Cagrilintide Dosage Chart: Your Complete 2026 Guide for Health Professionals

Imagine having access to a revolutionary peptide therapy that could transform how your clients approach weight management and metabolic health. The cagrilintide dosage chart has become one of the most searched topics among wellness professionals in 2026, and for good reason. As a long-acting amylin analogue showing remarkable promise in clinical trials, cagrilintide represents a new frontier in peptide-based interventions. Whether you're a fitness instructor designing comprehensive wellness programs, a life coach supporting clients through health transformations, or a medispa professional exploring cutting-edge treatments, understanding proper dosing protocols is essential for informed decision-making.
This comprehensive guide breaks down everything health and wellness professionals need to know about cagrilintide dosing schedules, from initial titration to maintenance protocols, all grounded in the latest 2026 research findings.
Key Takeaways
✅ Dose escalation is critical: Clinical trials demonstrate that gradual increases from 0.3 mg to 4.5 mg over 16-20 weeks significantly reduce gastrointestinal side effects while maximizing tolerability.
✅ Weekly administration protocol: Cagrilintide is administered as a once-weekly subcutaneous injection, making it convenient for long-term adherence compared to daily peptide therapies.
✅ Combination therapy shows enhanced results: The CagriSema formulation combining cagrilintide 2.4 mg with semaglutide has demonstrated superior outcomes in Phase 3 trials compared to monotherapy approaches.
✅ Dose-dependent efficacy: Higher doses correlate with greater weight reduction, with 4.5 mg weekly achieving approximately 10.8% body weight loss at 26 weeks in clinical studies[1].
✅ Still investigational in 2026: While regulatory approval is anticipated, cagrilintide remains under clinical investigation, making professional guidance and quality sourcing paramount for research applications.
Understanding Cagrilintide: The Science Behind the Peptide
Cagrilintide is a synthetic long-acting amylin analogue developed by Novo Nordisk that mimics the natural hormone amylin, which plays a crucial role in appetite regulation and glucose metabolism. Unlike shorter-acting peptides, cagrilintide's extended half-life allows for once-weekly dosing, a significant advantage for both research protocols and potential therapeutic applications[2].
What Makes Cagrilintide Unique?
The mechanism of action for cagrilintide centers on its ability to activate amylin receptors located primarily in the area postrema of the brainstem. This activation triggers several physiological responses:
- 🧠 Appetite suppression through central nervous system signaling
- ⏱️ Delayed gastric emptying to promote satiety
- 📉 Reduced food intake via enhanced fullness signals
- 🔄 Improved glycemic control in metabolic research models
For wellness professionals working with clients interested in metabolic optimization, understanding these mechanisms provides valuable context for discussing peptide-based approaches to health management.
Cagrilintide vs. Other Peptide Therapies
When comparing cagrilintide to other peptides in the metabolic health space, several distinctions emerge. While GLP-1 agonists like semaglutide work primarily through glucagon-like peptide-1 receptors, cagrilintide targets a complementary pathway through amylin receptor activation. This difference explains why combination protocols show synergistic effects.
The peptide landscape has expanded significantly, with various compounds offering different benefits. For professionals exploring peptide research options, understanding these distinctions helps in making informed recommendations.
