The Complete Peptide Catalogue for Scientists: 2026 Research Navigation Guide

The landscape of peptide research has transformed dramatically. In 2026, scientists no longer search for simple linear sequences—they demand engineered architectures, pre-conjugated structures, and stability-validated compounds that reflect the industry's shift into what experts now call the "Modified Peptide" era.[1] A comprehensive peptide catalogue for scientists must now address these elevated expectations, offering not just sequences but complete research solutions with documented stability profiles, conjugation options, and application-specific formulations.
This evolution means that researchers in fitness science, martial arts performance optimization, and weightlifting recovery protocols need access to catalogues that understand both the cutting-edge peptide engineering principles and the practical requirements of athletic research. The days of browsing basic amino acid sequences are over—today's peptide catalogue for scientists must provide cyclized peptides, disulfide-bridged structures, and glycan-conjugated variants that survive real-world research conditions.
Key Takeaways
- Modern peptide catalogues prioritize engineered structures over linear sequences, with cyclized and disulfide-bridged peptides becoming the new research baseline for stability studies[1]
- Pre-conjugated peptides with chelator-ready cages (DOTA, NOTA) and biopolymer conjugates are increasingly standard offerings rather than custom modifications[1]
- Quality documentation has evolved to include stability engineering data, half-life extension validation, and GI survival profiles for oral delivery research[1]
- Catalogue navigation now requires understanding of peptide architecture categories: constrained peptides, glycan-conjugated variants, and caged structures for targeted applications
- Integration of manufacturing transparency helps researchers select suppliers using advanced synthesis methods like Tag-Assisted Peptide Synthesis (TAPS) for scalability and sustainability[5]
Understanding the Modern Peptide Catalogue for Scientists
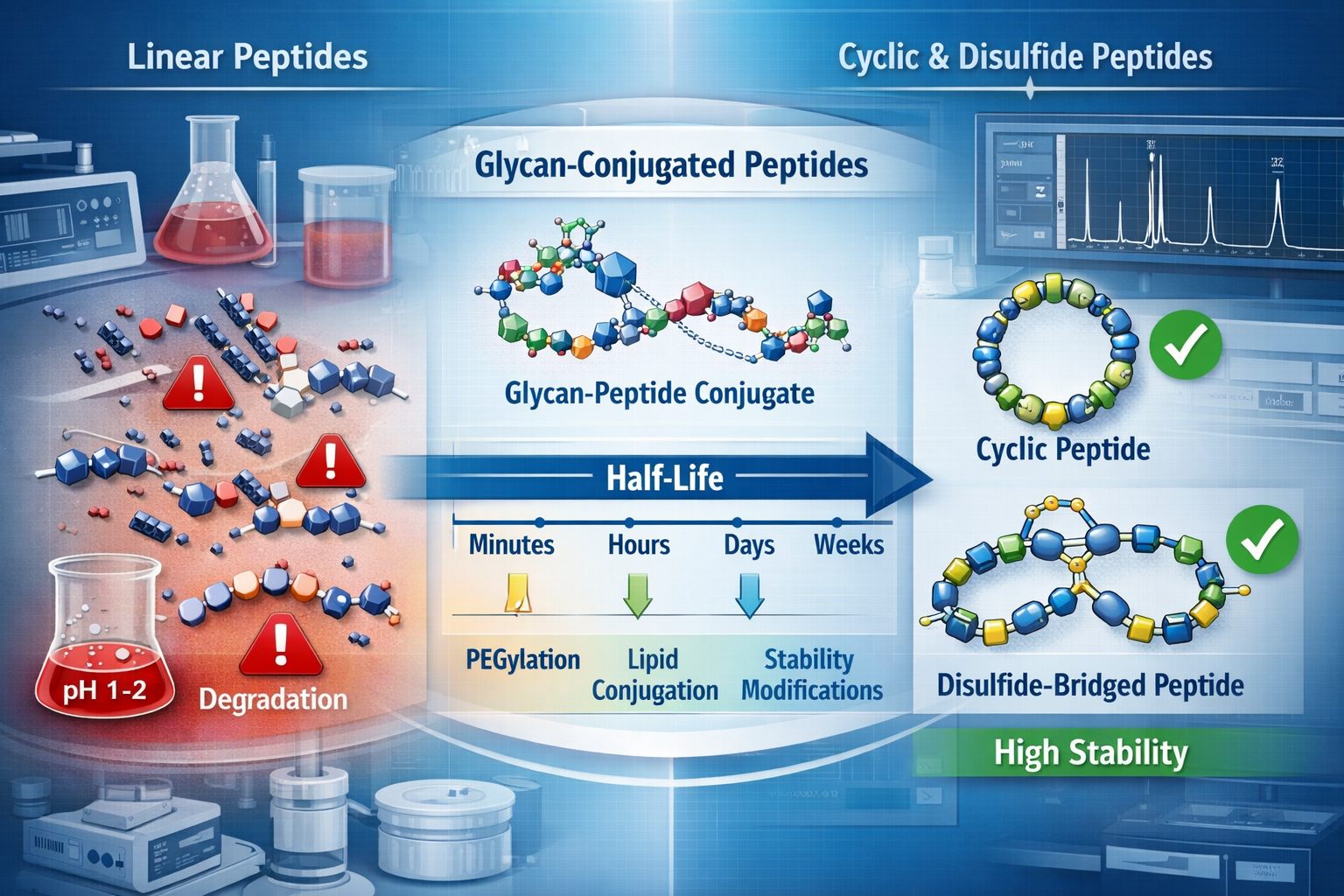
The transformation of peptide catalogues reflects fundamental shifts in research priorities. Traditional catalogues organized products by sequence length or alphabetical order. Modern scientific peptide catalogues categorize offerings by architectural class, stability profile, and delivery mechanism—reflecting the research community's focus on engineered solutions rather than raw sequences.
The Engineering Phase vs. Discovery Phase
