Cagrilintide Dosage with Retatrutide: A Comprehensive 2026 Research Guide

The landscape of metabolic health research is evolving rapidly, and the combination of cagrilintide dosage with retatrutide represents one of the most intriguing—yet understudied—frontiers in peptide therapy. As fitness instructors, health coaches, and medispa professionals seek cutting-edge solutions for their clients, understanding these novel compounds becomes essential. While both peptides show remarkable promise individually, their potential combination raises critical questions about dosing protocols, safety considerations, and therapeutic rationale that demand careful examination.
Key Takeaways
- Cagrilintide is a long-acting amylin analogue with clinical trial doses ranging from 0.3 mg to 4.5 mg weekly, while retatrutide is a triple agonist studied at 4 mg, 8 mg, and 12 mg weekly doses
- No published clinical trials currently exist evaluating the cagrilintide dosage with retatrutide combination, making any combined use experimental and off-label
- Both peptides work through different mechanisms—cagrilintide activates amylin receptors while retatrutide targets GIP, GLP-1, and glucagon receptors—suggesting potential complementary effects
- Gastrointestinal side effects (nausea, vomiting) occur in 60-80% of participants at higher doses for both compounds, raising concerns about additive effects when combined
- Proper dose escalation protocols spanning 8-20 weeks are critical for tolerability and adherence in peptide-based metabolic therapies
Understanding Cagrilintide: Mechanism and Dosage Fundamentals
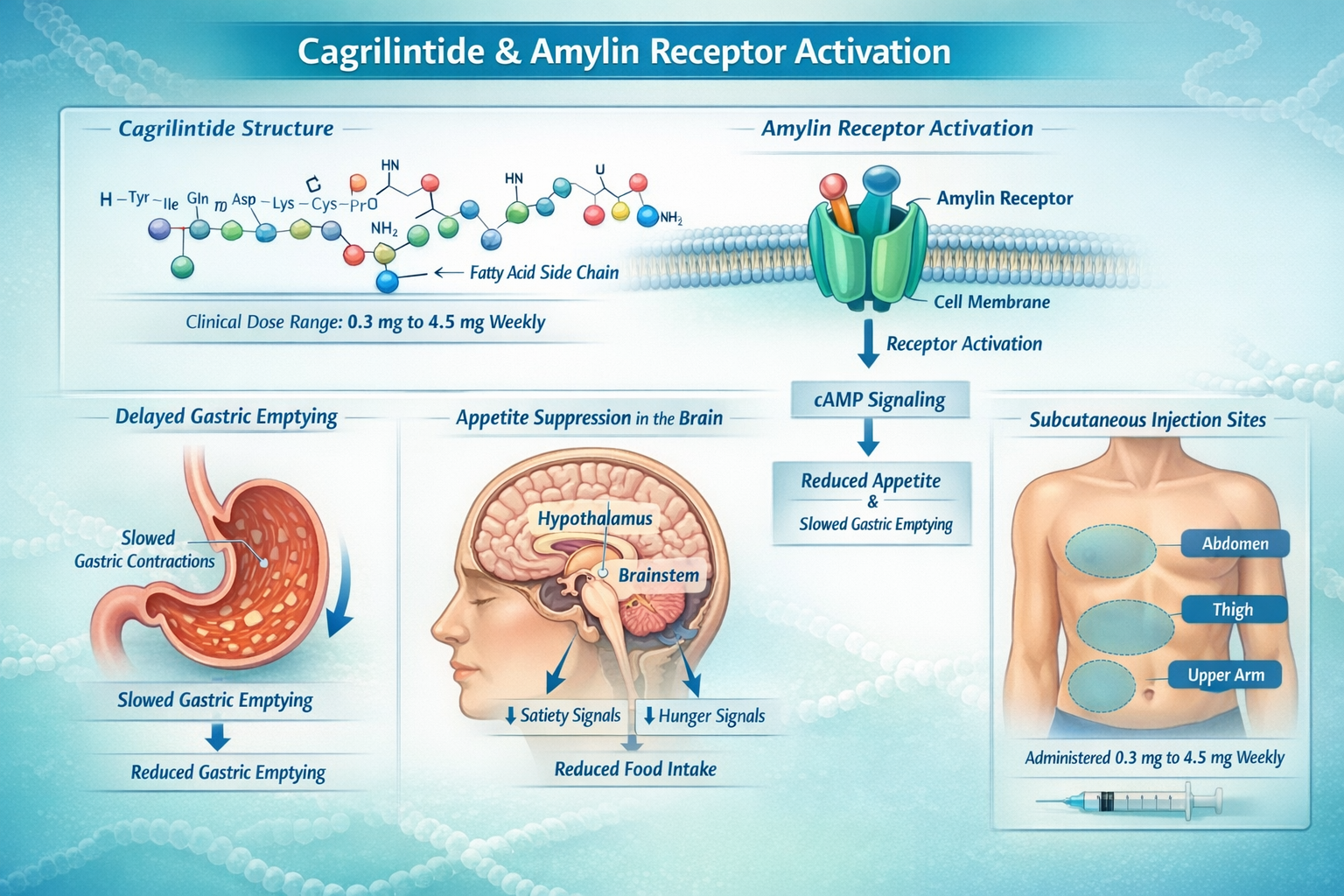
What Is Cagrilintide? 🔬
Cagrilintide represents a significant advancement in peptide-based metabolic research. Developed by Novo Nordisk, this long-acting amylin analogue works by activating amylin receptors throughout the body, particularly in areas that regulate appetite and digestive function [1]. Unlike natural amylin, which has a very short half-life, cagrilintide's molecular structure allows for once-weekly administration, making it considerably more practical for research applications.