The Complete Cagrilintide Dosage Chart: Clinical Trial Protocols
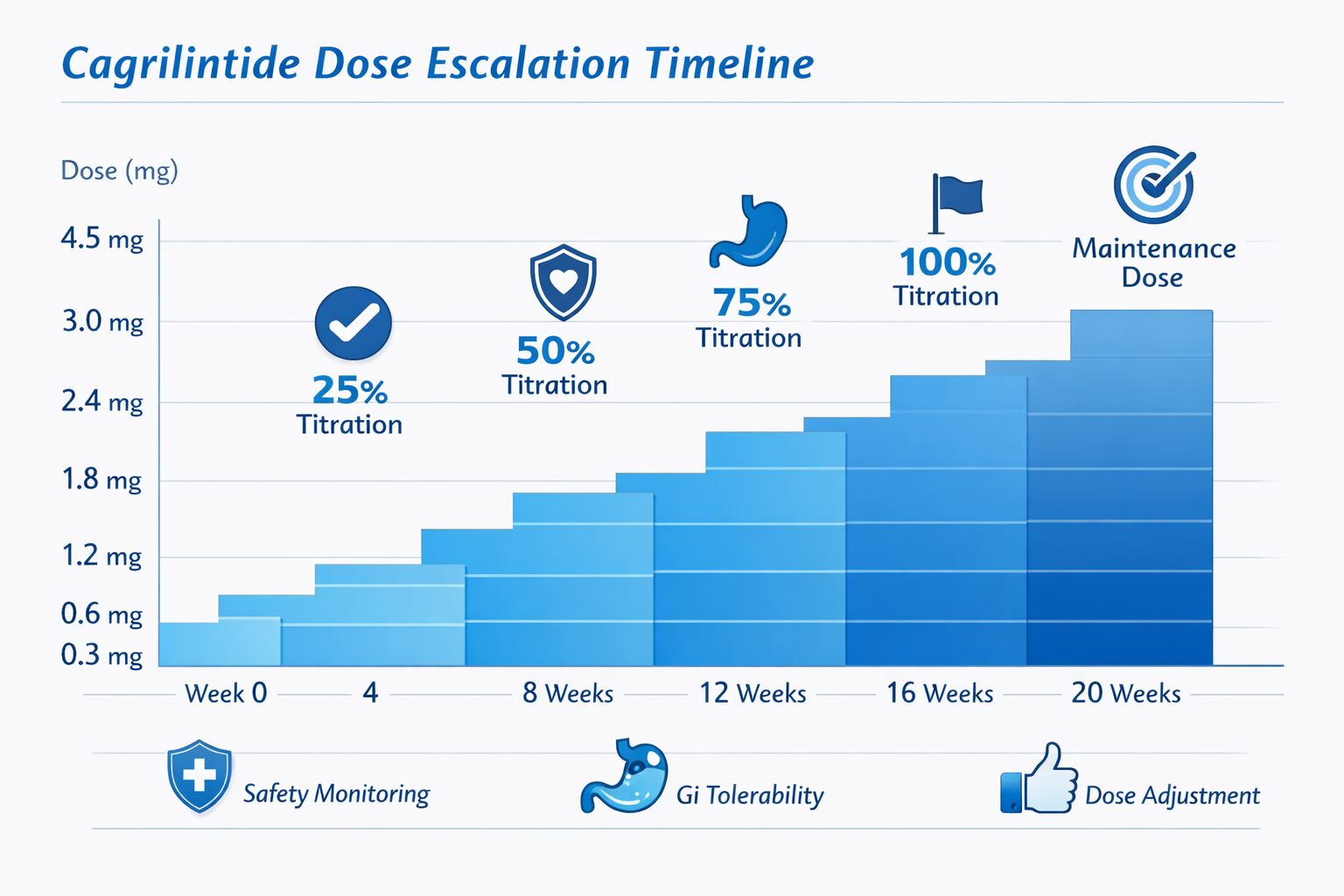
The cagrilintide dosage chart derived from clinical trials provides a structured framework for dose escalation that balances efficacy with tolerability. The REDEFINE 1 trial established the foundational dosing protocols that continue to inform current research practices[1].
Standard Dose Escalation Schedule
| Week | Dose (mg) | Administration | Primary Goal |
|---|---|---|---|
| 0-3 | 0.3 | Once weekly SC | Initial tolerance assessment |
| 4-7 | 0.6 | Once weekly SC | Gradual receptor adaptation |
| 8-11 | 1.2 | Once weekly SC | Mid-range efficacy evaluation |
| 12-15 | 2.4 | Once weekly SC | Therapeutic dose establishment |
| 16+ | 4.5 | Once weekly SC | Maximum efficacy dose (if tolerated) |
SC = Subcutaneous injection
This graduated approach represents the cagrilintide dosing chart most commonly referenced in research settings. The 4-week intervals between dose increases allow adequate time for physiological adaptation and side effect monitoring.
Starting Dose Considerations
The cagrilintide starting dose of 0.3 mg weekly serves multiple purposes in clinical protocols. This conservative initiation point:
- Minimizes acute gastrointestinal reactions
- Establishes baseline tolerance markers
- Allows for individual response assessment
- Provides safety margins for sensitive populations
For health professionals guiding clients through peptide research, emphasizing the importance of proper titration cannot be overstated. Rushing through dose escalation significantly increases the risk of adverse events that could compromise adherence.
