
Epithalon Risks and Safety: A Comprehensive Research-Based Analysis

The promise of cellular regeneration and extended lifespan has captivated researchers and health enthusiasts alike, but what happens when the science behind anti-aging peptides reveals significant safety gaps? Epithalon risks and safety concerns have emerged as critical considerations for anyone exploring this synthetic tetrapeptide, despite its decades of research and potential longevity benefits.
This comprehensive analysis examines the current state of epithalon safety data, regulatory concerns, and evidence-based recommendations for researchers and fitness enthusiasts considering this peptide for longevity research.
Key Takeaways
• Critical safety data gaps exist despite over 30 years of research, with limited long-term safety characterization
• FDA has identified immunogenicity risks that could trigger life-threatening immune responses
• No pharmaceutical-grade versions are approved for prescription use in the United States
• Medical supervision is essential for safe administration and proper dosing protocols
• Quality sourcing through licensed providers is crucial for minimizing contamination and dosing risks
Understanding Epithalon: The Foundation of Safety Assessment

Epithalon represents one of the most studied synthetic peptides in longevity research, yet understanding its safety profile requires examining both its mechanisms and limitations. This four-amino-acid sequence (Ala-Glu-Asp-Gly) mimics the natural peptide epithalamin, which is produced by the pineal gland and plays a role in circadian rhythm regulation and cellular aging processes.
The peptide's primary mechanism involves telomerase activation, which can potentially extend cellular lifespan by maintaining telomere length. However, this same mechanism raises important safety questions about uncontrolled cellular proliferation and cancer risk, particularly in individuals with existing malignancies or genetic predispositions.
Research has shown that epithalon can influence multiple physiological systems simultaneously, creating what scientists call "cascade effects." While these interconnected benefits may enhance overall health outcomes, they also complicate safety assessment since changes in one system can trigger unexpected responses in others.[2]
For those interested in exploring epithalon longevity research, understanding these foundational mechanisms is essential before considering any research applications. The complexity of epithalon's effects underscores why comprehensive safety evaluation remains challenging even after decades of study.
Current Regulatory Status and FDA Safety Concerns
FDA Immunogenicity Warnings
The U.S. Food and Drug Administration has specifically identified epithalon risks and safety concerns related to immunogenicity, where the body's immune system may mount potentially life-threatening responses against the peptide.[2] This classification places epithalon among substances that require careful monitoring and medical oversight.
Immunogenicity risks can manifest in several ways:
- Allergic reactions ranging from mild skin irritation to severe anaphylaxis
- Autoimmune responses where the immune system attacks healthy tissues
- Neutralizing antibodies that reduce peptide effectiveness over time
- Cross-reactivity with natural proteins in the body
Regulatory Gaps and Availability
Currently, only research-grade epithalon is available in the United States, with no FDA-approved pharmaceutical versions for clinical use.[2] This regulatory status creates important implications for safety and quality control:
Research-Grade Limitations:
- No standardized manufacturing requirements
- Variable purity levels between suppliers
- Limited quality control oversight
- Potential contamination risks
Clinical Implications:
- No established prescribing guidelines
- Lack of standardized dosing protocols
- Limited adverse event reporting systems
- Insufficient post-market surveillance
For researchers seeking high-quality materials, exploring most popular products for sale through verified suppliers becomes crucial for minimizing contamination and dosing accuracy risks.
International Regulatory Perspectives
While the FDA maintains strict oversight, other regulatory bodies have taken different approaches to epithalon. Some countries allow limited clinical use under specific conditions, while others have banned the substance entirely. This regulatory patchwork creates confusion and highlights the ongoing uncertainty about epithalon safety protocols worldwide.
