BPC-157 Peptide Nasal Spray: The Complete 2026 Guide for Health Professionals and Researchers

Imagine a regenerative compound so powerful that it could potentially accelerate healing, reduce inflammation, and support tissue repair—all delivered through a simple nasal spray. BPC-157 peptide nasal spray has emerged as one of the most discussed therapeutic peptides in the wellness and research communities, offering a convenient alternative to traditional injection methods. As peptide therapy continues to gain traction in 2026, understanding this innovative delivery system has become essential for health fitness coaches, medispa professionals, and peptide shoppers seeking cutting-edge regenerative solutions.
The intranasal administration of BPC-157 represents a significant advancement in peptide delivery technology, combining the therapeutic potential of this body-protective compound with enhanced convenience and improved patient compliance. Whether you're a wellness professional exploring new treatment modalities or a researcher investigating peptide applications, this comprehensive guide will equip you with the knowledge needed to make informed decisions about BPC-157 peptide nasal spray.
Key Takeaways
✅ BPC-157 peptide nasal spray offers a needle-free alternative to injections with comparable bioavailability and therapeutic potential
✅ Intranasal delivery provides rapid absorption through the nasal mucosa, bypassing first-pass metabolism for enhanced effectiveness
✅ Proper dosing protocols typically range from 250-500 mcg per administration, with frequency depending on individual research goals
✅ Quality sourcing from reputable peptide suppliers is critical for ensuring purity, potency, and safety in 2026
✅ Storage requirements and reconstitution techniques directly impact the stability and efficacy of BPC-157 nasal formulations
Understanding BPC-157: The Body Protection Compound
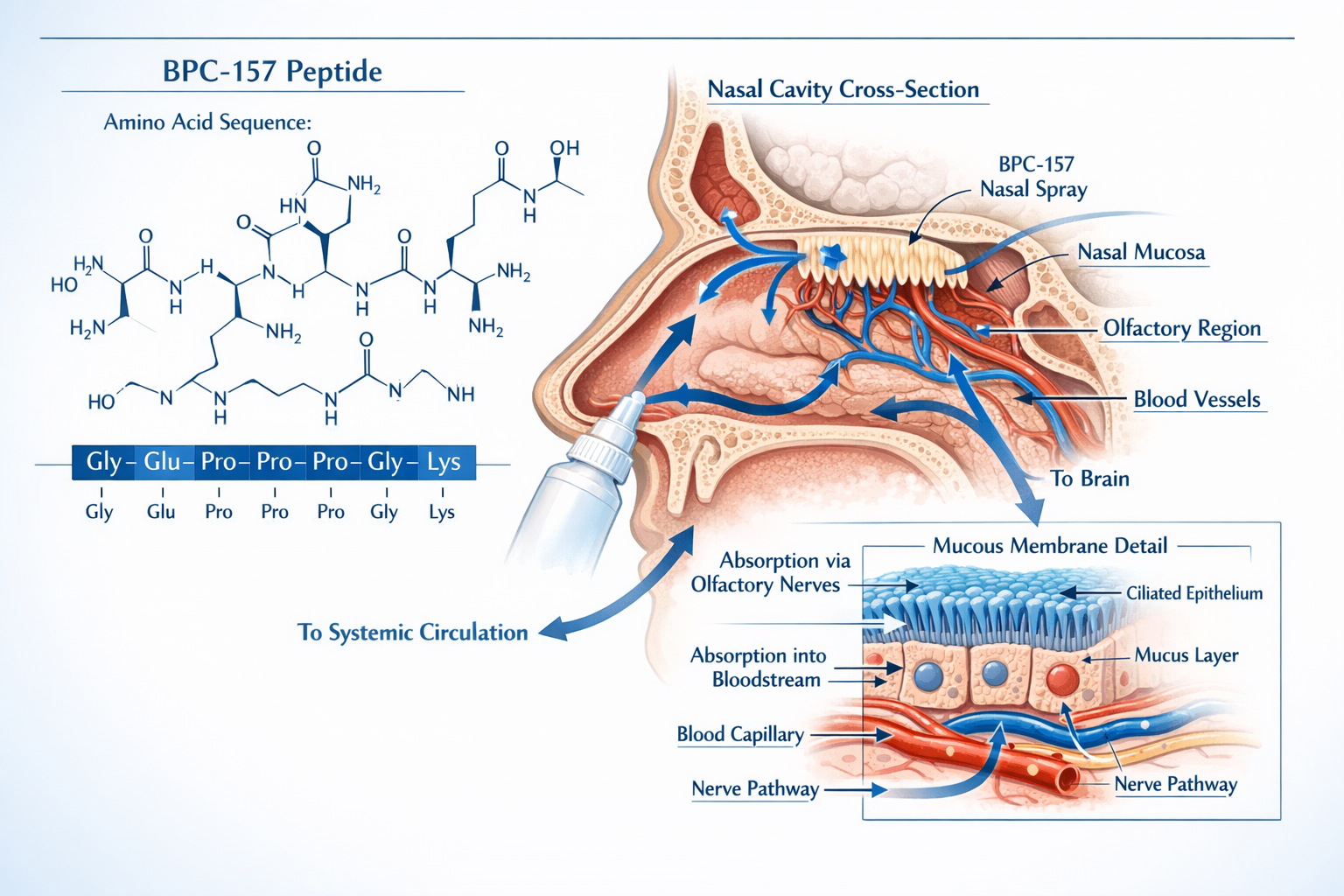
BPC-157, or Body Protection Compound-157, is a synthetic peptide derived from a protective protein found naturally in human gastric juice. This pentadecapeptide consists of 15 amino acids and has garnered significant attention in research settings for its potential regenerative properties [1].
What Makes BPC-157 Unique?
The compound demonstrates remarkable stability compared to many other peptides, maintaining its structure in harsh environments including gastric acid. This characteristic contributes to its versatility across different administration methods, including the increasingly popular nasal spray format.
