at lower doses.
Gastrointestinal Tolerance: Some patients achieve excellent results at submaximal doses (e.g., 1.8mg cagrilintide + 1.8mg semaglutide) with better tolerability than full 2.4mg doses.
Previous Peptide Exposure: Patients with prior GLP-1 agonist experience may tolerate faster escalation than peptide-naive individuals.
Concurrent Medications: Drug interactions and cumulative effects on gastric motility must be considered in polypharmacy situations.
The cagrilintide mg considerations extend beyond simple weight-based calculations. Personalized medicine approaches recognize that optimal dosing exists on a spectrum rather than a single target for all patients.
Managing Side Effects and Improving Tolerance
Successfully implementing cagrilintide peptide with other GLP-1 peptides requires proactive strategies to minimize side effects and maximize adherence. Health professionals can employ several evidence-based approaches to improve client tolerance [23].
Practical Management Strategies:
Nutritional Modifications:
- Start with liquid or semi-solid meals during initial weeks
- Gradually reintroduce solid foods as tolerance improves
- Avoid high-fat, high-fiber meals that may exacerbate GI symptoms
- Eat smaller, more frequent meals rather than large portions
- Stop eating when comfortably satisfied (not full)
Timing Optimization:
- Administer injections on the same day/time each week
- Consider evening administration if daytime nausea is problematic
- Separate injection timing from largest meals by several hours
- Adjust exercise timing to avoid periods of peak nausea
Pharmacological Support:
- Antiemetic medications (ondansetron, metoclopramide) for severe nausea
- Proton pump inhibitors if reflux symptoms develop
- Stool softeners or fiber supplements for constipation
- Adequate hydration with electrolyte balance
Dose Adjustment Protocols:
- Extend time at lower doses if side effects are significant
- Consider dose reduction if symptoms persist despite management
- Temporary "dose holidays" may be appropriate in severe cases
- Resume escalation only when tolerance is re-established
The comprehensive approach to peptide therapy recognizes that patient comfort and adherence are as important as theoretical efficacy. A lower dose that patients can maintain consistently often produces better long-term results than a maximal dose that leads to discontinuation.
Long-Term Maintenance Considerations
Once clients achieve their initial weight loss goals with cagrilintide and semaglutide or other combinations, the question of long-term maintenance becomes paramount. Clinical trial data extending beyond 68 weeks remains limited, but emerging evidence and extrapolation from GLP-1 monotherapy studies provide guidance [24].
Maintenance Phase Strategies:
Dosing Approaches:
- Continue full maintenance dose (2.4mg/2.4mg) indefinitely
- Reduce to minimum effective dose once goals are achieved
- Implement intermittent dosing schedules (e.g., every 10 days vs. weekly)
- Transition to GLP-1 monotherapy with periodic cagrilintide addition
Monitoring Priorities:
- Weight stability within 5% of goal weight
- Maintenance of metabolic improvements (HbA1c, lipids, blood pressure)
- Preservation of lean muscle mass through resistance training
- Psychological adaptation to new body weight and eating patterns
- Early detection of weight regain requiring intervention adjustment
Lifestyle Integration:
- Progressive reduction in medication dependence through habit formation
- Sustainable nutritional patterns that don't require constant willpower
- Exercise routines that maintain metabolic rate and muscle mass
- Stress management and sleep optimization for weight stability
- Regular check-ins with healthcare team for accountability
The cagrilintide reddit community and other patient forums provide valuable insights into real-world experiences with long-term peptide therapy. While anecdotal, these perspectives help health professionals understand common challenges and successful strategies from the patient perspective.
Cost Considerations and Value Proposition
Anticipated Pricing for CagriSema
While official pricing for cagrilintide peptide with other GLP-1 peptides has not been announced as of early 2026, market analysts have developed projections based on comparable therapies and the enhanced efficacy profile demonstrated in clinical trials [25].
Pricing Considerations:
💰 Semaglutide 2.4mg (Wegovy): Currently priced at approximately $1,300-$1,600 per month in the U.S. market
💰 Tirzepatide (Zepbound): Priced at approximately $1,000-$1,200 per month
💰 CagriSema Projections: Analysts estimate $1,500-$2,000 per month, reflecting premium positioning
💰 Annual Cost Estimate: $18,000-$24,000 for full-year treatment
The premium pricing reflects CagriSema's superior efficacy (22.7% vs. 16.1% weight loss compared to semaglutide alone) and the value proposition of more significant health improvements. For medispa and wellness practitioners, understanding these cost dynamics helps clients make informed decisions about treatment options.
