Cagrilintide 10mg: Comprehensive Research Guide for Health and Wellness Professionals

The landscape of peptide research is evolving rapidly, and one compound capturing significant attention in 2026 is cagrilintide 10mg—a long-acting amylin analogue that represents the cutting edge of metabolic research. For fitness instructors, life coaches, health and wellness practitioners, and medispa professionals seeking to understand emerging peptide therapies, cagrilintide offers a fascinating case study in how pharmaceutical innovation is approaching weight management through novel mechanisms. This comprehensive guide explores everything health professionals need to know about cagrilintide 10mg, from its mechanism of action to dosing protocols and research findings.
Key Takeaways
✅ Cagrilintide 10mg is a long-acting amylin receptor agonist developed by Novo Nordisk for weight management research, representing one of the higher dosing levels in clinical trials
✅ The compound works by activating amylin receptors in the brain, reducing appetite, slowing gastric emptying, and decreasing food intake through a mechanism distinct from GLP-1 receptor agonists
✅ Clinical trials show cagrilintide monotherapy produces approximately 10-11% weight loss, while combination therapy with semaglutide (CagriSema) achieves superior results of approximately 15.1% weight reduction
✅ Dose escalation protocols typically start at 0.3mg-0.6mg and gradually increase to minimize gastrointestinal side effects, which are the most common adverse events
✅ The cagrilintide 10mg formulation is administered via subcutaneous injection using pre-filled pen devices, similar to other injectable weight management medications
What Is Cagrilintide? Understanding the Amylin Analogue
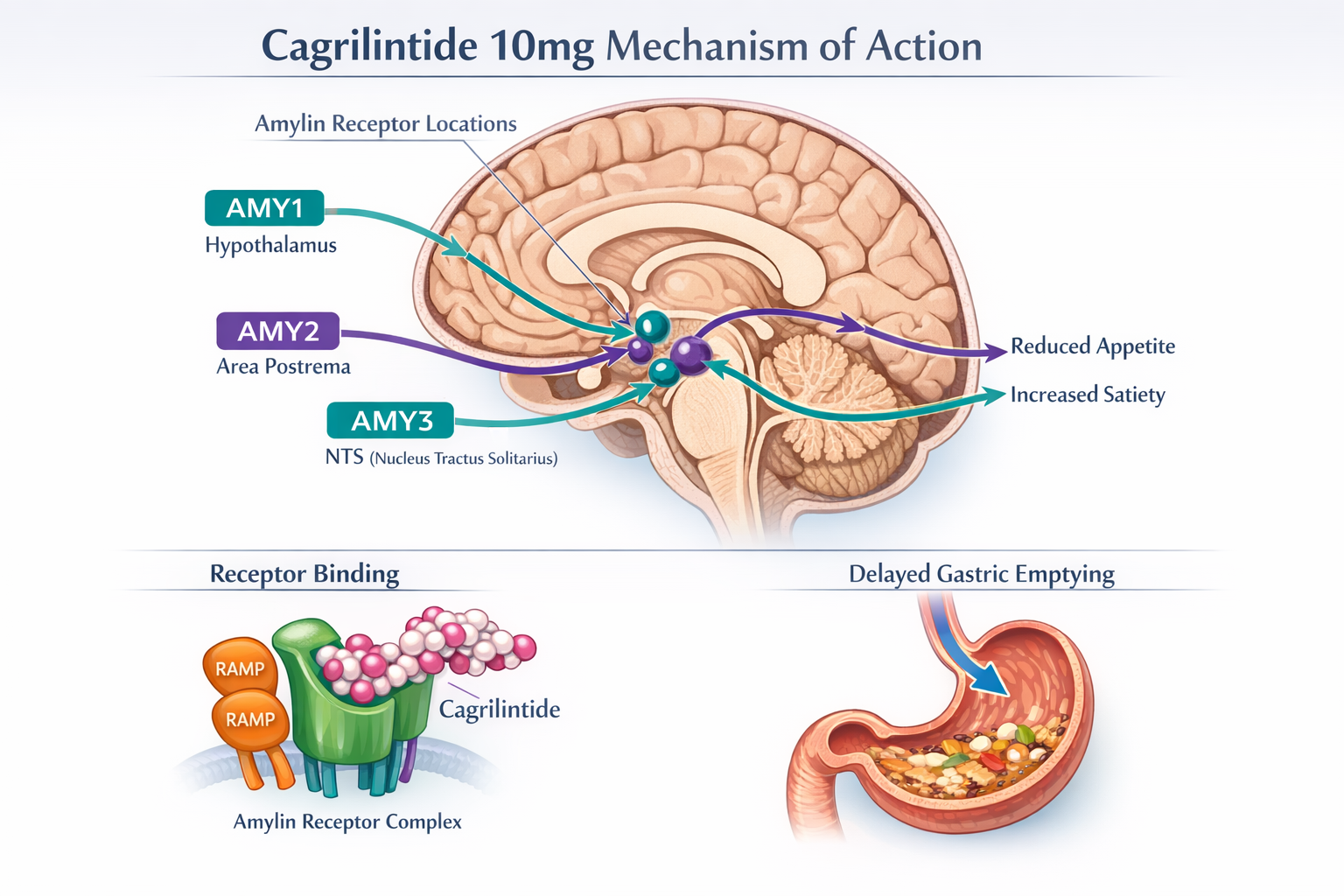
Cagrilintide is a synthetic long-acting amylin receptor agonist that mimics the effects of the naturally occurring hormone amylin. Developed by Novo Nordisk, this peptide compound has emerged as a promising candidate in metabolic research, particularly for weight management applications. Unlike traditional approaches that focus solely on GLP-1 receptor activation, cagrilintide works through the amylin pathway, offering a complementary mechanism that researchers believe could enhance overall efficacy when used in combination therapies.
The Science Behind Amylin Receptor Activation
Amylin is a hormone co-secreted with insulin from pancreatic beta cells in response to food intake. It plays a crucial role in glucose homeostasis and appetite regulation. Cagrilintide binds to amylin receptors—specifically AMY1, AMY2, and AMY3 receptors—which are complexes formed by calcitonin receptors and receptor activity-modifying proteins (RAMPs) [1]. These receptors are predominantly located in areas of the brain involved in appetite control and satiety signaling.