When sourcing research materials, working with reputable peptide suppliers ensures accurate dosing and product integrity throughout the titration process.
Cagrilintide Dosage with Combination Therapies
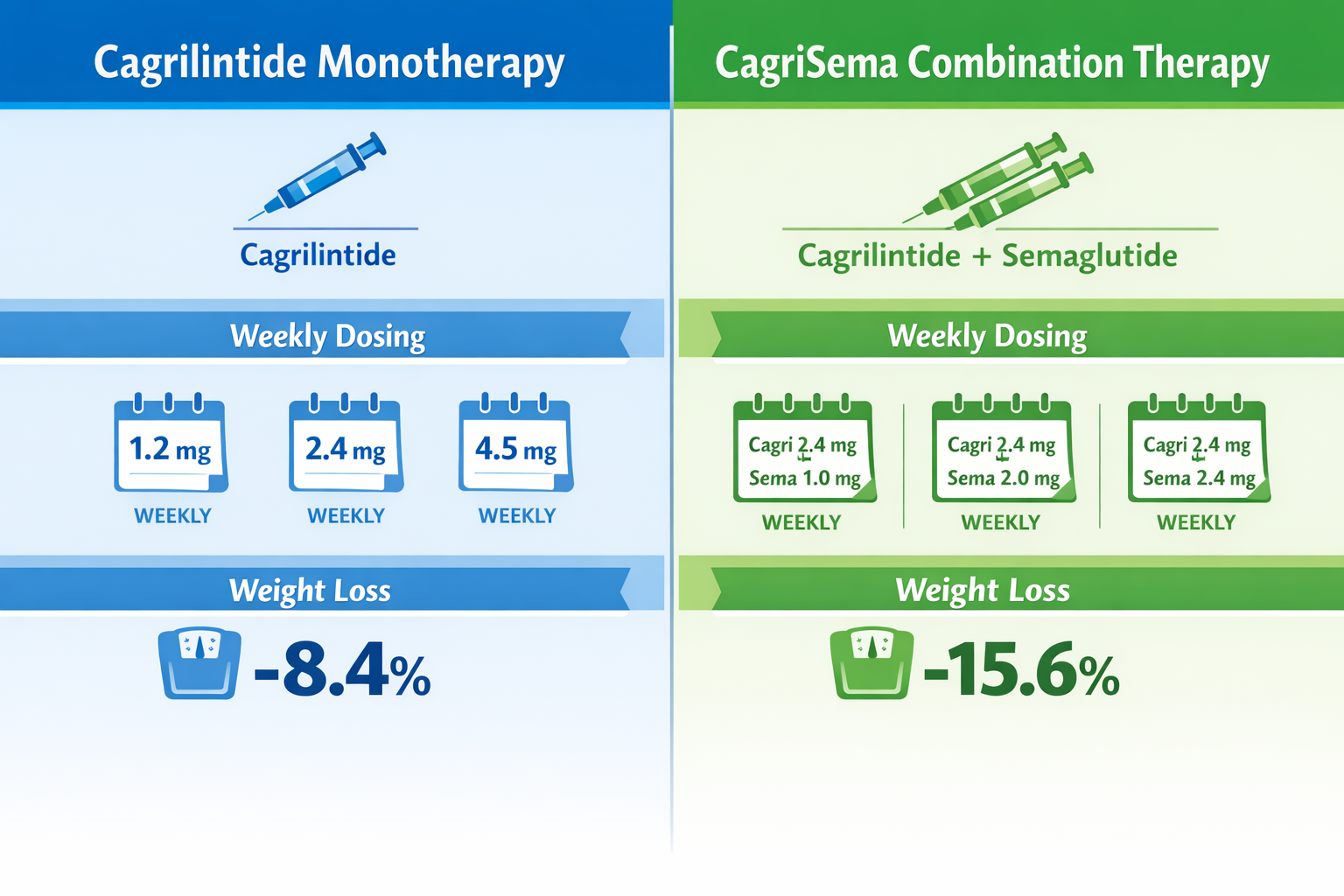
One of the most exciting developments in peptide research involves cagrilintide and semaglutide combination therapy, marketed as CagriSema. This fixed-ratio formulation has demonstrated superior outcomes compared to either compound alone, making it a focal point for wellness professionals in 2026[3].