According to data presented at the J.P. Morgan Healthcare Conference 2026, the peptide industry has officially transitioned from the "Discovery Phase" (characterized by breakthrough molecules like Semaglutide and Tirzepatide) into the "Engineering Phase."[1] This shift fundamentally changes what researchers expect from a peptide catalogue for scientists.
Discovery Phase Catalogues (pre-2025):
- Linear peptide sequences
- Basic purity specifications
- Standard lyophilized formats
- Minimal stability data
- Generic storage recommendations
Engineering Phase Catalogues (2026 forward):
- Cyclized and constrained architectures
- Stability engineering documentation
- Pre-conjugated delivery systems
- Half-life extension data
- Application-specific formulations
This evolution means that when fitness researchers, martial artists, or weightlifting scientists browse a modern peptide research catalogue, they encounter products designed with specific stability challenges in mind—whether that's surviving gastric acidity for oral administration studies or maintaining activity during extended storage periods.
Programmable Stability Engineering
MBX Biosciences has demonstrated that glycan-conjugation can extend peptide half-life from minutes to weeks, making traditional native sequences "obsolete as long-term controls."[1] This breakthrough has immediate implications for peptide catalogues designed for scientists conducting recovery research or performance optimization studies.
Researchers can now select peptides with engineered stability profiles matched to their experimental timeline:
| Stability Modification | Half-Life Extension | Primary Applications |
|---|---|---|
| Native sequence | Minutes to hours | Acute response studies |
| Single glycan conjugate | 12-24 hours | Daily administration protocols |
| Multi-glycan conjugate | 3-7 days | Weekly dosing research |
| Fc-fusion proteins | 7-14 days | Extended release studies |
| Elastin-like polypeptides | 14+ days | Long-term observation trials |
When evaluating a peptide catalogue for scientists, researchers should verify whether stability modifications are available as standard options rather than requiring custom synthesis. The comprehensive peptide catalogue should clearly indicate which architectural modifications are in stock versus special order.
Constrained Peptides as the New Baseline
Linear peptides are no longer considered relevant baselines for 2026 research. Cyclized and disulfide-bridged peptides—structures inspired by naturally occurring defensins and hepcidin—are now required for GI stability studies.[1] This represents a fundamental shift in how scientific peptide catalogues organize their inventory.
