Buy BPC Nasal Spray: Complete 2026 Research Guide for Peptide Enthusiasts

The peptide research landscape has transformed dramatically in 2026, with nasal spray delivery systems emerging as one of the most convenient and efficient methods for administering research compounds. Among the most sought-after formulations, those looking to buy BPC nasal spray are discovering a revolutionary approach to peptide research that combines convenience, bioavailability, and precise dosing. This comprehensive guide explores everything researchers, fitness professionals, and medispa owners need to know about acquiring and working with this innovative peptide delivery system.
Key Takeaways
- BPC nasal spray offers superior convenience and non-invasive administration compared to traditional injectable peptides
- Research indicates nasal delivery may provide enhanced bioavailability and faster systemic absorption for certain peptide compounds
- Quality verification through third-party testing and certificates of analysis remains essential when selecting peptide suppliers
- Proper storage, handling, and dosing protocols significantly impact research outcomes and peptide stability
- The 2026 peptide market offers diverse sourcing options, making supplier reputation and product purity critical selection factors
Understanding BPC-157 and Nasal Spray Delivery Systems
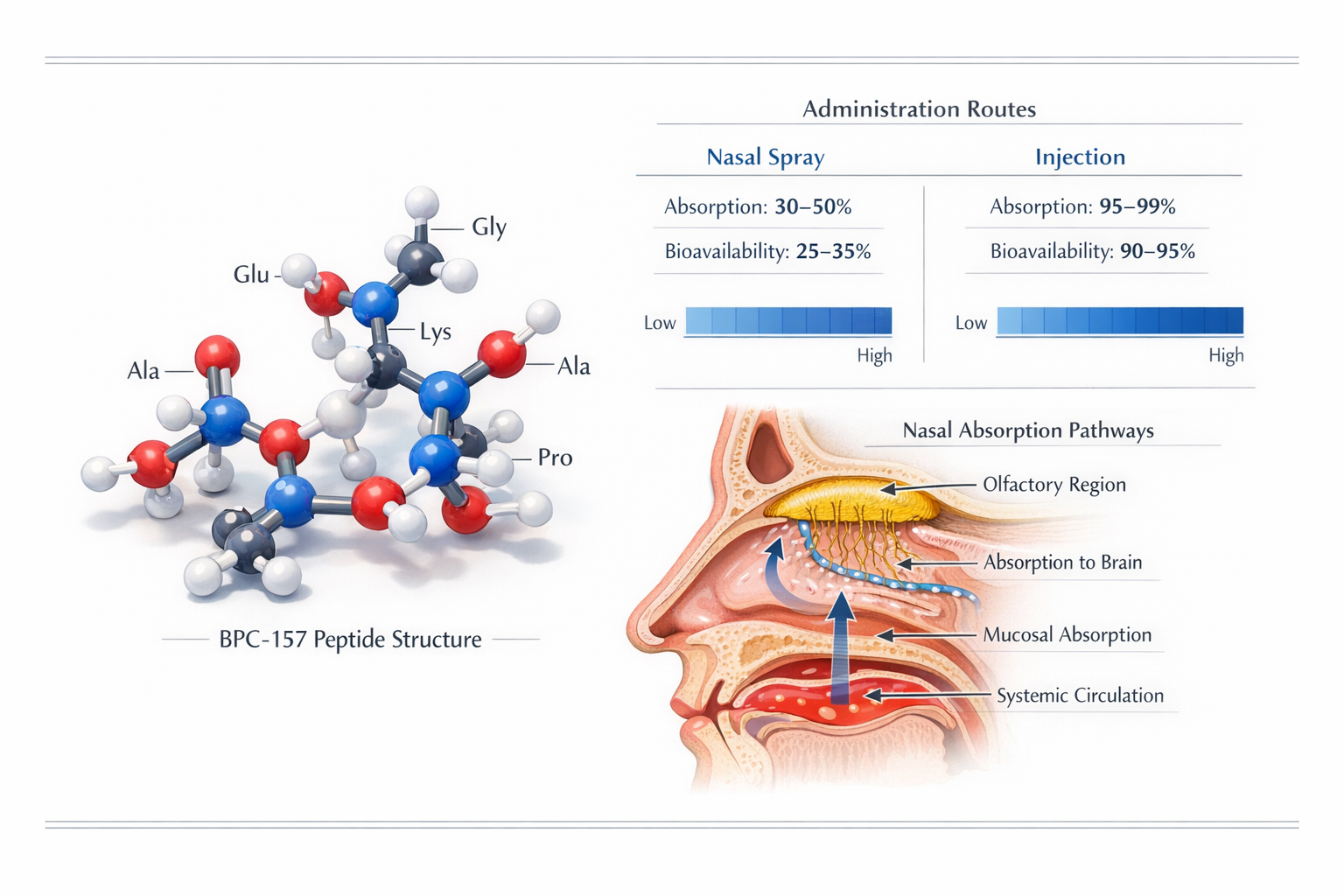
BPC-157, a synthetic peptide derived from a protective protein found in gastric juice, has captured significant attention in research communities worldwide. The compound consists of 15 amino acids and has been extensively studied in preclinical research models for its potential effects on tissue repair, inflammation modulation, and various physiological processes.
