Cagrilintide Dose: The Complete 2026 Guide for Health Professionals

Imagine a single weekly injection that could revolutionize weight management and metabolic health for millions of people worldwide. That's the promise behind cagrilintide dose protocols currently making waves in clinical research and the wellness industry. As a long-acting amylin analogue developed by Novo Nordisk, cagrilintide represents a breakthrough approach to appetite regulation and weight loss—but understanding the proper cagrilintide dose is essential for anyone working in health, wellness, or peptide research.
This comprehensive guide explores everything health professionals, fitness instructors, life coaches, and medispa practitioners need to know about cagrilintide dosing in 2026. Whether you're researching peptide therapies for your practice or helping clients navigate emerging weight management options, understanding the science behind cagrilintide dosing protocols is crucial.
Key Takeaways
✅ Standard cagrilintide dose: The maintenance dose is 2.4 mg administered subcutaneously once weekly, established through Phase 3 clinical trials
✅ Dose escalation matters: Gradual titration over 16-20 weeks minimizes gastrointestinal side effects and improves tolerability
✅ Combination therapy shows superior results: CagriSema (cagrilintide 2.4 mg + semaglutide 2.4 mg) demonstrated 22.7% mean weight loss at 68 weeks
✅ Dose-dependent effects observed: Phase 2 trials tested doses from 0.3 mg to 4.5 mg, with 2.4 mg offering optimal balance of efficacy and safety
✅ Proper administration technique: Injection sites include abdomen, thigh, or upper arm with rotation recommended to prevent lipodystrophy
What Is Cagrilintide and How Does It Work?
Cagrilintide is a synthetic long-acting amylin analogue peptide designed to mimic and enhance the effects of natural amylin, a hormone co-secreted with insulin from pancreatic beta cells. Unlike traditional weight loss medications, cagrilintide works through multiple receptor pathways to reduce appetite and food intake.
Mechanism of Action
Cagrilintide activates three key receptor types:
- Amylin receptors: Slow gastric emptying and promote satiety
- Calcitonin receptors: Contribute to metabolic regulation
- CGRP receptors (Calcitonin Gene-Related Peptide): Influence appetite control centers in the brain
This multi-receptor activation creates a powerful appetite-suppressing effect that goes beyond single-pathway medications. The peptide's long-acting formulation allows for convenient once-weekly dosing, making it practical for real-world use.
For professionals exploring various peptide research options, understanding cagrilintide's unique mechanism helps contextualize its role in the broader landscape of metabolic peptides.
Understanding Cagrilintide Dose: Clinical Trial Findings
The cagrilintide dose used in clinical practice has been carefully refined through extensive Phase 2 and Phase 3 research. Understanding this dose-finding process provides valuable context for health professionals.
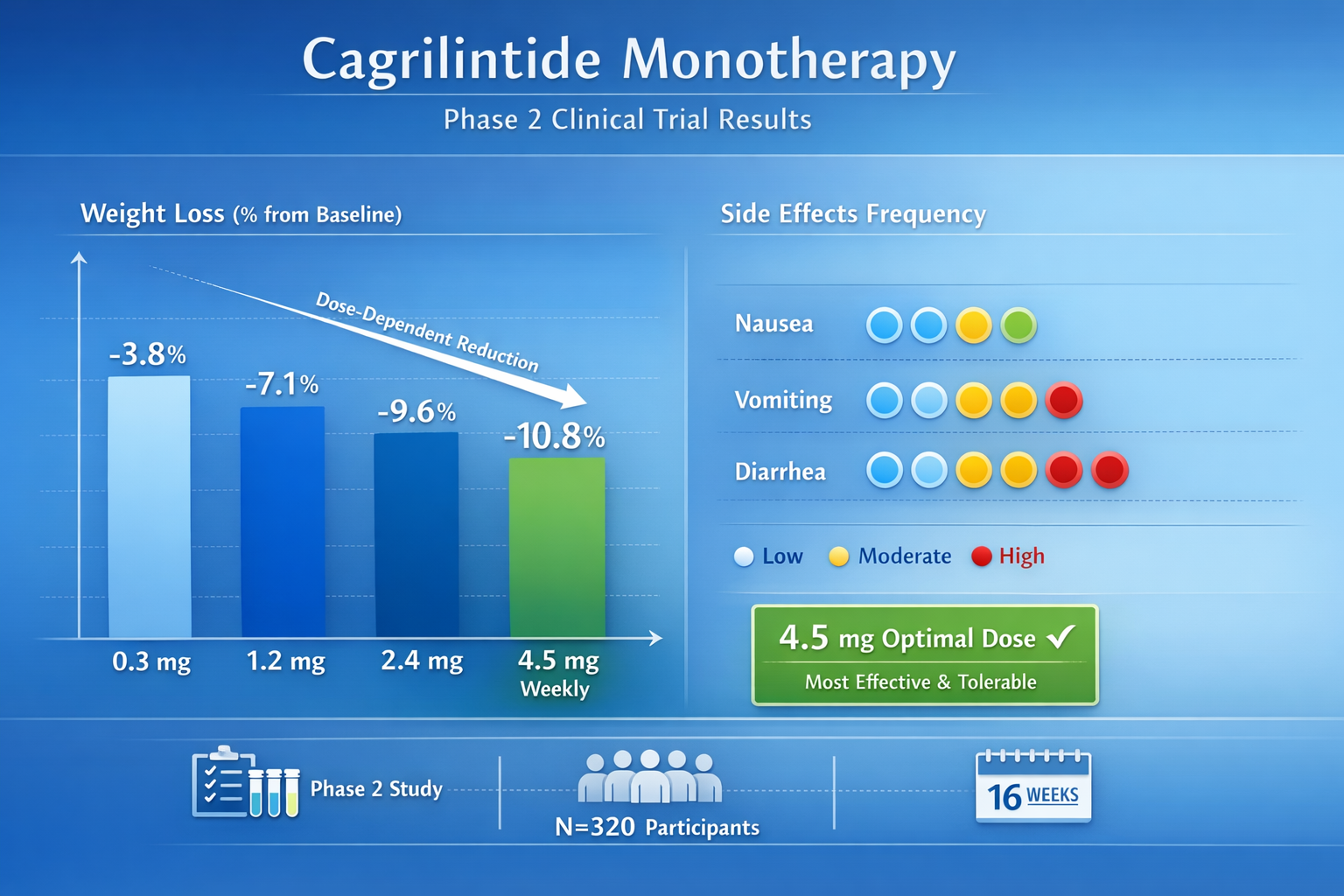
Phase 2 Dose-Ranging Studies
Early Phase 2 clinical trials evaluated cagrilintide monotherapy across a wide dose range:
| Dose Level | Administration | Key Findings |
|---|---|---|
| 0.3 mg | Weekly subcutaneous | Minimal weight loss, excellent tolerability |
| 1.2 mg | Weekly subcutaneous | Moderate weight loss, good tolerability |
| 2.4 mg | Weekly subcutaneous | Significant weight loss, acceptable side effects |
| 4.5 mg | Weekly subcutaneous | Maximum weight loss, higher GI adverse events |
Research demonstrated clear dose-dependent weight loss across this range. However, doses above 2.4 mg showed diminishing returns due to increased gastrointestinal side effects, including nausea, vomiting, and diarrhea.
