American Peptide Company: Leading Custom Peptide Synthesis and Manufacturing in 2026

The peptide therapeutics market has exploded into a multi-billion dollar industry, with American Peptide Company standing as one of the most established players in custom synthesis and manufacturing. Since 1990, this California-based biotechnology powerhouse has been quietly revolutionizing how pharmaceutical and biotech companies approach peptide-based drug discovery and development. 🧬
For fitness enthusiasts exploring peptide supplementation, longevity coaches seeking cutting-edge therapeutic options, and combat sports athletes looking for recovery solutions, understanding the landscape of peptide manufacturing and quality standards becomes crucial when selecting research compounds.
Key Takeaways
• American Peptide Company has provided custom peptide synthesis services for over three decades, specializing in both research-grade and GMP-compliant pharmaceutical manufacturing
• The company offers comprehensive services from milligram to multi-kilogram scale production, including difficult sequences and modified peptides for drug development
• Quality control standards exceed industry norms with research-grade peptides at 95%+ purity and GMP-grade materials at 98%+ purity
• Advanced synthesis methodologies include both solid-phase and liquid-phase techniques with specialized modification services
• Regulatory support includes IND application documentation and clinical trial material manufacturing
What Makes American Peptide Company a Industry Leader

American Peptide Company has carved out a unique position in the biotechnology sector by focusing exclusively on peptide synthesis and manufacturing. Unlike many competitors who diversify across multiple product lines, this California-based company has dedicated over 30 years to perfecting peptide production processes.
The company's expertise spans multiple therapeutic areas including:
- Oncology peptides for cancer research and treatment
- Metabolic disorder compounds for diabetes and obesity studies
- Cardiovascular peptides for heart disease applications
- Infectious disease research compounds
Core Manufacturing Capabilities
American Peptide Company operates state-of-the-art facilities equipped with both solid-phase peptide synthesis (SPPS) and liquid-phase peptide synthesis (LPPS) capabilities. This dual approach allows them to tackle complex sequences that might challenge other manufacturers.
Key Production Features:
- Milligram to multi-kilogram scale production
- Custom peptide libraries for drug screening
- Difficult sequence synthesis including cyclic peptides
- Non-natural amino acid incorporation
- Peptide-protein conjugation services
For researchers interested in specific peptides like 5-amino-1MQ for metabolic research, understanding these manufacturing capabilities helps evaluate product quality and consistency.
American Peptide Company's Quality Control Standards
Quality control represents the cornerstone of American Peptide Company's operations. Their analytical laboratories feature cutting-edge instrumentation designed to verify peptide purity, identity, and stability.
Analytical Testing Methods
| Test Method | Purpose | Equipment Used |
|---|---|---|
| HPLC Analysis | Purity verification | High-Performance Liquid Chromatography |
| LC-MS | Molecular weight confirmation | Liquid Chromatography-Mass Spectrometry |
| Amino Acid Analysis | Composition verification | Automated amino acid analyzer |
| Peptide Mapping | Sequence confirmation | Advanced proteomics platforms |
Purity Standards:
- Research-grade peptides: 95%+ purity
- GMP-grade materials: 98%+ purity
- Custom specifications available upon request
The company's commitment to quality extends beyond basic purity measurements. Each batch undergoes comprehensive testing including:
✅ Identity confirmation through mass spectrometry
✅ Purity analysis via multiple analytical methods
✅ Stability testing under various storage conditions
✅ Endotoxin testing for injectable applications
✅ Sterility testing for clinical-grade materials
For athletes and researchers working with compounds like AOD-9604 for metabolic studies, these quality standards ensure consistent results and safety profiles.