The Science Behind Nasal Peptide Administration
Nasal spray delivery represents a paradigm shift in peptide administration methodology. The nasal mucosa contains a rich vascular network that allows certain molecules to bypass first-pass hepatic metabolism, potentially enhancing bioavailability compared to oral administration. Research published in pharmaceutical sciences journals indicates that peptides administered nasally can achieve systemic circulation within minutes.
Key advantages of nasal delivery include:
- Direct absorption through the nasal mucosa into systemic circulation
- Avoidance of gastrointestinal degradation that affects oral peptides
- Non-invasive administration without needles or injections
- Rapid onset of systemic distribution
- Consistent dosing accuracy with metered spray devices
The molecular weight and hydrophilic properties of BPC-157 make it a suitable candidate for nasal administration, though individual absorption rates may vary based on formulation factors and nasal cavity conditions.
BPC-157 Research Applications and Study Areas
Scientific literature has explored BPC-157 across numerous research domains. Preclinical studies have investigated its potential role in:
- Tissue repair mechanisms: Animal studies have examined effects on tendon, ligament, muscle, and bone healing processes
- Gastrointestinal research: Investigation into protective effects on intestinal lining and inflammatory bowel conditions
- Vascular health: Research into blood vessel formation and endothelial function
- Neurological studies: Examination of potential neuroprotective properties and dopaminergic system interactions
- Anti-inflammatory pathways: Analysis of cytokine modulation and inflammatory response regulation
Researchers interested in these areas often seek to buy peptides from reputable suppliers who can provide documentation supporting compound identity and purity.
Where to Buy BPC Nasal Spray: Evaluating Peptide Suppliers
The decision of where to buy peptides significantly impacts research quality and outcomes. The 2026 peptide market includes numerous vendors, but not all maintain equivalent standards for purity, testing, and customer support.
Critical Supplier Selection Criteria
When evaluating potential sources to buy BPC nasal spray, researchers should prioritize several essential factors:
🔬 Third-Party Testing and Certificates of Analysis
Reputable peptide supplier US operations provide comprehensive certificates of analysis (COAs) from independent laboratories. These documents should include:
- High-performance liquid chromatography (HPLC) results showing purity percentages
- Mass spectrometry data confirming molecular identity
- Endotoxin testing results
- Sterility verification for injectable or nasal formulations
- Batch-specific identification numbers
📦 Product Formulation and Presentation
Quality nasal spray formulations require careful attention to:
- pH buffering: Proper pH levels (typically 5.5-7.0) prevent nasal irritation
- Osmolality: Isotonic solutions minimize discomfort during administration
- Preservative systems: Appropriate antimicrobial agents for multi-dose bottles
- Spray mechanism quality: Consistent metering devices ensure accurate dosing
- Concentration accuracy: Clearly labeled peptide content per spray actuation
💼 Vendor Reputation and Customer Service
The best peptide retail US suppliers distinguish themselves through:
- Transparent business practices and contact information
- Responsive customer support for technical questions
- Clear terms regarding research-only applications
- Secure payment processing and data protection
- Established track record within the research community
Comparing Peptide Supplier Options in 2026
| Supplier Characteristic | Premium Vendors | Budget Vendors | Red Flag Vendors |
|---|---|---|---|
| Third-Party Testing | Every batch with COA | Periodic testing | No testing documentation |
| Purity Standards | ≥98% verified | 95-98% claimed | Unverified claims |
| Customer Support | Dedicated research support | Email-only support | Minimal/no support |
| Product Information | Detailed technical specs | Basic descriptions | Vague or missing data |
| Pricing | Moderate to premium | Budget-focused | Suspiciously low |
| Shipping | Temperature-controlled | Standard shipping | Uncontrolled conditions |
Researchers seeking to buy peptides should recognize that exceptionally low pricing often correlates with compromised quality or inadequate testing protocols.
