AOD 9604 Peptide Oral: Complete Research Guide for 2026

The world of peptide research has witnessed remarkable advances in recent years, with AOD 9604 peptide oral formulations emerging as a significant area of scientific interest. This synthetic peptide fragment, derived from human growth hormone, represents a fascinating convergence of molecular biology and pharmaceutical innovation that continues to capture the attention of researchers worldwide.
Key Takeaways
• AOD 9604 is a synthetic peptide fragment consisting of the last 15 amino acids of human growth hormone with an additional tyrosine residue
• Oral formulations of AOD 9604 offer potential advantages in bioavailability and research applications compared to other delivery methods
• Laboratory studies have focused primarily on metabolic pathways and cellular mechanisms rather than direct therapeutic applications
• Research protocols typically involve careful dosing considerations and proper storage conditions for optimal peptide stability
• Quality sourcing from reputable suppliers is essential for maintaining research integrity and reproducible results
Understanding AOD 9604 Peptide Structure and Properties
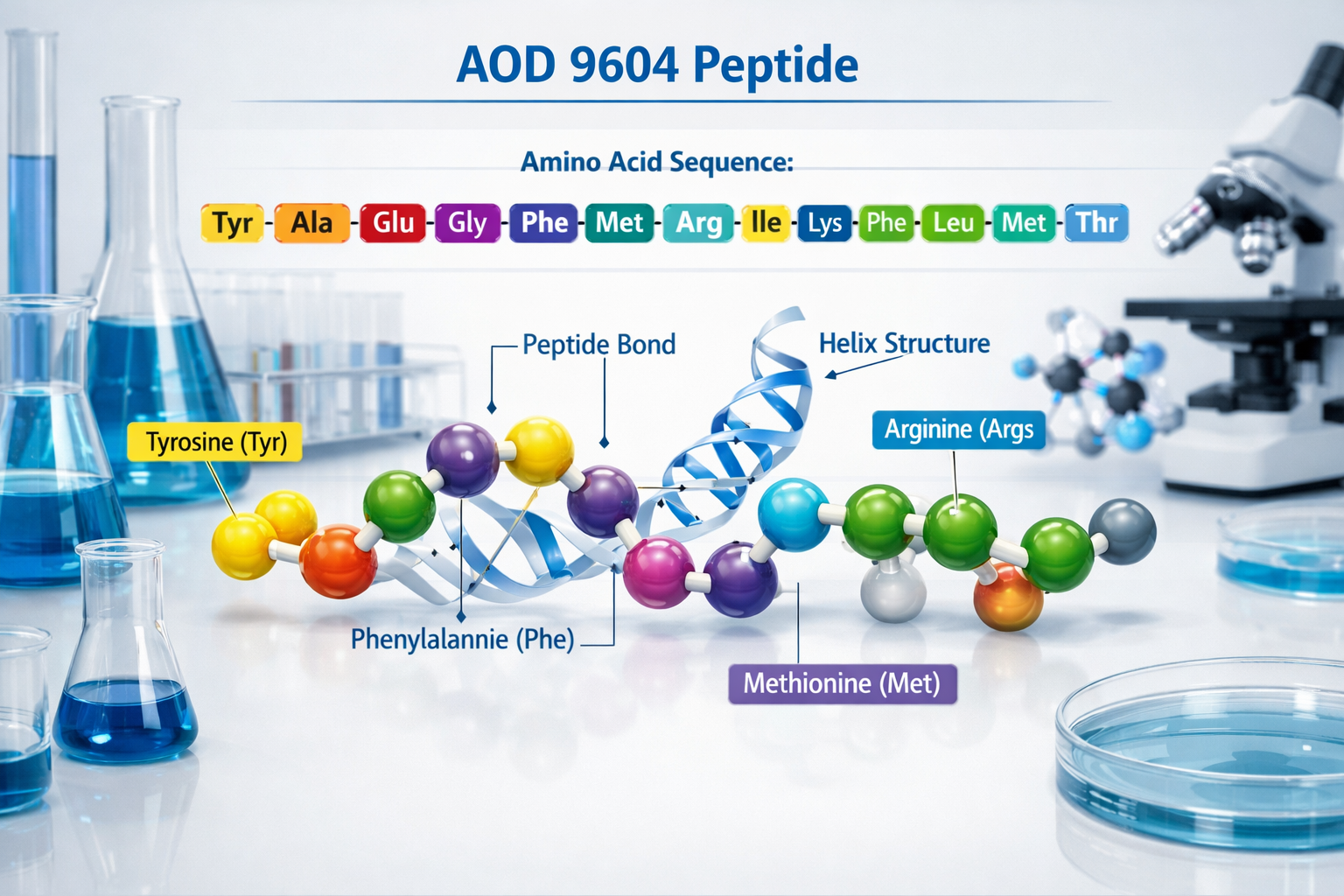
AOD 9604 peptide represents a carefully engineered fragment of human growth hormone (hGH), specifically comprising amino acids 177-191 with an additional tyrosine residue at the N-terminus. This modification creates a 16-amino acid sequence that maintains specific biological properties while exhibiting distinct characteristics from the parent hormone.