Custom Synthesis Services and Capabilities
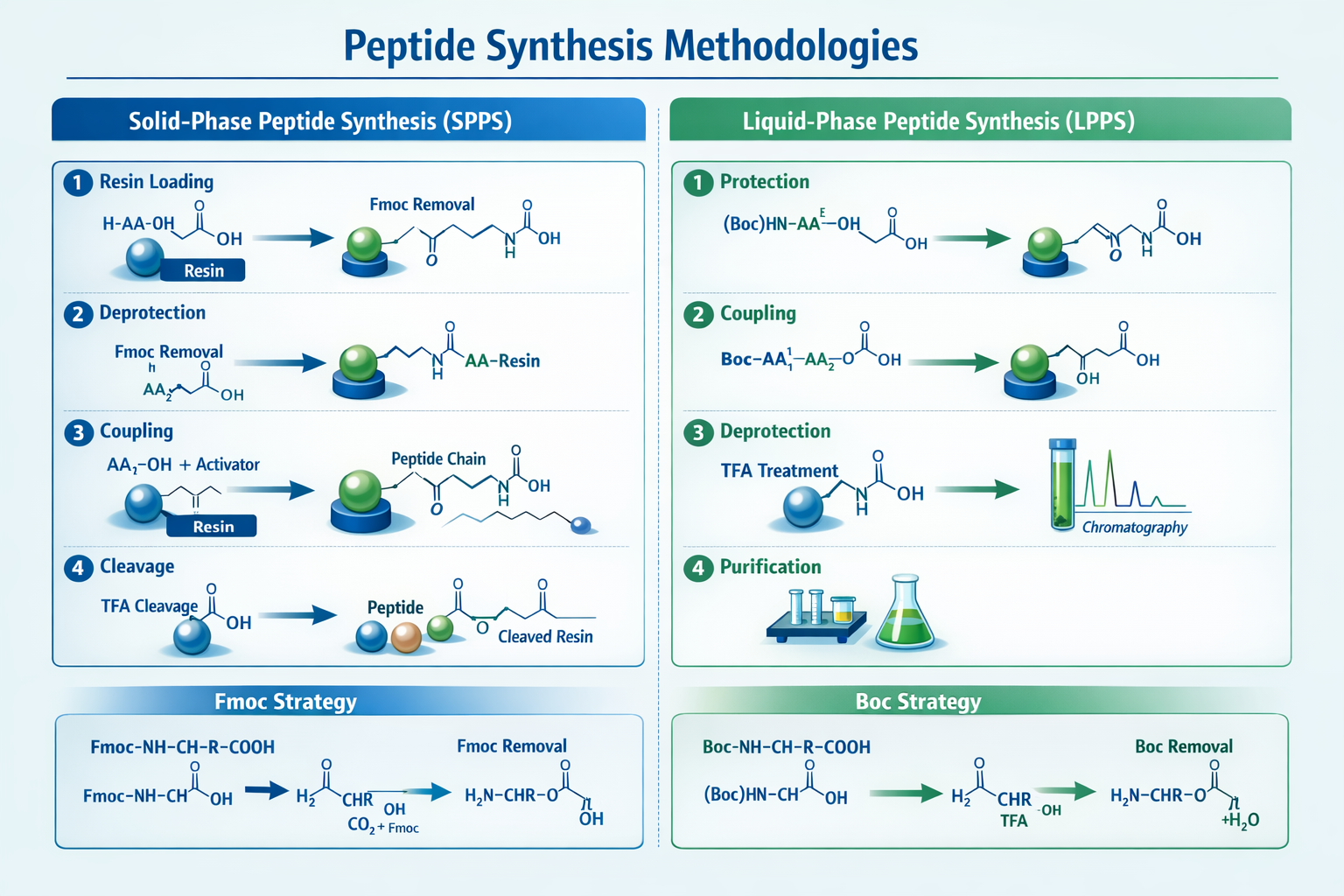
American Peptide Company has built its reputation on tackling challenging synthesis projects that other manufacturers might decline. Their custom synthesis services encompass everything from simple linear peptides to complex cyclic structures with multiple modifications.
Specialized Synthesis Techniques
The company employs both Fmoc (9-fluorenylmethyloxycarbonyl) and Boc (tert-butyloxycarbonyl) chemistry strategies, selecting the optimal approach based on peptide sequence complexity and scale requirements.