Constrained peptide architectures available in modern catalogues include:
✅ Disulfide-Rich Peptides (DRPs): Protagonist Therapeutics has validated that these structures survive gastric acidity, making them essential for oral delivery research[1]
✅ Bicyclic Peptides: Driving demand in targeted alpha therapy applications, particularly for chelator-ready precursors[1]
✅ Stapled Peptides: Enhanced membrane permeability for intracellular target access research
✅ Macrocyclic Peptides: Optimized for specific receptor binding with improved metabolic stability
Researchers working on mitochondrial peptide applications should prioritize catalogues offering these constrained architectures, as they provide superior stability profiles essential for athletic recovery and performance research.
Essential Categories in a Peptide Catalogue for Scientists
Modern peptide catalogues for scientists organize products by functional category and architectural class rather than simple alphabetical listings. Understanding these categories helps researchers navigate efficiently to compounds relevant for fitness, martial arts, and weightlifting applications.
Mitochondrial Function Peptides
Mitochondrial peptides represent a critical category for athletic performance research. These compounds target cellular energy production, oxidative stress management, and metabolic efficiency—all essential factors in recovery and endurance protocols.
SS-31 (Elamipretide) stands as the flagship mitochondrial peptide, with extensive documentation of its stability-engineered variants. A comprehensive SS-31 peptide catalogue should include:
- Standard sequence variants
- Cyclized versions for extended stability
- Pre-conjugated chelator-ready formats
- Combination formulations with complementary peptides
MOTS-C represents another essential mitochondrial peptide, particularly relevant for metabolic research in athletic populations. Quality catalogues provide MOTS-C with documented stability profiles and clear storage recommendations.
The emergence of synergistic peptide blends reflects advanced catalogue sophistication. Researchers can now access pre-formulated SS-31 and MOTS-C combinations designed for complementary mitochondrial support research.
Metabolic and Body Composition Peptides
The GLP-1 receptor agonist category has expanded significantly, reflecting the industry's transition through multiple "generations" of peptide engineering. A thorough peptide catalogue for scientists studying metabolic pathways should distinguish between these generations clearly.
First-generation GLP-1 peptides (Exenatide-like):
- Short half-life requiring frequent administration
- Limited stability modifications
- Basic receptor activation profiles
Second-generation compounds (Semaglutide-like):
- Extended half-life through fatty acid conjugation
- Improved stability profiles
- Enhanced receptor selectivity
Third-generation multi-agonists (Tirzepatide and beyond):
- Dual or triple receptor targeting
- Advanced stability engineering
- Optimized pharmacokinetic profiles
Researchers exploring these pathways should consult detailed GLP-1 generation comparisons and access next-generation multi-agonist compounds through catalogues offering comprehensive documentation.
Tesamorelin represents another critical category for body composition research, particularly relevant for martial artists and weightlifters studying growth hormone pathway modulation. Quality catalogues provide tesa with detailed dosage documentation and stability-engineered variants.
Immune Modulation and Recovery Peptides
Athletic recovery research increasingly focuses on immune system optimization and inflammation management. A complete scientific peptide catalogue should include well-documented immune modulators with stability profiles suitable for extended research protocols.
Thymosin Alpha-1 serves as a primary immune modulation peptide, with applications in recovery optimization and immune resilience research. Catalogues should provide thymosin with mechanism documentation and validated stability data.
Selank represents the cognitive and stress-management category, particularly relevant for martial artists studying mental performance factors. Access to Selank with comprehensive benefit documentation helps researchers design appropriate protocols.
BPC-157 remains a cornerstone of recovery research, though modern catalogues should offer cyclized variants with improved stability profiles. Researchers should seek BPC-157 documentation that includes architectural specifications and stability engineering details.
Longevity and Cellular Health Peptides
The longevity research category has expanded significantly, reflecting growing interest in peptides that address cellular aging mechanisms, telomere biology, and systemic rejuvenation pathways.
Epithalon leads this category as a primary telomerase-modulating peptide. Modern Epithalon catalogue entries should specify cyclization status and stability profiles, as these factors significantly impact research outcomes in extended protocols.
Bioregulator peptides including Thymogen, Crystagen, and Thymalin represent specialized subcategories within longevity research. A comprehensive peptide catalogue for scientists should organize these compounds with clear documentation of their tissue-specific targeting properties and recommended research applications.
The emergence of longevity-focused peptide blends reflects catalogue evolution toward application-specific formulations rather than individual compounds only.
