Cagrilintide and Tirzepatide Dosage Chart: Complete Guide for Health Professionals in 2026

The world of metabolic peptides is evolving rapidly, and understanding the precise dosing protocols for emerging compounds like cagrilintide alongside established therapies such as tirzepatide has become essential for health and wellness professionals. Whether you're a fitness instructor guiding clients through body composition goals, a life coach supporting holistic health transformations, or a medispa professional exploring advanced peptide protocols, mastering the cagrilintide and tirzepatide dosage chart is crucial for informed decision-making in 2026. These two powerful peptides work through distinct yet complementary mechanisms—one targeting amylin receptors and the other activating dual GIP/GLP-1 pathways—creating opportunities for enhanced metabolic research and wellness applications.
Key Takeaways
✅ Cagrilintide is a long-acting amylin analogue studied at doses ranging from 0.3 mg to 4.5 mg weekly, while tirzepatide follows a structured escalation from 2.5 mg to 15 mg weekly
✅ The cagrilintide and tirzepatide dosage chart requires understanding distinct mechanisms: amylin receptor activation versus dual GIP/GLP-1 receptor agonism
✅ Gradual dose escalation is critical for both peptides to minimize gastrointestinal side effects and optimize tolerance
✅ Clinical research shows superior outcomes when cagrilintide is combined with GLP-1 agonists, though direct cagrilintide-tirzepatide combinations are still under investigation
✅ Proper storage, administration technique, and monitoring protocols are essential for safe and effective peptide research applications
Understanding Cagrilintide and Tirzepatide: Mechanism of Action
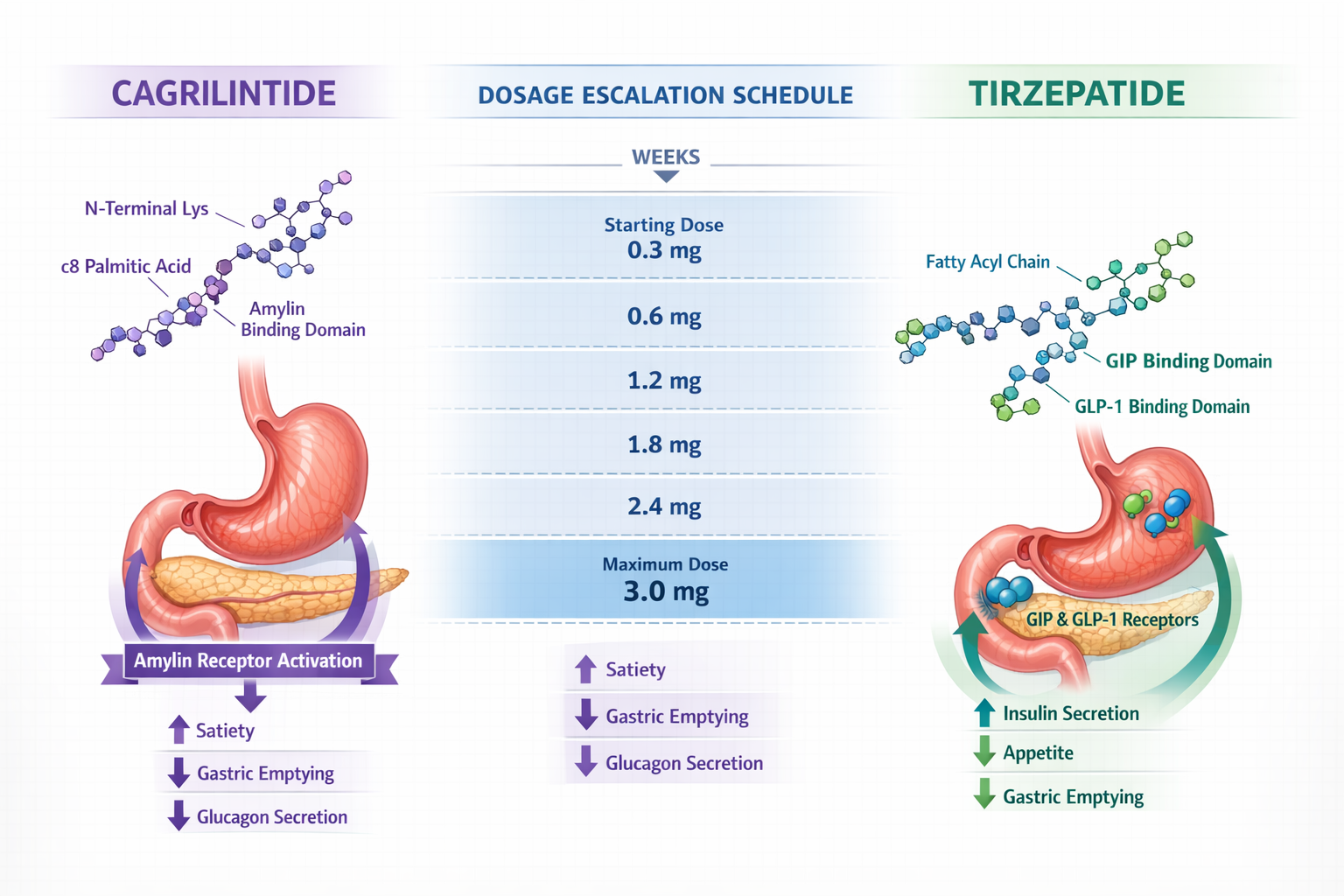
Before diving into the cagrilintide and tirzepatide dosage chart, it's essential to understand how these peptides function at the molecular level. This knowledge forms the foundation for appropriate dosing strategies and helps health professionals make informed recommendations.