Phase 3 Standard Cagrilintide Dose
Based on Phase 2 findings, the standard cagrilintide dose for Phase 3 trials was established at:
🎯 2.4 mg administered subcutaneously once weekly
This dose represents the optimal balance between:
- Efficacy: Substantial weight loss and metabolic improvements
- Safety: Manageable side effect profile
- Tolerability: Acceptable patient adherence rates
The REDEFINE 1 trial, which evaluated cagrilintide in combination with semaglutide (CagriSema), used this 2.4 mg dose and demonstrated remarkable results. Participants achieved a mean weight loss of 22.7% at 68 weeks when combining cagrilintide 2.4 mg with semaglutide 2.4 mg.
For those researching metabolic peptides, similar dose-finding principles apply across different compounds, as seen with AOD-9604 peptide dosage protocols.
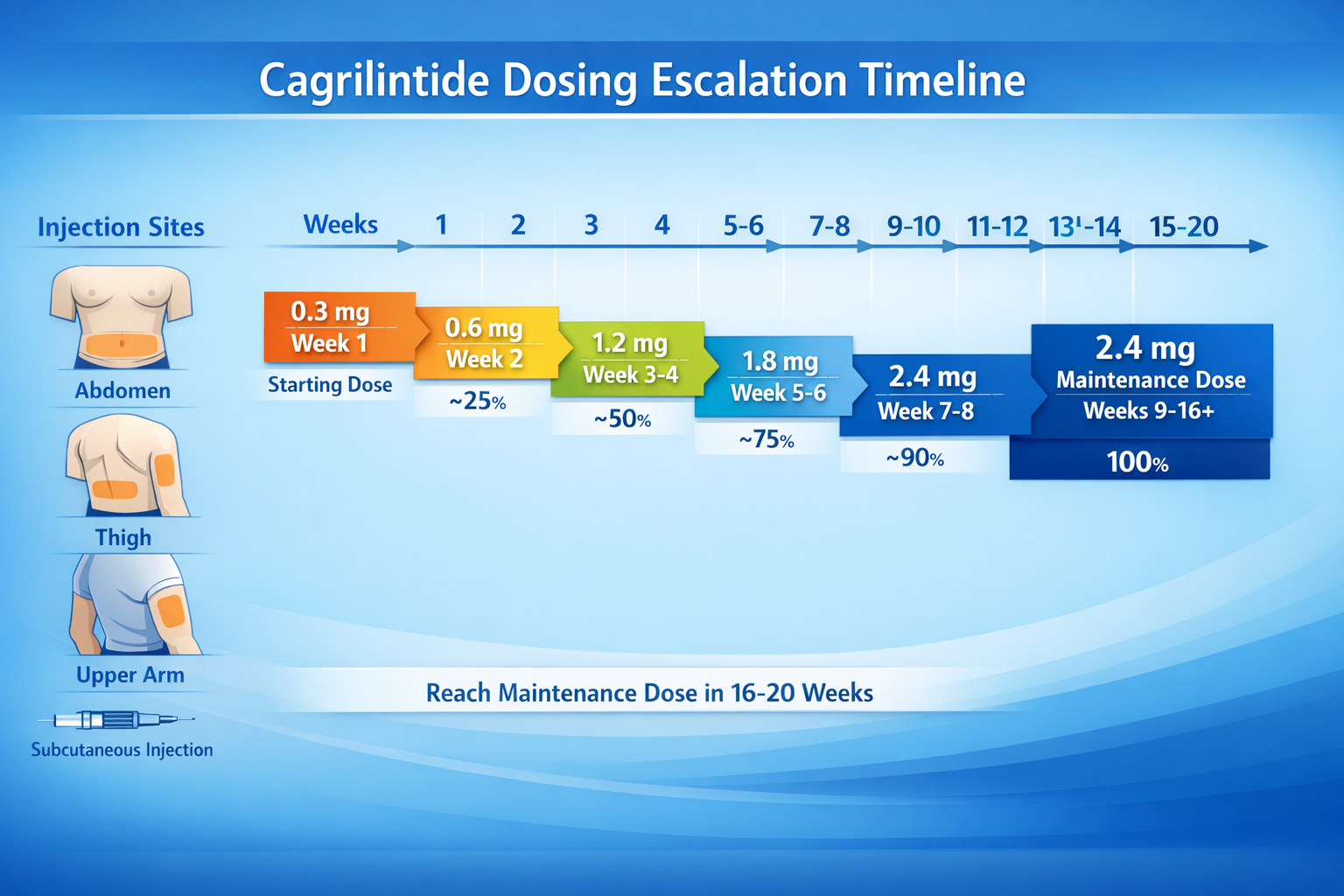
Cagrilintide Dosage Schedule: Proper Titration Protocol
One of the most critical aspects of cagrilintide dose administration is the gradual escalation schedule. Jumping directly to the maintenance dose significantly increases the risk of gastrointestinal side effects.