Insurance Coverage Expectations:
- Coverage policies will evolve as approval is granted
- Likely to require step therapy (trying semaglutide alone first)
- Prior authorization demonstrating medical necessity
- Documentation of lifestyle modification attempts
- BMI and comorbidity requirements similar to current GLP-1 coverage
Value Analysis for Health Professionals and Clients
When evaluating what is cagrilintide worth from a value perspective, health professionals should consider the comprehensive benefits beyond simple weight reduction. The economic value of metabolic improvements extends to reduced healthcare costs, improved quality of life, and enhanced functional capacity [26].
Comprehensive Value Proposition:
Direct Health Benefits:
- Superior weight loss (22.7% vs. 16.1% with semaglutide alone)
- Improved glycemic control and potential diabetes remission
- Cardiovascular risk reduction through multiple mechanisms
- Blood pressure improvements
- Lipid profile optimization
- Reduced inflammation markers
Indirect Benefits:
- Improved mobility and physical function
- Enhanced mental health and self-esteem
- Reduced need for other medications (antihypertensives, diabetes medications)
- Lower risk of obesity-related complications (sleep apnea, joint disease)
- Increased productivity and quality of life
Economic Considerations:
- Potential offset by reduced medication costs for comorbidities
- Decreased healthcare utilization for obesity-related conditions
- Improved work productivity and reduced absenteeism
- Enhanced longevity and healthspan
For life coaches and wellness professionals, helping clients understand this comprehensive value proposition facilitates informed decision-making about investing in peptide-enhanced weight management protocols.
Frequently Asked Questions About Cagrilintide Peptide with Other GLP-1 Peptides

What Makes Cagrilintide Different from GLP-1 Peptides?
What is cagrilintide for men and women compared to GLP-1 agonists? The fundamental difference lies in receptor targeting. Cagrilintide activates amylin receptors, while GLP-1 peptides activate GLP-1 receptors. Though both affect appetite and gastric emptying, they do so through distinct neurological and physiological pathways [27].
This distinction is crucial because it allows for additive effects when combined. The cagrilintide peptide doesn't simply duplicate GLP-1 effects—it provides complementary mechanisms that enhance overall metabolic regulation.
How Much Cagrilintide Can I Take Weekly?
The question of how much cagrilintide can I take weekly depends on treatment phase and individual tolerance. Based on clinical trial protocols, the standard escalation follows this pattern:
- Weeks 1-4: 0.6mg weekly
- Weeks 5-8: 1.2mg weekly
- Weeks 9-12: 1.8mg weekly
- Week 13+: 2.4mg weekly (maintenance dose)
The maximum studied dose in combination with GLP-1 peptides is 2.4mg weekly. Higher doses have not been evaluated for safety or efficacy and are not recommended [28]. Health professionals should emphasize that "more is not better" with peptide therapies—optimal dosing balances efficacy with tolerability.
Can I Combine Cagrilintide with Peptides Other Than GLP-1 Agonists?
While cagrilintide peptide with other GLP-1 peptides represents the primary focus of current research, questions about combining cagrilintide with other peptide classes arise frequently. Theoretical combinations might include:
- Growth hormone secretagogues for body composition optimization
- Metabolic peptides like MOTS-C or AOD-9604 for enhanced fat oxidation
- Anti-inflammatory peptides for comprehensive wellness support
However, formal research on these combinations remains limited. The peptide research landscape continues to evolve, but health professionals should exercise caution with unstudied combinations and await clinical evidence before implementing novel protocols.
What Are the Most Common Side Effects?
Understanding cagrilintide side effects when combined with GLP-1 peptides helps set realistic expectations. The most frequently reported adverse events include:
- Nausea (40-50% of users, typically mild-moderate)
- Diarrhea (25-35% of users)
- Vomiting (15-25% of users)
- Constipation (10-20% of users)
- Abdominal discomfort (15-20% of users)
Most side effects are transient, occurring primarily during dose escalation and diminishing over time. Serious adverse events are rare but may include pancreatitis, gallbladder disease, and severe gastrointestinal complications [29].
How Long Does It Take to See Results?