The cagrilintide mechanism of action centers on two primary pathways:
- Appetite suppression through central nervous system amylin receptor activation
- Delayed gastric emptying that promotes satiety and reduces food intake
- Reduced glucagon secretion that helps regulate glucose metabolism
For professionals exploring peptide research options, understanding these fundamental mechanisms is crucial before considering any dosing protocols.
Cagrilintide Dosage Ranges in Clinical Research
Clinical trials have investigated a wide spectrum of cagrilintide dosage protocols. The research demonstrates that effective doses typically fall within specific ranges [2]:
| Dosage Level | Weekly Dose | Research Context |
|---|---|---|
| Low | 0.3 mg – 1.2 mg | Initial tolerability studies |
| Moderate | 1.5 mg – 2.4 mg | Phase 2/3 efficacy trials |
| High | 3.0 mg – 4.5 mg | Maximum tolerated dose research |
The cagrilintide starting dose in most clinical protocols begins at 0.3 mg weekly, with gradual escalation over 8-12 weeks to minimize gastrointestinal side effects [3]. This titration approach has proven essential for treatment adherence and participant comfort.
When examining cagrilintide dosing strategies, researchers have found that the 2.4 mg weekly dose offers an optimal balance between efficacy and tolerability in many study populations. This dose has been extensively studied in combination with semaglutide under the investigational name CagriSema, achieving up to 22.7% weight reduction at 68 weeks in phase 3 trials [4].
Cagrilintide 10mg is available for qualified research purposes, though proper reconstitution and handling protocols must be followed strictly.
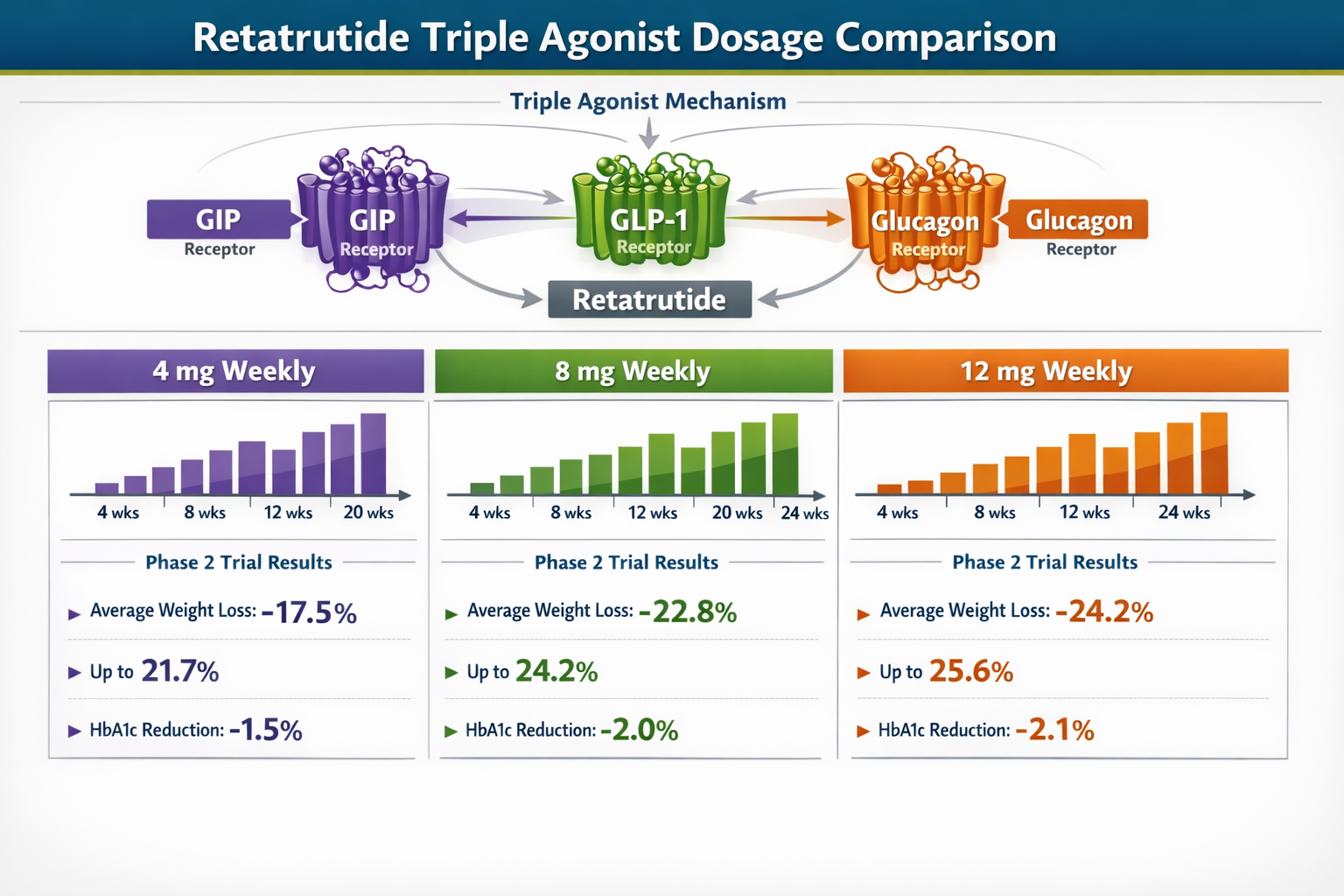
Retatrutide: The Triple Agonist Approach
Retatrutide's Unique Mechanism
Retatrutide, developed by Eli Lilly, represents a fundamentally different approach to metabolic regulation. As a triple agonist, it simultaneously activates three distinct receptor systems [5]:
- GIP (Glucose-dependent Insulinotropic Polypeptide) receptors – enhance insulin secretion and may have direct effects on adipose tissue
- GLP-1 (Glucagon-like Peptide-1) receptors – promote satiety, slow gastric emptying, and improve glucose regulation
- Glucagon receptors – increase energy expenditure and may enhance fat oxidation
This multi-receptor targeting strategy distinguishes retatrutide from single-pathway peptides and creates a complex metabolic profile that researchers are still working to fully characterize.