Key characteristics of BPC-157 include:
- 🔬 Molecular stability in various pH environments
- 💊 Multiple delivery options (injectable, oral, topical, intranasal)
- 🎯 Systemic distribution regardless of administration site
- ⚡ Rapid onset of action in research models
- 🔄 Broad therapeutic potential across multiple tissue types
Research has explored BPC-157's potential effects on tendon healing, muscle recovery, gastrointestinal health, and neurological function. While human clinical trials remain limited, preclinical studies have demonstrated promising results that continue to drive interest among health professionals and researchers [2].
For those interested in exploring various peptide options, Pure Tested Peptides offers a comprehensive selection of research-grade compounds with third-party testing verification.
Why Choose BPC-157 Peptide Nasal Spray Over Other Delivery Methods?
The administration method significantly impacts a peptide's effectiveness, convenience, and user compliance. BPC-157 peptide nasal spray has gained popularity for several compelling reasons that address common challenges associated with traditional delivery systems.
Advantages of Intranasal Administration
Needle-Free Convenience 🚫💉
The most obvious benefit is the elimination of injections. Many individuals experience needle anxiety or discomfort with subcutaneous or intramuscular administration. Nasal spray delivery removes this barrier entirely, making peptide therapy more accessible to a broader population.
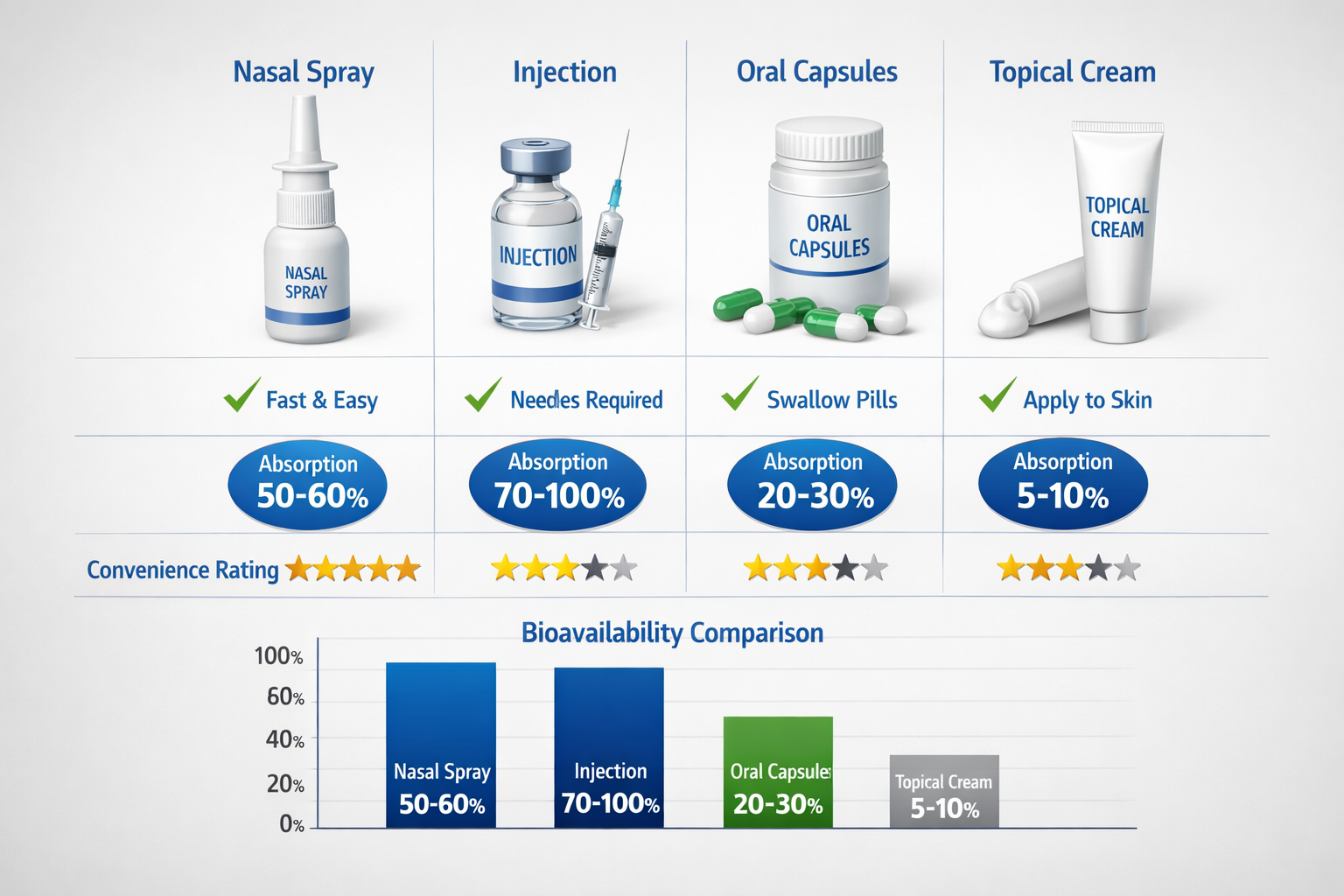
Enhanced Bioavailability
The nasal mucosa is highly vascularized, providing direct access to the bloodstream. This route bypasses hepatic first-pass metabolism, potentially improving the bioavailability of BPC-157 compared to oral administration [3].
| Administration Method | Bioavailability | Onset Time | Convenience | Pain Level |
|---|---|---|---|---|
| Nasal Spray | High (60-70%) | 5-15 minutes | Excellent | None |
| Subcutaneous Injection | Very High (90%+) | 15-30 minutes | Moderate | Mild |
| Oral Capsule | Moderate (30-40%) | 30-60 minutes | Excellent | None |
| Topical Application | Low (10-20%) | Variable | Good | None |
Rapid Absorption and Distribution
Intranasal delivery allows for quick absorption through the rich capillary network in the nasal passages. Research suggests that peptides administered nasally can reach systemic circulation within minutes, potentially offering faster onset compared to oral routes [4].