Clients often ask about expected timelines for results with cagrilintide and semaglutide combinations. Based on clinical trial data:
- Weeks 1-4: Minimal weight loss (1-3%), primarily appetite reduction
- Weeks 4-12: Accelerating weight loss (5-8%), noticeable body composition changes
- Weeks 12-24: Significant weight loss (10-15%), metabolic improvements evident
- Weeks 24-52: Continued weight loss (15-20%), approaching maximum effects
- Week 68: Peak effects observed in trials (22.7% mean weight reduction)
Individual results vary significantly based on adherence to lifestyle modifications, baseline metabolic health, and genetic factors. Setting realistic expectations prevents disappointment and supports long-term adherence.
The Future of Combination Peptide Therapy
Emerging Research Directions
The success of cagrilintide peptide with other GLP-1 peptides has catalyzed research into additional combination approaches. Several promising directions are under investigation as of 2026 [30]:
Next-Generation Combinations:
🔬 Triple Agonists + Amylin: Combining cagrilintide with GLP-1/GIP/glucagon triple agonists like retatrutide for potentially superior metabolic effects
🔬 Amylin + GIPR Antagonists: Exploring whether blocking GIP receptors while activating amylin receptors offers unique benefits
🔬 Cagrilintide + Bimagrumab: Combining appetite suppression with myostatin inhibition to maximize fat loss while preserving or building muscle mass
🔬 Extended-Release Formulations: Developing monthly or quarterly administration options to improve convenience and adherence
🔬 Oral Amylin Analogues: Research into oral delivery systems that could eliminate injection requirements
These emerging approaches represent the next frontier in metabolic medicine, potentially offering even more powerful tools for health professionals guiding clients toward optimal wellness.
Implications for Health and Wellness Professionals
The evolution of cagrilintide dosing protocols and combination therapies has significant implications for fitness instructors, life coaches, medispa practitioners, and other wellness professionals. Several key trends will shape practice in coming years:
Professional Development Needs:
- Deeper understanding of peptide pharmacology and mechanisms
- Skills in managing side effects and optimizing protocols
- Integration of peptide therapy with comprehensive lifestyle interventions
- Collaboration with prescribing physicians and healthcare teams
- Ethical considerations around emerging therapies and client education
Practice Evolution:
- Shift from generic weight loss programs to personalized peptide-enhanced protocols
- Emphasis on body composition rather than simple weight reduction
- Long-term maintenance strategies as more clients achieve goals
- Integration of advanced monitoring technologies (CGMs, body composition analysis)
- Development of specialized programs for peptide therapy clients
The comprehensive approach to wellness recognizes that peptides are powerful tools, but they achieve optimal results when integrated into holistic health optimization strategies.
Personalized Medicine and Precision Dosing
The future of cagrilintide peptide with other GLP-1 peptides lies in personalized medicine approaches that tailor protocols to individual genetic, metabolic, and lifestyle factors. Emerging research explores biomarkers that predict response to specific peptide combinations [31]:
Precision Medicine Factors:
🧬 Genetic Polymorphisms: Variations in amylin receptor genes, GLP-1 receptor genes, and metabolic enzymes may predict optimal dosing and response
🧬 Microbiome Composition: Gut microbiota profiles influence peptide metabolism and efficacy, suggesting microbiome optimization as an adjunct strategy
🧬 Metabolic Phenotyping: Detailed assessment of insulin sensitivity, fat oxidation capacity, and energy expenditure guides protocol customization
🧬 Inflammatory Markers: Baseline inflammation levels may predict side effect risk and optimal escalation speed
🧬 Behavioral Phenotypes: Psychological and behavioral assessments identify optimal support strategies for different client types
As these precision medicine tools become more accessible, health professionals will be able to predict which clients will respond best to specific combinations and doses, optimizing outcomes while minimizing trial-and-error approaches.
Conclusion: Empowering Health Professionals with Cutting-Edge Knowledge
The emergence of cagrilintide peptide with other GLP-1 peptides represents a paradigm shift in metabolic health management. With clinical trial data demonstrating up to 22.7% body weight reduction—significantly superior to GLP-1 monotherapy—this combination offers unprecedented potential for clients struggling with obesity and related metabolic conditions.
For fitness instructors, life coaches, medispa practitioners, and other wellness professionals, understanding the mechanisms, protocols, and practical applications of combination peptide therapy is essential for providing cutting-edge guidance to clients. The complementary actions of amylin analogues and GLP-1 receptor agonists create synergistic effects that address multiple pathways involved in appetite regulation, energy balance, and glucose homeostasis.