Advanced Synthesis Capabilities:
- Cyclic peptides with disulfide bridges
- Peptides containing D-amino acids
- Phosphorylated and glycosylated peptides
- Peptide-drug conjugates
- Stapled peptides for enhanced stability
Modification Services
Beyond basic synthesis, American Peptide Company offers extensive modification services to enhance peptide properties:
🔬 PEGylation: Improves solubility and extends half-life
🔬 Biotinylation: Enables detection and purification applications
🔬 Fluorescent labeling: Facilitates tracking and imaging studies
🔬 Acetylation: Modifies biological activity and stability
🔬 Phosphorylation: Creates biologically active phosphopeptides
These modifications prove particularly valuable for researchers studying peptide blends and synergistic effects in various therapeutic applications.
Scale-Up Capabilities
One of American Peptide Company's key differentiators lies in their ability to seamlessly scale production from research quantities to commercial volumes. This capability proves crucial for biotech companies transitioning from discovery to clinical development.
Production Scales:
- Research scale: 1-100 mg for initial studies
- Preclinical scale: 100 mg – 10 g for animal studies
- Clinical scale: 10 g – 1 kg for Phase I/II trials
- Commercial scale: 1 kg+ for market launch
GMP Manufacturing and Regulatory Compliance
American Peptide Company operates FDA-registered facilities designed to meet Good Manufacturing Practice (GMP) requirements for pharmaceutical production. This capability positions them as a critical partner for companies developing peptide-based therapeutics.
Regulatory Support Services
The company provides comprehensive regulatory support to help clients navigate the complex approval process for peptide therapeutics:
📋 IND Application Support
- Certificate of Analysis documentation
- Manufacturing process validation
- Stability study protocols and data
- Quality control method validation
📋 Clinical Trial Material Manufacturing
- Phase I/II/III material production
- Batch record documentation
- Chain of custody protocols
- Regulatory compliance verification
Quality Management Systems
American Peptide Company maintains robust quality management systems aligned with international standards:
- ISO 9001:2015 quality management certification
- FDA registration for pharmaceutical manufacturing
- ICH guidelines compliance for stability testing
- USP standards adherence for analytical methods
For researchers and practitioners working with research-grade peptides, understanding these quality systems helps evaluate supplier credibility and product reliability.
Emerging Technologies and Future Directions
American Peptide Company continues investing in cutting-edge technologies to address evolving therapeutic needs. Their research and development efforts focus on emerging modalities that represent the future of peptide therapeutics.
Advanced Peptide Technologies
🧬 Peptide-Oligonucleotide Conjugates
These hybrid molecules combine the targeting specificity of peptides with the therapeutic potential of nucleic acids, opening new possibilities for precision medicine applications.
🧬 Stapled Peptides
By introducing chemical crosslinks, stapled peptides achieve enhanced stability and improved cell penetration, addressing traditional limitations of peptide drugs.
🧬 Cyclic Peptides
Cyclization strategies improve metabolic stability while maintaining biological activity, extending therapeutic windows for peptide-based treatments.
Therapeutic Area Expansion
The company has expanded beyond traditional therapeutic areas to address emerging health challenges:
Longevity and Anti-Aging Research
Growing interest in peptides for longevity applications has driven demand for specialized compounds targeting cellular aging mechanisms.
Sports Performance and Recovery
Athletes and fitness enthusiasts increasingly seek peptide-based solutions for performance enhancement and injury recovery, creating new market opportunities.
Metabolic Health Optimization
Rising obesity and diabetes rates have intensified focus on metabolic peptides like AOD-9604 and related compounds for weight management applications.