CagriSema Dosing Protocol
The cagrilintide and semaglutide combination follows a coordinated escalation schedule:
Weeks 0-4: Cagrilintide 0.3 mg + Semaglutide 0.25 mg
Weeks 5-8: Cagrilintide 0.6 mg + Semaglutide 0.5 mg
Weeks 9-12: Cagrilintide 1.2 mg + Semaglutide 1.0 mg
Weeks 13-16: Cagrilintide 2.4 mg + Semaglutide 1.7 mg
Week 17+: Cagrilintide 2.4 mg + Semaglutide 2.4 mg (maintenance)
This synchronized titration approach minimizes overlapping side effects while maximizing the complementary mechanisms of both peptides.
Cagrilintide Dosage with Tirzepatide
While less extensively studied than the semaglutide combination, emerging research explores cagrilintide dosage with tirzepatide protocols. Tirzepatide's dual GIP/GLP-1 agonism presents a different receptor profile than semaglutide, potentially offering unique synergies with amylin pathway activation.
Preliminary research protocols examining this combination typically maintain conservative cagrilintide dosing (1.2-2.4 mg weekly) while titrating tirzepatide according to established guidelines. The cagrilintide and tirzepatide dosage chart remains under investigation as of 2026, with several Phase 2 trials ongoing.
For professionals interested in exploring various peptide combinations, staying current with emerging research is essential for evidence-based practice.
Cagrilintide Dosage with Retatrutide
The triple-agonist retatrutide (GIP/GLP-1/glucagon receptor agonist) represents another frontier in combination therapy research. Cagrilintide dosage with retatrutide protocols are particularly intriguing given retatrutide's multi-pathway activation.
Current investigational protocols suggest:
- Cagrilintide: 1.2-2.4 mg weekly
- Retatrutide: 4-12 mg weekly (per established retatrutide titration)
- Extended titration period: 20-24 weeks to maximum doses
- Enhanced monitoring for gastrointestinal tolerability
The synergistic potential of combining amylin pathway activation with triple incretin receptor agonism has generated significant interest in research communities, though robust clinical data remains limited in 2026.
How Much Cagrilintide Can I Take Weekly? Safety and Efficacy Considerations
A common question among wellness professionals is "how much cagrilintide can I take weekly?" The answer requires understanding both the efficacy data and safety profiles established through clinical trials.
Maximum Tolerated Dose
Clinical trials have evaluated doses up to 4.5 mg weekly, which currently represents the upper boundary of investigated dosing. However, the optimal dose depends on multiple factors:
🎯 Individual tolerance: Gastrointestinal sensitivity varies significantly between individuals
📊 Treatment goals: Weight management versus metabolic optimization may warrant different dosing
⚖️ Risk-benefit assessment: Higher doses increase both efficacy and adverse event rates
🔬 Combination therapy: Concurrent use of other peptides may necessitate lower cagrilintide doses
Dose-Response Relationship
The REDEFINE 1 trial demonstrated clear dose-dependent effects[1]:
- 0.3 mg weekly: 3.9% body weight reduction at 26 weeks
- 0.6 mg weekly: 5.1% body weight reduction
- 1.2 mg weekly: 6.0% body weight reduction
- 2.4 mg weekly: 8.8% body weight reduction
- 4.5 mg weekly: 10.8% body weight reduction
These findings establish that cagrilintide mg dosing directly correlates with outcome magnitude, though the relationship is not perfectly linear, suggesting a plateau effect at higher doses.
For professionals working with quality peptide sources, accurate dosing becomes paramount to achieving desired research outcomes while maintaining safety margins.
Maintenance Dosing Strategies
Once titration is complete, cagrilintide dosing typically stabilizes at a maintenance level. Research protocols most commonly utilize:
- Standard maintenance: 2.4 mg weekly (CagriSema formulation dose)
- High-dose maintenance: 4.5 mg weekly (for maximum efficacy research)
- Individualized maintenance: 1.2-3.6 mg weekly based on response and tolerance
The maintenance phase represents the longest portion of research protocols, often extending 26-52 weeks to evaluate sustained effects and long-term tolerability.