How to Buy BPC Nasal Spray: Step-by-Step Acquisition Process
Successfully acquiring research-grade BPC nasal spray requires methodical evaluation and proper documentation. This systematic approach ensures compliance with research protocols and maximizes the likelihood of obtaining authentic, high-quality compounds.
Step 1: Research Qualification and Documentation
Before attempting to buy BPC nasal spray, researchers should prepare appropriate documentation:
- Institutional affiliation: Academic, clinical, or commercial research facility information
- Research objectives: Clear statement of intended research applications
- Credentials: Relevant qualifications demonstrating research competency
- Compliance awareness: Understanding of research-only designations and regulatory frameworks
Legitimate suppliers typically require acknowledgment that peptides are intended exclusively for research purposes and not for human consumption or therapeutic use.
Step 2: Supplier Evaluation and Comparison
Conduct thorough due diligence on potential vendors:
- Review supplier websites for professional presentation and technical information
- Examine available COAs from recent batches to verify testing standards
- Check independent reviews from research community forums and professional networks
- Compare pricing across multiple suppliers while considering quality indicators
- Assess shipping practices including temperature control and delivery timeframes
- Evaluate customer service responsiveness through preliminary inquiries
When deciding where to buy peptides, researchers should create a comparison matrix weighing these factors against their specific research requirements.
Step 3: Product Selection and Specification
BPC nasal spray formulations vary across suppliers. Consider these specifications:
- Concentration: Common concentrations range from 250mcg to 1000mcg per spray
- Volume: Bottle sizes typically contain 30-60 doses
- Formulation type: Pre-mixed solutions versus lyophilized powder requiring reconstitution
- Preservative system: Benzyl alcohol, benzalkonium chloride, or preservative-free options
- Expiration dating: Shelf life and stability data
Researchers working with peptide supplier US vendors should request detailed formulation information to ensure compatibility with research protocols.
Step 4: Order Placement and Payment
Secure ordering processes protect both researchers and suppliers:
- Account creation: Establish verified accounts with complete contact information
- Secure payment methods: Credit cards, cryptocurrency, or institutional purchase orders
- Order confirmation: Detailed receipts specifying product, batch number, and pricing
- Privacy protection: Discreet packaging and secure data handling practices
Step 5: Receipt, Verification, and Storage
Upon delivery of BPC nasal spray:
✅ Immediate inspection: Check for package integrity, temperature excursions, and product condition
✅ COA verification: Match batch numbers on product labels with provided certificates of analysis
✅ Proper storage: Refrigerate at 2-8°C (36-46°F) unless otherwise specified; protect from light
✅ Documentation: Record receipt date, batch information, and storage conditions in research logs
✅ Visual inspection: Examine solution for clarity, color consistency, and absence of particulates
Researchers should never use peptide products that show signs of contamination, discoloration, or have been subjected to temperature extremes during shipping.
BPC Nasal Spray Research Protocols and Considerations
Establishing rigorous research protocols ensures data quality and reproducibility when working with BPC nasal spray formulations. The following guidelines reflect current best practices within the peptide research community as of 2026.
Dosing Considerations in Research Models
Research literature examining BPC-157 has employed various dosing strategies depending on the model system and research objectives. When designing protocols involving nasal spray administration:
Dosage variables to consider:
- Concentration per actuation: Typically 250-500mcg per spray
- Administration frequency: Once daily to multiple times daily in various studies
- Duration of administration: Research periods ranging from days to weeks
- Subject characteristics: Body weight, metabolic factors, and baseline conditions
Researchers should consult published literature relevant to their specific research questions and adjust protocols accordingly. Those looking to buy peptides for research should maintain detailed dosing logs to support data analysis and reproducibility.