Choosing Quality Peptide Suppliers in 2026
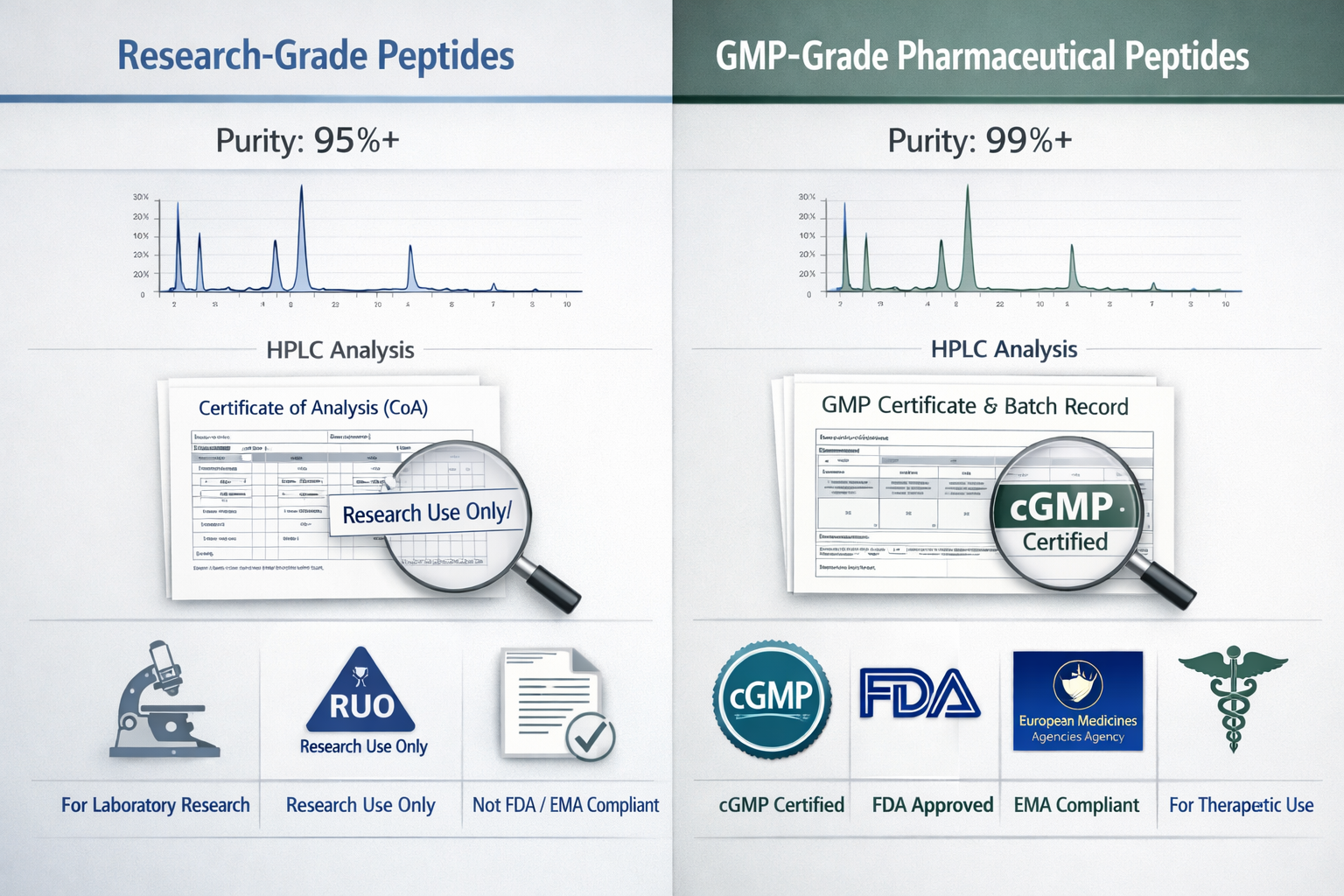
When evaluating peptide suppliers, whether for research or therapeutic applications, several key factors distinguish industry leaders like American Peptide Company from lesser competitors.