Clinical Evidence: What Research Reveals About Epithalon Risks and Safety
Favorable Short-Term Safety Data
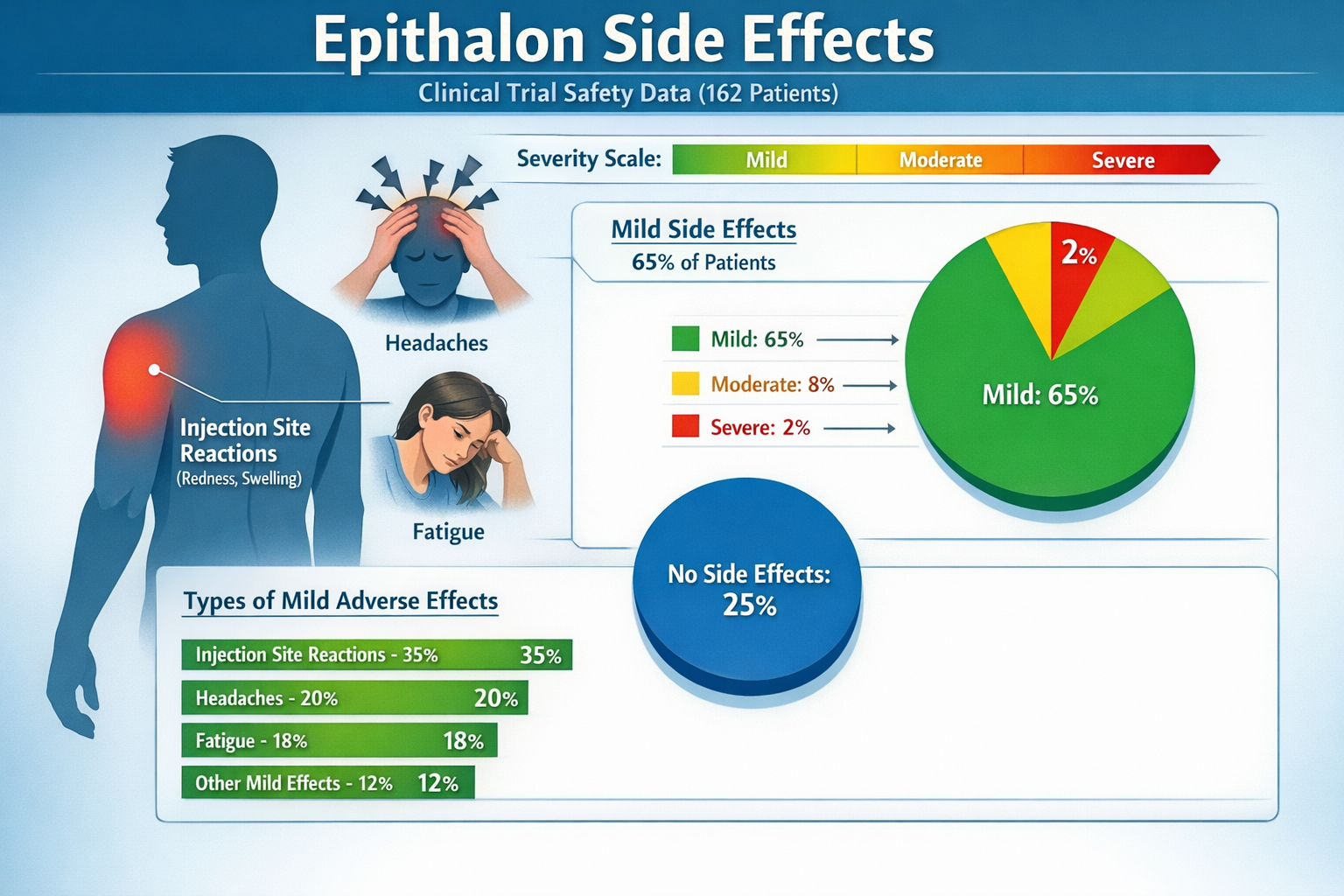
Despite regulatory concerns, clinical trials have provided some reassuring safety data. A significant 2002 study involving 162 patients found no serious side effects reported in the epithalon treatment group, suggesting that short-term use may be relatively well-tolerated in healthy populations.[2]
Additional encouraging findings include:
- 2015 Cognitive Vitality Report by the Alzheimer's Drug Discovery Foundation noted no severe adverse events in multi-year treatment trials involving older adults[2]
- Minimal dropout rates in clinical studies due to adverse effects
- Good tolerance across different age groups and health conditions
- Reversible side effects that typically resolve after treatment discontinuation
Documented Side Effects and Adverse Reactions
While serious adverse events appear rare, epithalon risks and safety profiles do include several documented side effects that researchers and users should understand:
Common Mild Side Effects:
- Injection site reactions (redness, swelling, mild pain)
- Transient headaches lasting 1-2 hours
- Mild fatigue or drowsiness
- Temporary changes in sleep patterns
- Minor gastrointestinal discomfort
Less Common Reactions:
- Dizziness or lightheadedness
- Mood changes or irritability
- Temporary changes in appetite
- Mild nausea
Most reported side effects are temporary and resolve within 24-48 hours of administration.[3] However, the limited scope of clinical trials means that rare or delayed adverse effects may not yet be fully characterized.
Critical Safety Data Gaps
A 2025 systematic review highlighted a concerning reality: "information regarding critical issues about this peptide's safety is missing," despite over 30 years of research.[2] These gaps include:
Long-Term Safety Unknowns:
- Effects of extended use beyond 6-12 months
- Potential cumulative toxicity
- Long-term cardiovascular impacts
- Reproductive system effects
- Interaction with age-related diseases
Population-Specific Risks:
- Safety in individuals with autoimmune conditions
- Effects in people with genetic predispositions to cancer
- Interactions with common medications
- Safety in different ethnic populations
For those comparing different longevity approaches, reviewing epithalon vs NAD evidence can provide valuable context for understanding relative safety profiles across different anti-aging interventions.