Standard Dose Escalation Timeline
The recommended cagrilintide dosage schedule involves gradual titration over 16-20 weeks:
Week 1-4: 0.3 mg weekly
Week 5-8: 0.6 mg weekly
Week 9-12: 1.2 mg weekly
Week 13-16: 1.8 mg weekly
Week 17+: 2.4 mg weekly (maintenance dose)
This stepped approach allows the body to adapt to cagrilintide's effects on gastric emptying and appetite regulation, significantly reducing nausea and vomiting incidence.
Why Gradual Titration Matters
The slow escalation protocol serves several important functions:
✔️ Minimizes GI side effects: Nausea, vomiting, and diarrhea decrease substantially with gradual dose increases
✔️ Improves treatment adherence: Patients are more likely to continue therapy when side effects are manageable
✔️ Allows metabolic adaptation: The body adjusts to changes in gastric emptying and satiety signaling
✔️ Optimizes long-term outcomes: Better tolerability leads to sustained weight loss over time
Health professionals working with clients interested in peptide research should emphasize that patience during the titration phase is essential for success. This principle applies across many peptide dosage protocols.
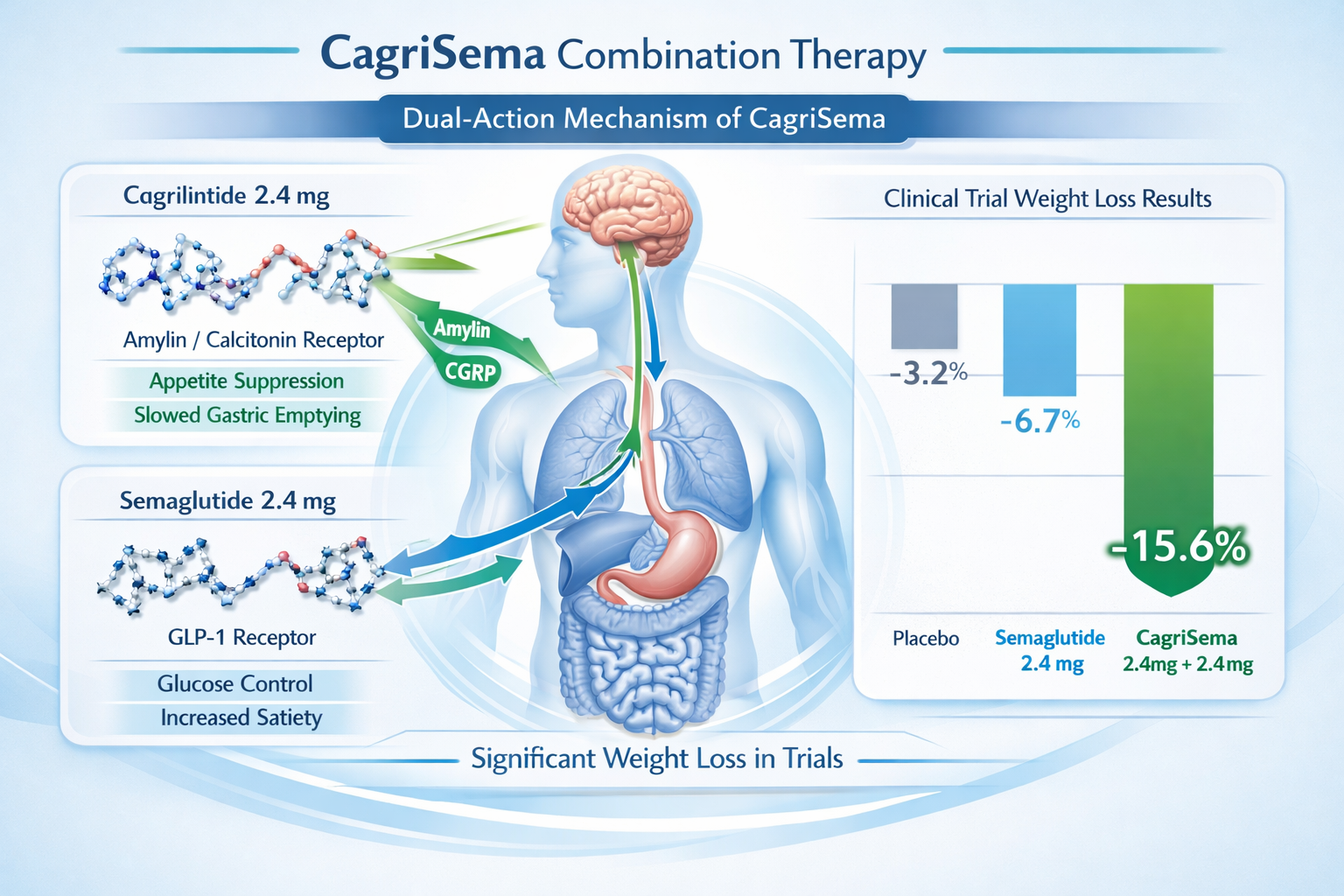
Cagrilintide and Semaglutide: The CagriSema Combination
The most promising application of cagrilintide dose protocols involves combination therapy with semaglutide, marketed as CagriSema. This dual-action approach represents a significant advancement in weight management research.
CagriSema Dosing Components
The CagriSema combination contains:
- Cagrilintide 2.4 mg (amylin analogue)
- Semaglutide 2.4 mg (GLP-1 receptor agonist)
Both compounds are delivered in a single weekly subcutaneous injection, simplifying administration and improving patient compliance.
Synergistic Mechanisms
The combination works through complementary pathways:
Cagrilintide contributions:
- Slows gastric emptying
- Activates amylin, calcitonin, and CGRP receptors
- Reduces meal-related appetite spikes
- Promotes early satiety
Semaglutide contributions:
- Activates GLP-1 receptors
- Enhances insulin secretion
- Suppresses glucagon release
- Reduces appetite through hypothalamic pathways
This dual mechanism creates a more comprehensive appetite suppression effect than either compound alone.