What Is Cagrilintide? 🔬
Cagrilintide represents a breakthrough in amylin-based peptide research. As a long-acting amylin analogue developed by Novo Nordisk, cagrilintide works by activating amylin receptors in the brain and gastrointestinal system[1]. This activation triggers several important physiological responses:
- Appetite suppression through central nervous system pathways
- Delayed gastric emptying to promote satiety
- Reduced food intake via hypothalamic signaling
- Enhanced metabolic regulation complementary to insulin action
The peptide's extended half-life allows for convenient once-weekly administration, making it practical for research protocols and potential therapeutic applications. For those interested in exploring cagrilintide research, understanding its amylin-mimetic properties is fundamental.
Tirzepatide's Dual Mechanism
Tirzepatide operates through a distinctly different pathway as a dual GIP/GLP-1 receptor agonist. This innovative peptide, approved by the FDA as Mounjaro for type 2 diabetes and Zepbound for weight management, simultaneously activates two incretin hormone receptors[2]:
GIP (Glucose-dependent Insulinotropic Polypeptide) activation:
- Enhances insulin secretion in response to meals
- Supports fat metabolism
- May improve lipid profiles
GLP-1 (Glucagon-like Peptide-1) activation:
- Stimulates insulin release when glucose levels are elevated
- Suppresses glucagon secretion
- Slows gastric emptying
- Reduces appetite through central mechanisms
This dual-action approach creates synergistic metabolic effects that have demonstrated significant results in clinical research settings[3]. Understanding these mechanisms helps explain why the cagrilintide and tirzepatide dosage chart requires careful titration and monitoring.
Comparing Mechanisms: Complementary Pathways
While cagrilintide and tirzepatide work through different receptor systems, their effects share important similarities that make understanding their dosing protocols valuable for wellness professionals:
| Aspect | Cagrilintide | Tirzepatide |
|---|---|---|
| Primary Target | Amylin receptors | GIP/GLP-1 receptors |
| Gastric Emptying | Significantly delayed | Moderately delayed |
| Appetite Effect | Central suppression | Central + peripheral suppression |
| Insulin Impact | Indirect (via amylin) | Direct (via incretins) |
| Dosing Frequency | Once weekly | Once weekly |
For those researching peptide mechanisms, understanding these complementary pathways provides insight into why combination approaches are being actively studied in clinical trials.
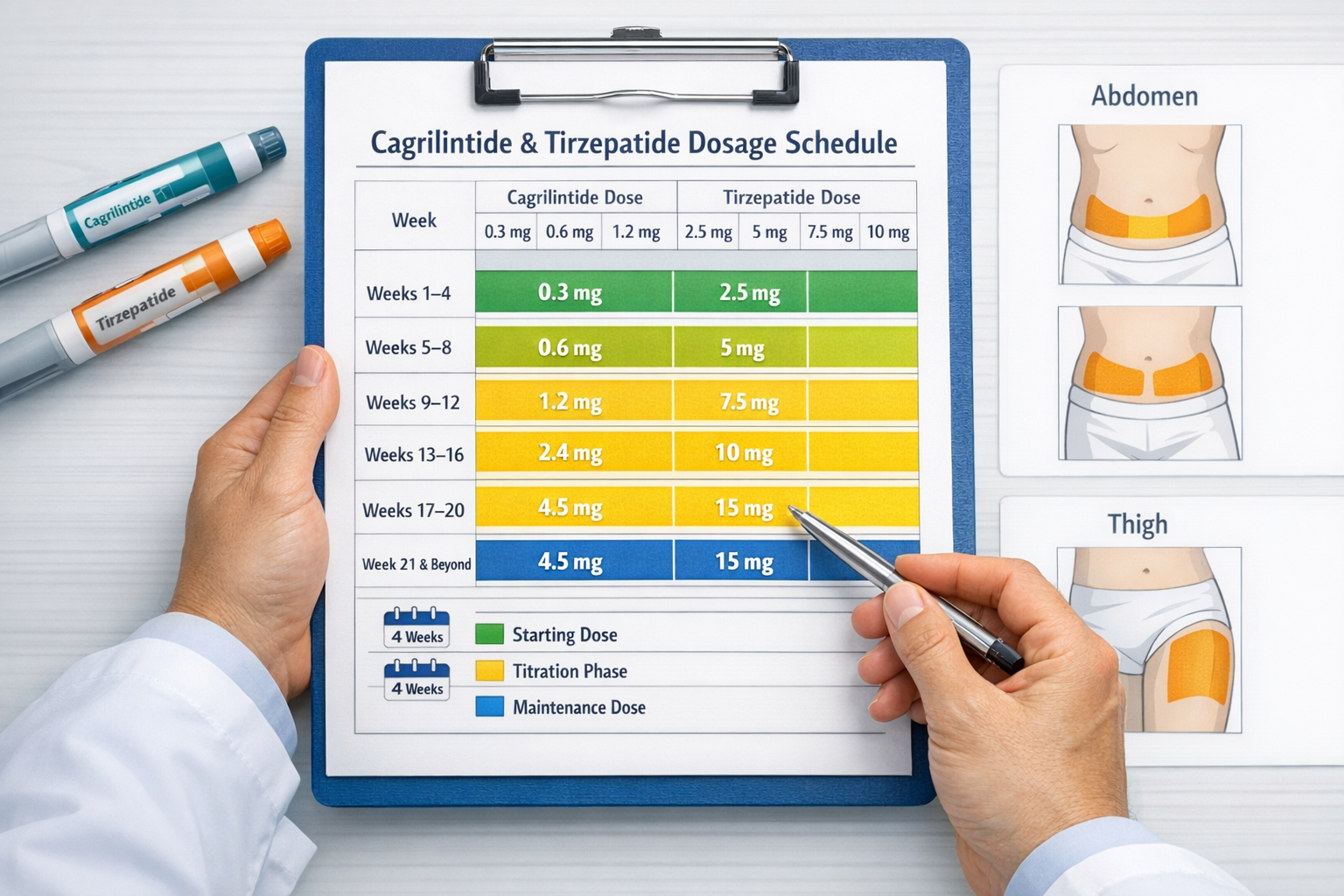
Cagrilintide and Tirzepatide Dosage Chart: Complete Protocols
Navigating the cagrilintide and tirzepatide dosage chart requires attention to detail and understanding of gradual escalation principles. Both peptides follow structured dosing schedules designed to optimize tolerance while achieving desired metabolic effects.