When cagrilintide activates these receptors, it triggers several physiological responses:
- Reduced appetite through central nervous system signaling
- Slowed gastric emptying, which prolongs feelings of fullness
- Decreased food intake at meals
- Modulation of glucose metabolism and insulin sensitivity
For health and wellness professionals working with clients interested in metabolic optimization, understanding this mechanism helps contextualize how cagrilintide differs from other peptide therapies. Those exploring peptide research options should recognize that amylin analogues represent a distinct class with unique properties.
Cagrilintide vs. Other Metabolic Peptides
The peptide landscape includes various compounds targeting different pathways. While GLP-1 receptor agonists like semaglutide and tirzepatide have dominated recent discussions, cagrilintide offers a different approach. The amylin pathway provides complementary effects that, when combined with GLP-1 activation, may produce synergistic benefits beyond either mechanism alone.
This is particularly relevant for professionals advising clients who may be researching various peptide options, including compounds like AOD-9604 peptide or other metabolic modulators. Each peptide class has distinct mechanisms, dosing requirements, and research profiles.
Cagrilintide 10mg Dosage: Clinical Research Protocols
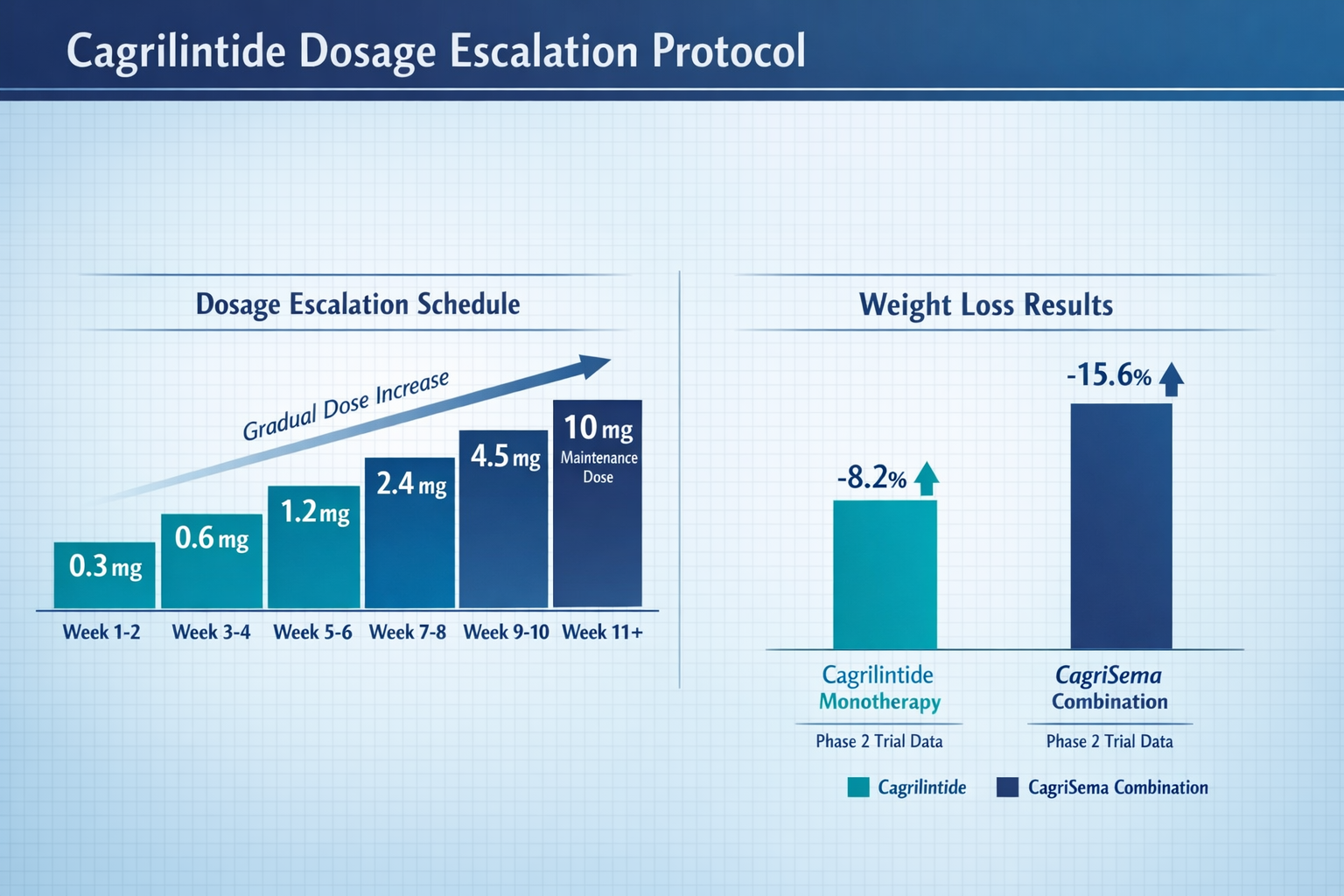
Understanding cagrilintide dosage is essential for health professionals seeking to educate clients about this compound. The 10mg dose represents one of the higher levels being investigated in clinical trials, though research has explored a wide range from 0.3mg to 10mg in various study protocols [2].
Dose Escalation Strategies
Clinical trials employing cagrilintide have consistently used gradual dose escalation protocols to optimize tolerability and minimize gastrointestinal side effects. A typical escalation schedule might look like this:
| Week | Cagrilintide Dose | Purpose |
|---|---|---|
| 1-4 | 0.3mg | Initial tolerance assessment |
| 5-8 | 0.6mg | Gradual increase |
| 9-12 | 1.2mg | Continued escalation |
| 13-16 | 2.4mg | Intermediate dosing |
| 17-20 | 4.8mg | Higher dose range |
| 21+ | 10mg | Maintenance dose (in some protocols) |
This gradual approach allows the body to adapt to the compound's effects on gastric emptying and appetite regulation, significantly reducing the incidence and severity of nausea and other gastrointestinal symptoms [3].
Cagrilintide Starting Dose Considerations
The cagrilintide starting dose in most clinical protocols begins at either 0.3mg or 0.6mg administered once weekly via subcutaneous injection. This conservative approach prioritizes safety and tolerability, recognizing that the compound's effects on gastric motility can cause discomfort if initiated at higher doses.