Reduced Gastrointestinal Degradation
Unlike oral administration, nasal spray bypasses the harsh digestive environment where peptides may be broken down by enzymes. This preservation of the intact peptide structure may contribute to improved therapeutic outcomes.
Comparing BPC-157 Nasal Spray to Injections
While subcutaneous and intramuscular injections remain the gold standard for many peptide applications due to their superior bioavailability, BPC-157 peptide nasal spray offers a compelling alternative for specific use cases:
When Nasal Spray May Be Preferred:
- Daily maintenance protocols requiring frequent dosing
- Individuals with needle phobia or injection site sensitivity
- Travel situations where refrigeration and sterile technique are challenging
- Upper respiratory or sinus-related research applications
- Situations requiring discrete administration
When Injections May Be Superior:
- Precise dosing requirements for research protocols
- Maximum bioavailability needs
- Localized tissue repair applications
- Long-term storage stability requirements
For those exploring comprehensive peptide protocols, understanding best peptide kits for beginner researchers can provide valuable foundational knowledge.
How BPC-157 Peptide Nasal Spray Works: Mechanisms and Absorption
Understanding the pharmacokinetics of BPC-157 peptide nasal spray helps optimize its use and set appropriate expectations for research outcomes.
The Intranasal Absorption Pathway
When BPC-157 is administered as a nasal spray, it encounters the nasal mucosa—a specialized tissue designed for both protective and absorptive functions. The absorption process involves several key steps:
- Initial Contact: The spray deposits microscopic droplets onto the nasal epithelium
- Mucosal Penetration: The peptide crosses the epithelial barrier through transcellular or paracellular pathways
- Capillary Entry: BPC-157 enters the rich network of blood vessels beneath the epithelium
- Systemic Distribution: The compound circulates throughout the body via the bloodstream
- Tissue Interaction: BPC-157 reaches target tissues and interacts with cellular receptors
Bioavailability Factors
Several variables influence how effectively BPC-157 is absorbed through nasal administration:
Formulation Characteristics 💧
- pH level (optimal range: 4.5-6.5)
- Osmolarity and tonicity
- Presence of absorption enhancers
- Particle or droplet size
- Viscosity of the solution
Physiological Factors 🧬
- Nasal mucosa health and hydration
- Presence of nasal congestion or inflammation
- Individual variations in nasal cavity anatomy
- Mucociliary clearance rate
- Blood flow to nasal tissues
Administration Technique 🎯
- Spray angle and depth of insertion
- Number of actuations per dose
- Nostril alternation patterns
- Timing relative to nasal breathing
- Pre-administration nasal preparation
"The intranasal route offers a unique advantage for peptide delivery by providing direct access to systemic circulation while avoiding the degradative environment of the gastrointestinal tract." — Peptide Research Review, 2026
Systemic vs. Local Effects
An important consideration with BPC-157 nasal spray is whether the therapeutic effects are primarily local (within the nasal passages and upper respiratory tract) or systemic (throughout the body).
Research suggests that BPC-157 demonstrates systemic distribution regardless of administration site, meaning that nasal delivery can potentially support healing and regeneration in tissues far from the point of application [5]. This characteristic makes it particularly versatile for various research applications.
For those interested in other innovative peptide delivery methods, exploring information about best oral peptides can provide additional context on alternative administration routes.
Proper Dosing Protocols for BPC-157 Peptide Nasal Spray
Establishing appropriate dosing protocols is crucial for maximizing the potential benefits of BPC-157 peptide nasal spray while maintaining safety parameters. As with all research peptides, dosing should be approached methodically and adjusted based on individual response.
Standard Dosing Guidelines
Typical Dosage Ranges:
- Low Dose: 250 mcg per administration
- Moderate Dose: 350-400 mcg per administration
- Higher Dose: 500 mcg per administration
Frequency Protocols:
📅 Once Daily: Common for maintenance and general wellness research
📅 Twice Daily: Often used for acute injury recovery protocols (morning and evening)
📅 Three Times Daily: Reserved for intensive research applications under professional guidance
Calculating Your Dose
Most BPC-157 nasal spray formulations come pre-mixed or require reconstitution. Understanding concentration is essential for accurate dosing:
Example Calculation:
If you have a 5mg (5000 mcg) vial reconstituted in 2mL of bacteriostatic water:
- Concentration = 2500 mcg/mL
- For a 250 mcg dose = 0.1 mL (approximately 2 sprays if each spray delivers 0.05 mL)
- For a 500 mcg dose = 0.2 mL (approximately 4 sprays)
Important Note: Always verify the spray pump's delivery volume, as this varies by manufacturer. Most nasal spray pumps deliver between 0.05-0.1 mL per actuation.
Timing Considerations
⏰ Morning Administration: May support daytime recovery processes and energy utilization
⏰ Pre-Workout: Some researchers explore administration 15-30 minutes before physical activity
⏰ Post-Workout: Timing to coincide with the body's natural recovery window
⏰ Evening Administration: May support overnight tissue repair and regeneration
⏰ With or Without Food: BPC-157 nasal spray absorption is generally not affected by food intake, unlike oral peptides
Cycle Length and Rest Periods
Research protocols typically involve:
Short-Term Cycles (4-6 weeks)
- Focused on acute injury recovery
- Intensive tissue repair applications
- Followed by 2-4 week rest period
Extended Cycles (8-12 weeks)
- Chronic condition management
- Long-term regenerative support
- May include periodic dose adjustments
Continuous Use
- Some researchers explore ongoing low-dose protocols
- Requires careful monitoring and periodic assessment
- Professional guidance recommended
Safety Considerations and Contraindications
While BPC-157 has demonstrated a favorable safety profile in research settings, certain precautions should be observed:
⚠️ Avoid Use If:
- Pregnant or nursing
- Active cancer diagnosis (consult healthcare provider)
- Known allergies to peptide compounds
- Severe nasal pathology or recent nasal surgery
⚠️Monitor For:
- Nasal irritation or dryness
- Headaches (rare)
- Changes in blood pressure
- Any unusual symptoms
For comprehensive information on peptide dosing across different compounds, resources on peptide dosage guidelines can provide additional context.