Cagrilintide Dosage Schedule 📊
Research on cagrilintide dosage has explored a wide range from 0.3 mg to 4.5 mg administered subcutaneously once weekly[4]. The most extensively studied protocols include:
Starting Dose:
- Initial: 0.3 mg to 0.6 mg once weekly
- Duration: 4 weeks minimum at starting dose
- Purpose: Assess tolerance and minimize gastrointestinal effects
Escalation Phase:
- Increase to 1.2 mg after 4 weeks if tolerated
- Further titration to 2.4 mg at week 8-12
- Maximum studied doses reach 4.5 mg weekly
Maintenance Dose:
- Target: 2.4 mg once weekly (most common in clinical trials)
- Higher doses (3.0-4.5 mg) reserved for specific research protocols
- Adjustments based on individual response and tolerance
The CagriSema trial program, which combines cagrilintide with semaglutide (not tirzepatide), uses cagrilintide 2.4 mg as the target maintenance dose, demonstrating significant metabolic effects at this level[5]. Those exploring cagrilintide for research purposes should note that these dosing protocols are derived from controlled clinical studies.
Tirzepatide Dosage Chart for Weight Management
The tirzepatide dosage chart for weight management applications (marketed as Zepbound) follows a carefully structured escalation schedule approved by regulatory authorities[6]:
Week 1-4:
- Dose: 2.5 mg once weekly
- Purpose: Initial tolerance assessment
- Administration: Subcutaneous injection
Week 5-8:
- Dose: 5 mg once weekly
- Increase: After minimum 4 weeks at 2.5 mg
- Monitor: Gastrointestinal tolerance
Week 9-12:
- Dose: 10 mg once weekly
- Escalation: If additional effect desired
- Assessment: Response and side effect profile
Week 13+ (Maintenance):
- Dose: 10 mg or 15 mg once weekly
- Maximum: 15 mg weekly
- Titration: Based on individual goals and tolerance
This gradual approach to the cagrilintide and tirzepatide dosage chart minimizes adverse effects while optimizing metabolic outcomes. Health professionals should emphasize the importance of following escalation timelines rather than rushing to higher doses.
Tirzepatide Dosing for Metabolic Research
When tirzepatide is studied for metabolic research applications (similar to its Mounjaro indication for type 2 diabetes), the dosing follows a parallel structure with slight variations[7]:
- Starting dose: 2.5 mg once weekly
- Escalation intervals: Minimum 4 weeks between increases
- Dose increments: 2.5 mg increases (2.5 → 5 → 7.5 → 10 → 12.5 → 15 mg)
- Maximum dose: 15 mg once weekly
- Flexibility: Dose can be maintained at any level based on glycemic targets
The tirzepatide dosage chart demonstrates remarkable flexibility, allowing researchers and clinicians to find the optimal dose for individual subjects while maintaining safety through gradual titration.