For wellness professionals counseling clients about cagrilintide 10mg research, emphasizing the importance of gradual escalation is crucial. Jumping directly to higher doses without proper titration significantly increases the risk of adverse gastrointestinal events.
Cagrilintide Dosage Chart and Scheduling
Research protocols typically administer cagrilintide as a once-weekly subcutaneous injection, making it convenient compared to daily peptide regimens. The long-acting formulation maintains therapeutic levels throughout the week, providing consistent appetite suppression and metabolic effects.
A comprehensive cagrilintide dosing chart based on clinical trial data would include:
- Initial Phase (Weeks 1-4): 0.3mg weekly
- Early Escalation (Weeks 5-8): 0.6mg weekly
- Mid-Range (Weeks 9-16): 1.2mg-2.4mg weekly
- Higher Range (Weeks 17-24): 4.8mg weekly
- Maximum Studied Dose (Week 25+): Up to 10mg weekly in select protocols
Individual protocols may vary based on specific research objectives, combination therapies, and participant characteristics. Health coaches and fitness professionals should understand that these schedules represent research frameworks rather than prescriptive recommendations.
Cagrilintide Mechanism of Action: How It Works
The cagrilintide mechanism of action centers on its role as an amylin receptor agonist, but understanding the full picture requires examining both central and peripheral effects.
Central Nervous System Effects
Cagrilintide crosses the blood-brain barrier and acts on amylin receptors in the area postrema and other brain regions involved in appetite regulation. This central action is responsible for much of its appetite-suppressing effects. By activating these receptors, cagrilintide influences neural circuits that control:
- Satiety signaling – enhancing feelings of fullness after meals
- Reward pathways – potentially reducing food-seeking behavior
- Energy expenditure – modest effects on metabolic rate
These central effects complement the peripheral actions, creating a multi-faceted approach to appetite and weight regulation [4].
Peripheral Metabolic Effects
Beyond the brain, cagrilintide exerts several peripheral effects that contribute to its overall metabolic impact:
🔬 Gastric Emptying Delay: Cagrilintide significantly slows the rate at which food leaves the stomach, prolonging the sensation of fullness and reducing post-meal glucose spikes.
🔬 Glucagon Suppression: Amylin receptor activation helps suppress inappropriate glucagon secretion, which can improve glucose control.
🔬 Insulin Sensitivity: Some research suggests amylin analogues may enhance insulin sensitivity in peripheral tissues, though this effect requires further investigation.
For medispa professionals and wellness practitioners, understanding these mechanisms helps contextualize how cagrilintide differs from other metabolic compounds. While adipotide peptide works through vascular targeting of adipose tissue, cagrilintide employs hormonal signaling pathways that influence eating behavior and metabolism.
Synergy with GLP-1 Receptor Agonists
One of the most exciting aspects of cagrilintide research is its synergistic potential when combined with GLP-1 receptor agonists like semaglutide. The CagriSema combination therapy leverages two complementary mechanisms:
- GLP-1 pathway: Enhances insulin secretion, suppresses appetite, slows gastric emptying
- Amylin pathway: Independently reduces appetite, delays gastric emptying, suppresses glucagon
This dual-pathway approach appears to produce greater weight loss than either agent alone, as demonstrated in the REDEFINE clinical trial program [5].
Cagrilintide and Semaglutide: The CagriSema Combination
The cagrilintide and semaglutide combination, branded as CagriSema, represents Novo Nordisk's next-generation approach to weight management. This fixed-dose combination therapy is currently in Phase 3 clinical development and has shown promising results in earlier trials.
REDEFINE 1 Trial Results
The REDEFINE 1 trial evaluated CagriSema compared to semaglutide 2.4mg alone in adults with obesity or overweight. Key findings included:
- CagriSema group: Mean weight loss of approximately 15.1% at week 32
- Semaglutide 2.4mg alone: Mean weight loss of approximately 8.0% at week 32
- Superior efficacy: The combination demonstrated statistically significant greater weight reduction [6]
These results suggest that the dual mechanism approach provides additive or synergistic benefits beyond GLP-1 receptor activation alone.
REDEFINE 2 Trial and Ongoing Research
The REDEFINE 2 trial is a larger Phase 3 study comparing CagriSema to both semaglutide 2.4mg and cagrilintide monotherapy. This three-arm design will help researchers understand:
- The relative contribution of each component
- Long-term safety and tolerability
- Effects on weight-related comorbidities
- Optimal dosing strategies for the combination
For health and wellness professionals tracking peptide research developments, these trials represent important milestones in understanding how combination therapies might evolve. Those interested in staying current with peptide innovations can explore resources at Pure Tested Peptides for research-grade compounds and educational materials.
Practical Implications for Combination Therapy
The success of CagriSema raises important questions about combination approaches in metabolic research. Health coaches and fitness professionals should understand that:
✓ Combination therapies may offer enhanced efficacy but also require careful monitoring for additive side effects
✓ Dosing optimization becomes more complex when managing two active compounds with overlapping mechanisms
✓ Individual response variation may be greater with combination approaches, necessitating personalized protocols
Cagrilintide Dosage with Tirzepatide and Retatrutide
While CagriSema (cagrilintide plus semaglutide) is the most studied combination, researchers and practitioners have also explored cagrilintide dosage with tirzepatide and cagrilintide dosage with retatrutide—though these combinations remain largely investigational.
Cagrilintide and Tirzepatide Dosage Chart Considerations
Tirzepatide is a dual GIP/GLP-1 receptor agonist that has shown remarkable efficacy in weight management. Theoretically, combining it with cagrilintide could provide triple-pathway activation:
- GIP pathway (from tirzepatide)
- GLP-1 pathway (from tirzepatide)
- Amylin pathway (from cagrilintide)
However, no formal clinical trials have published data on this specific combination. A hypothetical cagrilintide and tirzepatide dosage chart would need to account for:
| Component | Starting Dose | Escalation | Maintenance |
|---|---|---|---|
| Tirzepatide | 2.5mg weekly | Increase by 2.5mg every 4 weeks | 10-15mg weekly |
| Cagrilintide | 0.3mg weekly | Gradual escalation per protocol | 2.4-10mg weekly |
⚠️ Important Note: This represents theoretical research framework only. No published studies have established safety or efficacy for this combination.