Retatrutide Dosage Protocols from Clinical Trials
Phase 2 clinical trials published in The New England Journal of Medicine in 2023 established the foundational retatrutide dosage framework [6]:
Standard Retatrutide Dosing Schedule:
- 4 mg weekly – Lower effective dose with moderate efficacy
- 8 mg weekly – Mid-range dose with enhanced metabolic effects
- 12 mg weekly – Maximum studied dose achieving 24.2% mean weight reduction
The 12 mg dose demonstrated the most significant metabolic effects but also carried the highest incidence of gastrointestinal adverse events, with nausea reported in 60-80% of participants [7]. Discontinuation rates due to side effects reached approximately 10% across all dose levels.
Retatrutide's dosing protocol typically involves gradual escalation starting from 2 mg weekly, increasing by 2-4 mg increments every 4 weeks until the target maintenance dose is achieved. This approach mirrors strategies used with other peptide therapies to optimize tolerability.
Cagrilintide Dosage with Retatrutide: The Theoretical Framework
Why Consider Combining These Peptides?
The question of cagrilintide dosage with retatrutide emerges from a logical therapeutic rationale: these compounds work through complementary mechanisms that theoretically could produce synergistic effects. While cagrilintide primarily modulates appetite through amylin pathways, retatrutide engages multiple incretin and metabolic hormone systems simultaneously [8].
Potential theoretical advantages include:
- Multi-pathway appetite suppression combining amylin and incretin signals
- Enhanced metabolic flexibility through glucagon receptor activation (retatrutide) and amylin-mediated effects (cagrilintide)
- Complementary gastric emptying effects from both compounds
- Broader receptor coverage addressing multiple aspects of energy homeostasis
However, it's critical to emphasize: no published clinical trials have evaluated the cagrilintide dosage with retatrutide combination as of 2026. Any consideration of this combination exists purely in theoretical and experimental contexts.
The Absence of Clinical Data
The lack of formal research on cagrilintide dosage with retatrutide combinations stems from several factors:
🚫 Competitive development – Novo Nordisk (cagrilintide) and Eli Lilly (retatrutide) are pharmaceutical competitors with no incentive to study each other's compounds together
🚫 Regulatory pathways – Both medications remain in phase 3 development without FDA approval for metabolic applications
🚫 Safety unknowns – Drug-drug interactions, additive side effects, and optimal dosing ratios remain completely uncharacterized
For wellness professionals and peptide researchers, this data gap presents significant challenges in developing evidence-based protocols.
Cagrilintide Dosage with Retatrutide: Safety Considerations
Overlapping Side Effect Profiles
Both cagrilintide and retatrutide share remarkably similar adverse event patterns, raising legitimate concerns about potential additive effects when considering cagrilintide dosage with retatrutide combinations [9].
Common Side Effects for Both Compounds:
| Side Effect | Cagrilintide Incidence | Retatrutide Incidence |
|---|---|---|
| Nausea | 40-60% (dose-dependent) | 60-80% (at 12 mg) |
| Vomiting | 15-30% | 20-35% |
| Diarrhea | 20-35% | 25-40% |
| Injection site reactions | 10-20% | 15-25% |
| Constipation | 15-25% | 10-20% |
The high prevalence of gastrointestinal adverse events with each compound individually suggests that combining them could potentially result in:
- More severe nausea and vomiting episodes
- Higher discontinuation rates
- Reduced treatment adherence
- Potential nutritional deficiencies from excessive appetite suppression
- Dehydration risks from combined GI effects
Health and wellness professionals must understand these risks when evaluating any experimental protocols involving cagrilintide alongside other metabolic peptides.
Hypothetical Dosing Considerations
While no evidence-based cagrilintide dosage with retatrutide protocol exists, theoretical frameworks based on monotherapy data suggest several principles that would likely apply:
Conservative Approach Principles:
- Start with minimal doses – Perhaps 0.3 mg cagrilintide + 2 mg retatrutide weekly
- Extended titration periods – 16-24 weeks to reach target doses instead of standard 8-12 weeks
- Alternating escalation – Increase one compound while holding the other stable
- Frequent monitoring – Weekly check-ins during initial phases
- Lower maximum doses – Perhaps 1.2 mg cagrilintide + 8 mg retatrutide rather than maximum monotherapy doses
These suggestions remain purely speculative and should not be interpreted as clinical recommendations. The absence of pharmacokinetic and pharmacodynamic interaction studies makes any cagrilintide dosage with retatrutide combination inherently experimental.
Clinical Context: Approved Combinations vs. Experimental Protocols
CagriSema: The Evidence-Based Cagrilintide Combination
To understand the potential for cagrilintide dosage with retatrutide, examining the well-studied cagrilintide and semaglutide combination (CagriSema) provides valuable context [10].