Quality Markers in a Peptide Catalogue for Scientists
Not all peptide catalogues for scientists provide equal quality assurance. Understanding which documentation and specifications indicate superior sourcing helps researchers make informed decisions, particularly when working with limited budgets or strict protocol requirements.
Purity Testing and Documentation Standards
Modern peptide research demands purity levels exceeding historical standards. While 95% purity was once acceptable, 2026 research protocols increasingly specify 98%+ purity for critical applications.
Essential purity documentation in quality catalogues:
📊 HPLC chromatograms showing actual peak purity, not just percentage claims
📊 Mass spectrometry confirmation verifying molecular weight and sequence accuracy
📊 Certificate of Analysis (CoA) for each batch, not generic product specifications
📊 Endotoxin testing results for peptides intended for biological research
📊 Stability testing data documenting degradation profiles under various storage conditions
Researchers can verify catalogue quality by requesting detailed purity testing information before purchase. Catalogues that readily provide batch-specific documentation demonstrate superior quality control.
Reference Standards and Benchmarking
The integration of reference standards represents a critical quality marker in scientific peptide catalogues. Bachem, a leading peptide manufacturer, has established robust benchmarking protocols that quality catalogues should reference or exceed.
Reference standard indicators include:
✓ Comparison to pharmaceutical-grade benchmarks
✓ Stability validation against known degradation profiles
✓ Impurity profiling identifying common synthesis artifacts
✓ Batch-to-batch consistency documentation
Catalogues providing reference standard comparisons demonstrate commitment to quality that extends beyond basic purity claims. This becomes particularly important for researchers conducting long-term studies where batch consistency impacts data integrity.
Manufacturing Method Transparency
The emergence of Tag-Assisted Peptide Synthesis (TAPS) as a next-generation manufacturing technology highlights the importance of understanding production methods when evaluating peptide catalogues for scientists.[5]
Manufacturing transparency markers:
🔬 Synthesis method disclosure: SPPS (Solid-Phase Peptide Synthesis), liquid-phase synthesis, recombinant expression, or hybrid approaches
🔬 Scalability documentation: Indicating whether peptides are produced in research quantities only or scalable to larger batches
🔬 Sustainability practices: TAPS and other green chemistry approaches reduce environmental impact while improving consistency[5]
🔬 Quality control checkpoints: Documentation of purification steps, resin quality, and coupling efficiency
Catalogues that provide manufacturing transparency enable researchers to assess whether production methods align with their research requirements. For example, recombinant expression methods may offer advantages for larger peptides or those requiring specific post-translational modifications.[4]
Advanced Catalogue Features for 2026 Research
The peptide catalogue for scientists has evolved beyond simple product listings to include sophisticated features that support research design, protocol optimization, and data interpretation.
Pre-Conjugated and Delivery-Ready Formats
Research labs increasingly purchase peptides pre-conjugated with functional groups rather than performing conjugation in-house. This trend reflects both time efficiency and the specialized expertise required for consistent conjugation chemistry.
Standard pre-conjugated formats in advanced catalogues:
🧪 Chelator-ready peptides: DOTA and NOTA cages pre-attached for radiopharmaceutical research[1]
🧪 Fluorescent labels: FITC, rhodamine, or Cy5 conjugates for imaging studies
🧪 Biotin conjugation: For pull-down assays and binding studies
🧪 PEGylation variants: With defined PEG molecular weights for pharmacokinetic research
🧪 Cell-penetrating peptide fusions: Pre-conjugated delivery sequences for intracellular target research
The availability of these formats in a scientific peptide catalogue significantly accelerates research timelines and improves reproducibility by eliminating variable conjugation chemistry between experiments.
AI-Enabled Catalogue Navigation
The integration of artificial intelligence into catalogue navigation reflects presentations at the Peptide-Based Therapeutics Summit 2026, which featured workshops on "Artificial Intelligence for Peptide Design."[2] Advanced catalogues now incorporate AI-assisted search functions that understand research objectives rather than requiring exact sequence knowledge.
AI-enhanced catalogue features:
💡 Objective-based search: Entering "mitochondrial function in muscle tissue" returns relevant peptides ranked by research precedent
💡 Structure-activity predictions: AI models suggest architectural modifications likely to improve desired properties
💡 Protocol recommendations: Based on peptide characteristics and research objectives
💡 Literature integration: Automatic linking to relevant publications and research findings
These features transform the peptide catalogue for scientists from a passive product list into an active research tool that accelerates experimental design.