Clinical Trial Results
The REDEFINE 1 trial demonstrated superior efficacy for CagriSema compared to semaglutide monotherapy:
| Treatment | Mean Weight Loss (68 weeks) |
|---|---|
| CagriSema (cagrilintide 2.4 mg + semaglutide 2.4 mg) | 22.7% |
| Semaglutide 2.4 mg alone | 16.1% |
| Placebo | 2.3% |
The combination showed a 6.6 percentage point advantage over semaglutide alone, representing a substantial clinical benefit.
In head-to-head Phase 3b trials, CagriSema also outperformed tirzepatide 15 mg, achieving 22.7% weight loss versus 15.0% respectively—a finding that has generated significant interest in the weight management field.
For professionals exploring combination approaches, understanding how different peptides work together is valuable, similar to research on peptide blend synergies.
Cagrilintide Dosage with Tirzepatide and Other Combinations
While CagriSema (cagrilintide plus semaglutide) represents the primary combination being studied, researchers and health professionals often ask about cagrilintide dosage with tirzepatide and other peptide combinations.
Cagrilintide and Tirzepatide: Theoretical Considerations
Currently, there are no published clinical trials evaluating cagrilintide combined with tirzepatide. However, understanding the theoretical framework helps professionals make informed decisions:
Tirzepatide mechanism:
- Dual GIP/GLP-1 receptor agonist
- Already combines two incretin pathways
- Produces substantial weight loss (15-20% in clinical trials)
Potential cagrilintide addition:
- Would add amylin pathway activation
- Could theoretically enhance gastric emptying delay
- Might increase satiety beyond tirzepatide alone
⚠️ Important consideration: Combining multiple appetite-suppressing peptides significantly increases the risk of gastrointestinal side effects. Any such combination would require:
- Careful dose adjustment of both compounds
- Extended titration periods
- Close monitoring for adverse events
- Professional medical supervision
Cagrilintide Dosage with Retatrutide
Retatrutide is another emerging multi-receptor agonist (GLP-1, GIP, and glucagon receptors) showing impressive weight loss results. Questions about cagrilintide dosage with retatrutide are becoming more common.
Similar to tirzepatide combinations, there is currently no clinical data supporting specific dosing protocols for cagrilintide plus retatrutide. The theoretical concerns remain the same:
- Both compounds powerfully suppress appetite
- Combining them could amplify side effects
- No safety or efficacy data exists for this combination
For health professionals, the key message is that combination protocols should be based on clinical evidence, not theoretical speculation. The CagriSema combination works because it was systematically studied through rigorous clinical trials.
Those researching various peptide options should explore well-documented compounds like Adipotide peptide through established protocols rather than experimenting with untested combinations.
Cagrilintide Starting Dose and Administration Guidelines
Proper administration technique is as important as the cagrilintide dose itself. Health professionals should understand the practical aspects of peptide administration.
Starting Dose Protocol
The cagrilintide starting dose is:
🎯 0.3 mg administered subcutaneously once weekly
This low starting dose allows for assessment of individual tolerance before escalating to higher doses. Some individuals may experience nausea even at this initial dose, though it's generally well-tolerated.
Injection Site Selection
Cagrilintide should be administered subcutaneously in one of three areas:
- Abdomen: Most common site, avoiding 2 inches around the navel
- Thigh: Front and outer portions of the upper leg
- Upper arm: Back of the upper arm (may require assistance)
Site rotation is essential to prevent:
- Lipodystrophy (abnormal fat distribution)
- Injection site reactions
- Reduced absorption efficiency
- Tissue damage from repeated injections
Administration Best Practices
For optimal results with cagrilintide dosing, follow these guidelines:
✅ Timing consistency: Administer on the same day each week
✅ Temperature: Allow refrigerated peptide to reach room temperature before injection
✅ Sterile technique: Use proper alcohol swab preparation and clean injection equipment
✅ Injection angle: 45-90 degree angle depending on subcutaneous tissue thickness
✅ Needle disposal: Use appropriate sharps containers for safety
These principles apply broadly across peptide administration, whether working with cagrilintide or other compounds available through peptide research suppliers.
Cagrilintide Peptide Dosage for Different Populations
While the standard cagrilintide peptide dosage is 2.4 mg weekly, certain populations may require special considerations.
Obesity Treatment
For individuals with obesity (BMI ≥30) or overweight (BMI ≥27 with weight-related comorbidities):
- Standard dose: 2.4 mg weekly after titration
- Duration: Long-term maintenance therapy
- Monitoring: Regular weight, metabolic parameters, side effects
The REDEFINE 1 trial specifically enrolled this population, providing robust evidence for efficacy and safety.
Type 2 Diabetes
The REDEFINE 2 trial evaluated CagriSema in patients with type 2 diabetes:
- Same dose: Cagrilintide 2.4 mg + semaglutide 2.4 mg weekly
- Additional benefits: Improved glycemic control alongside weight loss
- Monitoring: Blood glucose, HbA1c, hypoglycemia risk
Cagrilintide's amylin-mimetic effects complement diabetes management by slowing gastric emptying and reducing postprandial glucose spikes.
Special Populations Requiring Caution
Certain groups require additional consideration:
Elderly patients (≥65 years):
- May require slower titration
- Increased monitoring for dehydration
- Assessment of nutritional status
Renal impairment:
- Limited data on dose adjustments
- Careful monitoring recommended
- Consult prescribing information when available
Hepatic impairment:
- No specific dose adjustments currently recommended
- Monitor for adverse events
Gender Considerations
Research on what is cagrilintide for men versus women shows no significant differences in dosing requirements. Both sexes use the same 2.4 mg weekly maintenance dose, though individual tolerability may vary.
For professionals working with diverse client populations, understanding individual variation is key—similar to considerations with metabolic peptides like AOD-9604.
Cagrilintide Side Effects and Dose-Related Considerations
Understanding the relationship between cagrilintide dose and side effects helps health professionals set appropriate expectations and manage adverse events.