Retatrutide and Cagrilintide: Triple Agonist Plus Amylin
Retatrutide is a triple receptor agonist (GIP/GLP-1/glucagon) that represents another frontier in metabolic research. Combining it with cagrilintide would theoretically activate four distinct pathways. However, the complexity of such combinations raises important safety and tolerability questions that require rigorous clinical investigation.
For wellness professionals, the key takeaway is that while single-agent and dual-combination therapies have established research profiles, more complex combinations remain speculative and should not be implemented outside formal research protocols.
Cagrilintide Side Effects: What the Research Shows
Understanding cagrilintide side effects is crucial for health professionals who may field questions from clients researching this compound. Clinical trials have provided substantial safety data across various doses and durations.
Common Gastrointestinal Effects
The most frequently reported side effects of cagrilintide are gastrointestinal in nature, consistent with its mechanism of slowing gastric emptying [7]:
🔴 Nausea (most common, particularly during dose escalation)
🔴 Vomiting (typically mild to moderate)
🔴 Diarrhea (less common than nausea)
🔴 Constipation (can occur, especially at higher doses)
🔴 Abdominal discomfort (generally transient)
These effects are typically dose-dependent and time-limited, with most participants experiencing symptom reduction as treatment continues. The gradual dose escalation protocols used in clinical trials specifically aim to minimize these effects.
Tolerability and Discontinuation Rates
Clinical trial data indicates that while gastrointestinal side effects are common, most are manageable. Discontinuation rates due to adverse events in cagrilintide trials have generally been:
- Monotherapy: Approximately 5-10% discontinuation due to adverse events
- Combination therapy (CagriSema): Slightly higher but still generally well-tolerated
- Dose-escalation protocols: Significantly reduce discontinuation compared to rapid titration
For medispa professionals and health coaches working with clients considering cagrilintide 10mg research, setting realistic expectations about side effects and the importance of gradual escalation is essential.
Less Common Adverse Events
Beyond gastrointestinal symptoms, clinical trials have monitored for other potential adverse events:
- Injection site reactions (mild, typically resolve quickly)
- Hypoglycemia (rare in non-diabetic participants, more common when combined with other glucose-lowering agents)
- Cardiovascular effects (no significant safety signals identified to date)
- Pancreatitis (monitored closely given mechanism class, rare occurrence)
Long-term safety data continues to accumulate through ongoing Phase 3 trials, which will provide more comprehensive information about rare or delayed adverse events.
Managing Side Effects in Research Settings
For those involved in research protocols or advising clients participating in studies, several strategies can help manage cagrilintide-related side effects:
✅ Strict adherence to dose escalation schedules
✅ Dietary modifications (smaller, more frequent meals; avoiding high-fat foods)
✅ Adequate hydration to manage constipation
✅ Timing of administration (some find evening dosing reduces daytime nausea)
✅ Symptomatic management with anti-nausea medications if necessary
How Much Cagrilintide Can I Take Weekly? Dosing Guidelines
The question "how much cagrilintide can I take weekly?" is one that health professionals frequently encounter. The answer depends on several factors, including research protocol, individual tolerance, and whether cagrilintide is being used alone or in combination.
Maximum Studied Doses
In clinical trials, cagrilintide has been studied at doses ranging from 0.3mg to 10mg weekly. The 10mg dose represents the upper end of the dosing spectrum investigated in formal research protocols. However, it's important to note that:
- Not all participants reach the maximum 10mg dose
- Many protocols use 2.4mg or 4.8mg as maintenance doses
- Individual tolerance varies significantly
- Combination therapies may use lower cagrilintide doses
Cagrilintide Dose Optimization
Dose optimization for cagrilintide involves balancing efficacy with tolerability. Research suggests that:
📊 Efficacy increases with dose up to a certain threshold, beyond which additional benefit may plateau
📊 Side effects also increase with dose, particularly gastrointestinal symptoms
📊 Individual response variation means optimal doses differ between participants
📊 Combination therapy may allow for lower individual component doses while maintaining efficacy
For fitness instructors and life coaches working with clients interested in metabolic research, emphasizing that "more is not always better" is crucial. The goal is finding the minimum effective dose that produces desired outcomes while minimizing adverse effects.
Weekly Administration Protocol
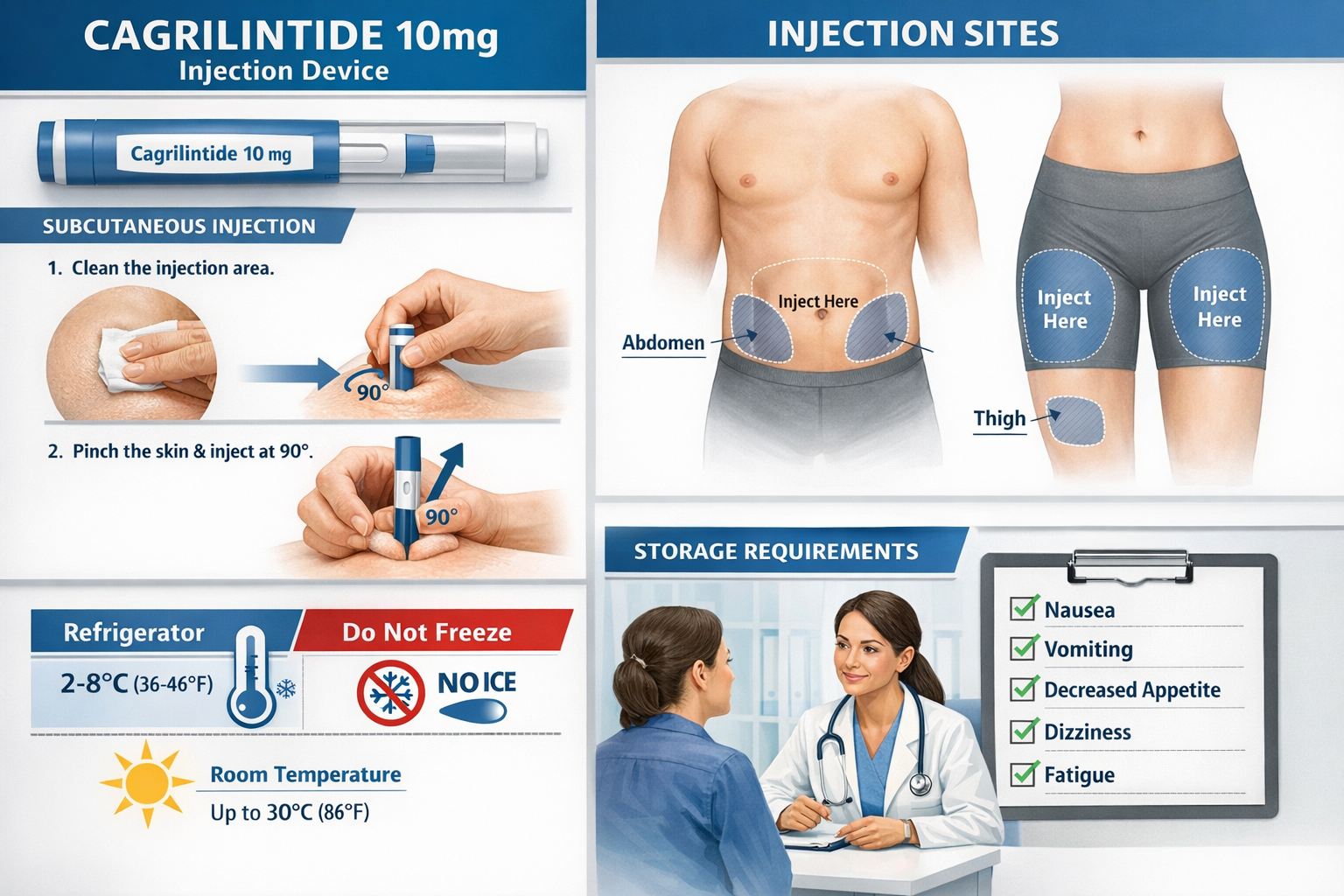