The molecular structure of this peptide fragment has been extensively studied in laboratory settings. Research indicates that the aod 9604 peptide retains certain functional domains of growth hormone while lacking others, creating a unique profile for scientific investigation. The peptide's molecular weight of approximately 1815 daltons makes it suitable for various research applications.
Molecular Characteristics
Laboratory analysis reveals several key structural features:
- Amino acid sequence: Tyr-hGH(177-191)
- Molecular formula: C78H123N23O23S2
- Stability profile: Requires specific storage conditions
- Solubility: Water-soluble with pH considerations
Research teams have documented that AOD 9604 peptide demonstrates stability under controlled laboratory conditions, making it suitable for extended research protocols. The peptide's structure allows for various formulation approaches, including oral delivery systems.
Research Applications in Laboratory Settings
Scientific investigations have explored multiple aspects of peptide aod 9604 functionality. Laboratory studies focus on cellular mechanisms, receptor interactions, and metabolic pathways. Researchers have particularly noted the peptide's unique properties compared to full-length growth hormone.
The comprehensive research on AOD 9604 has revealed interesting cellular interactions that continue to drive scientific inquiry. These studies form the foundation for understanding optimal research protocols and experimental design.
AOD 9604 Peptide Oral Delivery Systems and Bioavailability
The development of aod 9604 peptide oral formulations represents a significant advancement in peptide delivery technology. Traditional peptide administration faces challenges related to enzymatic degradation and absorption barriers, making oral delivery systems particularly noteworthy for research applications.
Oral Formulation Advantages
Research into oral peptide delivery has identified several potential benefits:
Enhanced Convenience 📋
- Simplified administration protocols
- Reduced handling complexity
- Improved research compliance
Bioavailability Considerations 🧬
- Protected peptide stability
- Controlled release mechanisms
- Optimized absorption profiles
Laboratory studies examining aod 9604 peptide oral formulations have investigated various encapsulation technologies and protective matrices. These approaches aim to preserve peptide integrity through the digestive environment while maintaining biological activity.
Research Protocol Considerations
When working with oral formulations, research teams must consider several factors:
- Timing protocols – Relationship to feeding schedules
- pH stability – Gastric environment interactions
- Absorption windows – Optimal sampling timeframes
- Storage requirements – Maintaining formulation integrity
The metabolic research applications of oral AOD 9604 have provided valuable insights into peptide behavior under various experimental conditions.
Quality Control in Oral Formulations
Researchers emphasize the importance of rigorous quality control when selecting aod 9604 peptide for sale in oral formulations. Key considerations include:
- Peptide purity verification through analytical testing
- Stability assays under storage conditions
- Dissolution profiles for oral formulations
- Contamination screening for research-grade materials
Pure Tested Peptides maintains comprehensive quality control protocols to ensure research-grade peptides meet stringent laboratory standards.