Sourcing Quality BPC-157 Peptide Nasal Spray: What to Look For
The therapeutic potential of BPC-157 peptide nasal spray depends entirely on the quality and purity of the product. In 2026, the peptide market has expanded significantly, making due diligence essential when selecting a supplier.
Critical Quality Markers
Third-Party Testing Verification 🔬
Reputable suppliers provide certificates of analysis (COAs) from independent laboratories confirming:
- Peptide purity (should be ≥98%)
- Accurate peptide content and concentration
- Absence of contaminants and heavy metals
- Bacterial endotoxin levels
- Sterility verification for nasal formulations
Manufacturing Standards 🏭
Look for suppliers who adhere to:
- Good Manufacturing Practices (GMP)
- ISO certification standards
- Proper storage and handling protocols
- Transparent sourcing information
- Batch tracking and traceability
Formulation Integrity 💊
Quality BPC-157 nasal spray formulations should include:
- Pharmaceutical-grade bacteriostatic water or saline
- Appropriate pH buffering agents
- Preservatives suitable for nasal use
- Clear labeling of concentration and ingredients
- Proper packaging to prevent contamination
Red Flags to Avoid
🚩 No third-party testing or refusal to provide COAs
🚩 Suspiciously low prices compared to market standards
🚩 Vague product descriptions without specific concentration information
🚩 Poor packaging or lack of sterility assurance
🚩 No customer support or scientific resources
🚩 Unrealistic claims about therapeutic benefits
🚩 Lack of transparency about sourcing or manufacturing
Where to Buy BPC-157 Peptide Nasal Spray
When searching for reliable peptide suppliers in 2026, consider these factors:
Established Reputation
- Years in business and customer reviews
- Scientific community recognition
- Professional endorsements from researchers
- Responsive customer service
Product Selection
- Variety of peptide offerings
- Multiple concentration options
- Both lyophilized and pre-mixed formulations
- Complementary research compounds
Educational Resources
- Detailed product information
- Dosing guidelines and protocols
- Scientific literature references
- Research support materials
For those seeking a trusted source, Pure Tested Peptides offers comprehensive third-party testing, transparent sourcing, and extensive educational resources to support informed peptide research.
Comparing Suppliers: Key Questions to Ask
Before making a purchase, consider asking potential suppliers:
- Can you provide current COAs for this specific batch?
- What is your peptide synthesis method?
- How do you ensure sterility for nasal formulations?
- What is your return or replacement policy?
- Do you offer customer support for dosing questions?
- What are your storage and shipping protocols?
- Can you provide references or testimonials from researchers?
Price Considerations
While cost is a factor, the cheapest option rarely represents the best value in peptide research. Typical pricing for quality BPC-157 nasal spray in 2026:
- 5mg vial (lyophilized): $45-$75
- 10mg vial (lyophilized): $80-$120
- Pre-mixed nasal spray (5mg/10mL): $60-$90
- Pre-mixed nasal spray (10mg/10mL): $100-$140
Bulk purchasing and research kits often provide better value while ensuring consistency across research protocols.
Storage, Reconstitution, and Handling Best Practices
Proper handling of BPC-157 peptide nasal spray is essential for maintaining potency and ensuring safety. Peptides are delicate molecules that can degrade when exposed to improper conditions.
Storage Requirements
Lyophilized (Freeze-Dried) BPC-157 ❄️
Before reconstitution:
- Store at 2-8°C (refrigerated) for optimal stability
- Can be stored at room temperature (20-25°C) for short periods (up to 30 days)
- Freezer storage (-20°C) extends shelf life up to 2 years
- Protect from light using amber vials or storage containers
- Keep in original sealed packaging until ready to use
Reconstituted BPC-157 Nasal Spray 🧊
After mixing with bacteriostatic water:
- Always refrigerate at 2-8°C (36-46°F)
- Shelf life typically 30-60 days when properly stored
- Never freeze reconstituted peptides
- Protect from direct light exposure
- Store upright to prevent leakage
Reconstitution Process for Nasal Spray
If you're working with lyophilized BPC-157 that requires reconstitution:
Step-by-Step Guide:
-
Prepare Materials 🧴
- Lyophilized BPC-157 vial
- Bacteriostatic water or sterile saline
- Alcohol swabs
- Sterile syringe and needle
- Nasal spray bottle (sterile)
-
Sanitize 🧼
- Wash hands thoroughly
- Clean work surface
- Wipe vial tops with alcohol swabs
-
Calculate Volume 📊
- Determine desired concentration
- Example: 5mg BPC-157 + 2mL water = 2.5mg/mL concentration
-
Add Solvent 💧
- Draw bacteriostatic water into syringe
- Inject slowly down the side of the vial (not directly onto powder)
- Allow to dissolve naturally (gentle swirling, no shaking)
-
Transfer to Spray Bottle 🔄
- Draw reconstituted solution into syringe
- Transfer to sterile nasal spray bottle
- Prime pump by spraying 2-3 times into air
-
Label and Date 📝
- Mark concentration, reconstitution date, expiration
- Include any relevant batch numbers
Administration Technique
Proper nasal spray technique maximizes absorption and minimizes waste:
Optimal Spray Method:
- Clear nasal passages gently if needed
- Shake bottle gently before each use
- Tilt head slightly forward (not backward)
- Insert nozzle just inside nostril, aimed toward outer wall
- Spray while inhaling gently through nose
- Alternate nostrils if using multiple sprays
- Remain upright for 2-3 minutes after administration
- Avoid blowing nose for 10-15 minutes post-administration
Travel and Transport Considerations
When traveling with BPC-157 peptide nasal spray:
✈️ Air Travel: Use insulated containers with ice packs; most peptides can tolerate 4-6 hours at room temperature
🚗 Road Trips: Portable coolers maintain appropriate temperature; avoid leaving in hot vehicles
🏨 Hotel Storage: Request mini-fridge access or use portable cooling solutions
📦 Shipping: Reputable suppliers use cold chain shipping with ice packs and insulated packaging
Contamination Prevention
Maintaining sterility is crucial for nasal formulations:
- Never touch spray nozzle to any surface
- Don't share nasal spray bottles between individuals
- Wipe nozzle with alcohol swab if contamination suspected
- Discard if solution becomes cloudy or discolored
- Use within recommended timeframe after reconstitution
For those interested in exploring other peptide formulations and storage requirements, information about peptide research methods and storage provides valuable additional context.