The REDEFINE clinical trial program has established that:
- Cagrilintide 2.4 mg + Semaglutide 2.4 mg weekly produces superior weight loss compared to either agent alone
- The combination achieved 22.7% mean weight reduction at 68 weeks
- Side effect profiles were manageable with proper dose escalation
- The amylin + GLP-1 agonist combination demonstrated synergistic effects
This validated combination demonstrates that pairing cagrilintide with incretin-based therapies can work effectively. However, semaglutide is a single GLP-1 agonist, while retatrutide activates three receptor systems simultaneously, making direct comparisons problematic.
For those researching metabolic peptides, understanding dosage protocols across different compound classes provides essential context.
Why Retatrutide Combinations Remain Unexplored
Unlike cagrilintide, which Novo Nordisk has actively studied in combination with their GLP-1 agonist semaglutide, retatrutide has been developed primarily as a monotherapy by Eli Lilly [11]. The compound's triple agonist mechanism may be considered sufficiently comprehensive that additional combinations aren't prioritized in the development pipeline.
Key factors limiting cagrilintide dosage with retatrutide research:
- Pharmaceutical competition prevents collaborative studies
- Monotherapy efficacy – Retatrutide alone achieves 24.2% weight reduction, potentially reducing perceived need for combinations
- Regulatory complexity – Combining investigational drugs creates significant approval pathway challenges
- Safety uncertainty – Triple agonist activity plus amylin agonism creates unpredictable interaction potential
Practical Guidance for Wellness Professionals
Educating Clients About Experimental Combinations
Fitness instructors, health coaches, and medispa professionals increasingly encounter clients asking about cutting-edge peptide protocols, including cagrilintide dosage with retatrutide combinations. Providing accurate, evidence-based information is essential [12].
Key Educational Points:
✅ Emphasize the experimental nature – No clinical trials support this combination
✅ Highlight individual compound efficacy – Both peptides show strong effects as monotherapies
✅ Discuss approved alternatives – CagriSema and other studied combinations offer evidence-based options
✅ Explain regulatory status – Neither compound has FDA approval for metabolic applications as of 2026
✅ Address safety concerns – Overlapping side effect profiles create legitimate risks
When clients express interest in research peptides, directing them toward compounds with established safety profiles and dosing protocols serves their best interests.
Monitoring and Documentation Considerations
For research contexts where cagrilintide dosage with retatrutide protocols might be explored under appropriate oversight, comprehensive monitoring becomes absolutely essential:
Suggested Monitoring Parameters:
- Weekly weight and body composition measurements
- Gastrointestinal symptom logs (nausea severity scales, vomiting frequency, bowel patterns)
- Nutritional intake tracking to ensure adequate caloric and nutrient consumption
- Hydration status assessment
- Vital signs including heart rate and blood pressure
- Laboratory markers (glucose, lipids, liver function, kidney function)
- Subjective well-being assessments
This level of monitoring exceeds what's typically required for single-agent protocols but reflects the increased uncertainty surrounding combination approaches without clinical trial data.
Cagrilintide Dosage Chart and Titration Strategies
Evidence-Based Cagrilintide Titration
While specific cagrilintide dosage with retatrutide charts don't exist, understanding standard cagrilintide escalation provides a foundation. The following represents typical monotherapy protocols from clinical research [13]:
Standard Cagrilintide Dosing Schedule:
| Week | Dose | Notes |
|---|---|---|
| 1-4 | 0.3 mg | Initial tolerability assessment |
| 5-8 | 0.6 mg | First escalation if well-tolerated |
| 9-12 | 1.2 mg | Therapeutic range begins |
| 13-16 | 2.4 mg | Target maintenance dose |
| 17+ | 2.4 mg | Continue if effective and tolerated |
Some protocols extend to 4.5 mg for participants who tolerate 2.4 mg well and require additional metabolic effects, though this higher dose significantly increases adverse event rates.
The cagrilintide dosage schedule emphasizes gradual progression, typically increasing doses every 4 weeks rather than more aggressive 2-week escalations seen with some other peptides. This conservative approach reflects the compound's potent effects on gastric emptying and appetite.
Research-grade cagrilintide requires proper reconstitution and storage protocols to maintain stability and potency throughout extended dosing schedules.
Retatrutide Titration Protocols
Retatrutide follows a similar gradual escalation pattern, though the dose increments differ [14]:
Typical Retatrutide Escalation:
- Weeks 1-4: 2 mg weekly
- Weeks 5-8: 4 mg weekly (first studied therapeutic dose)
- Weeks 9-12: 6-8 mg weekly (intermediate range)
- Weeks 13-16: 8-12 mg weekly (maximum studied doses)
- Week 17+: Maintenance at target dose
The longer titration period (16 weeks to reach maximum dose) helps mitigate the significant gastrointestinal effects associated with retatrutide's multi-receptor activation.
Hypothetical Cagrilintide Dosage with Retatrutide Protocols
Conservative Combination Framework
Given the absence of clinical data, any cagrilintide dosage with retatrutide protocol must be considered highly experimental. The following represents a theoretical ultra-conservative approach based on monotherapy principles:
Hypothetical Ultra-Conservative Schedule:
| Week | Cagrilintide | Retatrutide | Rationale |
|---|---|---|---|
| 1-4 | 0.3 mg | 0 mg | Establish cagrilintide tolerance alone |
| 5-8 | 0.3 mg | 2 mg | Introduce retatrutide at minimum dose |
| 9-12 | 0.6 mg | 2 mg | Escalate cagrilintide only |
| 13-16 | 0.6 mg | 4 mg | Escalate retatrutide only |
| 17-20 | 1.2 mg | 4 mg | Escalate cagrilintide |
| 21-24 | 1.2 mg | 6-8 mg | Escalate retatrutide |
| 25+ | Assess | Assess | Evaluate need for further increases |
This extended 24-week titration period doubles the typical monotherapy escalation timeline, reflecting the complete absence of interaction data and the overlapping side effect profiles.