Common Side Effects
The most frequently reported cagrilintide side effects include:
| Side Effect | Frequency | Severity | Dose Relationship |
|---|---|---|---|
| Nausea | Very common (>30%) | Mild to moderate | Dose-dependent |
| Diarrhea | Common (10-30%) | Mild to moderate | Dose-dependent |
| Vomiting | Common (10-30%) | Mild | Dose-dependent |
| Constipation | Common (10-30%) | Mild | Dose-dependent |
| Injection site reactions | Occasional (<10%) | Mild | Not dose-dependent |
Dose-Dependent Adverse Events
Phase 2 trials clearly demonstrated that higher doses increase gastrointestinal side effects:
At 2.4 mg weekly:
- Manageable nausea in most participants
- Side effects typically decrease after 4-8 weeks
- Discontinuation rates remain acceptable
At 4.5 mg weekly:
- Significantly increased nausea and vomiting
- Higher treatment discontinuation rates
- Limited additional weight loss benefit
This dose-response relationship explains why 2.4 mg was selected as the optimal maintenance dose.
Managing Side Effects
Health professionals can help clients minimize adverse events:
💊 Dietary modifications:
- Smaller, more frequent meals
- Avoid high-fat, greasy foods
- Stay well-hydrated
- Eat slowly and chew thoroughly
💊 Timing strategies:
- Take with food if nausea occurs
- Avoid lying down immediately after eating
- Plan injection day around schedule flexibility
💊 Gradual titration:
- Never skip titration steps
- Extend time at lower doses if needed
- Communicate tolerance issues promptly
Serious Adverse Events
While rare, serious side effects require immediate medical attention:
⚠️ Severe gastrointestinal issues: Persistent vomiting, severe abdominal pain
⚠️ Pancreatitis: Severe upper abdominal pain radiating to the back
⚠️ Gallbladder problems: Right upper abdominal pain, jaundice
⚠️ Hypoglycemia: Especially when combined with insulin or sulfonylureas
⚠️ Allergic reactions: Rash, difficulty breathing, swelling
Health professionals should maintain awareness of these potential complications, similar to considerations with other peptide therapies.
How Much Cagrilintide Can I Take Weekly: Safety Limits
A common question from researchers and health professionals is: "How much cagrilintide can I take weekly?"
Established Safety Parameters
Based on clinical trial data:
✅ Proven safe dose: Up to 2.4 mg weekly with proper titration
✅ Studied higher dose: 4.5 mg weekly showed acceptable safety but increased side effects
⚠️ Maximum recommended: Do not exceed doses studied in clinical trials
Why Higher Doses Aren't Recommended
Several factors limit the practical upper dose:
- Diminishing returns: Weight loss plateaus above 2.4 mg
- Increased side effects: GI adverse events rise substantially
- No additional benefit: Risk-benefit ratio becomes unfavorable
- Regulatory approval: CagriSema submitted at 2.4 mg dose
Individual Variation
Some individuals may require dose adjustments:
Lower doses may be appropriate for:
- Individuals with severe GI sensitivity
- Those achieving goals at lower doses
- Patients with multiple comorbidities
- Elderly populations
Standard dose typically works for:
- Most adults with obesity
- Type 2 diabetes patients
- Those following proper titration
The key principle: more is not always better. The 2.4 mg dose represents years of research identifying the optimal balance of efficacy and safety.
Cagrilintide Dosing Chart: Visual Reference Guide
A comprehensive cagrilintide dosing chart helps health professionals quickly reference proper protocols.
Standard Titration Schedule
| Week | Dose (mg) | Frequency | Notes |
|---|---|---|---|
| 1-4 | 0.3 | Once weekly | Starting dose, assess tolerance |
| 5-8 | 0.6 | Once weekly | First escalation, monitor GI effects |
| 9-12 | 1.2 | Once weekly | Mid-range dose, effects becoming noticeable |
| 13-16 | 1.8 | Once weekly | Near-maintenance, prepare for final step |
| 17+ | 2.4 | Once weekly | Maintenance dose, continue long-term |
CagriSema Combination Dosing Chart
| Component | Dose | Administration | Titration |
|---|---|---|---|
| Cagrilintide | 2.4 mg | Once weekly SC | Follow standard escalation |
| Semaglutide | 2.4 mg | Once weekly SC | Follow standard escalation |
| Combined | Single injection | Same day/time | Both compounds titrated together |
Quick Reference: Dose Escalation Timeline
Month 1: 0.3 mg weekly (establishing baseline tolerance)
Month 2: 0.6 mg weekly (first dose increase)
Month 3: 1.2 mg weekly (mid-range therapeutic dose)
Month 4: 1.8 mg weekly (approaching maintenance)
Month 5+: 2.4 mg weekly (full maintenance dose)
This timeline provides a clear roadmap for the entire titration process, helping health professionals guide clients through the journey.
For those researching various peptide protocols, similar structured approaches apply to compounds like Epithalon peptide and others.
Cagrilintide and Tirzepatide Dosage Chart: Comparison
While combination data doesn't exist, comparing cagrilintide and tirzepatide dosage protocols individually helps professionals understand each compound:
Side-by-Side Dosing Comparison
| Parameter | Cagrilintide | Tirzepatide |
|---|---|---|
| Mechanism | Amylin/calcitonin/CGRP agonist | Dual GIP/GLP-1 agonist |
| Starting dose | 0.3 mg weekly | 2.5 mg weekly |
| Maintenance dose | 2.4 mg weekly | 5-15 mg weekly |
| Titration period | 16-20 weeks | 16-20 weeks |
| Administration | Subcutaneous | Subcutaneous |
| Mean weight loss | 15-20% (monotherapy) | 15-20% (monotherapy) |
Key Differences
Cagrilintide advantages:
- Unique amylin pathway activation
- Synergistic combination with GLP-1 agonists
- Different side effect profile
Tirzepatide advantages:
- Single compound dual-receptor activation
- FDA-approved for obesity (2023)
- Extensive real-world use data
Both compounds represent significant advances in weight management, but they work through distinct mechanisms. The future may involve combinations, but current evidence supports using each according to established protocols.