Special Populations and Contraindications
High-Risk Groups
Healthcare providers strongly advise that certain populations should avoid epithalon or consult extensively with medical professionals before considering use due to insufficient safety data:[2]
Absolute Contraindications:
- Pregnant women – No safety data exists for fetal development
- Breastfeeding mothers – Unknown effects on infant health through breast milk
- Active cancer patients – Telomerase activation could potentially accelerate tumor growth
- Suspected cancer cases – May interfere with early detection or treatment
Relative Contraindications (Require Medical Consultation):
- History of cancer or strong family history
- Autoimmune disorders
- Severe cardiovascular disease
- Liver or kidney dysfunction
- Immunocompromised states
Age-Related Considerations
While epithalon research has focused primarily on older adults, age-specific safety considerations remain important:
Younger Adults (18-40):
- Limited research data in this population
- Potential interference with natural hormonal development
- Unknown effects on natural telomerase activity
- Risk-benefit ratio less favorable due to existing cellular health
Older Adults (65+):
- Most clinical trial data available for this group
- Generally better tolerance reported
- Higher potential benefits may justify risks
- Increased medication interaction potential
Understanding these population-specific factors becomes essential when reviewing longevity peptide research options and making informed decisions about research participation.
Dosing Protocols and Administration Safety
Standard Research Protocols
Research settings typically employ specific dosing protocols designed to maximize benefits while minimizing epithalon risks and safety concerns:
Typical Dosing Parameters:
- Dose range: 5-10 mg per administration
- Route: Subcutaneous injection
- Duration: 10-20 days per cycle
- Frequency: 1-2 cycles per year
- Rest periods: Several months between cycles
Injection Safety Considerations
Proper injection technique is crucial for minimizing adverse reactions and ensuring optimal absorption:
Injection Site Management:
- Rotate injection sites to prevent tissue damage
- Use sterile technique and new needles for each injection
- Monitor for signs of infection or adverse reactions
- Apply appropriate post-injection care
Common Injection Errors:
- Reusing needles or syringes
- Inadequate site sterilization
- Incorrect injection depth
- Failure to rotate injection sites
Cycle Timing and Medical Supervision
Functional medicine providers typically recommend epithalon use in carefully planned cycles under licensed medical oversight.[1] This supervised approach helps ensure:
- Appropriate candidate selection through comprehensive health screening
- Proper dosing calculations based on individual factors
- Ongoing monitoring for adverse effects or complications
- Adjustment protocols if side effects occur
For researchers interested in understanding how epithalon compares to other peptide combinations, exploring information about peptide safety protocols can provide valuable insights into best practices for multiple peptide research.
Quality Control and Sourcing Safety
Critical Importance of Verified Suppliers
Epithalon risks and safety considerations extend significantly beyond the peptide itself to include quality control and sourcing factors. Self-sourcing carries substantial risks related to purity, contamination, and dosing accuracy.[1]
Quality Control Standards:
- Third-party purity testing and verification
- Certificate of analysis (COA) documentation
- Proper storage and handling protocols
- Sterile manufacturing environments
- Batch tracking and quality assurance
Contamination Risks
Unverified or low-quality epithalon sources may contain:
- Bacterial endotoxins that can cause severe inflammatory reactions
- Heavy metals leading to toxic accumulation
- Residual solvents from manufacturing processes
- Incorrect peptide sequences reducing effectiveness or causing adverse effects
- Variable concentrations leading to under- or overdosing
Professional Sourcing Recommendations
Healthcare providers emphasize sourcing epithalon through qualified medical professionals to ensure safety and quality.[1] This approach provides:
- Medical screening to identify contraindications
- Proper dosing guidance based on individual health status
- Ongoing monitoring throughout treatment cycles
- Emergency protocols if adverse reactions occur
- Quality assurance through verified pharmaceutical suppliers
For those researching quality sourcing options, examining quality testing protocols can provide essential information about proper verification standards and safety measures.