Application-Specific Formulation Guides
Modern catalogues recognize that researchers need more than just peptides—they need complete formulation guidance for their specific applications. This is particularly valuable for fitness researchers, martial artists, and weightlifting scientists who may have deep expertise in their performance domains but limited peptide chemistry backgrounds.
Formulation documentation in quality catalogues includes:
📋 Recommended reconstitution protocols for different research applications
📋 Stability data in various buffer systems and pH ranges
📋 Compatibility information for common research additives
📋 Storage optimization based on intended use timeline
📋 Handling precautions for sensitive peptide architectures
Access to application-specific peptide guides helps researchers avoid common formulation errors that compromise experimental outcomes.
Evaluating Peptide Catalogue Suppliers for Scientific Research
Selecting the right peptide catalogue for scientists requires evaluation beyond product listings. Supplier characteristics significantly impact research success, particularly for long-term studies requiring consistent peptide quality across multiple batches.
Batch Consistency and Supply Reliability
Research protocols spanning months or years require confidence that peptide quality remains consistent across batches. This becomes critical for studies comparing baseline and intervention periods or tracking longitudinal changes in athletic populations.
Batch consistency indicators:
✅ Documented batch-to-batch variability: Quality suppliers provide statistical analysis of purity and composition across production runs
✅ Reserved batch programs: Option to reserve sufficient material from a single batch for entire study duration
✅ Retest dating: Clear guidance on stability limits and recommended use timelines
✅ Supply chain transparency: Information about raw material sourcing and manufacturing location stability
Catalogues offering wholesale peptide options often provide better batch consistency for larger research projects, as wholesale production typically involves more rigorous quality control systems.
Technical Support and Research Consultation
The complexity of modern peptide architectures means that researchers benefit significantly from supplier technical support. The best peptide catalogues for scientists connect to teams with deep peptide chemistry expertise who can address formulation questions, stability concerns, and protocol optimization.
Technical support quality markers:
🎯 Responsive communication: Clear timelines for inquiry responses
🎯 Chemistry expertise: Staff with advanced degrees in peptide chemistry or related fields
🎯 Protocol assistance: Willingness to review research designs and suggest optimizations
🎯 Troubleshooting support: Help diagnosing unexpected results or stability issues
🎯 Literature access: Provision of relevant research publications and data
Researchers working on novel applications—such as combining peptide protocols with specific martial arts training regimens or weightlifting periodization—particularly benefit from suppliers offering consultation services.
Regulatory Compliance and Documentation
While research peptides exist in a different regulatory space than pharmaceutical products, quality suppliers maintain documentation standards that support rigorous scientific work and potential future translation.
Compliance documentation in professional catalogues:
📑 Good Manufacturing Practice (GMP) adherence: Even for research-grade materials
📑 Chain of custody documentation: Tracking from synthesis through delivery
📑 Storage and handling records: Demonstrating proper conditions throughout supply chain
📑 Regulatory status clarity: Clear labeling of research-only status and appropriate disclaimers
📑 Export documentation: For international research collaborations
These standards become particularly important for researchers at institutions with strict procurement requirements or those planning to publish findings in high-impact journals requiring detailed methods documentation.
Navigating Peptide Catalogues by Research Theme
Efficient catalogue navigation requires understanding how to filter vast product selections to identify peptides relevant for specific research themes. Modern peptide catalogues for scientists increasingly organize by research objective rather than only by chemical structure.
Performance and Recovery Research Navigation
Researchers studying athletic performance, martial arts training optimization, or weightlifting recovery protocols should navigate catalogues using functional categories rather than browsing alphabetically.
Performance research peptide categories:
🏋️ Mitochondrial function: SS-31, MOTS-C, and humanin for cellular energy research
🏋️ Growth hormone pathways: Tesamorelin, CJC-1295, Ipamorelin for body composition studies
🏋️ Recovery acceleration: BPC-157, TB-500, and thymosin compounds for tissue repair research
🏋️ Metabolic optimization: GLP-1 agonists and multi-receptor agonists for energy utilization studies
🏋️ Cognitive performance: Selank, Semax, and nootropic peptides for mental aspects of athletic performance
Quality catalogues provide thematic navigation tools that organize peptides by these functional categories, significantly reducing search time.