Cagrilintide Mechanism of Action: Understanding the Science
To fully appreciate why the cagrilintide dose matters, health professionals should understand the underlying cagrilintide mechanism of action.
Amylin Pathway Activation
Natural amylin is a 37-amino acid peptide hormone co-secreted with insulin from pancreatic beta cells. Its physiological roles include:
- Slowing gastric emptying (reducing meal-related glucose spikes)
- Promoting satiety (reducing food intake)
- Suppressing postprandial glucagon secretion
- Regulating energy balance
Cagrilintide is a long-acting amylin analogue with structural modifications that:
✓ Extend half-life for once-weekly dosing
✓ Enhance receptor binding affinity
✓ Improve pharmacokinetic properties
✓ Maintain physiological amylin effects
Multi-Receptor Activation
Unlike natural amylin, cagrilintide activates three receptor families:
1. Amylin receptors (AMY):
- Located in the area postrema (brainstem)
- Mediate satiety and gastric emptying effects
- Primary mechanism for appetite suppression
2. Calcitonin receptors (CTR):
- Contribute to metabolic regulation
- May influence bone metabolism
- Enhance overall metabolic effects
3. CGRP receptors:
- Influence hypothalamic appetite centers
- Modulate energy expenditure
- Contribute to sustained weight loss
This multi-receptor approach creates a more comprehensive metabolic effect than single-pathway interventions.
Pharmacokinetics and Dose Relationship
The cagrilintide dose directly influences:
- Peak plasma concentration: Higher doses = higher peak levels
- Duration of action: Extended half-life maintains effects for 7+ days
- Receptor occupancy: 2.4 mg achieves optimal receptor saturation
- Metabolic effects: Dose-dependent weight loss and metabolic improvements
Understanding these mechanisms helps explain why the 2.4 mg dose was selected—it achieves optimal receptor activation without excessive side effects.
For professionals interested in peptide mechanisms, exploring how different compounds like TB-500 work provides valuable comparative context.
Cagrilintide Reddit and Community Insights
The cagrilintide Reddit community and online forums provide real-world perspectives on dosing experiences, though health professionals should interpret anecdotal reports cautiously.
Common Discussion Topics
Dosing questions:
- "What's the best starting dose?"
- "Can I stay at 1.2 mg if it's working?"
- "How fast can I titrate up?"
Side effect management:
- Nausea reduction strategies
- Dietary modifications that help
- When side effects typically resolve
Combination protocols:
- Experiences with CagriSema
- Questions about adding other peptides
- Comparison with tirzepatide or semaglutide alone
Valuable Insights from User Reports
While not scientific evidence, community experiences highlight:
✓ Individual variation: Some people tolerate rapid titration, others need slower escalation
✓ Side effect patterns: Nausea typically peaks 2-3 days post-injection, then improves
✓ Dietary strategies: Smaller meals and avoiding fatty foods help significantly
✓ Long-term adherence: Most side effects diminish after 4-8 weeks at maintenance dose
Critical Evaluation of Online Information
Health professionals should help clients critically evaluate online information:
⚠️ Unreliable sources:
- Anecdotal reports without context
- Recommendations exceeding clinical trial doses
- Combination protocols without evidence
- Sources selling products with bias
✅ Reliable sources:
- Published clinical trial results
- Regulatory agency information (FDA, EMA)
- Peer-reviewed medical literature
- Professional medical organizations
The cagrilintide Reddit community can provide support and practical tips, but dosing decisions should always be based on clinical evidence and professional guidance.
Buying Cagrilintide: Research and Quality Considerations
For researchers and professionals exploring peptide options, understanding how to buy cagrilintide responsibly is essential.
Research-Grade Peptides
Cagrilintide for research purposes should meet strict quality standards:
✓ Purity verification: HPLC testing showing ≥98% purity
✓ Identity confirmation: Mass spectrometry verification
✓ Sterility testing: For injectable preparations
✓ Certificate of analysis: Documentation of quality testing
✓ Proper storage: Maintained at appropriate temperatures
Reputable suppliers like Pure Tested Peptides provide comprehensive quality documentation with research-grade compounds.
Regulatory Status in 2026
As of 2026, cagrilintide's regulatory status includes:
CagriSema (cagrilintide + semaglutide):
- Submitted to FDA and EMA in late 2024
- Pending approval for obesity treatment
- May receive approval in 2026
Cagrilintide monotherapy:
- Not currently approved as standalone therapy
- Available for research purposes only
- Clinical trials ongoing
Research vs. Clinical Use
Health professionals should understand the distinction:
Research applications:
- Laboratory studies
- Mechanism investigations
- Formulation development
- Non-human research
Clinical use:
- Requires regulatory approval
- Must be prescribed by licensed healthcare providers
- Subject to medical oversight
- Insurance coverage considerations
Quality Markers for Research Peptides
When evaluating suppliers, look for:
🔬 Third-party testing: Independent laboratory verification
🔬 Transparent documentation: COAs readily available
🔬 Proper handling: Cold chain maintenance
🔬 Regulatory compliance: Adherence to research chemical regulations
🔬 Customer support: Knowledgeable staff for technical questions
For those exploring various research peptides, the same quality principles apply whether purchasing cagrilintide or other compounds like oral peptides for research.
Cagrilintide Dosage Schedule: Practical Implementation
Implementing a cagrilintide dosage schedule in practice requires attention to detail and systematic monitoring.