Administration Technique and Best Practices
Proper nasal spray administration technique significantly impacts absorption consistency:
- Pre-administration preparation: Clear nasal passages if necessary; allow refrigerated product to reach room temperature
- Positioning: Upright head position with slight forward tilt
- Spray technique: Insert nozzle into nostril, aim toward outer nasal wall (not septum)
- Actuation: Depress spray mechanism while inhaling gently through nose
- Post-administration: Remain upright for 2-3 minutes to prevent drainage into throat
- Alternating nostrils: Rotate between nostrils if multiple sprays are required
Storage and Stability Management
Peptide stability directly affects research outcomes. BPC nasal spray formulations require careful attention to storage conditions:
| Storage Condition | Unopened Product | Opened Product | Room Temperature Exposure |
|---|---|---|---|
| Optimal Temperature | 2-8°C (refrigerated) | 2-8°C (refrigerated) | Minimize to <30 minutes |
| Light Exposure | Protect from direct light | Store in original container | Avoid prolonged exposure |
| Typical Stability | 12-24 months | 30-60 days | Reduced stability |
| Freeze-Thaw Cycles | Avoid freezing | Never freeze liquid formulations | N/A |
Researchers should always verify specific storage requirements with their peptide supplier US vendor, as formulation differences may necessitate adjusted storage protocols.
Research Documentation and Data Management
Comprehensive documentation supports research integrity and reproducibility:
📋 Essential records to maintain:
- Supplier information and batch numbers
- Receipt dates and storage conditions
- COA documentation with purity and identity verification
- Administration logs with dates, times, and dosages
- Subject identifiers and baseline characteristics
- Observational data and measurement protocols
- Adverse observations or protocol deviations
- Statistical analysis plans and results
This documentation becomes particularly important when researchers buy BPC nasal spray from multiple suppliers or compare formulations across studies.
Comparing BPC Nasal Spray to Other Peptide Delivery Methods
Understanding the advantages and limitations of nasal spray delivery compared to alternative administration routes helps researchers select optimal methodologies for their specific research objectives.
Nasal Spray vs. Injectable Peptides
Injectable peptides have traditionally dominated research applications, but nasal spray formulations offer distinct characteristics:
Advantages of nasal spray delivery:
- ✨ Non-invasive administration eliminates injection-related complications
- ✨ Rapid absorption through nasal mucosa into systemic circulation
- ✨ Consistent dosing with metered spray devices
- ✨ Reduced training requirements for administration technique
- ✨ Lower contamination risk compared to reconstituted injectables
- ✨ Improved compliance in research protocols requiring frequent administration
Potential limitations:
- ⚠️ Bioavailability variability based on nasal cavity conditions
- ⚠️ Nasal irritation potential with certain formulations
- ⚠️ Limited volume capacity per administration
- ⚠️ Preservative requirements for multi-dose formulations
- ⚠️ Higher cost per dose compared to some injectable formulations
Nasal Spray vs. Oral Peptide Formulations
Oral peptide administration faces significant challenges due to gastrointestinal degradation. Researchers comparing options to buy BPC nasal spray versus oral formulations should consider:
Nasal spray advantages over oral administration:
- Superior bioavailability bypassing first-pass hepatic metabolism
- Protection from gastric acid and proteolytic enzyme degradation
- Faster onset of systemic distribution
- More predictable pharmacokinetic profiles
- Lower dosing requirements for equivalent systemic exposure
Integration with Other Research Peptides
Many researchers work with multiple peptide compounds simultaneously. Those who buy peptides for comprehensive research protocols often combine BPC-157 with complementary compounds.
Commonly researched peptide combinations include:
- BPC-157 + TB-500: Both studied for tissue repair mechanisms
- BPC-157 + Growth hormone secretagogues: Examining synergistic effects on recovery processes
- BPC-157 + Anti-inflammatory peptides: Investigating combined effects on inflammatory pathways
When designing multi-peptide protocols, researchers should consider potential interactions, administration timing, and appropriate controls. The best peptide retail US suppliers can provide technical guidance on compatibility and protocol design.
Quality Assurance and Safety Considerations When You Buy BPC Nasal Spray
Quality assurance represents the foundation of reliable peptide research. Researchers must implement rigorous verification procedures to ensure compound authenticity, purity, and stability throughout the research lifecycle.