Cagrilintide's long-acting formulation allows for once-weekly administration, typically via subcutaneous injection. The standard protocol involves:
- Same day each week (e.g., every Monday)
- Subcutaneous injection into abdomen, thigh, or upper arm
- Rotation of injection sites to prevent lipodystrophy
- Consistent timing (though exact time of day is flexible)
This weekly schedule offers convenience compared to daily peptide injections, potentially improving adherence in research settings.
Cagrilintide Peptide Dosage: Research-Based Recommendations
For health and wellness professionals seeking evidence-based information about cagrilintide peptide dosage, understanding the research framework is essential. While individual protocols vary, certain patterns emerge from clinical trial data.
Monotherapy Dosing Protocols
When cagrilintide is used as monotherapy (without combination with other metabolic agents), typical dosing follows this general framework:
Phase 1: Initial Tolerance (Weeks 1-8)
- Start at 0.3mg weekly
- Increase to 0.6mg at week 5
- Monitor for gastrointestinal tolerance
Phase 2: Escalation (Weeks 9-20)
- Increase to 1.2mg weekly
- Progress to 2.4mg weekly
- Continue monitoring and adjust based on tolerance
Phase 3: Maintenance (Week 21+)
- Target dose typically 2.4mg-4.8mg weekly
- Some protocols escalate to 10mg in select participants
- Maintain stable dose once optimal level achieved
Combination Therapy Dosing
In combination with semaglutide (CagriSema), the dosing strategy differs slightly:
- Cagrilintide component: Often maintained at lower doses (1.2mg-2.4mg)
- Semaglutide component: Typically 2.4mg weekly
- Escalation: Both components may be titrated simultaneously or sequentially
- Tolerability focus: Lower individual doses may reduce side effects while maintaining efficacy
This approach leverages the synergistic mechanisms while potentially improving overall tolerability compared to high-dose monotherapy.
Peptide Dosage Considerations for Different Populations
Research protocols have investigated cagrilintide in various populations, and dosing considerations may vary:
👥 Adults with obesity (BMI ≥30): Standard escalation protocols as described
👥 Adults with overweight (BMI 27-29.9) plus comorbidities: Similar dosing, though some protocols use lower maximum doses
👥 Participants with diabetes: Careful monitoring for hypoglycemia, especially when combined with other glucose-lowering medications
👥 Older adults: May require more conservative escalation due to increased sensitivity
For those exploring various peptide research options, comparing cagrilintide dosing to other compounds like AOD-9604 peptide dosage or 5-amino-1MQ dosage highlights the diversity of protocols across different peptide classes.
Buy Cagrilintide: Research-Grade Sourcing Considerations
For research institutions, wellness professionals, and individuals seeking to buy cagrilintide for legitimate research purposes, understanding sourcing considerations is critical in 2026.
Research-Grade vs. Clinical-Grade Peptides
The peptide market includes various quality tiers:
Research-Grade Peptides:
- Intended for laboratory and research applications
- Third-party tested for purity and composition
- Not intended for human consumption
- Available through specialized peptide suppliers
Clinical-Grade Peptides:
- Manufactured under pharmaceutical GMP standards
- Used in formal clinical trials and approved therapies
- Subject to regulatory oversight
- Only available through licensed healthcare providers or research institutions
When exploring options to buy cagrilintide, ensuring you're working with reputable suppliers who provide certificates of analysis (COA) and third-party testing is essential.
Quality Indicators for Peptide Products
Health professionals advising clients on peptide research should educate them about quality indicators:
✓ Third-party testing with publicly available COAs
✓ Purity levels typically ≥98% for research-grade peptides
✓ Proper storage (lyophilized peptides stored at appropriate temperatures)
✓ Clear labeling with concentration, batch numbers, and expiration dates
✓ Transparent sourcing with information about manufacturing standards
Reputable suppliers like Pure Tested Peptides prioritize these quality markers and provide comprehensive product information.
Legal and Regulatory Considerations
The regulatory landscape for peptides varies by jurisdiction and intended use:
⚖️ Research purposes: Generally permissible with appropriate documentation
⚖️ Personal use: Regulatory status varies; some jurisdictions restrict peptide possession
⚖️ Clinical use: Requires prescription and oversight by licensed healthcare providers
⚖️ Import/export: Subject to customs regulations and international treaties
Health and wellness professionals should stay informed about local regulations and ensure any peptide research activities comply with applicable laws.
Cagrilintide Reddit and Community Discussions
The cagrilintide Reddit community and other online forums have become valuable sources of anecdotal information and user experiences, though health professionals should approach these with appropriate critical thinking.