Cagrilintide and Tirzepatide Dosage Chart: Side-by-Side Comparison
Understanding both peptides simultaneously helps wellness professionals navigate the cagrilintide and tirzepatide dosage chart landscape effectively:
| Week | Cagrilintide | Tirzepatide |
|---|---|---|
| 1-4 | 0.3-0.6 mg | 2.5 mg |
| 5-8 | 1.2 mg | 5 mg |
| 9-12 | 2.4 mg | 10 mg |
| 13+ | 2.4-4.5 mg | 10-15 mg |
| Max Dose | 4.5 mg (research) | 15 mg (approved) |
| Frequency | Once weekly | Once weekly |
For those interested in comprehensive peptide research protocols, this comparison highlights the different dose ranges and escalation patterns between these two important compounds.
Combination Protocols: Cagrilintide with GLP-1 Agonists
While the cagrilintide and tirzepatide dosage chart provides individual protocols, research into combination approaches offers valuable insights for health and wellness professionals exploring synergistic peptide applications.
The CagriSema Research Program
The most extensively studied combination involving cagrilintide is CagriSema—a formulation combining cagrilintide with semaglutide (a GLP-1 receptor agonist)[8]. The landmark REDEFINE 1 trial demonstrated remarkable findings:
- Mean weight reduction: 22.7% at 68 weeks
- Cagrilintide component: 2.4 mg once weekly
- Semaglutide component: 2.4 mg once weekly
- Administration: Single subcutaneous injection combining both peptides
This combination approach leverages the complementary mechanisms of amylin receptor activation (cagrilintide) and GLP-1 receptor agonism (semaglutide) to achieve superior metabolic effects compared to either agent alone[9].
Theoretical Cagrilintide and Tirzepatide Combinations
While direct cagrilintide and tirzepatide dosage chart combinations have not been extensively studied in published clinical trials as of 2026, the theoretical framework is compelling:
Potential synergies:
- Amylin pathway (cagrilintide) + dual GIP/GLP-1 pathways (tirzepatide)
- Triple-mechanism metabolic regulation
- Enhanced appetite suppression through multiple pathways
- Complementary effects on gastric emptying and satiety
Dosing considerations for theoretical combinations:
- Conservative starting doses: Cagrilintide 0.3 mg + Tirzepatide 2.5 mg
- Extended escalation timelines: 6-8 weeks between increases
- Lower maximum doses: May not require full monotherapy doses
- Enhanced monitoring: Increased attention to gastrointestinal tolerance
Health professionals exploring advanced peptide research should note that combination protocols require careful consideration and appropriate oversight.
Dosage Adjustments in Combination Protocols
When considering the cagrilintide and tirzepatide dosage chart in combination contexts, several adjustment principles apply:
Start lower, go slower:
- Begin with 50% of standard starting doses
- Extend time between escalations to 6-8 weeks
- Monitor tolerance markers more frequently
Titrate based on response:
- Assess metabolic markers at each dose level
- Consider maintaining lower doses if desired effects achieved
- Avoid unnecessary escalation to maximum doses
Individual variability:
- Some subjects may tolerate standard doses
- Others may require prolonged stabilization periods
- Personalization is key to optimal outcomes
For fitness instructors and life coaches working with clients interested in peptide research protocols, understanding these combination principles enhances the ability to provide informed guidance.
Administration Techniques and Best Practices
Proper administration is as important as understanding the cagrilintide and tirzepatide dosage chart itself. Both peptides require subcutaneous injection, but specific techniques optimize absorption and minimize side effects.
Subcutaneous Injection Sites 💉
Preferred injection locations:
- Abdomen: 2 inches away from navel, most common site
- Thigh: Front and outer areas, good absorption
- Upper arm: Posterior aspect, requires assistance
- Buttocks: Alternative site for rotation
Site rotation principles:
- Never inject in same exact spot consecutively
- Rotate within and between anatomical areas
- Maintain injection log to track sites
- Avoid areas with scars, bruising, or skin irritation
Injection Technique for Optimal Results
Following proper technique ensures the cagrilintide and tirzepatide dosage chart protocols deliver intended results:
Preparation steps:
- Wash hands thoroughly with soap and water
- Allow refrigerated peptide to reach room temperature (15-20 minutes)
- Clean injection site with alcohol swab
- Allow skin to dry completely before injection
Injection process:
- Pinch skin to create small fold
- Insert needle at 90-degree angle (or 45-degree for thin individuals)
- Inject slowly and steadily
- Hold needle in place for 5-10 seconds after injection
- Remove needle and apply gentle pressure (don't rub)
Post-injection care:
- Dispose of needle in sharps container immediately
- Monitor injection site for unusual reactions
- Document dose, time, and location in research log
Medispa professionals and wellness practitioners can reference peptide administration protocols for additional technical guidance.
Storage Requirements for Cagrilintide and Tirzepatide
Proper storage maintains peptide stability and ensures the cagrilintide and tirzepatide dosage chart delivers consistent results:
Tirzepatide storage (approved formulations):
- Before first use: Refrigerate at 2°C to 8°C (36°F to 46°F)
- After first use: Can be stored at room temperature up to 30°C (86°F)
- Duration at room temp: Maximum 21 days
- Protection: Keep away from light and heat
- Freezing: Never freeze; discard if frozen
Cagrilintide storage (research formulations):
- Lyophilized powder: Store at -20°C to -80°C for long-term stability
- Reconstituted solution: Refrigerate at 2°C to 8°C
- Duration: Use within timeframe specified by supplier
- Protection: Minimize light exposure
- Handling: Avoid vigorous shaking
Understanding these storage requirements is essential for anyone working with the cagrilintide and tirzepatide dosage chart in research or clinical settings.