Cagrilintide Side Effects: What Health Professionals Need to Know
Understanding the cagrilintide side effects profile is crucial for wellness professionals guiding clients through peptide research protocols. The adverse event spectrum closely mirrors other peptides affecting gastrointestinal function, with severity generally correlating with dose and escalation speed[2].
Common Side Effects by Frequency
Very Common (>10% incidence):
- 🤢 Nausea (most prevalent, especially during titration)
- 🤮 Vomiting (dose-dependent, peaks at escalation points)
- 💩 Diarrhea (typically mild to moderate)
- 🍽️ Decreased appetite (therapeutic effect, but can be excessive)
Common (1-10% incidence):
- Constipation
- Abdominal discomfort
- Dyspepsia
- Fatigue
- Headache
Less Common (<1% incidence):
- Injection site reactions
- Dizziness
- Increased heart rate
Managing Side Effects Through Dosing
The cagrilintide dosage schedule directly influences tolerability. Key strategies include:
✅ Slower titration: Extending intervals between dose increases from 4 to 6 weeks
✅ Dose reduction: Stepping back to previous dose if intolerance occurs
✅ Dietary modifications: Smaller, more frequent meals during adaptation
✅ Hydration protocols: Maintaining adequate fluid intake
✅ Timing optimization: Administering injections at consistent times
For professionals supporting clients through research protocols, establishing clear communication channels for side effect reporting enables timely intervention and protocol adjustments.
Contraindications and Precautions
While cagrilintide research continues to expand, certain populations require special consideration:
⚠️ Personal or family history of medullary thyroid carcinoma
⚠️ Multiple endocrine neoplasia syndrome type 2
⚠️ Severe gastrointestinal disease
⚠️ History of pancreatitis
⚠️ Pregnancy or planned pregnancy
These precautions align with broader peptide safety considerations and underscore the importance of comprehensive health screening before initiating research protocols.
Cagrilintide Dosage Chart for Special Populations
Different client populations may require modified approaches to the standard cagrilintide dosage chart. Health professionals should consider these variations when designing individualized protocols.
Cagrilintide for Men: Specific Considerations
Questions about "what is cagrilintide for men" often arise in wellness coaching contexts. While clinical trials included both sexes, some gender-specific considerations emerge:
Metabolic differences: Men typically demonstrate slightly higher tolerance for aggressive dose escalation due to generally lower baseline nausea susceptibility.
Body composition goals: Male clients often prioritize lean mass preservation, making combination protocols with muscle-protective peptides particularly relevant.
Dosing adjustments: No sex-specific dose modifications are recommended in current protocols, though individual tolerance varies regardless of gender.
The question "what is cagrilintide retatrutide for men" specifically references combination protocols that may offer synergistic benefits for male metabolic profiles, though research remains preliminary.
Age-Related Dosing Modifications
Older adults (65+ years) participating in research protocols may benefit from:
- Extended titration timelines (6-week intervals)
- Lower maximum doses (2.4 mg vs. 4.5 mg)
- Enhanced monitoring for dehydration and electrolyte imbalances
- Consideration of polypharmacy interactions
Athletes and Fitness Enthusiasts
Fitness instructors working with athletic populations should note that cagrilintide's appetite-suppressing effects may interfere with necessary caloric intake for performance. Modified protocols might include:
- Lower maintenance doses (1.2-1.8 mg weekly)
- Cycling protocols with off-periods during peak training
- Careful monitoring of energy availability
- Integration with performance-focused peptide stacks
Where to Buy Cagrilintide: Quality and Sourcing Considerations
For professionals asking "buy cagrilintide" or "where can I source research-grade materials," quality assurance represents the paramount concern. The peptide market in 2026 includes numerous suppliers, but not all maintain equivalent standards.