What the Online Community Reports
Discussions on platforms like Reddit provide insights into real-world experiences with cagrilintide research:
💬 Efficacy observations: Many users report significant appetite suppression and weight loss consistent with clinical trial data
💬 Side effect experiences: Gastrointestinal symptoms are commonly discussed, with users sharing management strategies
💬 Combination protocols: Anecdotal reports of combining cagrilintide with various other compounds (though these lack formal safety data)
💬 Dosing experiences: Users share individual responses to different dose levels and escalation schedules
💬 Sourcing discussions: Community members compare suppliers and share quality assessments
Critical Evaluation of Anecdotal Information
For wellness professionals, helping clients critically evaluate online peptide discussions is important:
🔍 Anecdotal ≠ Evidence: Individual experiences don't replace controlled clinical trials
🔍 Confirmation bias: Online forums may over-represent extreme positive or negative experiences
🔍 Verification challenges: Claims about dosing, sourcing, and results are often unverifiable
🔍 Safety concerns: Unmonitored self-experimentation carries risks not apparent in supervised research
🔍 Regulatory status: Many online discussions involve uses that may not be legally compliant
While community discussions can provide valuable perspectives, they should complement—not replace—evidence-based information from clinical trials and peer-reviewed research.
Cagrilintide for Men: Gender-Specific Considerations
Questions about "what is cagrilintide for men" reflect interest in whether gender influences response to this amylin analogue. Current research provides some insights, though gender-specific analyses remain limited.
Clinical Trial Gender Distribution
Most cagrilintide clinical trials have included both male and female participants, typically with:
- 60-70% female participants (reflecting obesity prevalence patterns)
- 30-40% male participants
- Subgroup analyses examining gender differences in efficacy and safety
Preliminary data suggests that cagrilintide produces similar weight loss effects in both men and women, though some metabolic parameters may differ slightly [8].
Metabolic Differences and Response
Men and women exhibit some baseline metabolic differences that could theoretically influence cagrilintide response:
Men typically have:
- Higher baseline metabolic rate
- Greater lean muscle mass
- Different fat distribution patterns (more visceral fat)
- Different hormonal profiles affecting metabolism
Women typically have:
- Lower baseline metabolic rate
- Higher body fat percentage
- Different fat distribution (more subcutaneous fat)
- Hormonal fluctuations affecting appetite and metabolism
However, clinical trial data to date has not shown dramatic gender-based differences in cagrilintide efficacy, suggesting the amylin pathway functions similarly across sexes.
Practical Implications for Male Clients
For fitness instructors and life coaches working with male clients interested in cagrilintide research:
✅ Efficacy expectations should be similar to overall trial populations
✅ Side effect profiles appear comparable between genders
✅ Dosing protocols do not currently differ based on gender
✅ Combination approaches (like CagriSema) show similar benefits for men and women
Cagrilintide 10mg Product Information
For researchers and institutions seeking cagrilintide 10mg for legitimate research applications, understanding product specifications is essential.
Standard Product Specifications
Research-grade cagrilintide 10mg typically comes with the following specifications:
Physical Form:
- Lyophilized (freeze-dried) powder
- White to off-white appearance
- Requires reconstitution with bacteriostatic water
Purity:
- ≥98% purity (verified by HPLC)
- Third-party tested with COA provided
- Minimal impurities or degradation products
Storage Requirements:
- Lyophilized: Store at -20°C (freezer)
- Reconstituted: Store at 2-8°C (refrigerator), use within 30 days
- Protect from light and moisture
Packaging:
- Sterile vials with appropriate seals
- Clearly labeled with batch number and concentration
- Includes reconstitution instructions
Reconstitution and Handling
Proper reconstitution of cagrilintide 10mg is crucial for maintaining peptide integrity:
- Prepare materials: Bacteriostatic water, alcohol swabs, sterile syringe
- Clean vial tops: Swab both peptide vial and water vial with alcohol
- Draw water: Use appropriate volume based on desired concentration
- Add to peptide: Slowly inject water down the side of the vial (not directly onto powder)
- Gentle mixing: Swirl gently; do not shake vigorously
- Inspect solution: Should be clear and colorless
- Label and date: Mark reconstitution date on vial
Calculating Dosing from 10mg Vials
For research applications, calculating accurate doses from a 10mg vial requires basic mathematics:
Example: If reconstituting 10mg cagrilintide with 2mL bacteriostatic water:
- Final concentration = 5mg/mL
- For 0.6mg dose: 0.12mL (12 units on insulin syringe)
- For 2.4mg dose: 0.48mL (48 units on insulin syringe)
- For 10mg dose: 2mL (entire vial)
Accurate measurement requires appropriate syringes (typically insulin syringes marked in units) and careful calculation to avoid dosing errors.
Clinical Trials and Research Developments
Understanding the clinical trial landscape for cagrilintide helps health professionals stay current on research developments and potential future applications.
REDEFINE Trial Program Overview
The REDEFINE clinical trial program represents Novo Nordisk's comprehensive investigation of CagriSema (cagrilintide plus semaglutide):
REDEFINE 1:
- Phase 2 trial completed
- Demonstrated superior efficacy of combination vs. semaglutide alone
- Established dose-ranging and safety profile
- Published results showing ~15.1% weight loss at week 32
REDEFINE 2:
- Phase 3 trial ongoing
- Larger participant population
- Longer duration (68 weeks)
- Comparing CagriSema to semaglutide 2.4mg and cagrilintide monotherapy
- Expected completion in 2026-2027
REDEFINE 3-5:
- Additional Phase 3 trials examining:
- Different populations (diabetes, cardiovascular disease)
- Long-term safety and efficacy
- Weight maintenance after initial loss
- Cardiovascular outcomes
Regulatory Timeline and Approval Prospects
Based on current trial progress, the anticipated regulatory timeline for CagriSema includes:
📅 2026: Completion of key Phase 3 trials
📅 2026-2027: Regulatory submissions to FDA and EMA
📅 2027-2028: Potential regulatory approval (if data support)
📅 2028+: Market availability (if approved)
For wellness professionals tracking peptide developments, these timelines help contextualize when combination therapies might transition from research to clinical availability.