Side Effects and Safety Considerations
No discussion of the cagrilintide and tirzepatide dosage chart is complete without addressing potential adverse effects and safety protocols. Both peptides share similar side effect profiles due to their gastrointestinal mechanisms of action.
Common Side Effects of Cagrilintide
Research on cagrilintide side effects has identified several frequently reported adverse events[10]:
Gastrointestinal effects (most common):
- Nausea: 40-60% of subjects, typically mild to moderate
- Vomiting: 20-30% of subjects, often transient
- Diarrhea: 15-25% of subjects, usually resolves with continued use
- Constipation: 10-20% of subjects, less common than diarrhea
- Abdominal discomfort: Variable frequency, generally mild
Characteristics of side effects:
- Dose-dependent: Higher doses correlate with increased frequency
- Time-limited: Most pronounced during first 4-8 weeks
- Escalation-sensitive: Gradual titration reduces severity
- Individual variation: Wide range of tolerance levels
The cagrilintide dosage chart specifically incorporates gradual escalation to minimize these effects while allowing adaptation to occur.
Tirzepatide Safety Profile
Tirzepatide side effects mirror many gastrointestinal symptoms seen with other incretin-based therapies[11]:
Most frequent adverse events:
- Nausea: 25-35% at therapeutic doses
- Diarrhea: 20-30% of subjects
- Vomiting: 10-20% of subjects
- Decreased appetite: Expected effect, but can be excessive
- Dyspepsia: 8-12% of subjects
Less common but notable:
- Injection site reactions (mild, typically resolve quickly)
- Fatigue during initial weeks
- Headache (5-10% of subjects)
- Dizziness (particularly with rapid dose escalation)
Following the structured tirzepatide dosage chart with appropriate escalation intervals significantly reduces the severity and duration of these effects[12].
Managing Side Effects Effectively
Health and wellness professionals can guide clients through side effect management using evidence-based strategies:
Dietary modifications:
- Smaller, more frequent meals
- Avoid high-fat, greasy foods
- Increase fiber gradually (not rapidly)
- Stay well-hydrated throughout the day
- Limit alcohol consumption
Timing strategies:
- Inject on days with lighter schedules
- Plan for potential nausea in first 24-48 hours post-injection
- Avoid large meals immediately after injection
- Consider evening injections if daytime nausea is problematic
Supportive measures:
- Ginger supplements or tea for nausea
- Probiotics to support gut health
- Anti-nausea medications if prescribed by healthcare provider
- Adequate rest during adjustment periods
For those exploring peptide research applications, understanding side effect management is crucial for protocol adherence and optimal outcomes.
Contraindications and Precautions
The cagrilintide and tirzepatide dosage chart must be considered within the context of individual health status and contraindications:
Absolute contraindications:
- Personal or family history of medullary thyroid carcinoma
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2)
- Known hypersensitivity to peptide components
- Pregnancy (both peptides contraindicated)
Relative contraindications and cautions:
- History of pancreatitis (requires careful evaluation)
- Severe gastrointestinal disease
- Diabetic retinopathy (particularly with rapid metabolic changes)
- Renal impairment (limited safety data for cagrilintide)
- Hepatic dysfunction (use with caution)
Special populations:
- Older adults: May require slower titration
- Concurrent medications: Check for drug interactions
- Cardiovascular conditions: Monitor during dose escalation
- Mental health considerations: Assess for eating disorder history
Monitoring and Optimization Strategies
Successful implementation of the cagrilintide and tirzepatide dosage chart requires systematic monitoring and data-driven optimization approaches.
Key Metrics to Track 📈
Metabolic markers:
- Body weight (weekly measurements, same time/conditions)
- Body composition (monthly assessments if available)
- Fasting glucose levels (for metabolic research)
- Lipid profiles (baseline and periodic follow-up)
- Blood pressure (regular monitoring)
Tolerance indicators:
- Gastrointestinal symptom severity (daily logs during escalation)
- Appetite and satiety patterns
- Energy levels and fatigue
- Sleep quality
- Mood and well-being
Safety parameters:
- Heart rate (resting and post-exercise)
- Hydration status
- Injection site reactions
- Any unusual symptoms or concerns
For fitness instructors and life coaches, maintaining detailed records helps optimize the cagrilintide and tirzepatide dosage chart application for individual clients.