Understanding Certificates of Analysis (COAs)
Certificates of analysis serve as the primary quality documentation for research peptides. When evaluating suppliers to buy BPC nasal spray, researchers should understand COA components:
Essential COA Elements
1. Product Identification
- Peptide name and sequence
- Batch or lot number
- Manufacturing date
- Expiration or retest date
- Supplier identification
2. Purity Analysis
- HPLC chromatogram showing purity percentage (typically ≥98% for research-grade)
- Peak identification and integration
- Method specifications and column details
- Retention time data
3. Identity Confirmation
- Mass spectrometry results (MALDI-TOF or ESI-MS)
- Observed molecular weight vs. theoretical
- Fragmentation patterns if applicable
4. Contamination Testing
- Endotoxin levels (typically <1.0 EU/mg)
- Bacterial contamination screening
- Heavy metal analysis
- Residual solvent testing
5. Physical Characteristics
- Appearance description
- pH measurement for solutions
- Osmolality for nasal formulations
- Moisture content for lyophilized products
Researchers should request COAs before finalizing purchases from any peptide supplier US vendor and verify that testing was performed by independent, accredited laboratories.
Red Flags and Quality Concerns
⚠️ Warning signs indicating potentially compromised products:
- Absence of batch-specific COAs
- Generic or template COAs without actual test data
- Purity claims without supporting chromatography
- Suspiciously low pricing compared to market standards
- Lack of proper business registration or contact information
- Vague product descriptions without technical specifications
- No cold-chain shipping for temperature-sensitive products
- Marketing claims suggesting human therapeutic use
Researchers should exercise extreme caution with vendors exhibiting these characteristics, regardless of pricing advantages.
Implementing Internal Quality Controls
Beyond supplier verification, research facilities should establish internal quality assurance protocols:
Recommended internal controls:
- Visual inspection protocols: Document appearance, color, clarity, and particulate matter
- Storage monitoring: Temperature logging for refrigeration units
- Expiration tracking: Inventory management systems preventing use of expired materials
- Cross-contamination prevention: Dedicated storage and handling procedures
- Batch segregation: Clear labeling and separation of different lots
- Usage logs: Documentation of when products are opened and used
- Periodic verification: Consider independent testing of critical batches
Handling and Disposal Considerations
Proper handling protects research integrity and laboratory safety:
Best practices for BPC nasal spray handling:
- Use appropriate personal protective equipment (gloves, lab coat)
- Work in clean, designated areas away from food or drink
- Avoid direct skin contact with peptide solutions
- Clean work surfaces before and after use
- Store away from incompatible materials
- Follow institutional biosafety guidelines
Disposal procedures:
- Never dispose of peptides through regular trash or drains
- Follow institutional chemical waste protocols
- Deactivate biological activity if required by local regulations
- Maintain disposal records for regulatory compliance
- Consult environmental health and safety departments for specific requirements
The 2026 Peptide Research Landscape: Market Trends and Future Directions
The peptide research sector has experienced remarkable evolution, with 2026 marking a period of unprecedented accessibility, quality standardization, and scientific advancement. Understanding current market dynamics helps researchers make informed decisions when they buy BPC nasal spray and other research compounds.
Market Growth and Accessibility
The global peptide therapeutics market has expanded significantly, with research-grade peptides becoming increasingly available to qualified researchers. Key trends include:
📈 Market expansion factors:
- Increased manufacturing capacity from specialized peptide synthesis facilities
- Improved purification technologies yielding higher-quality products
- Competitive pricing driven by multiple suppliers entering the market
- Enhanced distribution networks with proper cold-chain logistics
- Greater awareness of peptide research applications across disciplines
This expansion has made it easier for researchers to identify reliable sources where to buy peptides while maintaining quality standards.
Regulatory Environment and Compliance
The regulatory landscape surrounding research peptides continues to evolve. In 2026, researchers must navigate:
Regulatory considerations:
- Research-only designations prohibiting human consumption
- Import/export regulations for international peptide shipments
- Institutional review board (IRB) requirements for certain research applications
- Good Laboratory Practice (GLP) standards for commercial research
- State and federal regulations governing peptide possession and use
Reputable suppliers who serve as a peptide supplier US source maintain awareness of these regulatory frameworks and implement appropriate compliance measures.