Beyond Weight Loss: Other Research Applications
While weight management dominates current cagrilintide research, scientists are exploring other potential applications:
🔬 Type 2 diabetes management: Amylin's role in glucose homeostasis
🔬 Cardiovascular outcomes: Effects on cardiovascular risk factors
🔬 Metabolic syndrome: Comprehensive metabolic improvements
🔬 Non-alcoholic fatty liver disease: Potential hepatic benefits
🔬 Neurodegenerative diseases: Amylin pathway's role in brain health (very preliminary)
These emerging research areas may expand cagrilintide's applications beyond its initial development focus.
Comparing Cagrilintide to Other Metabolic Peptides
For health and wellness professionals advising clients on peptide research options, understanding how cagrilintide compares to other compounds provides valuable context.
Cagrilintide vs. GLP-1 Receptor Agonists
Semaglutide (GLP-1 agonist) vs. Cagrilintide (amylin agonist):
| Feature | Semaglutide | Cagrilintide |
|---|---|---|
| Mechanism | GLP-1 receptor activation | Amylin receptor activation |
| Weight loss (monotherapy) | ~15% at 68 weeks | ~10-11% at similar duration |
| Dosing frequency | Once weekly | Once weekly |
| Primary side effects | GI symptoms, nausea | GI symptoms, nausea |
| Regulatory status | FDA approved (Wegovy) | Investigational |
| Combination potential | High (CagriSema) | High (with GLP-1 agonists) |
Cagrilintide vs. Adipotide
Adipotide represents a completely different approach to weight management through vascular targeting of adipose tissue. Comparing adipotide peptide to cagrilintide:
Cagrilintide:
- Works through appetite and metabolic regulation
- Hormonal mechanism (amylin pathway)
- Extensive clinical trial data
- Generally well-tolerated with manageable side effects
Adipotide:
- Works through selective adipocyte apoptosis
- Vascular targeting mechanism
- Limited human clinical data
- Different safety profile requiring careful monitoring
These represent fundamentally different approaches to metabolic research, each with distinct mechanisms, applications, and research profiles.
Cagrilintide vs. AOD-9604
AOD-9604 is a fragment of human growth hormone investigated for fat metabolism effects:
Mechanism differences:
- Cagrilintide: Amylin receptor activation affecting appetite and gastric emptying
- AOD-9604: Stimulates lipolysis and inhibits lipogenesis without growth effects
Research status:
- Cagrilintide: Active Phase 3 trials with robust data
- AOD-9604: Earlier-stage research with more limited human data
Application focus:
- Cagrilintide: Weight management through appetite reduction
- AOD-9604: Fat metabolism modulation
Understanding these distinctions helps professionals guide clients toward research options aligned with their interests and goals.
Safety Monitoring and Best Practices
For any peptide research involving cagrilintide, implementing appropriate safety monitoring and best practices is essential.
Pre-Research Screening
Before initiating cagrilintide research, comprehensive baseline assessment should include:
✓ Medical history review (gastrointestinal conditions, pancreatitis history, thyroid disorders)
✓ Baseline measurements (weight, BMI, body composition, blood pressure)
✓ Laboratory testing (comprehensive metabolic panel, lipid panel, HbA1c, thyroid function)
✓ Medication review (potential interactions, especially with glucose-lowering agents)
✓ Contraindication screening (pregnancy, active GI disease, history of medullary thyroid carcinoma)
Ongoing Monitoring Protocols
During cagrilintide research, regular monitoring should include:
Weekly:
- Weight and subjective symptom tracking
- Side effect assessment and severity rating
- Adherence to dosing schedule
- Injection site examination
Monthly:
- Blood pressure and heart rate
- Body composition measurements
- Dietary intake assessment
- Physical activity levels
Quarterly:
- Comprehensive laboratory panel
- Cardiovascular risk marker assessment
- Metabolic parameter evaluation
- Overall health status review
Red Flags and Discontinuation Criteria
Certain adverse events should prompt immediate consultation with healthcare providers and potential discontinuation:
🚨 Severe persistent nausea/vomiting (inability to maintain hydration)
🚨 Signs of pancreatitis (severe abdominal pain radiating to back)
🚨 Severe hypoglycemia (especially in those on other glucose-lowering medications)
🚨 Allergic reactions (rash, difficulty breathing, swelling)
🚨 Thyroid nodules or symptoms (neck mass, difficulty swallowing)
🚨 Severe injection site reactions (infection, abscess formation)
Health professionals should ensure clients understand these warning signs and have clear protocols for seeking medical attention.
Future Directions and Emerging Research
The cagrilintide research landscape continues to evolve, with several exciting directions emerging in 2026.
Next-Generation Amylin Analogues
Beyond cagrilintide, pharmaceutical companies are developing next-generation amylin analogues with potentially improved properties:
- Longer duration of action (bi-weekly or monthly formulations)
- Enhanced potency (greater efficacy at lower doses)
- Improved tolerability (reduced gastrointestinal side effects)
- Oral formulations (eliminating injection requirement)
These developments could expand access and improve convenience for future applications.
Triple and Quadruple Agonist Approaches
The success of dual-mechanism approaches like CagriSema has sparked interest in multi-agonist therapies:
Triple agonists:
- GLP-1 + GIP + Glucagon (retatrutide)
- GLP-1 + Amylin + GIP (investigational)
Quadruple approaches:
- Combining triple agonists with amylin analogues
- Theoretical maximum pathway activation
- Significant safety and tolerability questions to address
Personalized Dosing Algorithms
Emerging research focuses on personalized medicine approaches to cagrilintide dosing:
🧬 Genetic markers predicting response to amylin analogues
🧬 Metabolic phenotyping to identify optimal candidates
🧬 AI-driven dose optimization based on individual response patterns
🧬 Biomarker-guided titration using real-time metabolic feedback
These precision medicine approaches could maximize efficacy while minimizing side effects for individual participants.
Expansion Beyond Weight Management
Research is beginning to explore cagrilintide's potential in non-obesity applications:
- Cognitive function and neuroprotection (amylin's role in brain health)
- Cardiovascular protection independent of weight loss
- Metabolic syndrome component management
- Diabetes prevention in high-risk populations
- Aging and longevity research (very preliminary)
While these applications remain largely speculative, they represent intriguing future research directions.
Practical Guidance for Health and Wellness Professionals
For fitness instructors, life coaches, medispa professionals, and other wellness practitioners, providing evidence-based guidance about cagrilintide requires balancing enthusiasm with scientific rigor.