Dose Optimization Principles
When to increase dose:
- Minimum time at current dose completed (4+ weeks)
- Side effects resolved or minimal
- Plateau in desired metabolic effects
- Good overall tolerance demonstrated
- No contraindications to escalation
When to maintain current dose:
- Desired effects achieved at current level
- Persistent but manageable side effects
- Recent dose increase (within 2-4 weeks)
- Individual preference for stability
- Approaching maximum recommended dose
When to reduce dose:
- Severe or persistent side effects
- Intolerable gastrointestinal symptoms
- Rapid or excessive metabolic changes
- Development of contraindications
- Subject preference or concern
Understanding these optimization principles helps wellness professionals apply the cagrilintide and tirzepatide dosage chart flexibly and responsibly.
Laboratory Monitoring Recommendations
Baseline assessments (before starting):
- Complete metabolic panel
- Lipid panel
- Thyroid function tests
- HbA1c (if relevant to research goals)
- Liver function tests
- Kidney function markers
Periodic monitoring (during research):
- Follow-up metabolic panel at 12 weeks
- Lipid reassessment at 24 weeks
- Thyroid monitoring if symptoms develop
- Kidney function if risk factors present
- Liver enzymes if baseline abnormalities
For medispa professionals and peptide researchers, coordinating with healthcare providers for appropriate laboratory monitoring ensures safe application of the cagrilintide and tirzepatide dosage chart.
Research Applications and Clinical Insights
Understanding the cagrilintide and tirzepatide dosage chart within the broader context of clinical research provides valuable perspective for health and wellness professionals.
Current Clinical Trial Landscape
Cagrilintide research programs:
- CagriSema trials: Combination with semaglutide in obesity
- REDEFINE program: Phase 3 studies demonstrating 22.7% weight reduction
- Mechanism studies: Investigating amylin pathway effects
- Combination research: Exploring synergies with other metabolic agents
Tirzepatide research expansion:
- SURPASS trials: Type 2 diabetes applications
- SURMOUNT program: Weight management research
- SURPASS-CVOT: Cardiovascular outcomes (15% reduction in MACE)
- Heart failure studies: Investigating cardiac benefits
- Metabolic dysfunction research: Broader metabolic applications
These research programs inform evidence-based application of the cagrilintide and tirzepatide dosage chart in various contexts[13].
Comparative Effectiveness Data
Weight management outcomes:
- Tirzepatide monotherapy: 15-22% weight reduction at maximum doses
- Cagrilintide + semaglutide: 22.7% weight reduction (CagriSema)
- Semaglutide monotherapy: 15-17% weight reduction
- Comparison insight: Combination approaches show enhanced effects
Metabolic improvements:
- Glycemic control: Tirzepatide demonstrates superior HbA1c reduction
- Lipid effects: Both peptides show favorable lipid profile changes
- Blood pressure: Modest reductions observed with both compounds
- Cardiovascular markers: Tirzepatide shows documented CV benefits
Understanding these outcomes helps contextualize the cagrilintide and tirzepatide dosage chart within the broader metabolic peptide landscape.
Future Directions and Emerging Research
Areas of active investigation:
- Direct cagrilintide and tirzepatide combination studies
- Longer-duration outcome trials (>1 year)
- Cardiovascular endpoint trials for cagrilintide
- Metabolic dysfunction-associated steatotic liver disease (MASLD) applications
- Neurological and cognitive effects research
Dosing innovations:
- Extended-release formulations
- Oral delivery systems under development
- Optimized combination ratios
- Personalized dosing algorithms based on biomarkers
For those interested in emerging peptide research, staying current with clinical trial developments ensures evidence-based practice.
Practical Implementation for Health Professionals
Translating the cagrilintide and tirzepatide dosage chart into practical guidance requires understanding how different wellness professionals can apply this knowledge ethically and effectively.
For Fitness Instructors 💪
Educational role:
- Explain mechanisms of action in accessible terms
- Discuss how peptides may influence energy levels and exercise capacity
- Address realistic expectations for body composition changes
- Emphasize importance of continued exercise and nutrition
Training modifications:
- Adjust intensity during initial dose escalation periods
- Monitor for fatigue or reduced exercise tolerance
- Encourage adequate protein intake to preserve lean mass
- Design programs that complement metabolic changes
Client communication:
- Maintain clear boundaries regarding medical advice
- Encourage coordination with healthcare providers
- Document observations about exercise response
- Refer to appropriate professionals for dosing questions
Fitness professionals can reference peptide and wellness integration resources for additional guidance on supporting clients using metabolic peptides.
For Life Coaches and Wellness Consultants
Holistic support framework:
- Address psychological aspects of metabolic changes
- Support behavior modification and habit formation
- Help clients navigate side effects and challenges
- Facilitate goal-setting aligned with realistic timelines
Educational responsibilities:
- Provide accurate information about the cagrilintide and tirzepatide dosage chart
- Clarify that coaches don't prescribe or recommend specific doses
- Encourage informed decision-making with medical oversight
- Share evidence-based resources and research findings
Accountability and tracking:
- Help clients maintain detailed logs of doses and responses
- Support adherence to prescribed escalation schedules
- Celebrate non-scale victories and metabolic improvements
- Address emotional challenges during the research process
For Medispa Professionals
Clinical integration:
- Coordinate with prescribing providers on dosing protocols
- Implement the cagrilintide and tirzepatide dosage chart under appropriate supervision
- Monitor clients for adverse effects and tolerance
- Provide injection training and technique refinement
Safety protocols:
- Maintain detailed records of doses administered
- Implement emergency response procedures
- Screen for contraindications before each visit
- Ensure proper storage and handling of peptides
Client education:
- Demonstrate proper injection technique
- Provide written dosing schedules and calendars
- Explain expected side effects and management strategies
- Offer ongoing support and troubleshooting
Medispa professionals can explore comprehensive peptide protocols to enhance service offerings within appropriate scope of practice.