Emerging Peptide Research Areas
Scientific interest in peptide research continues expanding across multiple domains:
🔬 Active research frontiers:
- Metabolic research: Compounds like those available when researchers buy MOTS C peptide for mitochondrial function studies
- Tissue engineering: Peptides supporting regenerative medicine applications
- Neuroprotection: Compounds like those researchers find when they where to buy epithalon for aging research
- Metabolic optimization: Products including options to buy Lipo C peptide for lipid metabolism studies
- Cognitive research: Formulations such as those available to buy Selank spray for anxiolytic research
Technology Advances in Peptide Delivery
Delivery system innovation represents a major focus area:
Novel delivery technologies:
- Sustained-release formulations extending peptide activity
- Transdermal patches for continuous peptide delivery
- Oral formulations with enhanced bioavailability
- Targeted delivery systems using nanoparticle carriers
- Improved nasal spray formulations with absorption enhancers
These advances complement traditional options to buy BPC nasal spray, providing researchers with expanding methodological choices.
Quality Standardization Initiatives
The research peptide industry has increasingly embraced quality standardization:
Quality improvement trends:
- Industry-wide adoption of third-party testing protocols
- Standardized COA formats improving transparency
- Collaborative efforts establishing purity benchmarks
- Enhanced supplier verification processes
- Greater emphasis on supply chain integrity
Researchers benefit from these initiatives when selecting the best peptide retail US suppliers for their research programs.
Frequently Asked Questions About Buying BPC Nasal Spray
What should I look for when I buy BPC nasal spray?
Prioritize suppliers providing batch-specific certificates of analysis from independent laboratories, showing ≥98% purity via HPLC. Verify proper formulation specifications including pH, osmolality, and sterility testing. Ensure the supplier implements temperature-controlled shipping and provides clear storage instructions.
How does nasal spray compare to injectable BPC-157?
Nasal spray offers non-invasive administration with rapid absorption through nasal mucosa, bypassing gastrointestinal degradation. While injectable formulations may offer more precise dosing control, nasal delivery provides convenience and consistent administration without injection-related complications. Bioavailability may vary between individuals based on nasal cavity conditions.
Where to buy peptides with reliable quality assurance?
Reputable peptide supplier US vendors distinguish themselves through comprehensive third-party testing, transparent business practices, responsive customer support, and established track records within research communities. Verify that suppliers provide batch-specific COAs, implement proper storage and shipping protocols, and maintain clear communication channels.
What is the typical shelf life of BPC nasal spray?
Unopened BPC nasal spray stored at proper refrigeration temperatures (2-8°C) typically maintains stability for 12-24 months from manufacture date. Once opened, most formulations remain stable for 30-60 days when refrigerated and protected from light. Always verify specific stability data with your supplier and follow expiration dating on product labels.
Can I combine BPC nasal spray with other research peptides?
Many research protocols incorporate multiple peptides with complementary mechanisms. When researchers buy peptides for combination studies, they should consider potential interactions, appropriate dosing intervals, and establish proper controls. Consult published literature and supplier technical support for guidance on specific combinations.
What storage conditions are required for BPC nasal spray?
Store unopened and opened BPC nasal spray at refrigeration temperatures (2-8°C/36-46°F). Protect from direct light by keeping in original packaging. Avoid freezing, which can damage formulation integrity. Minimize room temperature exposure during administration. Monitor storage temperatures with calibrated thermometers or data loggers.
How do I verify the authenticity of BPC nasal spray?
Request and review batch-specific certificates of analysis showing HPLC purity analysis and mass spectrometry identity confirmation. Verify COAs are from independent, accredited laboratories rather than in-house testing. Check for proper product labeling including batch numbers, concentration, expiration dates, and supplier information. Consider periodic independent testing of critical batches.
What concentration of BPC nasal spray is appropriate for research?
Research literature has examined various concentrations depending on study objectives and model systems. Common nasal spray formulations range from 250-500mcg per actuation. Researchers should base concentration selection on published protocols relevant to their research questions and adjust based on preliminary findings. Maintain detailed documentation of dosing protocols for reproducibility.
Are there any contraindications for using BPC nasal spray in research models?