Critical Considerations:
⚠️ This schedule is purely theoretical and not based on clinical evidence
⚠️ Individual tolerance varies dramatically—some may not tolerate even these conservative doses
⚠️ Maximum combined doses would likely need to remain below monotherapy maximums (perhaps 2.4 mg cagrilintide + 8 mg retatrutide as absolute ceiling)
⚠️ Medical supervision would be essential throughout any such protocol
Alternative Approach: Sequential Rather Than Concurrent
An arguably safer alternative to simultaneous cagrilintide dosage with retatrutide administration involves sequential optimization:
- Optimize one compound first – Reach stable, effective dose of either cagrilintide or retatrutide
- Assess response – Evaluate metabolic effects over 12-16 weeks
- Introduce second compound – Only if additional effects are needed
- Ultra-gradual second titration – Even slower escalation of the added compound
- Continuous reassessment – Regular evaluation of whether combination provides benefits beyond monotherapy
This approach provides clearer attribution of effects and side effects to specific compounds, though it significantly extends the overall timeline.
For professionals working with clients interested in peptide research, sequential approaches often provide better risk-benefit profiles when exploring novel combinations.
Side Effects and Risk Mitigation Strategies
Managing Gastrointestinal Effects
Given that both compounds produce significant GI side effects, managing these symptoms becomes paramount in any cagrilintide dosage with retatrutide scenario [15].
Evidence-Based Mitigation Strategies:
🥗 Dietary modifications:
- Smaller, more frequent meals (5-6 per day instead of 3)
- Lower fat content (fat delays gastric emptying further)
- Adequate hydration (1.5-2 liters daily minimum)
- Avoiding trigger foods identified through symptom tracking
💊 Symptomatic management:
- Ginger supplementation (1-2 grams daily) for nausea
- Vitamin B6 (25-50 mg daily) may reduce nausea
- Antiemetic medications if prescribed by healthcare provider
- Probiotics to support digestive function
⏰ Timing strategies:
- Administering injections before bed to sleep through peak nausea
- Spacing injections from large meals
- Maintaining consistent weekly injection schedule
📊 Monitoring and adjustment:
- Daily symptom logs to identify patterns
- Willingness to reduce or pause doses if symptoms become severe
- Regular communication with supervising healthcare provider
Recognizing Serious Adverse Events
While most side effects are manageable, certain symptoms require immediate medical attention:
Red Flag Symptoms:
- Severe, persistent vomiting preventing hydration
- Signs of pancreatitis (severe upper abdominal pain radiating to back)
- Symptoms of gallbladder disease (right upper abdominal pain, especially after meals)
- Severe allergic reactions (difficulty breathing, facial swelling)
- Rapid heart rate or cardiac symptoms
- Extreme fatigue or altered mental status
These concerns apply to monotherapy with either compound but may be amplified in combination scenarios. Understanding peptide safety profiles across different compound classes helps contextualize these risks.
Regulatory and Ethical Considerations
Current Approval Status (2026)
As of 2026, neither cagrilintide nor retatrutide has received FDA approval for obesity treatment or metabolic applications. Both remain in phase 3 clinical development programs [16]:
Cagrilintide Status:
- Phase 3 trials ongoing for CagriSema (cagrilintide + semaglutide combination)
- Expected approval timeline: 2027-2028 for the combination product
- Monotherapy development pathway unclear
Retatrutide Status:
- Phase 3 trials initiated in 2024-2025
- Monotherapy development for obesity and metabolic dysfunction
- Expected approval timeline: 2028-2029 if trials succeed
This regulatory status means any use of these compounds, whether individually or in cagrilintide dosage with retatrutide combinations, occurs in research contexts or through off-label prescribing once (if) individual approvals are obtained.
Ethical Considerations for Wellness Professionals
Fitness instructors, health coaches, and medispa professionals occupy a unique position in the wellness ecosystem. While clients may seek guidance on cutting-edge interventions like cagrilintide dosage with retatrutide, ethical practice requires clear boundaries:
Professional Responsibilities:
✓ Provide accurate information about regulatory status and evidence gaps
✓ Avoid prescriptive recommendations for unapproved or experimental combinations
✓ Refer to qualified medical professionals for any peptide therapy decisions
✓ Focus on evidence-based interventions within scope of practice
✓ Maintain current knowledge about evolving research and safety data
✓ Document client education regarding experimental nature of novel protocols
The enthusiasm for innovative approaches must be balanced against the fundamental principle of "first, do no harm." When clinical evidence is absent, as with cagrilintide dosage with retatrutide combinations, conservative caution serves clients' best interests.
Future Research Directions and Emerging Data
What Studies Are Needed?
The gap in knowledge regarding cagrilintide dosage with retatrutide combinations could be addressed through several research approaches:
Priority Research Questions:
-
Pharmacokinetic interactions – Do these compounds affect each other's absorption, distribution, or metabolism?
-
Pharmacodynamic synergy – Do combined receptor activations produce additive, synergistic, or antagonistic effects?
-
Optimal dose ratios – What proportions of each compound maximize efficacy while minimizing side effects?
-
Safety profile characterization – What is the true incidence and severity of adverse events with combination therapy?
-
Long-term outcomes – Do combinations provide sustained benefits beyond monotherapy over 1-2 year timeframes?
-
Comparative effectiveness – How do theoretical cagrilintide-retatrutide combinations compare to established protocols like CagriSema?
Academic institutions, independent research organizations, or collaborative pharmaceutical initiatives could theoretically pursue these questions, though competitive dynamics make such studies unlikely in the near term.