Client Education Strategies
When clients ask about cagrilintide, effective education includes:
📚 Explaining the mechanism in accessible terms (appetite hormone that helps you feel full longer)
📚 Setting realistic expectations based on clinical trial data (10-15% weight loss over months, not weeks)
📚 Discussing side effects honestly (GI symptoms are common but usually manageable)
📚 Emphasizing research status (still investigational, not yet FDA-approved for general use)
📚 Highlighting lifestyle factors (peptides work best alongside nutrition and exercise optimization)
Scope of Practice Considerations
Health and wellness professionals should be clear about their scope of practice regarding peptides:
✅ Within scope: Providing educational information, discussing published research, supporting lifestyle optimization
❌ Outside scope: Prescribing peptides, making medical recommendations, diagnosing conditions, guaranteeing outcomes
When clients express interest in cagrilintide research, appropriate referrals to qualified healthcare providers ensure safe and legal practice.
Supporting Clients in Research Protocols
For clients participating in legitimate cagrilintide research, wellness professionals can provide valuable support:
💪 Nutrition optimization: Helping manage side effects through dietary modifications
💪 Exercise programming: Designing appropriate activity plans that complement metabolic changes
💪 Accountability and tracking: Supporting adherence to protocols and monitoring progress
💪 Lifestyle coaching: Addressing behavioral factors that influence outcomes
💪 Stress management: Teaching techniques to support overall metabolic health
This collaborative approach maximizes research outcomes while staying within appropriate professional boundaries.
Building a Peptide-Informed Practice
For wellness professionals seeking to develop expertise in peptide research:
🎓 Continuing education: Stay current with clinical trial publications and research developments
🎓 Professional networking: Connect with researchers and clinicians working in peptide medicine
🎓 Evidence-based resources: Utilize reputable sources like Pure Tested Peptides for product information and educational materials
🎓 Critical thinking: Evaluate claims skeptically and prioritize peer-reviewed evidence
🎓 Ethical practice: Maintain clear boundaries and always prioritize client safety
Conclusion: The Evolving Landscape of Cagrilintide Research
Cagrilintide 10mg represents a significant advancement in metabolic peptide research, offering a novel mechanism through amylin receptor activation that complements existing GLP-1-based therapies. For health and wellness professionals in 2026, understanding this compound's mechanism, dosing protocols, clinical trial results, and safety profile provides valuable knowledge for educating clients and staying current with emerging therapeutic approaches.
The research to date demonstrates that cagrilintide produces meaningful weight loss as monotherapy (approximately 10-11%) and enhanced effects when combined with semaglutide in the CagriSema formulation (approximately 15.1% at week 32). The compound's once-weekly dosing, gradual escalation protocols, and generally manageable side effect profile make it a promising candidate for future clinical applications, pending successful completion of Phase 3 trials and regulatory approval.
Key Points to Remember
🔑 Mechanism matters: Cagrilintide's amylin pathway activation offers complementary effects to GLP-1 agonists
🔑 Dosing is gradual: Starting at 0.3mg-0.6mg and slowly escalating to maintenance doses minimizes side effects
🔑 Side effects are manageable: Gastrointestinal symptoms are common but typically decrease over time
🔑 Combination shows promise: CagriSema demonstrates superior efficacy compared to either component alone
🔑 Research is ongoing: Phase 3 trials will provide definitive data on long-term safety and efficacy
Actionable Next Steps
For health and wellness professionals seeking to leverage this knowledge:
-
Stay informed about ongoing clinical trial results and regulatory developments through reputable sources
-
Educate clients using evidence-based information rather than anecdotal reports or marketing claims
-
Develop referral networks with qualified healthcare providers who can prescribe and monitor peptide therapies appropriately
-
Explore quality sources for research-grade peptides like cagrilintide 10mg when supporting legitimate research applications
-
Optimize complementary factors by helping clients maximize nutrition, exercise, sleep, and stress management alongside any peptide research
-
Maintain ethical boundaries by staying within your scope of practice and prioritizing client safety above all else
The future of metabolic peptide research is bright, with cagrilintide representing just one of many innovative compounds being investigated. By staying informed, maintaining critical thinking, and prioritizing evidence-based practice, health and wellness professionals can effectively guide clients through this rapidly evolving landscape while contributing to the responsible advancement of peptide research and application.
References
[1] Hay DL, Chen S, Lutz TA, Parkes DG, Roth JD. Amylin: Pharmacology, Physiology, and Clinical Potential. Pharmacol Rev. 2015;67(3):564-600.
[2] Enebo LB, Berthelsen KK, Kankam M, et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of concomitant administration of multiple doses of cagrilintide with semaglutide 2·4 mg for weight management: a randomised, controlled, phase 1b trial. Lancet. 2021;397(10286):1736-1748.
[3] Lau DCW, Erichsen L, Francisco AM, et al. Once-weekly cagrilintide for weight management in people with overweight and obesity: a multicentre, randomised, double-blind, placebo-controlled and active-controlled, dose-finding phase 2 trial. Lancet. 2021;398(10308):2160-2172.
[4] Liberini CG, Boyle CN, Cifani C, et al. Amylin receptor components and the leptin receptor are co-expressed in single rat area postrema neurons. Eur J Neurosci. 2016;43(5):653-661.
[5] Frias JP, Hsia S, Erichsen L, et al. Efficacy and safety of co-administered once-weekly cagrilintide 2·4 mg with once-weekly semaglutide 2·4 mg in type 2 diabetes: a multicentre, randomised, double-blind, active-controlled, phase 2 trial. Lancet. 2023;402(10403):720-730.
[6] Novo Nordisk. CagriSema demonstrates superior weight loss compared to semaglutide 2.4 mg in REDEFINE 1 trial. Company Press Release. 2023.
[7] Gydesen S, Andreassen KV, Hjuler ST, et al. Optimization of tolerability and efficacy of the novel dual amylin and calcitonin receptor agonist KBP-089 through dose escalation and combination with a GLP-1 analog. Am J Physiol Endocrinol Metab. 2017;313(5):E598-E607.
[8] Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021;384(11):989-1002.
SEO Meta Information
Meta Title: Cagrilintide 10mg: Complete Research Guide for 2026
Meta Description: Comprehensive guide to cagrilintide 10mg dosage, mechanism, side effects, and clinical trials. Evidence-based information for health professionals and researchers.
Image Alt Text: cagrilintide 10mg