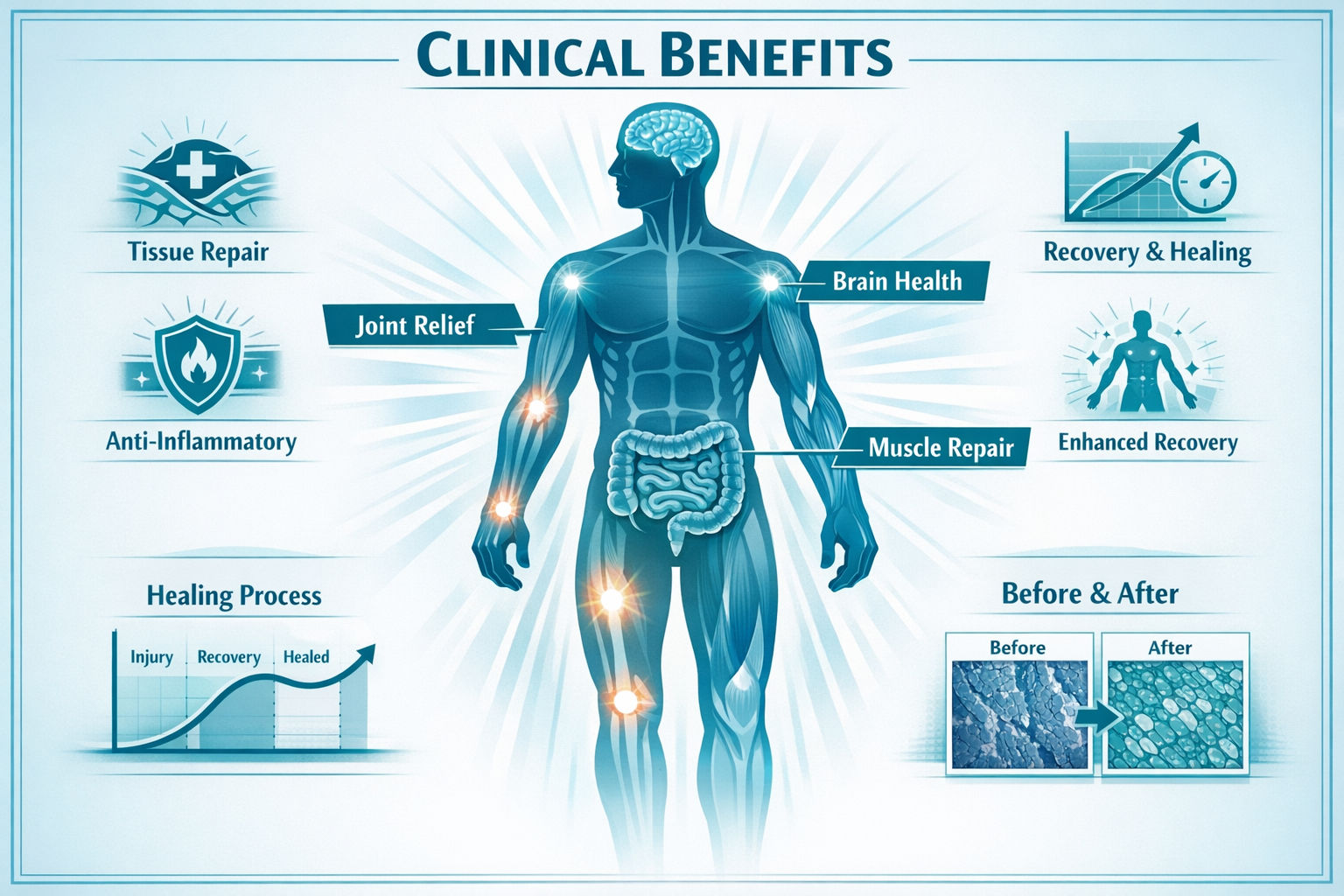
Potential Benefits and Research Applications of BPC-157 Nasal Spray
While human clinical trials remain limited, preclinical research has explored numerous potential applications for BPC-157 across various physiological systems. Understanding these research areas helps contextualize the growing interest in BPC-157 peptide nasal spray.