Longevity and Cellular Health Research
The intersection of athletic performance and longevity research represents a growing area of interest, particularly for martial artists and fitness enthusiasts interested in sustainable, long-term training capacity.
Longevity research peptide organization:
🧬 Telomere biology: Epithalon and related epigenetic modulators
🧬 Cellular senescence: Peptides targeting aging cell clearance and renewal
🧬 Tissue-specific bioregulation: Organ-targeted peptides for systemic optimization
🧬 Oxidative stress management: Antioxidant peptides and mitochondrial protectants
🧬 Immune system optimization: Thymic peptides and immune modulators
Catalogues offering dedicated longevity research sections recognize this growing research area and organize products accordingly.
Combination Protocols and Synergistic Formulations
Advanced research protocols increasingly employ multiple peptides with complementary mechanisms. Modern peptide catalogues for scientists respond by offering pre-formulated combinations validated for synergistic activity.
Combination formulation categories:
💊 Growth hormone secretagogue blends: CJC-1295 with Ipamorelin for pulsatile release research
💊 Mitochondrial support stacks: SS-31 with MOTS-C for comprehensive cellular energy studies
💊 Recovery complexes: BPC-157 with TB-500 for multi-pathway tissue repair research
💊 Metabolic optimization blends: Tesamorelin with AOD9604 for body composition research
💊 Longevity formulations: Epithalon with bioregulator peptides for systemic anti-aging studies
Access to validated peptide blends provides researchers with formulations that have undergone compatibility testing and stability validation, reducing experimental variables.
Emerging Trends Shaping Peptide Catalogues in 2026
The peptide catalogue for scientists continues to evolve rapidly, reflecting both technological advances in peptide engineering and changing research priorities across multiple disciplines.
Oral Delivery Peptides Becoming Standard
Protagonist Therapeutics' validation of Disulfide-Rich Peptides (DRPs) that survive gastric acidity has triggered a fundamental shift in catalogue organization.[1] Oral delivery capability is no longer a specialized feature—it's becoming an expected standard for many peptide categories.
Oral delivery architecture categories now appearing in catalogues:
🔄 Disulfide-bridged peptides: Multiple disulfide bonds creating gastric acid-resistant structures
🔄 Cyclized peptides: Head-to-tail cyclization improving proteolytic stability
🔄 Caged structures: Protective molecular cages that release active peptide after absorption
🔄 Permeation enhancers: Peptides pre-formulated with absorption-enhancing excipients
The Oral & Macrocyclic Peptides Conference (April 15-16, 2026) highlighted membrane permeability optimization and intracellular target access as priority research areas,[3] suggesting that catalogues will increasingly organize peptides by delivery route capability.
Researchers interested in oral delivery applications should seek catalogues providing innovative delivery system documentation that includes GI stability data and absorption profiles.
Integration of Recombinant Expression Products
The PepTalk Conference (January 19-22, 2026) emphasized the integration of recombinant expression methods alongside traditional chemical synthesis.[4] This reflects a broader industry trend toward hybrid manufacturing approaches that combine the precision of chemical synthesis with the scalability and sustainability of biological production.
Recombinant expression advantages driving catalogue integration:
🧫 Scalability: Biological production scales more economically for larger peptides
🧫 Sustainability: Reduced solvent use and waste generation compared to SPPS
🧫 Post-translational modifications: Natural glycosylation and other modifications difficult to achieve chemically
🧫 Cost efficiency: Lower production costs for peptides over 50 amino acids
Modern peptide catalogues for scientists should indicate production method, allowing researchers to select based on their specific requirements. Some applications benefit from the absolute sequence control of chemical synthesis, while others may prefer the natural modifications and cost advantages of recombinant production.
AI-Designed Peptides Entering Catalogues
The Peptide-Based Therapeutics Summit 2026 featured workshops on artificial intelligence for peptide design,[2] and the Oral & Macrocyclic Peptides Conference highlighted AI-enabled design methods including AfCycDesign and RFpeptides.[3] These tools are beginning to generate novel peptide sequences that appear in commercial catalogues.