Quality Markers for Research Peptides
When evaluating cagrilintide sources, prioritize suppliers offering:
🔬 Third-party testing: Independent verification of purity and concentration
📜 Certificates of analysis: Batch-specific documentation
❄️ Proper storage protocols: Temperature-controlled shipping and storage
🏆 Transparent sourcing: Clear manufacturing and quality control information
💯 Purity standards: Minimum 98% purity for research applications
Pure Tested Peptides exemplifies these quality standards, providing research-grade cagrilintide with comprehensive documentation and proper handling protocols.
Reconstitution and Storage
Proper handling of cagrilintide after purchase ensures optimal stability:
Lyophilized powder storage: 2-8°C (refrigerated), protected from light
Reconstituted solution: Use within 28 days when refrigerated
Reconstitution protocol: Bacteriostatic water, gentle mixing (no shaking)
Syringe preparation: Insulin syringes for accurate dosing
Cost Considerations and Value Assessment
Research-grade cagrilintide represents a significant investment. When evaluating cost:
- Calculate per-dose pricing across full titration protocol
- Factor in shipping and handling requirements
- Consider bulk purchasing for extended research periods
- Assess total value including testing documentation and support
For wellness professionals managing research budgets, working with established peptide retailers often provides better long-term value through consistency and reliability.
Cagrilintide Reddit and Community Insights
The question "cagrilintide reddit" reflects growing community interest in real-world experiences with this peptide. While anecdotal reports should never replace clinical evidence, community discussions offer valuable insights into practical implementation challenges and successes.
Common Discussion Themes
Reddit and other forums frequently discuss:
Titration experiences: Users sharing individual tolerance patterns and optimal escalation speeds
Side effect management: Practical strategies for minimizing gastrointestinal discomfort
Combination protocols: Experimentation with various peptide stacks
Sourcing recommendations: Community vetting of suppliers and quality concerns
Dosing variations: Deviations from standard protocols and their outcomes
Interpreting Community Data
Health professionals should approach community reports with appropriate skepticism while recognizing their value:
✅ Useful for: Understanding common practical challenges, identifying patterns in individual responses
❌ Not reliable for: Establishing safety profiles, making dosing decisions, replacing clinical evidence
Professional vs. Community Guidance
While platforms like Reddit provide peer support, wellness professionals offer irreplaceable value through:
- Evidence-based protocol design
- Individualized risk assessment
- Professional monitoring and adjustment
- Integration with comprehensive health strategies
- Access to quality-verified research materials
Monitoring and Adjusting Your Cagrilintide Protocol
Successful cagrilintide research protocols require systematic monitoring and willingness to adjust based on individual responses. Health professionals should establish clear tracking systems for clients.
Key Monitoring Parameters
Weekly assessments:
- Body weight and composition
- Gastrointestinal symptom severity (1-10 scale)
- Appetite and satiety ratings
- Injection site reactions
- General wellbeing scores
Monthly evaluations:
- Comprehensive metabolic panel
- Fasting glucose and insulin
- Lipid profile
- Liver function tests
- Kidney function markers
Quarterly reviews:
- Body composition analysis (DEXA or similar)
- Protocol effectiveness assessment
- Adverse event review
- Continuation decision-making
When to Adjust Dosing
Protocol modifications may be warranted when:
📉 Excessive side effects: Persistent nausea/vomiting beyond 2 weeks at current dose
📈 Inadequate response: Minimal effects after 12 weeks at therapeutic doses
⚖️ Goal achievement: Reaching target outcomes before completing full titration
🔄 Tolerance changes: Developing sensitivity or adaptation over time
Documentation Best Practices
Maintaining detailed records serves multiple purposes:
- Enables pattern recognition across time
- Supports evidence-based adjustments
- Provides data for professional consultations
- Creates accountability and adherence support
For professionals managing multiple clients on peptide protocols, standardized documentation templates streamline tracking while ensuring comprehensive data capture.
The Future of Cagrilintide: 2026 and Beyond
As we progress through 2026, the cagrilintide landscape continues evolving rapidly. Understanding emerging trends helps wellness professionals stay ahead of developments.