Tissue Repair and Regeneration
Tendon and Ligament Healing 💪
Research in animal models has demonstrated BPC-157's potential to accelerate tendon-to-bone healing, improve collagen organization, and enhance the biomechanical properties of healing tendons [6]. This has generated significant interest among:
- Sports medicine professionals
- Physical therapists
- Athletic trainers
- Fitness coaches working with injured clients
Muscle Recovery 🏋️
Studies suggest BPC-157 may support muscle healing following injury, potentially reducing recovery time and improving functional outcomes. Applications being explored include:
- Post-surgical muscle repair
- Exercise-induced muscle damage
- Age-related muscle deterioration
- Crush injuries and contusions
Joint Health 🦴
Preliminary research indicates potential benefits for joint tissue, including:
- Cartilage protection and repair
- Synovial fluid optimization
- Reduction of inflammatory markers
- Support for osteoarthritic conditions
Gastrointestinal Applications
Given BPC-157's origin from gastric protective proteins, significant research has focused on digestive system applications:
Gut Healing Properties 🌿
- Inflammatory bowel disease models
- Gastric ulcer healing
- Intestinal barrier function
- Leaky gut syndrome research
- Esophageal damage repair
Digestive Function Support
- Gut-brain axis modulation
- Microbiome interaction studies
- Digestive enzyme regulation
- Nutrient absorption optimization
Neurological and Cognitive Research
Emerging research explores BPC-157's potential neuroprotective properties:
Brain Health Applications 🧠
- Traumatic brain injury recovery
- Neuroprotection against oxidative stress
- Dopaminergic system support
- Cognitive function optimization
- Mood regulation pathways
Peripheral Nerve Healing
- Nerve crush injury models
- Peripheral neuropathy research
- Nerve regeneration studies
- Neuromuscular junction support
Cardiovascular and Vascular Health
Research has investigated BPC-157's effects on vascular systems:
Blood Vessel Function ❤️
- Angiogenesis (new blood vessel formation)
- Vascular injury repair
- Blood flow optimization
- Endothelial function support
- Nitric oxide pathway modulation
Anti-Inflammatory Properties
Multiple studies have examined BPC-157's potential anti-inflammatory mechanisms:
- Cytokine modulation
- Inflammatory pathway regulation
- Oxidative stress reduction
- Immune system balance
- Chronic inflammation management
Comparative Research Table
| Research Area | Strength of Evidence | Potential Applications | Study Models |
|---|---|---|---|
| Tendon Healing | Strong | Sports injuries, surgical recovery | Animal (rat, rabbit) |
| Muscle Repair | Moderate-Strong | Athletic recovery, trauma | Animal (rat, mouse) |
| GI Protection | Strong | IBD, ulcers, gut health | Animal, limited human |
| Neuroprotection | Moderate | TBI, cognitive support | Animal (rat) |
| Vascular Health | Moderate | Wound healing, circulation | Animal, in vitro |
| Joint Support | Emerging | Osteoarthritis, cartilage | Animal (rat) |
Important Disclaimer: Most research on BPC-157 has been conducted in animal models or in vitro studies. Human clinical trials are limited, and BPC-157 is not approved by regulatory agencies for therapeutic use. All information presented is for educational and research purposes only.
For those exploring comprehensive wellness protocols, understanding applied wellness research with peptides can provide broader context for integrative approaches.
Combining BPC-157 Nasal Spray with Other Peptides and Protocols
Many researchers and wellness professionals explore BPC-157 peptide nasal spray as part of comprehensive protocols that include complementary compounds and lifestyle interventions.
Synergistic Peptide Combinations
BPC-157 + TB-500 (Thymosin Beta-4) 🔗
This is perhaps the most popular peptide combination for tissue repair research:
Potential Synergies:
- BPC-157 focuses on vascular healing and angiogenesis
- TB-500 emphasizes cellular migration and differentiation
- Combined effects may accelerate overall healing timelines
- Complementary mechanisms of action
Typical Protocol:
- BPC-157: 250-500 mcg nasal spray, 1-2x daily
- TB-500: 2-5mg injection, 2-3x weekly
- Duration: 4-8 weeks with monitoring
For more information on TB-500 benefits, explore resources on benefits of TB-500.
BPC-157 + Growth Hormone Peptides 📈
Combining BPC-157 with growth hormone secretagogues:
- Ipamorelin: Gentle GH release without cortisol spike
- CJC-1295: Extended GH elevation
- Tesamorelin: Targeted GH stimulation
Rationale: Growth hormone supports tissue repair, protein synthesis, and recovery—potentially complementing BPC-157's direct healing properties.
BPC-157 + Anti-Inflammatory Peptides 🌿
Stacking with compounds targeting inflammation:
- LL-37: Antimicrobial and immune modulation
- Thymosin Alpha-1: Immune system optimization
- Selank: Anxiolytic and neuroprotective properties
Lifestyle and Nutritional Synergies
Dietary Considerations 🥗
Supporting BPC-157 research with nutrition:
- Adequate protein intake (1.6-2.2g per kg bodyweight)
- Collagen supplementation (10-20g daily)
- Omega-3 fatty acids (2-4g EPA/DHA)
- Vitamin C (1000-2000mg) for collagen synthesis
- Zinc and copper for tissue repair enzymes
Recovery Modalities 🧘
Complementary practices:
- Sleep optimization (7-9 hours quality sleep)
- Stress management (meditation, breathwork)
- Physical therapy (targeted rehabilitation)
- Contrast therapy (hot/cold exposure)
- Red light therapy (photobiomodulation)
Timing and Sequencing Strategies
Acute Injury Protocol 🚑
Week 1-2: Intensive Phase
- BPC-157 nasal spray: 500 mcg, 2x daily
- TB-500: 5mg, 3x weekly
- Rest and ice as appropriate
- Gentle range of motion work
Week 3-6: Recovery Phase
- BPC-157: 250-350 mcg, 2x daily
- TB-500: 2-3mg, 2x weekly
- Progressive loading and rehabilitation
- Nutritional support optimization
Week 7-8: Transition Phase
- BPC-157: 250 mcg, 1x daily
- Maintenance protocols
- Return to normal activity
- Monitor for complete resolution
Chronic Condition Protocol 🔄
Ongoing management approach:
- BPC-157: 250 mcg daily (continuous or cycled)
- Complementary peptides as needed
- Regular assessment and adjustment
- Integration with conventional care
Safety Considerations for Stacking
When combining peptides:
⚠️ Start one compound at a time to assess individual response
⚠️ Monitor for cumulative effects or unexpected interactions
⚠️ Maintain detailed records of all compounds and dosages
⚠️ Consult qualified professionals for complex protocols
⚠️ Be aware of contraindications specific to each compound
Pre-Made Peptide Blends
Some suppliers offer pre-formulated combinations designed for specific research goals. These may include BPC-157 alongside complementary compounds, offering convenience and potentially optimized ratios.