AI-designed peptide characteristics:
🤖 Optimized binding affinity: Computational design targeting specific receptors
🤖 Enhanced stability profiles: AI-predicted modifications improving half-life
🤖 Reduced immunogenicity: Sequence optimization minimizing immune response risk
🤖 Improved membrane permeability: Designed for intracellular target access
As these peptides enter catalogues, researchers should look for documentation explaining the design rationale, validation data, and comparison to naturally occurring or traditionally designed alternatives.
Targeted Alpha Therapy Peptide Precursors
The expansion of Targeted Alpha Therapy, supported by major supply deals for Lead-212,[1] is driving demand for bicyclic peptides and chelator-ready precursors. While this application area may seem distant from fitness and athletic research, the underlying peptide architectures offer advantages for other applications.
Bicyclic peptide advantages relevant beyond radiopharmaceuticals:
⚛️ Extreme stability: Constrained structures resist degradation
⚛️ Precise targeting: Designed for specific receptor binding
⚛️ Reduced off-target effects: Structural constraints improve selectivity
⚛️ Predictable pharmacokinetics: Constrained structures show consistent behavior
Researchers working on targeted delivery applications—such as directing recovery peptides specifically to muscle tissue or joint structures—may benefit from exploring bicyclic architectures even when not working with radioisotopes.
Practical Steps for Using a Peptide Catalogue for Scientists

Translating catalogue knowledge into successful research requires systematic approaches to peptide selection, validation, and protocol development.
Defining Research Objectives Before Catalogue Search
The sophistication of modern peptide catalogues for scientists means that effective searching requires clear objective definition before browsing. Vague searches return overwhelming results, while precisely defined objectives enable efficient navigation.
Objective definition framework:
1️⃣ Primary research question: What specific biological process or outcome is being studied?
2️⃣ Target tissue or system: Where should the peptide act (muscle, mitochondria, immune system, etc.)?
3️⃣ Timeline requirements: Acute effects vs. chronic administration studies?
4️⃣ Delivery route preferences: Oral, injectable, topical, or other administration routes?
5️⃣ Stability requirements: Storage duration and conditions relevant to research design?
6️⃣ Budget constraints: Research-grade vs. pharmaceutical-grade requirements?
7️⃣ Regulatory considerations: Institutional requirements or publication standards?
With clear objectives defined, researchers can efficiently navigate comprehensive peptide catalogues using filters and category systems rather than browsing entire inventories.
Validating Peptide Selection Through Literature Review
Once candidate peptides are identified in a scientific peptide catalogue, validation through literature review ensures that selections align with established research precedent and avoid common pitfalls.
Literature validation checklist:
📚 Mechanism documentation: Published research confirming proposed mechanism of action
📚 Dosage precedents: Established concentration ranges in similar research contexts
📚 Stability considerations: Published data on storage, handling, and formulation requirements
📚 Known interactions: Documentation of synergies or conflicts with other compounds
📚 Safety profiles: Research-context safety data relevant to study design
📚 Methodological guidance: Published protocols for similar applications
Resources like the latest peptide research findings help researchers stay current with emerging evidence that may not yet be reflected in catalogue descriptions.
Requesting Technical Documentation
Quality peptide catalogues for scientists provide extensive technical documentation, but researchers often need to specifically request certain information rather than assuming it's automatically provided.
Essential technical documents to request:
📄 Batch-specific Certificate of Analysis: Not generic product specifications
📄 HPLC and mass spectrometry data: Actual chromatograms and spectra
📄 Stability testing results: Data on degradation under various conditions
📄 Handling and storage protocols: Detailed guidance for maintaining peptide integrity
📄 Reconstitution recommendations: Solvent selection and concentration guidance
📄 Compatibility information: Data on buffer systems, pH ranges, and common additives
Researchers should establish communication with catalogue suppliers before purchase to ensure all necessary documentation will be available. This becomes particularly important when institutional procurement systems require specific documentation formats.
Pilot Testing Before Full Protocol Implementation
Even when using high-quality peptide catalogues for scientists with extensive documentation, pilot testing before full protocol implementation helps identify unexpected issues and optimize procedures.