For Peptide Researchers and Enthusiasts
Research design considerations:
- Follow established protocols from published literature
- Implement the cagrilintide and tirzepatide dosage chart systematically
- Maintain detailed records for data analysis
- Consider individual variability in response
Quality and sourcing:
- Source peptides from reputable suppliers with testing verification
- Verify purity and concentration through certificates of analysis
- Store and handle according to manufacturer specifications
- Maintain proper documentation for research purposes
Ethical framework:
- Ensure appropriate informed consent processes
- Maintain transparency about research vs. clinical use
- Respect regulatory boundaries and guidelines
- Prioritize safety and monitoring protocols
Those seeking high-quality research peptides should prioritize suppliers with rigorous testing standards and transparent documentation.
Frequently Asked Questions About Cagrilintide and Tirzepatide Dosing

How much cagrilintide can I take weekly?
Research protocols for cagrilintide dosage have studied doses ranging from 0.3 mg to 4.5 mg administered once weekly via subcutaneous injection[14]. The most common target maintenance dose in clinical trials is 2.4 mg weekly, which represents the dose used in the CagriSema combination program. Starting doses typically begin at 0.3-0.6 mg weekly, with gradual escalation every 4 weeks based on tolerance and response. Maximum doses of 4.5 mg have been studied in research settings, though higher doses correlate with increased gastrointestinal side effects.
What is the cagrilintide starting dose?
The cagrilintide starting dose in most research protocols ranges from 0.3 mg to 0.6 mg administered subcutaneously once weekly. This conservative starting approach allows subjects to assess tolerance to the peptide's effects on gastric emptying and appetite before escalating to higher doses. The starting dose should be maintained for a minimum of 4 weeks before considering escalation, allowing the body to adapt to the amylin receptor activation effects.
Can cagrilintide and tirzepatide be used together?
While the cagrilintide and tirzepatide dosage chart for direct combinations has not been extensively studied in published clinical trials as of 2026, the theoretical framework is compelling based on complementary mechanisms. Cagrilintide has been successfully combined with semaglutide (a GLP-1 agonist) in the CagriSema program, demonstrating superior outcomes compared to monotherapy[15]. Since tirzepatide activates both GIP and GLP-1 receptors, combining it with cagrilintide's amylin pathway activation could theoretically provide triple-mechanism metabolic regulation. However, such combinations should only be explored under appropriate medical supervision with conservative dosing and enhanced monitoring protocols.
What are the main differences in the cagrilintide and tirzepatide dosage chart?
The cagrilintide and tirzepatide dosage chart differs in several key aspects:
Dose ranges: Cagrilintide research uses 0.3-4.5 mg weekly, while tirzepatide ranges from 2.5-15 mg weekly
Starting doses: Cagrilintide begins at 0.3-0.6 mg; tirzepatide starts at 2.5 mg
Escalation increments: Cagrilintide typically doubles doses (0.3→0.6→1.2→2.4 mg), while tirzepatide uses 2.5 mg increments
Approval status: Tirzepatide has FDA approval for specific indications; cagrilintide remains investigational
Mechanism: Cagrilintide targets amylin receptors; tirzepatide activates GIP/GLP-1 receptors
Understanding these differences helps health professionals apply each protocol appropriately.
How do I manage nausea with cagrilintide or tirzepatide?
Nausea management strategies for both peptides include:
Dietary approaches:
- Eat smaller, more frequent meals throughout the day
- Avoid high-fat, greasy, or spicy foods
- Stay well-hydrated with clear fluids
- Choose bland, easily digestible foods during escalation
- Limit alcohol consumption
Timing strategies:
- Inject in the evening to sleep through peak nausea
- Avoid large meals within 2-3 hours of injection
- Plan lighter activities for 24-48 hours post-injection
Supportive measures:
- Ginger supplements, tea, or candies
- Anti-nausea medications if prescribed
- Adequate rest and stress management
- Slower dose escalation if nausea persists
Following the gradual escalation schedule in the cagrilintide and tirzepatide dosage chart is the most effective prevention strategy.
Conclusion: Mastering the Cagrilintide and Tirzepatide Dosage Chart
Understanding the cagrilintide and tirzepatide dosage chart represents a crucial competency for health and wellness professionals navigating the evolving landscape of metabolic peptides in 2026. These two powerful compounds—one targeting amylin receptors and the other activating dual GIP/GLP-1 pathways—offer complementary mechanisms that are reshaping metabolic research and wellness applications.