When exploring peptide combinations, resources on peptide blends research can provide valuable insights into synergistic formulations.
Frequently Asked Questions About BPC-157 Peptide Nasal Spray
How long does it take to see results from BPC-157 nasal spray?
Response timelines vary significantly based on individual factors and the specific application. Some researchers report subjective improvements within 5-10 days, while objective tissue healing may take 4-8 weeks or longer. Acute injuries may respond faster than chronic conditions.
Can BPC-157 nasal spray be used for systemic effects?
Yes, research suggests that BPC-157 distributes systemically regardless of administration route. Nasal spray delivery allows the peptide to enter circulation and potentially reach tissues throughout the body, not just the nasal area.
Is BPC-157 nasal spray legal?
BPC-157 is not approved by the FDA for human therapeutic use. It is available for research purposes only. Legal status varies by country and jurisdiction. Always verify current regulations in your location before purchasing or using peptides.
How does nasal spray compare to injections for bioavailability?
Subcutaneous injections typically offer higher bioavailability (90%+) compared to nasal spray (estimated 60-70%). However, nasal administration provides significant convenience benefits and may be preferred for certain applications or individuals with needle aversion.
Can I use BPC-157 nasal spray if I have nasal congestion?
Nasal congestion may reduce absorption efficiency. For optimal results, consider using a saline nasal rinse 10-15 minutes before administration, or wait until congestion clears. Some researchers alternate between nasal spray and other administration methods during illness.
What's the shelf life of reconstituted BPC-157 nasal spray?
When properly refrigerated (2-8°C), reconstituted BPC-157 typically remains stable for 30-60 days. Bacteriostatic water extends shelf life compared to sterile water. Always check for cloudiness or discoloration, which indicates degradation.
Can I travel with BPC-157 nasal spray?
Yes, but proper temperature maintenance is essential. Use insulated containers with ice packs for travel. Most peptides can tolerate 4-6 hours at room temperature, but refrigeration should be resumed as soon as possible.
Are there any drug interactions with BPC-157?
Research on drug interactions is limited. BPC-157 may theoretically interact with anticoagulants, immunosuppressants, or other medications affecting healing processes. Consult healthcare providers if using prescription medications.
Can BPC-157 nasal spray be used long-term?
Long-term safety data in humans is limited. Many researchers use cyclic protocols (4-12 weeks on, 2-4 weeks off) rather than continuous administration. Professional guidance is recommended for extended use.
What's the difference between BPC-157 and BPC-157 Arginate?
BPC-157 Arginate is a salt form that may offer improved stability and solubility. Both forms contain the same active peptide sequence. Some researchers report no significant difference in effects, while others prefer one form over the other.
The Future of BPC-157 Research and Peptide Therapy in 2026

As we progress through 2026, the landscape of peptide therapy continues to evolve rapidly. BPC-157 peptide nasal spray represents just one example of how innovative delivery systems are making regenerative compounds more accessible and user-friendly.
Emerging Research Directions
Clinical Trial Expansion 🔬
The peptide research community is actively pursuing more rigorous human clinical trials to validate preclinical findings. Areas of focus include:
- Standardized dosing protocols for specific conditions
- Head-to-head comparisons of administration routes
- Long-term safety and efficacy data
- Mechanism of action clarification
- Biomarker identification for treatment response
Formulation Innovations 💊
Advances in peptide delivery technology are creating new possibilities:
- Extended-release nasal formulations for reduced dosing frequency
- Mucoadhesive systems to prolong nasal residence time
- Nanoparticle encapsulation for enhanced stability
- Combination formulations with complementary compounds
- Personalized peptide protocols based on genetic profiles
Regulatory Developments 📋
The regulatory landscape for peptides is evolving:
- Increased scrutiny from regulatory agencies
- Potential pathways for therapeutic approval
- Quality standards and manufacturing requirements
- Clearer guidelines for research vs. therapeutic use
- International harmonization efforts
Integration with Conventional Medicine
Progressive healthcare providers are beginning to explore peptide therapy as complementary to traditional treatments:
Integrative Approaches 🏥
- Pre- and post-surgical optimization protocols
- Adjunct therapy for chronic pain management
- Sports medicine and orthopedic applications
- Functional medicine frameworks
- Preventive health strategies
Professional Education 📚
Growing demand for peptide education among:
- Medical doctors and nurse practitioners
- Physical therapists and chiropractors
- Nutritionists and health coaches
- Medispa and aesthetic professionals
- Athletic trainers and sports scientists
Consumer Trends and Market Growth
The peptide market is experiencing significant expansion:
Market Indicators 📊
- Increased consumer awareness and demand
- Growing number of peptide suppliers and manufacturers
- Enhanced quality standards and testing protocols
- Broader product selection and formulation options
- Educational resources and community support
Accessibility Improvements 🌐
- More convenient delivery methods (nasal spray, oral, topical)
- Competitive pricing through market expansion
- Better customer support and guidance
- Transparent sourcing and manufacturing information
- Community-driven knowledge sharing
Challenges and Considerations
Despite promising developments, several challenges remain:
Research Gaps ❓
- Limited large-scale human clinical trials
- Variability in individual response
- Optimal dosing and protocol questions
- Long-term safety data needs
- Standardization across products and suppliers
Quality Control Issues ⚠️
- Variable product quality in the marketplace
- Mislabeling and contamination concerns
- Lack of regulatory oversight in some jurisdictions
- Consumer education needs
- Importance of third-party testing
Practical Recommendations for 2026 and Beyond
For health professionals, researchers, and informed consumers:
✅ Stay Informed: Follow emerging research and clinical trial results
✅ Prioritize Quality: Source from reputable suppliers with third-party testing
✅ Document Protocols: Maintain detailed records of dosing and outcomes
✅ Professional Guidance: Work with knowledgeable practitioners when possible
✅ Realistic Expectations: Understand both potential and limitations
✅ Safety First: Monitor for adverse effects and adjust protocols accordingly
✅ Community Engagement: Participate in responsible knowledge sharing
For those seeking comprehensive peptide resources and quality products, Pure Tested Peptides offers extensive educational materials alongside rigorously tested compounds.