Research models with compromised nasal mucosa, active nasal infections, or anatomical abnormalities may show altered absorption patterns. Certain preservatives in multi-dose formulations may affect sensitive model systems. Researchers should consider baseline characteristics of their research subjects and potential formulation interactions when designing protocols.
How quickly does nasally administered BPC-157 reach systemic circulation?
Pharmacokinetic studies of nasally administered peptides generally show rapid absorption with detectable systemic levels within 5-15 minutes. Peak concentrations typically occur within 30-60 minutes, though specific timing varies based on formulation characteristics, nasal cavity conditions, and administration technique. Researchers should consult relevant pharmacokinetic literature for detailed absorption profiles.
Practical Tips for Researchers Working with BPC Nasal Spray
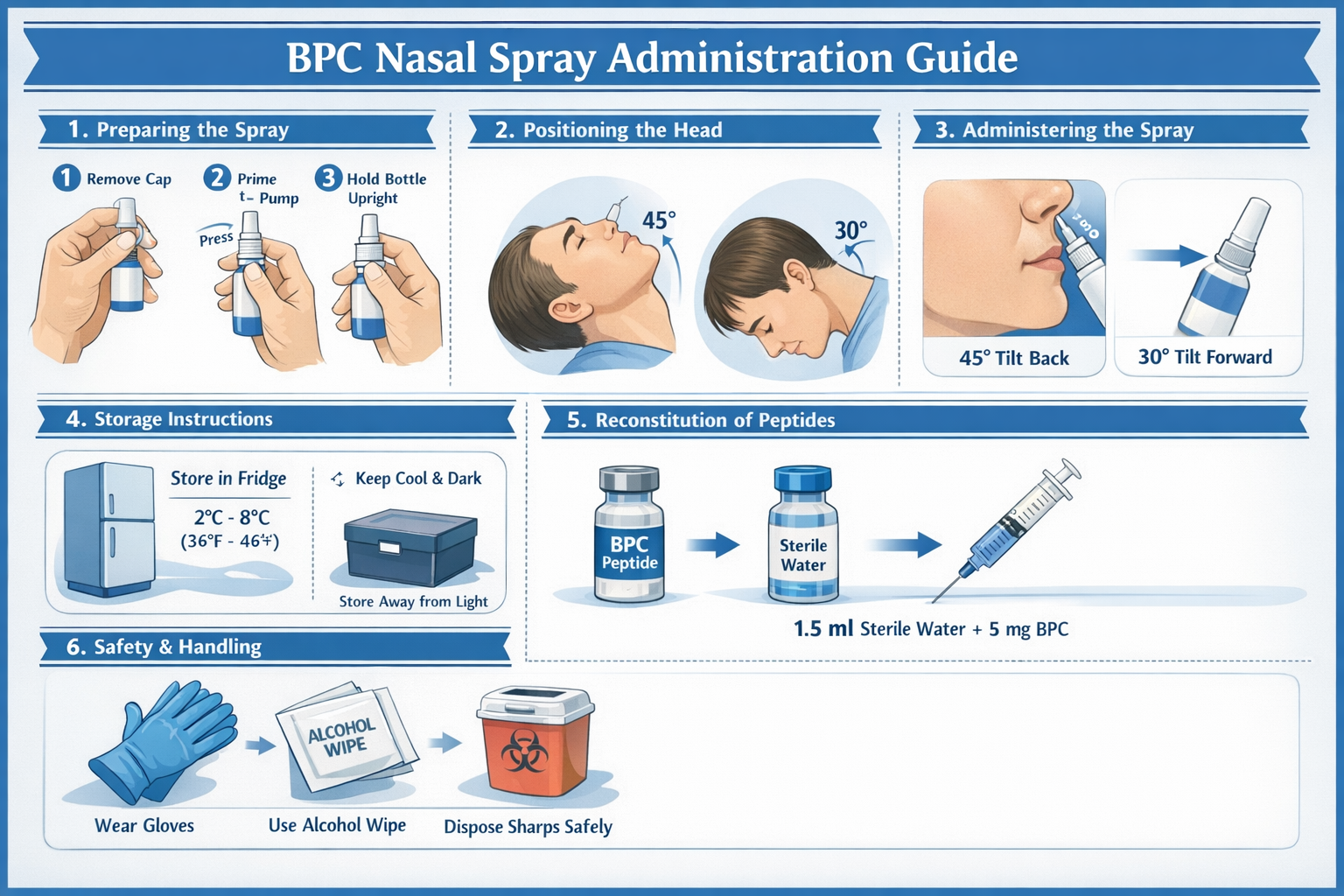
Successful peptide research requires attention to numerous practical details beyond initial acquisition. These field-tested recommendations help researchers maximize data quality and experimental reproducibility.