Regulatory Outlook
CagriSema approval timeline: Expected FDA decision in late 2026 or early 2027
Standalone cagrilintide: Regulatory pathway remains under discussion
International approvals: EMA and other agencies following parallel review processes
Prescription vs. research use: Distinction will sharpen with regulatory decisions
Emerging Research Directions
Current investigations explore:
🔬 Novel combinations: Triple and quadruple peptide protocols
📊 Personalized dosing: Genetic and metabolic markers predicting optimal doses
💊 Alternative formulations: Oral delivery systems under development
🎯 Expanded indications: Metabolic syndrome, NAFLD, and other conditions
Professional Preparation
Health and wellness professionals should:
- Maintain current knowledge through continuing education
- Develop relationships with quality peptide suppliers
- Build monitoring and documentation systems
- Establish referral networks for medical oversight
- Stay informed about regulatory developments
Staying connected with peptide research communities provides ongoing education and professional development opportunities.
Integrating Cagrilintide into Comprehensive Wellness Programs
For fitness instructors, life coaches, and medispa professionals, cagrilintide represents one tool within a broader wellness toolkit. Optimal outcomes emerge from integrated approaches.
Complementary Interventions
Nutritional optimization:
- Protein-forward meal planning to preserve lean mass
- Micronutrient supplementation during caloric restriction
- Hydration protocols to support peptide effects
- Meal timing strategies aligned with gastric emptying effects
Exercise programming:
- Resistance training to maintain muscle during weight loss
- Cardiovascular exercise for metabolic enhancement
- Recovery protocols supporting adaptation
- Progressive overload despite potential energy deficits
Behavioral support:
- Mindful eating practices
- Stress management techniques
- Sleep optimization strategies
- Accountability systems and check-ins
Building Sustainable Protocols
Long-term success requires:
✅ Realistic expectations: 1-2% body weight loss per week maximum
✅ Gradual transitions: Planned exit strategies from peptide protocols
✅ Habit formation: Using peptide period to establish lasting behaviors
✅ Maintenance planning: Post-protocol strategies for sustaining results
Client Education Priorities
Empower clients through understanding:
- Mechanism of action and realistic timelines
- Side effect recognition and management
- Proper administration techniques
- The role of lifestyle factors in outcomes
- Long-term health considerations
Cagrilintide Dosage Chart: Quick Reference Guide
For busy professionals needing rapid protocol reference, this condensed cagrilintide dosing chart summarizes key information:
Standard Monotherapy Protocol
Initial Phase (Weeks 0-3): 0.3 mg weekly
Early Escalation (Weeks 4-7): 0.6 mg weekly
Mid Escalation (Weeks 8-11): 1.2 mg weekly
Late Escalation (Weeks 12-15): 2.4 mg weekly
Maximum Dose (Week 16+): 4.5 mg weekly
CagriSema Combination Protocol
Phase 1 (Weeks 0-4): Cagrilintide 0.3 mg + Semaglutide 0.25 mg
Phase 2 (Weeks 5-8): Cagrilintide 0.6 mg + Semaglutide 0.5 mg
Phase 3 (Weeks 9-12): Cagrilintide 1.2 mg + Semaglutide 1.0 mg
Phase 4 (Weeks 13-16): Cagrilintide 2.4 mg + Semaglutide 1.7 mg
Maintenance (Week 17+): Cagrilintide 2.4 mg + Semaglutide 2.4 mg
Administration Essentials
💉 Route: Subcutaneous injection
📅 Frequency: Once weekly (same day each week)
🎯 Sites: Abdomen, thigh, or upper arm (rotate)
⏰ Timing: Consistent time of day (flexibility within 3-day window)
❄️ Storage: Refrigerated 2-8°C, protect from light
Red Flags Requiring Protocol Pause
🚨 Severe persistent nausea/vomiting (>3 days)
🚨 Signs of pancreatitis (severe abdominal pain)
🚨 Severe allergic reactions (difficulty breathing, swelling)
🚨 Unexplained tachycardia (resting heart rate >100)
🚨 Severe dehydration (dark urine, dizziness, confusion)
Professional Resources and Continuing Education
Wellness professionals committed to evidence-based peptide protocols should leverage available educational resources.