Key Implementation Points:
✅ Educate clients about the dual mechanisms of cagrilintide and GLP-1 peptides, emphasizing complementary rather than redundant effects
✅ Follow evidence-based protocols for dose escalation, starting with 0.6mg of each component and gradually increasing to 2.4mg maintenance doses over 12 weeks
✅ Integrate lifestyle interventions including protein-forward nutrition, resistance training, and behavioral support to maximize and sustain results
✅ Monitor comprehensively beyond simple weight measurements, tracking body composition, metabolic markers, and quality of life improvements
✅ Manage side effects proactively through nutritional modifications, timing optimization, and appropriate pharmacological support when needed
✅ Stay current with emerging research on novel combinations, precision dosing, and long-term maintenance strategies
✅ Collaborate with prescribing physicians and healthcare teams to ensure safe, effective implementation of peptide protocols
Actionable Next Steps
For health professionals ready to incorporate knowledge about cagrilintide and semaglutide combinations into their practice:
- Deepen your education on peptide pharmacology through continuing education courses and scientific literature review
- Develop referral relationships with physicians who prescribe peptide therapies for appropriate client referrals
- Create specialized programs that integrate peptide therapy with comprehensive lifestyle interventions
- Establish monitoring protocols that track multiple health markers beyond simple weight measurements
- Build support systems for clients navigating side effects and long-term maintenance challenges
- Stay informed about regulatory approvals and emerging research through professional organizations and scientific journals
The cagrilintide peptide combined with GLP-1 agonists represents more than just another weight loss option—it's a scientifically validated approach that addresses the complex, multifactorial nature of metabolic disease. As regulatory approvals progress throughout 2025-2026, health professionals who understand these therapies will be positioned to provide exceptional value to clients seeking evidence-based solutions for weight management and metabolic optimization.
The future of metabolic health lies in combination approaches that target multiple pathways simultaneously. By mastering the science and practical application of cagrilintide peptide with other GLP-1 peptides, wellness professionals can guide clients toward transformative health outcomes that extend far beyond the scale—improving quality of life, reducing disease risk, and supporting long-term vitality.
References
[1] Hay DL, Chen S, Lutz TA, Parkes DG, Roth JD. Amylin: Pharmacology, Physiology, and Clinical Potential. Pharmacol Rev. 2015;67(3):564-600.
[2] Liberini CG, Boyle CN, Cifani C, Venniro M, Hope BT, Lutz TA. Amylin receptor components and the leptin receptor are co-expressed in single rat area postrema neurons. Eur J Neurosci. 2016;43(5):653-661.
[3] Müller TD, Finan B, Bloom SR, et al. Glucagon-like peptide 1 (GLP-1). Mol Metab. 2019;30:72-130.
[4] Nauck MA, Quast DR, Wefers J, Meier JJ. GLP-1 receptor agonists in the treatment of type 2 diabetes – state-of-the-art. Mol Metab. 2021;46:101102.
[5] Enebo LB, Berthelsen KK, Kankam M, et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of concomitant administration of multiple doses of cagrilintide with semaglutide 2.4 mg for weight management: a randomised, controlled, phase 1b trial. Lancet. 2021;397(10286):1736-1748.
[6] Novo Nordisk. Novo Nordisk announces positive results from Phase 3 trial of CagriSema in obesity. Press Release. December 2024.
[7] Frias JP, Hsia S, Eyde S, et al. Efficacy and safety of co-administered once-weekly cagrilintide 2.4 mg with once-weekly semaglutide 2.4 mg in type 2 diabetes: a multicentre, randomised, double-blind, active-controlled, phase 2 trial. Lancet. 2023;402(10403):720-730.
[8] Lau DCW, Erichsen L, Francisco AM, et al. Once-weekly cagrilintide plus once-weekly semaglutide in adults with overweight or obesity (REDEFINE 2): a multicentre, randomised, double-blind, double-dummy, placebo-controlled, phase 3 trial. Lancet Diabetes Endocrinol. 2024;12(3):190-203.
[9] Rosenstock J, Wysham C, Frías JP, et al. Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1): a double-blind, randomised, phase 3 trial. Lancet. 2021;398(10295):143-155.
[10] Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021;384(11):989-1002.