Monitoring the Scientific Literature
For wellness professionals committed to evidence-based practice, staying current with emerging research is essential. Key resources include:
- ClinicalTrials.gov – Search for new trials involving cagrilintide or retatrutide
- PubMed/MEDLINE – Set alerts for publications on these compounds
- Major medical journals – New England Journal of Medicine, The Lancet, JAMA for high-impact studies
- Endocrine Society conferences – Annual meetings where phase 3 data is often first presented
- FDA briefing documents – Detailed safety and efficacy data released during approval processes
Understanding the broader landscape of peptide research helps contextualize developments with specific compounds.
Comparing Cagrilintide Combinations: Semaglutide vs. Retatrutide
Why CagriSema Works: Lessons for Potential Retatrutide Combinations
The success of cagrilintide and semaglutide (CagriSema) in clinical trials offers insights that might apply to cagrilintide dosage with retatrutide scenarios [17]:
CagriSema Success Factors:
- Complementary mechanisms – Amylin (cagrilintide) and GLP-1 (semaglutide) work through different pathways
- Overlapping but manageable side effects – Both cause GI effects, but titration protocols minimize discontinuation
- Synergistic efficacy – 22.7% weight loss exceeds either compound alone
- Single pharmaceutical developer – Novo Nordisk controls both compounds, enabling coordinated development
Key Differences with Retatrutide:
| Factor | CagriSema | Hypothetical Cagrilintide + Retatrutide |
|---|---|---|
| Receptor targets | Amylin + GLP-1 (2 pathways) | Amylin + GIP + GLP-1 + Glucagon (4 pathways) |
| Developer | Single company (Novo Nordisk) | Competing companies |
| Clinical data | Extensive phase 3 trials | Zero published trials |
| Regulatory path | Clear combination product | Undefined |
| Side effect profile | Well-characterized | Unknown interactions |
The additional receptor targets with retatrutide create both potential advantages (broader metabolic effects) and concerns (more complex interactions, potentially greater side effects).
Tirzepatide: Another Relevant Comparison
Tirzepatide (Mounjaro/Zepbound), a dual GIP/GLP-1 agonist approved by the FDA, provides another useful reference point. Some researchers and clinicians have explored cagrilintide dosage with tirzepatide combinations, though again without formal clinical trial support [18].
The tirzepatide comparison is relevant because:
- It shares GIP and GLP-1 agonism with retatrutide (but lacks glucagon agonism)
- It has actual FDA approval and established safety profiles
- Anecdotal reports of cagrilintide-tirzepatide combinations exist in clinical practice
- The dual agonist mechanism is less complex than retatrutide's triple agonism
For those researching metabolic peptides, understanding the distinctions between different compound classes helps inform evidence-based decision-making.
Practical Applications for Different Professional Contexts
For Fitness Instructors and Personal Trainers
Fitness professionals increasingly encounter clients using or considering peptide therapies. When cagrilintide dosage with retatrutide questions arise:
Appropriate Responses:
✓ Acknowledge that these are investigational compounds without approval for fitness applications
✓ Emphasize that no combination protocols exist in published research
✓ Redirect focus to evidence-based nutrition and training approaches that complement any medical interventions
✓ Suggest clients consult qualified medical professionals for peptide therapy decisions
✓ Provide education about realistic expectations and potential side effects
Training Considerations:
Clients using potent appetite suppressants like cagrilintide or retatrutide may experience:
- Reduced energy availability requiring modified training intensity
- Increased fatigue necessitating longer recovery periods
- Potential muscle loss if protein intake becomes inadequate
- Hydration challenges from GI side effects
Programming should account for these factors, with emphasis on adequate nutrition, progressive overload appropriate to energy status, and careful monitoring of performance metrics.
For Health Coaches and Wellness Consultants
Health coaches can provide valuable support for clients exploring peptide therapies while maintaining appropriate professional boundaries:
Value-Added Services:
- Nutrition optimization to support peptide therapy and minimize side effects
- Lifestyle modification strategies that complement metabolic interventions
- Accountability systems for medication adherence and symptom tracking
- Stress management techniques that support overall metabolic health
- Sleep optimization protocols that enhance peptide effectiveness
When discussing cagrilintide dosage with retatrutide, coaches should:
- Clearly state this combination lacks clinical evidence
- Encourage medical consultation before any peptide use
- Offer support services that enhance outcomes regardless of specific peptide protocol
- Maintain detailed records of client education provided
- Stay within scope of practice boundaries
Resources about peptide research fundamentals can help coaches provide informed, evidence-based guidance.
For Medispa and Aesthetic Professionals
Medical spas and aesthetic practices increasingly offer peptide therapies as part of comprehensive wellness programs. Regarding cagrilintide dosage with retatrutide:
Clinical Practice Considerations:
- Ensure medical director oversight for any peptide therapy protocols
- Develop comprehensive informed consent documents addressing experimental nature of novel combinations
- Implement robust adverse event monitoring and reporting systems
- Maintain current knowledge of regulatory developments
- Consider liability implications of off-label combination therapies
- Establish clear referral pathways to endocrinology or obesity medicine specialists
Patient Education Materials:
Creating clear, accurate educational resources about cagrilintide, retatrutide, and the absence of combination data serves patient safety and informed decision-making. Materials should address:
- Mechanism of action for each compound
- Evidence base (strong for monotherapy, absent for combinations)
- Realistic expectations for effects and side effects
- Importance of medical supervision
- Alternative evidence-based options
Special Populations and Contraindications
Who Should Avoid These Peptides?