Risk Mitigation Strategies and Best Practices

Pre-Treatment Assessment
Comprehensive health evaluation before beginning epithalon research should include:
Essential Health Screening:
- Complete blood count and metabolic panel
- Cancer screening appropriate for age and risk factors
- Cardiovascular assessment
- Immune system evaluation
- Medication and supplement review
Risk Factor Assessment:
- Family history of cancer or autoimmune disease
- Previous adverse reactions to peptides or biologics
- Current health conditions and medications
- Lifestyle factors affecting immune function
Monitoring Protocols During Treatment
Active monitoring during epithalon cycles helps identify potential issues early:
Weekly Monitoring:
- Injection site inspection
- Symptom tracking and documentation
- Sleep pattern and energy level assessment
- Any new or unusual symptoms
Monthly Evaluation:
- Blood work monitoring for key markers
- Comprehensive symptom review
- Dose adjustment if necessary
- Assessment of treatment response
Emergency Preparedness
Given the FDA's immunogenicity warnings, having emergency protocols in place is essential:
Emergency Indicators:
- Severe allergic reactions (difficulty breathing, widespread rash)
- Unusual swelling or inflammation
- Persistent or worsening side effects
- Signs of infection at injection sites
Response Protocols:
- Immediate medical contact information
- Emergency medication availability (if prescribed)
- Clear instructions for treatment discontinuation
- Healthcare provider emergency procedures
For researchers exploring multiple peptide options, understanding how different compounds interact is crucial. Resources about most popular products for sale can help identify safer combination approaches and evidence-based protocols.
Future Research Directions and Safety Evolution
Ongoing Safety Studies
Current research efforts focus on addressing the identified safety gaps in epithalon risks and safety profiles:
Priority Research Areas:
- Long-term safety studies extending beyond 2-3 years
- Population-specific safety profiles
- Optimal dosing strategies for different age groups
- Interaction studies with common medications
- Biomarker development for safety monitoring
Regulatory Development
As research continues, regulatory frameworks are evolving:
Expected Developments:
- Enhanced safety reporting requirements
- Standardized manufacturing guidelines
- Improved quality control standards
- Clearer prescribing protocols
- Better adverse event tracking systems
Technology Integration
Emerging technologies may improve epithalon safety monitoring:
Innovative Approaches:
- Wearable devices for real-time health monitoring
- Genetic testing for personalized risk assessment
- Advanced biomarker panels for safety tracking
- Telemedicine integration for remote monitoring
- AI-powered adverse event prediction
Understanding these evolving safety landscapes helps researchers make informed decisions about current and future epithalon research participation.
Conclusion
Epithalon risks and safety considerations present a complex landscape that requires careful evaluation and professional guidance. While clinical trials have shown generally favorable short-term safety profiles with minimal serious adverse events, significant data gaps remain regarding long-term use, population-specific risks, and rare adverse effects.
The FDA's immunogenicity warnings underscore the importance of medical supervision and proper risk assessment before considering epithalon research. Key safety principles include comprehensive pre-treatment screening, adherence to established dosing protocols, quality sourcing through verified suppliers, and ongoing monitoring throughout treatment cycles.
Actionable Next Steps:
- Consult with qualified healthcare providers experienced in peptide therapy before considering epithalon research
- Undergo comprehensive health screening to identify potential contraindications or risk factors
- Source only through verified, quality-controlled suppliers with proper testing documentation
- Implement robust monitoring protocols throughout any research participation
- Stay informed about evolving safety research and regulatory developments
For researchers and fitness enthusiasts interested in longevity peptides, prioritizing safety through professional guidance and evidence-based approaches remains essential. The potential benefits of epithalon must be carefully weighed against individual risk factors and current safety limitations.
As research continues to evolve, maintaining a conservative, medically-supervised approach to epithalon risks and safety will help ensure that the pursuit of longevity benefits doesn't compromise immediate health and safety.
References
[1] Epithalon Safety Guide – https://holisticmedicalwellness.com/peptides/epithalon-safety-guide/
[2] Epitalon – https://www.innerbody.com/epitalon
[3] Epithalon The Peptide Linked To Longevity And Cellular Renewal – https://pulseandremedy.com/anti-aging/epithalon-the-peptide-linked-to-longevity-and-cellular-renewal/
[4] Certain Bulk Drug Substances Use Compounding May Present Significant Safety Risks – https://www.fda.gov/drugs/human-drug-compounding/certain-bulk-drug-substances-use-compounding-may-present-significant-safety-risks
[5] Watch – https://www.youtube.com/watch?v=cM9kNK9llfE
Meta Title: Epithalon Risks and Safety: 2026 Research-Based Analysis
Meta Description: Comprehensive guide to epithalon risks and safety. Learn about FDA warnings, clinical evidence, contraindications, and best practices for safe peptide research in 2026.
{“@context”:”https://schema.org”,”@type”:”Article”,”headline”:”epithalon risks and safety”,”description”:”Comprehensive guide to epithalon risks and safety. Learn about FDA warnings, clinical evidence, contraindications, and best practices for safe peptide research “,”image”:”https://zsxkvszxbhpwnvzxdydv.supabase.co/storage/v1/object/public/generated-images/kie/dbb691a4-e81f-4e88-b195-867c3fa4f5f3/slot-0-1770077507377.png”,”datePublished”:”2026-02-03T00:09:10.477807+00:00″,”dateModified”:”2026-02-03T00:13:22.954Z”,”author”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″},”publisher”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″}}