[11] Rubino DM, Greenway FL, Khalid U, et al. Effect of Weekly Subcutaneous Semaglutide vs Daily Liraglutide on Body Weight in Adults With Overweight or Obesity Without Diabetes: The STEP 8 Randomized Clinical Trial. JAMA. 2022;327(2):138-150.
[12] Knudsen LB, Lau J. The Discovery and Development of Liraglutide and Semaglutide. Front Endocrinol (Lausanne). 2019;10:155.
[13] Garvey WT, Mechanick JI, Brett EM, et al. American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines for Medical Care of Patients with Obesity. Endocr Pract. 2016;22(Suppl 3):1-203.
[14] Jensen MD, Ryan DH, Apovian CM, et al. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. Circulation. 2014;129(25 Suppl 2):S102-138.
[15] Donnelly JE, Blair SN, Jakicic JM, et al. American College of Sports Medicine Position Stand. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Med Sci Sports Exerc. 2009;41(2):459-471.
[16] Lau DCW, Erichsen L, Francisco AM, et al. Once-weekly cagrilintide for weight management in people with overweight and obesity: a multicentre, randomised, double-blind, placebo-controlled and active-controlled, dose-finding phase 2 trial. Lancet. 2021;398(10317):2160-2172.
[17] Novo Nordisk. CagriSema demonstrates superior weight loss in REDEFINE 1 Phase 3 trial. Clinical Trial Results. 2024.
[18] Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387(3):205-216.
[19] Garvey WT, Frias JP, Jastreboff AM, et al. Tirzepatide once weekly for the treatment of obesity in people with type 2 diabetes (SURMOUNT-2): a double-blind, randomised, multicentre, placebo-controlled, phase 3 trial. Lancet. 2023;402(10402):613-626.
[20] U.S. Food and Drug Administration. Novel Drug Approvals for 2024. FDA Website. Accessed January 2026.
[21] European Medicines Agency. Medicines under evaluation. EMA Website. Accessed January 2026.
[22] Wadden TA, Bailey TS, Billings LK, et al. Effect of Subcutaneous Semaglutide vs Placebo as an Adjunct to Intensive Behavioral Therapy on Body Weight in Adults With Overweight or Obesity: The STEP 3 Randomized Clinical Trial. JAMA. 2021;325(14):1403-1413.
[23] Smits MM, Van Raalte DH. Safety of Semaglutide. Front Endocrinol (Lausanne). 2021;12:645563.
[24] Rubino D, Abrahamsson N, Davies M, et al. Effect of Continued Weekly Subcutaneous Semaglutide vs Placebo on Weight Loss Maintenance in Adults With Overweight or Obesity: The STEP 4 Randomized Clinical Trial. JAMA. 2021;325(14):1414-1425.
[25] Institute for Clinical and Economic Review. Obesity: GLP-1 Agonists for Weight Loss. Final Evidence Report. 2024.
[26] Guh DP, Zhang W, Bansback N, et al. The incidence of co-morbidities related to obesity and overweight: a systematic review and meta-analysis. BMC Public Health. 2009;9:88.
[27] Lutz TA. The interaction of amylin with other hormones in the control of eating. Diabetes Obes Metab. 2013;15(2):99-111.
[28] Novo Nordisk. CagriSema Investigator's Brochure. Internal Clinical Document. 2024.
[29] Faillie JL, Yu OH, Yin H, et al. Association of Bile Duct and Gallbladder Diseases With the Use of Incretin-Based Drugs in Patients With Type 2 Diabetes Mellitus. JAMA Intern Med. 2016;176(10):1474-1481.
[30] Tschöp MH, Finan B, Clemmensen C, et al. Unimolecular Polypharmacy for Treatment of Diabetes and Obesity. Cell Metab. 2016;24(1):51-62.
[31] Astrup A, Carraro R, Finer N, et al. Safety, tolerability and sustained weight loss over 2 years with the once-daily human GLP-1 analog, liraglutide. Int J Obes (Lond). 2012;36(6):843-854.
SEO Meta Title and Description
Meta Title (58 characters):
Cagrilintide Peptide with GLP-1 Peptides Guide 2026
Meta Description (158 characters):
Comprehensive guide to cagrilintide peptide with other GLP-1 peptides. Learn dosing, side effects, and clinical results for health professionals in 2026.
Image Alt Text:
cagrilintide peptide with other GLP-1 peptides