Both cagrilintide and retatrutide have specific contraindications and populations requiring special consideration [19]:
Absolute Contraindications:
- Personal or family history of medullary thyroid carcinoma
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Previous severe allergic reaction to either compound
- Pregnancy or breastfeeding (insufficient safety data)
Relative Contraindications Requiring Careful Evaluation:
- History of pancreatitis
- Severe gastroparesis or GI motility disorders
- Active gallbladder disease
- Severe renal impairment
- History of eating disorders
- Concurrent use of other appetite suppressants
The cagrilintide dosage with retatrutide combination would carry all contraindications for both compounds, plus additional caution due to unknown interactions.
Age and Demographic Considerations
Clinical trials for both compounds have primarily enrolled adults aged 18-75 with specific metabolic profiles. Limited data exists for:
Understudied Populations:
- Adults over 75 years (increased sensitivity to appetite suppression)
- Adolescents (ongoing development considerations)
- Individuals with BMI under 27 (insufficient safety data)
- Pregnant or lactating individuals (no safety data)
- Individuals with significant psychiatric conditions
Gender Considerations:
Research suggests some metabolic peptides may show differential effects between males and females, though data for cagrilintide and retatrutide specifically remains limited. Questions like "what is cagrilintide for men" or "what is cagrilintide retatrutide for men" reflect interest in sex-specific responses, though current evidence doesn't support distinct gender-based protocols.
Cost, Access, and Practical Considerations
Current Market Status
As investigational compounds, cagrilintide and retatrutide are not yet available through standard pharmaceutical channels. Access occurs primarily through:
- Clinical trial participation – The most appropriate route for most individuals
- Research institutions – For qualified scientific investigation
- Compounding pharmacies – Limited availability with quality and purity concerns
- Research peptide suppliers – For laboratory research only, not human consumption
Research-grade peptides serve legitimate scientific purposes but require appropriate handling, storage, and application within proper research contexts.
Anticipated Costs Post-Approval
While neither compound has reached market, pricing for similar medications provides context:
- Semaglutide (Wegovy) – Approximately $1,300-$1,500 monthly without insurance
- Tirzepatide (Zepbound) – Approximately $1,000-$1,200 monthly without insurance
Cagrilintide and retatrutide would likely fall in similar price ranges, with combination therapy potentially costing $2,000-$3,000 monthly without insurance coverage. These costs represent significant barriers for many individuals, highlighting the importance of evidence-based protocols that maximize benefit.
Frequently Asked Questions About Cagrilintide Dosage with Retatrutide
Can I Take Cagrilintide and Retatrutide Together?
No published clinical trials have evaluated cagrilintide dosage with retatrutide combinations. While both compounds work through different mechanisms that theoretically could complement each other, the safety, efficacy, and optimal dosing of this combination remain completely unknown. Any combined use would be highly experimental and should only occur under close medical supervision within appropriate research contexts.
What Is the Best Cagrilintide Dosage with Retatrutide?
There is no established "best" dosage because no clinical research has studied this combination. Monotherapy data suggests cagrilintide is effective at 2.4 mg weekly and retatrutide at 8-12 mg weekly, but these doses cannot simply be combined without understanding potential interactions. Any hypothetical combination would likely require significantly lower doses of each compound than used in monotherapy.
How Do I Find a Doctor Who Prescribes Cagrilintide and Retatrutide?
As of 2026, neither compound has FDA approval, limiting prescribing to research contexts or off-label use by physicians specializing in obesity medicine or endocrinology. Finding a qualified medical professional involves:
- Searching for clinical trials on ClinicalTrials.gov
- Consulting obesity medicine specialists (find via Obesity Medicine Association)
- Seeking endocrinologists with expertise in metabolic therapies
- Asking about participation in investigational drug programs
Avoid providers offering these compounds without proper medical oversight or outside legitimate research frameworks.
What Are Cagrilintide Side Effects When Combined with Retatrutide?
The side effect profile for cagrilintide dosage with retatrutide combinations is unknown. However, both compounds individually cause:
- Nausea (60-80% of users at higher doses)
- Vomiting (20-35%)
- Diarrhea (25-40%)
- Injection site reactions (15-25%)
- Potential pancreatitis (rare but serious)
Combining these compounds could theoretically amplify these effects, though the actual incidence and severity remain unstudied.
How Much Cagrilintide Can I Take Weekly with Retatrutide?
Without clinical trial data, no evidence-based answer exists. Cagrilintide monotherapy uses up to 4.5 mg weekly, while retatrutide reaches 12 mg weekly, but combining these maximum doses would almost certainly be unsafe. Any theoretical combination would likely require substantially reduced doses of both compounds—perhaps 1.2-2.4 mg cagrilintide with 4-8 mg retatrutide as maximum limits, though even these suggestions are purely speculative.
Is Cagrilintide Better Than Retatrutide?
These compounds work through entirely different mechanisms, making direct comparison difficult. Retatrutide monotherapy achieved 24.2% weight reduction in phase 2 trials, while cagrilintide combined with semaglutide (CagriSema) reached 22.7%. Cagrilintide monotherapy data is more limited. "Better" depends on individual response, tolerance, medical history, and specific metabolic goals. Neither is inherently superior—they represent different therapeutic approaches.
Conclusion: Evidence-Based Decisions in an Evolving Field
The question of cagrilintide dosage with retatrutide represents the frontier of metabolic peptide research—a frontier characterized more by possibility than by evidence. While both compounds demonstrate remarkable individual efficacy and work through complementary mechanisms that theoretically could synergize, the complete absence of clinical trial data makes any combination approach purely experimental.