Establishing Research Protocols
Protocol development checklist:
✓ Define clear research objectives and outcome measures
✓ Review relevant literature for comparable methodologies
✓ Determine appropriate dosing based on published research
✓ Establish administration schedules and frequency
✓ Design appropriate control groups
✓ Create detailed standard operating procedures (SOPs)
✓ Implement data collection and analysis plans
✓ Prepare necessary documentation and approvals
Optimizing Administration Technique
Consistent administration technique reduces variability:
Training recommendations:
- Practice with saline solution before using research peptides
- Develop standardized positioning and spray technique
- Document administration procedures with photographs or video
- Train all personnel involved in administration to identical standards
- Implement regular technique audits to ensure consistency
- Consider using spray actuation counters to track doses
Troubleshooting Common Issues
Problem: Inconsistent spray actuation
- Solution: Prime spray mechanism before first use; ensure bottle is upright; check for clogged nozzles
Problem: Nasal irritation in research models
- Solution: Verify pH and osmolality are within physiological ranges; consider preservative-free formulations; reduce administration frequency
Problem: Suspected product degradation
- Solution: Review storage temperature logs; check for discoloration or particulates; request replacement from supplier if within stability period
Problem: Variable research outcomes
- Solution: Audit administration technique consistency; verify product batch numbers; assess baseline subject characteristics; review environmental factors
Maximizing Research Value
Researchers who buy BPC nasal spray can optimize their investment through:
Cost-effective research strategies:
- Purchase quantities aligned with stability periods to prevent waste
- Implement proper storage to maximize shelf life
- Share resources with collaborating research groups when appropriate
- Maintain detailed records enabling publication of findings
- Consider pilot studies before large-scale experiments
- Build relationships with reliable suppliers for consistent quality
Building Supplier Relationships
Long-term relationships with trusted suppliers provide advantages:
Benefits of established supplier partnerships:
- Priority access during supply shortages
- Technical support for protocol development
- Advance notification of new products or formulations
- Potential volume discounts for ongoing research programs
- Streamlined ordering and shipping processes
- Collaborative problem-solving for research challenges
When researchers consistently buy peptides from reputable vendors, they establish credibility that facilitates these beneficial relationships.
Conclusion: Making Informed Decisions When You Buy BPC Nasal Spray
The decision to buy BPC nasal spray represents a significant commitment to research quality and scientific rigor. As the peptide research landscape continues evolving in 2026, informed researchers must navigate an increasingly complex marketplace while maintaining unwavering standards for product quality, supplier reliability, and methodological excellence.
The Future of Peptide Research
The peptide research field stands at an exciting juncture. Advances in synthesis technology, delivery systems, and quality assurance protocols continue expanding research possibilities. Nasal spray formulations represent just one innovation among many emerging methodologies that promise to enhance research accessibility and data quality.
Researchers who buy peptides from reputable suppliers, implement rigorous quality controls, and maintain detailed documentation contribute to the growing body of scientific knowledge surrounding these fascinating compounds. Whether investigating tissue repair mechanisms, metabolic pathways, neuroprotective effects, or other research domains, peptides offer unique tools for exploring fundamental biological processes.
Final Recommendations
The decision of where to buy peptides should never be made hastily or based solely on pricing considerations. Research quality depends fundamentally on compound purity, formulation integrity, and supplier reliability. By prioritizing these factors, implementing comprehensive quality assurance protocols, and maintaining rigorous research standards, investigators position themselves for successful outcomes and meaningful scientific contributions.
For researchers seeking to buy BPC nasal spray in 2026, the marketplace offers unprecedented access to high-quality research compounds. Success requires diligence, attention to detail, and unwavering commitment to scientific integrity—but the potential rewards in advancing peptide research make these efforts worthwhile investments in scientific progress.