Research Protocols and Laboratory Applications
Scientific investigation of aod 9604 peptides requires carefully designed protocols that account for the peptide's unique properties and research objectives. Laboratory teams have developed standardized approaches that maximize data quality while ensuring experimental reproducibility.
Standard Research Protocols
Research teams typically follow established protocols when working with what is aod 9604 peptide:
Preparation Phase
- Reconstitution procedures using sterile techniques
- Concentration calculations based on molecular weight
- pH adjustment protocols for optimal stability
- Storage condition verification
Experimental Design
- Control group establishment
- Dosing schedule development
- Sampling timepoint determination
- Data collection methodology
Dosing Considerations in Research
Laboratory studies examining aod 9604 peptide dosage parameters have established several key principles:
| Research Parameter | Typical Range | Considerations |
|---|---|---|
| Concentration | 0.1-10 mg/mL | Solubility limits |
| pH Range | 6.0-7.4 | Stability optimization |
| Storage Temp | 2-8°C | Peptide preservation |
| Reconstitution | Sterile water | Contamination prevention |
Research protocols often incorporate peptide blend approaches to investigate synergistic effects and comparative mechanisms.
Laboratory Safety and Handling
Working with aod 9604 peptide therapy research requires adherence to established safety protocols:
- Personal protective equipment requirements
- Proper waste disposal procedures
- Contamination prevention measures
- Documentation and traceability systems
Research teams emphasize that proper handling ensures both researcher safety and data integrity throughout experimental procedures.
Data Collection and Analysis
Scientific investigation of peptides aod 9604 generates multiple data streams requiring systematic analysis:
Quantitative Measurements
- Concentration-response relationships
- Time-course profiles
- Comparative efficacy data
- Statistical significance testing
Qualitative Observations
- Morphological changes
- Behavioral modifications
- Cellular characteristics
- Tissue responses
The AOD 9604 research applications continue to expand as laboratory techniques become more sophisticated and analytical capabilities improve.
Comparative Analysis and Research Considerations

Understanding what is aod 9604 peptide for men and broader research applications requires examining the peptide within the context of related compounds and alternative approaches. Laboratory studies have conducted extensive comparative analyses to characterize AOD 9604's unique properties.
Peptide Comparison Studies
Research teams have investigated how aod 9604 peptide compares to other growth hormone fragments and related peptides:
Structural Comparisons
- Amino acid sequence variations
- Molecular weight differences
- Stability profile comparisons
- Receptor binding characteristics
Functional Analyses
- Cellular mechanism studies
- Pathway activation profiles
- Duration of activity measurements
- Dose-response relationships
Laboratory investigations have revealed that AOD 9604 vs somatotropin studies provide valuable insights into peptide selectivity and mechanism specificity.
Research Design Considerations
When planning studies with buy aod 9604 peptide, researchers must consider several experimental variables:
Study Duration
- Short-term acute studies (hours to days)
- Medium-term investigations (weeks)
- Long-term stability assessments (months)
Experimental Models
- In vitro cellular systems
- Ex vivo tissue preparations
- Biochemical assay platforms
- Analytical chemistry methods
Quality Sourcing for Research
Selecting appropriate aod 9604 peptide where to buy sources significantly impacts research quality and reproducibility. Key evaluation criteria include:
Analytical Documentation
- Certificate of analysis (COA) completeness
- Purity verification methods
- Identity confirmation testing
- Stability data availability
Supplier Qualifications
- Manufacturing standards compliance
- Quality management systems
- Research support capabilities
- Technical documentation quality
Research-grade peptide sourcing requires careful vendor evaluation to ensure experimental validity and data reliability.
Future Research Directions
The scientific community continues to explore new applications for aod 9604 peptide before and after studies across various research domains:
Emerging Research Areas
- Combination peptide studies
- Novel delivery system development
- Mechanism elucidation projects
- Comparative efficacy investigations
Technological Advances
- Improved analytical methods
- Enhanced formulation approaches
- Advanced cellular models
- Sophisticated measurement techniques
Research teams increasingly recognize that peptide research applications benefit from interdisciplinary collaboration and technological innovation.