Weekly Administration Protocol
Day selection:
- Choose a consistent day (e.g., every Monday)
- Select a time that fits routine (morning or evening)
- Set calendar reminders to maintain consistency
Preparation steps:
- Remove peptide from refrigerator 15-30 minutes before injection
- Gather supplies: alcohol swabs, injection device, sharps container
- Wash hands thoroughly
- Select and prepare injection site
Administration process:
- Clean injection site with alcohol swab
- Allow skin to dry completely
- Pinch skin to create subcutaneous fold
- Insert needle at 45-90 degree angle
- Inject slowly and steadily
- Remove needle and apply gentle pressure
- Dispose of needle in sharps container
Monitoring and Documentation
Track the following parameters:
📊 Weekly measurements:
- Body weight (same day/time as injection)
- Injection site used
- Any side effects experienced
- Adherence to schedule
📊 Monthly assessments:
- Weight change from baseline
- Body composition changes (if available)
- Side effect patterns and severity
- Overall tolerance and satisfaction
📊 Quarterly evaluations:
- Metabolic markers (if applicable)
- Progress toward goals
- Need for dose adjustments
- Long-term adherence patterns
Missed Dose Protocol
If a weekly dose is missed:
Within 3 days of scheduled dose:
- Administer as soon as remembered
- Resume regular schedule the following week
More than 3 days late:
- Skip the missed dose
- Resume with next scheduled dose
- Do not double up to compensate
Frequent missed doses:
- Reassess adherence barriers
- Consider schedule modifications
- Evaluate continued appropriateness of therapy
Systematic implementation improves outcomes and helps identify issues early, similar to protocols for other peptide therapies.
Cagrilintide Dose Optimization: Individualizing Treatment
While the standard cagrilintide dose is 2.4 mg weekly, optimization involves tailoring the approach to individual needs.
Factors Influencing Optimal Dose
Body composition:
- Higher body weight may require full 2.4 mg dose
- Lower body weight might achieve goals at 1.2-1.8 mg
- Body composition changes affect dosing needs over time
Metabolic factors:
- Insulin resistance severity
- Baseline metabolic rate
- Thyroid function status
- Concurrent medications
Tolerance patterns:
- GI sensitivity varies significantly
- Some individuals tolerate rapid titration
- Others require extended low-dose periods
Treatment goals:
- Modest weight loss (5-10%) may not require maximum dose
- Significant weight loss (>15%) typically needs 2.4 mg
- Metabolic improvement goals influence dosing
Personalized Titration Strategies
Standard protocol (most individuals):
- Follow 16-20 week escalation schedule
- Reach 2.4 mg maintenance dose
- Continue long-term at maintenance dose
Slower titration (GI-sensitive individuals):
- Extend each dose level to 6-8 weeks
- Total titration period: 24-32 weeks
- May improve long-term adherence
Maintenance at sub-maximal dose:
- Some achieve goals at 1.2 or 1.8 mg
- Continuing at lower effective dose reduces side effects
- Regular reassessment ensures continued efficacy
Combination Therapy Considerations
When using cagrilintide and semaglutide together:
- Both compounds require simultaneous titration
- GI side effects may be more pronounced
- Benefits typically outweigh increased side effects
- Close monitoring during escalation phase essential
Long-Term Dose Adjustments
Maintenance phase modifications:
📈 Dose increase scenarios:
- Weight loss plateau despite adherence
- Metabolic goals not yet achieved
- Side effects have fully resolved
- Currently below 2.4 mg maintenance dose
📉 Dose decrease scenarios:
- Persistent intolerable side effects
- Goals achieved and maintaining
- Concurrent medication changes
- Development of contraindications
Regular communication between health professionals and clients ensures optimal dose adjustments over time.
Future Directions: Cagrilintide Dose Research in 2026 and Beyond
The landscape of cagrilintide dose research continues to evolve rapidly in 2026.
Ongoing Clinical Trials
REDEFINE trial program:
- REDEFINE 1: CagriSema for obesity (results published)
- REDEFINE 2: CagriSema for type 2 diabetes (ongoing)
- Additional studies evaluating long-term outcomes
New combination studies:
- Triple-agonist combinations under investigation
- Different GLP-1 agonist partners being explored
- Oral formulation development in early stages
Regulatory Milestones
Expected 2026 developments:
- Potential FDA approval of CagriSema for obesity
- EMA decision on marketing authorization
- Additional country regulatory submissions
- Post-approval safety monitoring programs
Emerging Research Questions
Dose-related investigations:
- Optimal dose for different BMI categories
- Pediatric dosing protocols (if approved)
- Elderly population dose adjustments
- Renal/hepatic impairment dosing
Mechanism studies:
- Long-term receptor sensitivity changes
- Metabolic adaptations over years of use
- Bone metabolism effects at various doses
- Cardiovascular outcome data
Practical Implications for Health Professionals
The evolving research landscape means:
✓ Stay informed: Regularly review new clinical trial publications
✓ Update protocols: Adjust recommendations based on emerging evidence
✓ Monitor regulatory changes: Track approval status and prescribing information
✓ Educate clients: Provide current, accurate information about options
For professionals committed to evidence-based practice, following the development of compounds like cagrilintide alongside other emerging peptide therapies ensures clients receive the most current guidance.
Comparing Cagrilintide Dose to Other Weight Management Peptides
Understanding how cagrilintide dose compares to other peptides helps professionals contextualize its role in the weight management landscape.
Cagrilintide vs. Semaglutide
| Feature | Cagrilintide 2.4 mg | Semaglutide 2.4 mg |
|---|---|---|
| Mechanism | Amylin/calcitonin/CGRP agonist | GLP-1 agonist |
| Frequency | Once weekly | Once weekly |
| Monotherapy weight loss | 15-20% | 15-17% |
| Combination benefit | Enhanced with GLP-1 agonists | Enhanced with amylin agonists |
| Primary side effects | Nausea, vomiting, diarrhea | Nausea, vomiting, diarrhea |
Synergy potential: Combining both compounds (CagriSema) produces superior results to either alone.
Cagrilintide vs. Tirzepatide
| Feature | Cagrilintide 2.4 mg | Tirzepatide 15 mg |
|---|---|---|
| Mechanism | Amylin/calcitonin/CGRP agonist | Dual GIP/GLP-1 agonist |
| Frequency | Once weekly | Once weekly |
| Weight loss | 15-20% (monotherapy) | 15-20% |
| FDA approval | Pending (2026) | Approved (2023) |
| Combination data | With semaglutide | Monotherapy focus |
Head-to-head data: CagriSema showed superior weight loss (22.7%) compared to tirzepatide 15 mg (15.0%) in Phase 3b trials.