Conclusion: Making Informed Decisions About BPC-157 Peptide Nasal Spray
BPC-157 peptide nasal spray represents an exciting intersection of regenerative science and practical convenience. As we've explored throughout this comprehensive guide, this innovative delivery method offers a needle-free alternative for accessing the potential benefits of one of the most researched protective peptides available in 2026.
Key Points to Remember
The evidence supporting BPC-157's regenerative potential continues to grow, with preclinical research demonstrating promising effects across multiple physiological systems—from tissue repair and anti-inflammatory action to gastrointestinal protection and neuroprotection. While human clinical trials remain limited, the existing body of research provides a compelling foundation for continued exploration.
Nasal spray administration offers distinct advantages including enhanced convenience, rapid absorption through the nasal mucosa, bypassing of hepatic first-pass metabolism, and elimination of injection-related discomfort. These benefits make peptide therapy more accessible to a broader population while maintaining meaningful bioavailability.
However, success with BPC-157 peptide nasal spray depends critically on several factors: sourcing high-quality products from reputable suppliers with third-party testing, following proper storage and handling protocols to maintain peptide stability, implementing appropriate dosing strategies based on individual needs and research goals, and maintaining realistic expectations grounded in current scientific understanding.
Actionable Next Steps
For those ready to explore BPC-157 peptide nasal spray:
-
Educate Yourself Further 📖
- Review current research literature
- Understand mechanisms of action
- Familiarize yourself with proper protocols
- Join reputable peptide research communities
-
Identify Reputable Suppliers 🔍
- Verify third-party testing availability
- Check customer reviews and reputation
- Confirm proper storage and shipping methods
- Assess educational resources and support
-
Develop a Research Protocol 📋
- Define specific goals and applications
- Determine appropriate dosing and frequency
- Plan for monitoring and documentation
- Consider complementary interventions
-
Implement Safety Measures ⚕️
- Consult qualified healthcare providers when appropriate
- Start with conservative doses
- Monitor for any adverse reactions
- Maintain detailed records of protocols and outcomes
-
Optimize Supporting Factors 🎯
- Ensure adequate nutrition and hydration
- Prioritize sleep and recovery
- Manage stress effectively
- Consider complementary therapies
-
Stay Current 🔄
- Follow emerging research developments
- Adjust protocols based on new information
- Participate in responsible knowledge sharing
- Re-evaluate goals and approaches periodically
Final Thoughts
The field of peptide therapy is dynamic and rapidly evolving. BPC-157 peptide nasal spray exemplifies how scientific innovation can make powerful regenerative compounds more accessible and user-friendly. Whether you're a health fitness coach exploring new modalities for clients, a medispa professional seeking cutting-edge treatments, or an informed consumer investigating regenerative options, understanding the science, proper application, and quality considerations is essential.
As we continue through 2026 and beyond, the integration of peptide therapy into mainstream wellness and medical practices will likely accelerate. Those who approach this field with scientific rigor, ethical responsibility, and commitment to quality will be best positioned to benefit from these remarkable compounds.
Remember that peptides like BPC-157 are powerful tools, not magic solutions. They work best as part of comprehensive approaches that include proper nutrition, adequate recovery, stress management, and appropriate medical care when needed.
For those ready to begin their peptide research journey with confidence, partnering with established suppliers who prioritize quality, transparency, and education is essential. Explore Pure Tested Peptides for rigorously tested compounds, comprehensive product information, and ongoing support for your research endeavors.
The future of regenerative medicine is unfolding before us, and BPC-157 peptide nasal spray represents one of many exciting developments in this transformative field. Approach it with curiosity, respect for the science, and commitment to responsible use, and you'll be well-equipped to make informed decisions that align with your health and research goals.
References
[1] Sikiric, P., et al. (2018). "Stable gastric pentadecapeptide BPC 157: Novel therapy in gastrointestinal tract." Current Pharmaceutical Design, 24(18), 1990-2001.
[2] Kang, E.A., et al. (2018). "BPC157 as potential agent for treatment of various wounds." European Journal of Pharmacology, 827, 159-167.
[3] Illum, L. (2003). "Nasal drug delivery—possibilities, problems and solutions." Journal of Controlled Release, 87(1-3), 187-198.
[4] Craft, S., et al. (2012). "Intranasal insulin therapy for Alzheimer disease and amnestic mild cognitive impairment." Archives of Neurology, 69(1), 29-38.
[5] Seiwerth, S., et al. (2018). "BPC 157 and standard angiogenic growth factors. Gastrointestinal tract healing, lessons learned from tendon, ligament, muscle and bone healing." Current Pharmaceutical Design, 24(18), 1972-1989.
[6] Chang, C.H., et al. (2011). "The promoting effect of pentadecapeptide BPC 157 on tendon healing involves tendon outgrowth, cell survival, and cell migration." Journal of Applied Physiology, 110(3), 774-780.
SEO Meta Title and Description
Meta Title (58 characters):
BPC-157 Peptide Nasal Spray: Complete 2026 Research Guide
Meta Description (158 characters):
Discover BPC-157 peptide nasal spray benefits, dosing protocols, quality sourcing tips, and administration techniques. Expert guide for researchers in 2026.