Storage, Handling, and Best Practices
Proper management of aod 9604 peptide dosage chart requirements extends beyond simple concentration calculations to encompass comprehensive storage and handling protocols that preserve peptide integrity throughout research investigations.
Optimal Storage Conditions
Laboratory studies have established specific requirements for maintaining aod 9604 peptide dosing materials:
Temperature Control 🌡️
- Lyophilized peptide: -20°C to -80°C
- Reconstituted solutions: 2-8°C
- Working solutions: 4°C maximum
- Avoid freeze-thaw cycles
Environmental Factors
- Light protection requirements
- Humidity control considerations
- Container material compatibility
- Atmospheric exposure limitations
Research teams report that proper storage significantly impacts experimental reproducibility and peptide stability over time.
Reconstitution Protocols
Working with aod 9604 peptide what is it requires standardized reconstitution procedures:
Step-by-Step Process
- Allow peptide vial to reach room temperature
- Add sterile water slowly down vial wall
- Gentle swirling motion (avoid vigorous mixing)
- Complete dissolution verification
- Immediate refrigeration of solution
Critical Considerations
- Sterile technique maintenance throughout
- Accurate volume measurements
- pH monitoring if required
- Documentation of preparation details
Research Documentation Requirements
Scientific investigations using aod 9604 peptide what is aod 9604 must maintain comprehensive records:
Batch Information
- Supplier identification
- Lot number documentation
- Expiration date tracking
- Certificate of analysis filing
Preparation Records
- Reconstitution date and time
- Solvent type and volume used
- Final concentration calculations
- Storage condition verification
The best practices for peptide storage emphasize systematic documentation as essential for research integrity and regulatory compliance.
Quality Assurance Measures
Laboratory teams implement multiple quality control checkpoints when working with research peptides:
Incoming Material Verification
- Visual inspection for physical damage
- Weight verification against specifications
- Documentation review and filing
- Storage condition establishment
Ongoing Monitoring
- Regular temperature log maintenance
- Solution clarity assessments
- Contamination screening protocols
- Stability indicator tracking
Research facilities often establish comprehensive peptide libraries with standardized management protocols to ensure consistent research quality across multiple projects.
Conclusion
The comprehensive examination of aod 9604 peptide oral formulations reveals a sophisticated landscape of research opportunities and technical considerations that continue to evolve with advancing scientific understanding. Laboratory investigations have established that this synthetic growth hormone fragment offers unique properties for research applications, particularly when formulated for oral delivery systems.
Research teams working with AOD 9604 must navigate multiple technical considerations, from proper storage and handling protocols to experimental design optimization. The peptide's stability characteristics, bioavailability profiles, and cellular mechanisms provide rich opportunities for scientific investigation across various research domains.
Key success factors for AOD 9604 research include rigorous quality control measures, standardized protocols, and careful attention to storage requirements. The selection of research-grade materials from qualified suppliers significantly impacts experimental outcomes and data reliability.
Next Steps for Researchers
Immediate Actions:
- Evaluate current research objectives and protocol requirements
- Assess laboratory capabilities and equipment needs
- Review supplier qualifications and quality documentation
- Establish proper storage and handling procedures
Long-term Planning:
- Develop comprehensive experimental designs
- Consider combination studies with related peptides
- Plan for advanced analytical capabilities
- Build collaborative research networks
For researchers seeking high-quality aod 9604 peptide for sale, Pure Tested Peptides provides research-grade materials with comprehensive analytical documentation and technical support to advance scientific discovery.
The future of AOD 9604 research appears promising, with ongoing technological advances in delivery systems, analytical methods, and experimental approaches continuing to expand the boundaries of scientific understanding in this fascinating area of peptide research.
SEO Meta Information:
Meta Title: AOD 9604 Peptide Oral: Research Guide & Applications 2026
Meta Description: Comprehensive guide to AOD 9604 peptide oral formulations, research protocols, dosing considerations, and laboratory applications for scientific investigation.