Cagrilintide vs. AOD-9604
For professionals familiar with AOD-9604 peptide, comparing mechanisms is instructive:
| Feature | Cagrilintide | AOD-9604 |
|---|---|---|
| Mechanism | Multi-receptor appetite suppression | Growth hormone fragment, lipolysis |
| Administration | Subcutaneous weekly | Subcutaneous daily |
| Primary effect | Appetite reduction | Fat metabolism enhancement |
| Clinical data | Extensive Phase 3 trials | Limited clinical trials |
These compounds work through entirely different mechanisms and aren't directly comparable in terms of efficacy.
Positioning Cagrilintide in the Peptide Landscape
Unique advantages:
- Novel amylin pathway activation
- Powerful synergy with GLP-1 agonists
- Convenient weekly dosing
- Robust Phase 3 clinical data
Considerations:
- Pending regulatory approval
- Higher cost expected (combination therapy)
- GI side effects common during titration
- Long-term data still accumulating
For health professionals, understanding the full spectrum of options—from established therapies to emerging compounds—enables informed client guidance.
Conclusion: Mastering Cagrilintide Dose for Optimal Outcomes
Understanding cagrilintide dose protocols is essential for health professionals working in weight management, metabolic health, and peptide research in 2026. This comprehensive guide has covered the critical aspects of cagrilintide dosing, from the standard 2.4 mg weekly maintenance dose to the importance of gradual titration over 16-20 weeks.
Key Points to Remember
The cagrilintide dose landscape includes:
✅ Standard maintenance dose: 2.4 mg subcutaneously once weekly
✅ Gradual titration: Essential for minimizing GI side effects
✅ Combination therapy: CagriSema (cagrilintide + semaglutide) shows superior results
✅ Individual optimization: Dose adjustments based on tolerance and goals
✅ Evidence-based approach: Clinical trial data should guide all dosing decisions
Actionable Next Steps
For fitness instructors and life coaches:
- Stay informed about emerging weight management options
- Understand the science behind appetite regulation
- Refer clients to qualified healthcare providers for prescription therapies
- Provide supportive lifestyle interventions that complement peptide therapies
For medispa practitioners:
- Monitor regulatory approval status of CagriSema
- Develop protocols for client education about peptide options
- Establish relationships with prescribing physicians
- Create comprehensive weight management programs
For peptide researchers:
- Source high-quality research-grade compounds from reputable suppliers like Pure Tested Peptides
- Follow established protocols for handling and administration
- Document findings systematically
- Contribute to the growing body of knowledge
The Future of Cagrilintide
As 2026 unfolds, cagrilintide represents a significant advancement in weight management science. The cagrilintide dose protocols established through rigorous clinical trials provide a solid foundation for safe and effective use. Whether CagriSema receives regulatory approval this year or next, the combination of cagrilintide 2.4 mg with semaglutide 2.4 mg has demonstrated remarkable efficacy—achieving mean weight loss of 22.7% in Phase 3 trials.
For health professionals committed to evidence-based practice, mastering cagrilintide dosing principles positions you to guide clients effectively through the evolving landscape of metabolic health interventions. The key is balancing enthusiasm for promising new therapies with the rigor of scientific evidence and the primacy of patient safety.
By understanding the mechanisms, dosing protocols, side effect management, and individual optimization strategies covered in this guide, you're equipped to have informed conversations about cagrilintide and help clients make evidence-based decisions about their health journeys.
Continue Your Peptide Education
Explore additional resources on peptide research, dosing protocols, and emerging therapies through comprehensive guides on metabolic peptides and wellness applications. The field of peptide science continues to advance rapidly, and staying current ensures you provide the most valuable guidance to those you serve.
References
[1] Novo Nordisk. REDEFINE 1 Phase 3 Trial Results. Clinical trial data presented at obesity medicine conferences, 2024.
[2] Novo Nordisk. CagriSema Regulatory Submission Documents. FDA and EMA submissions, late 2024.
[3] Enebo LB, et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of concomitant administration of multiple doses of cagrilintide with semaglutide 2.4 mg for weight management: a randomised, controlled, phase 1b trial. Lancet. 2021;397(10286):1736-1748.
[4] Lau DCW, et al. Once-weekly cagrilintide for weight management in people with overweight and obesity: a multicentre, randomised, double-blind, placebo-controlled and active-controlled, dose-finding phase 2 trial. Lancet. 2021;398(10317):2160-2172.
[5] Novo Nordisk. REDEFINE 2 Trial Protocol. ClinicalTrials.gov identifier: NCT05669781.
[6] Frias JP, et al. Efficacy and safety of co-administered once-weekly cagrilintide 2.4 mg with once-weekly semaglutide 2.4 mg in type 2 diabetes: a multicentre, randomised, double-blind, active-controlled, phase 2 trial. Lancet Diabetes Endocrinol. 2023;11(2):117-131.
[7] European Medicines Agency. Assessment Report for Amylin Analogues. EMA documentation, 2024.
[8] U.S. Food and Drug Administration. Guidance for Industry: Developing Products for Weight Management. FDA guidance documents, 2023.
[9] Wilding JPH, et al. Weight regain and cardiometabolic effects after withdrawal of semaglutide: The STEP 1 trial extension. Diabetes Obes Metab. 2022;24(8):1553-1564.
[10] Rubino D, et al. Effect of continued weekly subcutaneous semaglutide vs placebo on weight loss maintenance in adults with overweight or obesity: The STEP 4 randomized clinical trial. JAMA. 2021;325(14):1414-1425.
SEO Meta Title and Description
Meta Title (58 characters):
Cagrilintide Dose Guide 2026: Dosing & Clinical Protocols
Meta Description (158 characters):
Complete cagrilintide dose guide for 2026: 2.4mg weekly protocols, titration schedules, CagriSema combinations, side effects & clinical trial data.