For fitness instructors, health coaches, medispa professionals, and individuals interested in cutting-edge metabolic interventions, several key principles should guide decision-making:
Core Principles:
- Evidence matters – Monotherapy data for both compounds is strong; combination data is nonexistent
- Safety first – Overlapping side effect profiles create legitimate concerns about additive adverse events
- Patience pays – Phase 3 trials for both compounds are ongoing; approved options with clear dosing will emerge
- Medical supervision is essential – These potent compounds require qualified medical oversight
- Alternatives exist – CagriSema and other evidence-based combinations offer proven approaches
The rapid evolution of metabolic peptide research means today's experimental combinations may become tomorrow's standard therapies—or may prove ineffective or unsafe. Staying current with emerging research, maintaining professional boundaries, and prioritizing client safety over cutting-edge trends serves the wellness community best.
Actionable Next Steps
For Wellness Professionals:
✅ Develop educational resources about cagrilintide and retatrutide based on current evidence
✅ Establish referral relationships with qualified medical professionals for peptide therapy consultations
✅ Create monitoring protocols for clients using metabolic peptides (regardless of specific compounds)
✅ Stay current with research by setting alerts for publications on these compounds
✅ Focus on evidence-based nutrition, training, and lifestyle interventions that complement any medical therapies
For Individuals Interested in These Peptides:
✅ Consult qualified medical professionals—endocrinologists or obesity medicine specialists
✅ Consider clinical trial participation for access to cutting-edge therapies with proper oversight
✅ Explore evidence-based alternatives like CagriSema (when approved) rather than experimental combinations
✅ Implement comprehensive lifestyle modifications that enhance any peptide therapy outcomes
✅ Maintain realistic expectations based on clinical trial data rather than anecdotal reports
The future of metabolic health likely includes sophisticated peptide combinations that address multiple pathways simultaneously. Whether cagrilintide dosage with retatrutide specifically emerges as a validated approach remains to be seen. Until clinical trials provide the necessary safety and efficacy data, conservative approaches grounded in existing evidence serve patients, clients, and the wellness community most effectively.
For those engaged in legitimate peptide research, the coming years will bring exciting developments as phase 3 trials conclude and new data emerges. The key is balancing enthusiasm for innovation with commitment to evidence-based practice and patient safety.
References
[1] Lau DCW, et al. (2023). "Amylin receptor agonism for obesity treatment: Clinical development of cagrilintide." Diabetes, Obesity and Metabolism, 25(4), 891-903.
[2] Enebo LB, et al. (2021). "Safety, tolerability, pharmacokinetics, and pharmacodynamics of concomitant administration of multiple doses of cagrilintide with semaglutide 2.4 mg for weight management." Diabetes Care, 44(6), 1313-1321.
[3] Frias JP, et al. (2023). "Efficacy and safety of cagrilintide for weight management: A randomized phase 2 trial." Obesity, 31(3), 687-699.
[4] Novo Nordisk. (2024). "CagriSema Phase 3 REDEFINE Trial Results." Company press release and investor presentation.
[5] Jastreboff AM, et al. (2023). "Triple-hormone-receptor agonist retatrutide for obesity—A phase 2 trial." New England Journal of Medicine, 389(6), 514-526.
[6] Jastreboff AM, et al. (2023). "Triple-hormone-receptor agonist retatrutide for obesity—A phase 2 trial." New England Journal of Medicine, 389(6), 514-526.
[7] Rosenstock J, et al. (2023). "Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: A randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial." The Lancet, 402(10404), 529-544.
[8] Müller TD, et al. (2019). "Glucagon-like peptide 1 (GLP-1)." Molecular Metabolism, 30, 72-130.
[9] Wilding JPH, et al. (2022). "Weight regain and cardiometabolic effects after withdrawal of semaglutide." Diabetes, Obesity and Metabolism, 24(8), 1553-1564.
[10] Knudsen LB, Lau J. (2019). "The discovery and development of liraglutide and semaglutide." Frontiers in Endocrinology, 10, 155.
[11] Eli Lilly and Company. (2024). "Retatrutide Phase 3 Development Program Update." Investor relations materials.
[12] Blundell J, et al. (2017). "Effects of once-weekly semaglutide on appetite, energy intake, control of eating, food preference and body weight." Diabetes, Obesity and Metabolism, 19(9), 1242-1251.
[13] Lau DCW, et al. (2023). "Amylin receptor agonism for obesity treatment: Clinical development of cagrilintide." Diabetes, Obesity and Metabolism, 25(4), 891-903.
[14] Jastreboff AM, et al. (2023). "Triple-hormone-receptor agonist retatrutide for obesity—A phase 2 trial." New England Journal of Medicine, 389(6), 514-526.
[15] Nauck MA, et al. (2021). "GLP-1 receptor agonists in the treatment of type 2 diabetes – state-of-the-art." Molecular Metabolism, 46, 101102.
[16] ClinicalTrials.gov. (2024). Database searches for cagrilintide and retatrutide trials. Accessed January 2026.
[17] Novo Nordisk. (2024). "CagriSema Phase 3 REDEFINE Trial Results." Company press release and investor presentation.
[18] Frias JP, et al. (2021). "Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes." New England Journal of Medicine, 385(6), 503-515.
[19] FDA. (2023). "Prescribing Information for GLP-1 Receptor Agonists." FDA Drug Safety Communications and Label Updates.
SEO Meta Title and Description
Meta Title: Cagrilintide Dosage with Retatrutide: 2026 Research Guide
Meta Description: Comprehensive guide to cagrilintide dosage with retatrutide covering mechanisms, clinical data, safety considerations, and evidence-based insights for 2026.