Pilot testing protocol:
🔬 Reconstitution validation: Verify that peptide dissolves as expected and remains stable
🔬 Assay compatibility: Confirm that peptide doesn't interfere with detection or measurement methods
🔬 Dose-response exploration: Establish appropriate concentration ranges for main study
🔬 Timeline optimization: Determine optimal administration schedules and measurement timepoints
🔬 Storage stability confirmation: Validate that working solutions maintain activity over study duration
Small-scale pilot studies using limited quantities from a research peptide catalogue prevent costly errors in full-scale experiments and provide valuable preliminary data for protocol refinement.
Conclusion: Maximizing Research Success with Modern Peptide Catalogues
The transformation of the peptide catalogue for scientists from simple product lists to sophisticated research tools reflects the broader evolution of peptide science into the Engineering Phase. Researchers in 2026 have access to stability-engineered architectures, pre-conjugated delivery systems, and AI-designed sequences that would have been custom synthesis projects just a few years ago.
For fitness researchers, martial artists, and weightlifting scientists exploring peptide applications in performance optimization and recovery, this evolution offers unprecedented opportunities. Modern catalogues provide not just peptides but complete research solutions with documented stability profiles, application-specific formulations, and technical support that accelerates protocol development.
Key success factors for maximizing research outcomes:
✨ Prioritize architectural sophistication: Choose cyclized, disulfide-bridged, or otherwise constrained peptides over linear sequences for improved stability
✨ Demand comprehensive documentation: Insist on batch-specific certificates of analysis, stability data, and reference standard comparisons
✨ Leverage pre-conjugated formats: Save time and improve reproducibility by using peptides with pre-attached functional groups
✨ Utilize thematic navigation: Organize searches by research objective rather than browsing alphabetically
✨ Validate through multiple sources: Cross-reference catalogue information with published literature and technical consultation
✨ Establish supplier relationships: Build connections with catalogue technical support teams for ongoing protocol optimization
✨ Plan for batch consistency: For long-term studies, reserve sufficient material from single batches or establish supply agreements
Actionable Next Steps
Researchers ready to leverage modern peptide catalogues for scientists should take these immediate actions:
-
Audit current peptide sources against 2026 quality standards—are suppliers providing stability-engineered architectures and comprehensive documentation?
-
Review existing protocols to identify opportunities for upgrading from linear to constrained peptide architectures that may improve research outcomes
-
Explore pre-conjugated options for peptides currently requiring in-house conjugation chemistry
-
Establish technical support relationships with catalogue suppliers before urgent research needs arise
-
Create a peptide selection framework based on research objectives, delivery requirements, and stability needs rather than defaulting to familiar sequences
-
Investigate combination formulations that may provide synergistic benefits and reduce experimental complexity
-
Stay informed about emerging peptide engineering technologies through conference proceedings and industry publications
The peptide catalogue for scientists in 2026 represents far more than a purchasing portal—it's a gateway to cutting-edge research tools that can significantly accelerate discovery and optimize experimental outcomes. By understanding catalogue organization, demanding appropriate documentation, and leveraging advanced features like AI-enabled navigation and pre-conjugated formats, researchers position themselves to conduct higher-quality studies with greater efficiency and reproducibility.
Whether investigating mitochondrial function in athletic recovery, exploring metabolic optimization for body composition research, or studying immune modulation in training adaptation, the modern comprehensive peptide catalogue provides the tools and support necessary for rigorous scientific investigation. The key lies in approaching catalogue navigation with the same systematic rigor applied to experimental design itself—defining clear objectives, validating selections through multiple sources, and establishing relationships with suppliers who view their role as research partners rather than mere vendors.
References
[1] Jpm 2026 Peptide Trends – https://www.peptidesystems.com/post/jpm-2026-peptide-trends
[2] New For 2026 – https://peptide-based-therapeutics-summit.com/new-for-2026/
[3] Oral Peptides And Macrocyclics 2 – https://www.drugdiscoverychemistry.com/oral-peptides-and-macrocyclics-2
[4] Peptide – https://www.chi-peptalk.com/peptide
[5] Learn About Peptide Drug Substance Manufacturing At Dcat Week 2026 – https://cordenpharma.com/learn-about-peptide-drug-substance-manufacturing-at-dcat-week-2026/
SEO Meta Title and Description
Meta Title: Peptide Catalogue for Scientists: 2026 Research Guide
Meta Description: Comprehensive guide to navigating modern peptide catalogues for scientists. Discover stability-engineered peptides, quality markers, and research navigation strategies.