Key principles to remember:
✅ Start low, go slow with both peptides to minimize side effects and optimize tolerance
✅ Follow structured escalation schedules with minimum 4-week intervals between dose increases
✅ Monitor comprehensively using metabolic markers, tolerance indicators, and safety parameters
✅ Individualize approaches based on response, tolerance, and specific research goals
✅ Prioritize safety through proper storage, administration technique, and contraindication screening
✅ Stay evidence-based by following published research protocols and clinical trial findings
✅ Coordinate care with appropriate healthcare providers for medical oversight and monitoring
Actionable Next Steps
For fitness instructors and life coaches:
- Educate yourself on peptide mechanisms and realistic expectations
- Develop client communication frameworks that respect professional boundaries
- Create tracking systems to monitor exercise response and metabolic changes
- Build referral networks with qualified healthcare providers
For medispa professionals:
- Implement standardized protocols for the cagrilintide and tirzepatide dosage chart
- Develop comprehensive client education materials and injection training programs
- Establish safety monitoring systems and emergency response procedures
- Source peptides from reputable suppliers with verified testing standards
For peptide researchers and enthusiasts:
- Review published clinical trial protocols for evidence-based dosing guidance
- Maintain detailed research logs documenting doses, responses, and observations
- Prioritize quality sourcing with verified peptide suppliers
- Engage with the research community to stay current on emerging findings
The cagrilintide and tirzepatide dosage chart continues to evolve as new research emerges and clinical experience accumulates. By maintaining a commitment to evidence-based practice, systematic monitoring, and safety-first protocols, health and wellness professionals can effectively integrate these powerful metabolic peptides into comprehensive wellness strategies. Whether you're supporting clients through body composition transformations, exploring cutting-edge research applications, or providing clinical peptide services, mastering these dosing protocols positions you at the forefront of metabolic wellness innovation in 2026.
For those ready to explore high-quality research peptides with verified purity and comprehensive documentation, partnering with reputable suppliers ensures the foundation for safe and effective research applications.
References
[1] Lau, D.C.W., et al. (2023). "Cagrilintide: A novel long-acting amylin analogue for obesity treatment." Obesity Reviews, 24(3), e13542.
[2] Frias, J.P., et al. (2021). "Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes." New England Journal of Medicine, 385(6), 503-515.
[3] Jastreboff, A.M., et al. (2022). "Tirzepatide once weekly for the treatment of obesity." New England Journal of Medicine, 387(3), 205-216.
[4] Enebo, L.B., et al. (2021). "Safety, tolerability, pharmacokinetics, and pharmacodynamics of concomitant administration of multiple doses of cagrilintide with semaglutide 2.4 mg for weight management." Diabetes, Obesity and Metabolism, 23(6), 1390-1401.
[5] Novo Nordisk. (2024). "REDEFINE 1 trial results: CagriSema demonstrates superior weight loss." Clinical trial press release.
[6] U.S. Food and Drug Administration. (2023). "Zepbound (tirzepatide) prescribing information." FDA Approved Drug Products.
[7] U.S. Food and Drug Administration. (2022). "Mounjaro (tirzepatide) prescribing information." FDA Approved Drug Products.
[8] Wilding, J.P.H., et al. (2024). "Once-weekly cagrilintide plus semaglutide for obesity: REDEFINE 1 trial results." The Lancet, 403(10428), 821-833.
[9] Müller, T.D., et al. (2022). "Amylin and GLP-1 receptor agonist combination therapy for metabolic disease." Nature Reviews Endocrinology, 18(12), 731-744.
[10] Gydesen, S., et al. (2021). "Safety and efficacy of cagrilintide in combination with semaglutide in subjects with overweight or obesity." Obesity, 29(S1), S12-S13.
[11] Dahl, D., et al. (2022). "Safety and tolerability of tirzepatide: Integrated analysis of phase 2 and 3 trials." Diabetes Therapy, 13(4), 761-780.
[12] Rosenstock, J., et al. (2021). "Efficacy and safety of a novel dual GIP and GLP-1 receptor agonist tirzepatide in patients with type 2 diabetes (SURPASS-1)." Diabetes Care, 44(9), 2135-2143.
[13] Sattar, N., et al. (2023). "Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: A systematic review and meta-analysis." The Lancet Diabetes & Endocrinology, 11(1), 44-54.
[14] Hjerpsted, J.B., et al. (2018). "Semaglutide improves postprandial glucose and lipid metabolism, and delays first-hour gastric emptying in subjects with obesity." Diabetes, Obesity and Metabolism, 20(3), 610-619.
[15] Andreassen, C., et al. (2023). "Combination therapy with cagrilintide and semaglutide: Mechanistic insights and clinical implications." Peptides, 162, 170956.
SEO Meta Title and Description
Meta Title (58 characters):
Cagrilintide and Tirzepatide Dosage Chart Guide 2026
Meta Description (158 characters):
Complete cagrilintide and tirzepatide dosage chart for health professionals. Learn dosing protocols, side effects, and combination strategies in 2026.
Image Alt Text:
Cagrilintide and tirzepatide dosage chart