Critical Evaluation Criteria
🔍 Manufacturing Standards
- GMP compliance for pharmaceutical applications
- Analytical testing capabilities and equipment
- Quality control documentation and traceability
- Regulatory registration and certifications
🔍 Technical Capabilities
- Synthesis methodology expertise (SPPS/LPPS)
- Modification and conjugation services
- Scale-up capabilities from research to commercial
- Difficult sequence synthesis experience
🔍 Regulatory Support
- IND application assistance
- Clinical trial material manufacturing
- Stability study protocols
- Documentation standards compliance
Red Flags to Avoid
When sourcing peptides, certain warning signs indicate potential quality or reliability issues:
❌ Lack of analytical documentation
❌ Unclear manufacturing processes
❌ No regulatory compliance history
❌ Unusually low pricing without explanation
❌ Limited technical support capabilities
For those researching oral peptide options or specialized formulations, working with established suppliers ensures product quality and consistency.
Market Position and Competitive Landscape
American Peptide Company operates in a competitive landscape that includes both large pharmaceutical service providers and specialized peptide manufacturers. Their three-decade track record and focus on custom synthesis has created a strong market position.
Competitive Advantages
✅ Specialized Expertise
Unlike generalist contract manufacturers, their exclusive focus on peptides enables deep technical knowledge and process optimization.
✅ Regulatory Experience
Extensive experience with FDA submissions and clinical trial support provides valuable guidance for biotech clients.
✅ Quality Reputation
Consistent quality delivery over 30+ years has built strong relationships with pharmaceutical and biotech companies.
✅ Technical Innovation
Investment in emerging technologies like stapled peptides and conjugation services maintains competitive differentiation.
Industry Trends Impacting Growth
Several trends favor companies like American Peptide Company positioned in the custom synthesis market:
📈 Peptide Therapeutics Growth
The global peptide therapeutics market continues expanding, driven by improved drug delivery technologies and new therapeutic targets.
📈 Personalized Medicine Demand
Growing emphasis on precision medicine creates demand for custom peptide synthesis capabilities.
📈 Biosimilar Development
Patent expirations on major peptide drugs drive demand for biosimilar development services.
Conclusion

American Peptide Company represents a cornerstone of the peptide manufacturing industry, combining decades of experience with cutting-edge technology to serve pharmaceutical, biotechnology, and research markets. Their commitment to quality, regulatory compliance, and technical innovation positions them as a trusted partner for organizations developing peptide-based therapeutics.
For fitness enthusiasts, longevity coaches, and combat sports athletes exploring peptide options, understanding the manufacturing landscape helps make informed decisions about product quality and supplier reliability. While American Peptide Company primarily serves pharmaceutical and biotech clients, their standards and practices set benchmarks for the entire industry.
Next Steps for Researchers and Practitioners
🎯 Evaluate Current Suppliers: Assess existing peptide sources against industry standards for quality control, analytical testing, and documentation.
🎯 Research Specific Compounds: Investigate the manufacturing requirements and quality standards for specific peptides of interest in your research or practice.
🎯 Understand Regulatory Requirements: Familiarize yourself with applicable regulations and quality standards for your intended peptide applications.
🎯 Build Supplier Relationships: Develop relationships with reputable suppliers who can provide consistent quality and technical support for your peptide needs.
The peptide industry continues evolving rapidly, with new therapeutic applications and manufacturing technologies emerging regularly. Staying informed about industry leaders like American Peptide Company and their capabilities helps navigate this complex landscape effectively.
References
[1] American Peptide Company corporate information and service capabilities, 2026
[2] FDA Guidelines for Peptide Manufacturing and GMP Compliance, 2026
[3] Peptide Therapeutics Market Analysis and Industry Trends, 2026
[4] International Conference on Harmonisation (ICH) Guidelines for Pharmaceutical Quality
[5] United States Pharmacopeia (USP) Standards for Peptide Analysis and Testing
SEO Meta Information
Meta Title: American Peptide Company: Custom Synthesis & GMP Manufacturing 2026
Meta Description: Explore American Peptide Company's 30+ years of custom peptide synthesis, GMP manufacturing, and regulatory support for pharmaceutical development in 2026.