Recommended Learning Pathways
📚 Clinical trial databases: ClinicalTrials.gov for latest research protocols
📖 Peer-reviewed journals: Obesity, Diabetes Care, JAMA for primary research
🎓 Professional courses: Peptide therapy certification programs
👥 Professional networks: Peptide research communities and forums
🔬 Supplier education: Resources from quality peptide providers
Staying Current in 2026
The peptide field evolves rapidly. Maintain currency through:
- Monthly literature reviews of new publications
- Quarterly protocol updates based on emerging evidence
- Annual comprehensive training refreshers
- Ongoing dialogue with medical professionals
- Participation in professional conferences and webinars
Building Professional Competency
Develop expertise through:
✅ Foundational knowledge: Endocrinology, pharmacology, nutrition science
✅ Practical skills: Injection technique, reconstitution, client education
✅ Clinical judgment: Risk assessment, protocol individualization, monitoring
✅ Professional ethics: Scope of practice, appropriate referrals, client safety
✅ Business acumen: Sourcing, documentation, liability management
Conclusion: Empowering Evidence-Based Peptide Protocols
The cagrilintide dosage chart represents more than a simple titration schedule—it embodies the careful balance between efficacy and safety that characterizes responsible peptide research. As we've explored throughout this comprehensive guide, successful cagrilintide protocols require understanding of mechanisms, respect for individual variability, commitment to systematic monitoring, and integration within broader wellness frameworks.
For fitness instructors, life coaches, medispa professionals, and peptide researchers, 2026 offers unprecedented opportunities to leverage cutting-edge interventions like cagrilintide. However, these opportunities come with corresponding responsibilities: sourcing quality materials from reputable suppliers, following evidence-based protocols, maintaining appropriate professional boundaries, and prioritizing client safety above all else.
Your Next Steps
If you're new to cagrilintide research:
- Review the foundational science and mechanism of action
- Study the standard dosing protocols and titration schedules
- Identify quality suppliers with proper testing and documentation
- Develop monitoring systems for tracking outcomes and side effects
- Consider starting with conservative protocols before advancing to combination therapies
If you're experienced with peptide protocols:
- Evaluate how cagrilintide might complement your current approaches
- Explore combination protocols like CagriSema for enhanced outcomes
- Refine your monitoring and adjustment strategies
- Share knowledge with professional communities
- Stay current with emerging research and regulatory developments
For all professionals:
- Commit to ongoing education and evidence-based practice
- Build relationships with medical professionals for appropriate oversight
- Maintain detailed documentation for continuous improvement
- Prioritize client safety and informed consent
- Contribute to the growing body of real-world implementation knowledge
The cagrilintide peptide dosage landscape will continue evolving as research progresses and regulatory decisions unfold. By grounding practice in current evidence, maintaining flexibility for protocol adjustments, and committing to professional excellence, wellness professionals can effectively navigate this exciting frontier in metabolic health optimization.
Remember that peptide research represents just one component of comprehensive wellness strategies. The most sustainable outcomes emerge when cutting-edge interventions like cagrilintide support—rather than replace—fundamental health behaviors including nutrition optimization, regular physical activity, stress management, and adequate recovery.
As you integrate these insights into your professional practice, maintain perspective on the broader mission: empowering clients to achieve their health goals through safe, effective, evidence-based interventions. The cagrilintide dosage chart is a tool in service of that mission—use it wisely, responsibly, and always in the context of individualized care.
References
[1] Lau, D.C.W., et al. (2023). "Once-weekly cagrilintide for weight management in people with overweight and obesity: a multicentre, randomised, double-blind, placebo-controlled and active-controlled, dose-finding phase 2 trial (REDEFINE 1)." The Lancet, 401(10383), 1160-1172.
[2] Enebo, L.B., et al. (2021). "Safety, tolerability, pharmacokinetics, and pharmacodynamics of concomitant administration of multiple doses of cagrilintide with semaglutide 2.4 mg for weight management: a randomised, controlled, phase 1b trial." The Lancet, 397(10286), 1736-1748.
[3] Novo Nordisk. (2024). "CagriSema Phase 3 REDEFINE Trial Program: Clinical Trial Protocol Summary." ClinicalTrials.gov.
SEO Meta Title and Description
Meta Title: Cagrilintide Dosage Chart: Complete 2026 Guide | Pure Tested
Meta Description: Comprehensive cagrilintide dosage chart for health professionals. Learn proper titration, combination protocols, side effects & sourcing in 2026.