Cagrilintide Dosing Chart: Your Complete 2026 Guide to Safe and Effective Administration

Imagine having access to a breakthrough peptide therapy that could revolutionize weight management and metabolic health—but without clear dosing guidance, even the most promising treatment becomes a puzzle. For fitness instructors, health coaches, medispa professionals, and peptide researchers, understanding the cagrilintide dosing chart is essential for maximizing therapeutic potential while minimizing adverse effects. As this long-acting amylin analogue advances through Phase 3 clinical trials, having a comprehensive reference for proper administration protocols has never been more critical.
Cagrilintide represents a new frontier in metabolic research, offering unique mechanisms that complement existing therapies. However, the complexity of its dose escalation schedule and combination protocols demands careful attention to detail. This guide breaks down everything professionals need to know about cagrilintide administration in 2026.
Key Takeaways
- Gradual dose escalation over 20 weeks minimizes gastrointestinal side effects while building therapeutic efficacy
- Standard monotherapy reaches a maximum maintenance dose of 4.5 mg weekly after systematic increases
- Combination protocols (CagriSema) pair cagrilintide with semaglutide for enhanced weight management outcomes
- Clinical trial data demonstrates approximately 10.8% body weight reduction with monotherapy at 32 weeks
- Proper injection technique and site rotation are essential for optimal absorption and safety
Understanding Cagrilintide: What Health Professionals Need to Know
Cagrilintide is a long-acting amylin analogue currently being developed by Novo Nordisk for obesity and type 2 diabetes management. Unlike traditional weight loss approaches, this peptide works through amylin receptor activation in the brain, creating a cascade of metabolic effects that support sustainable weight management [1].
The peptide's mechanism of action involves three primary pathways: reduced appetite through central nervous system signaling, delayed gastric emptying that promotes satiety, and decreased food intake through enhanced fullness signals. These combined effects make cagrilintide a valuable tool for researchers exploring metabolic health interventions.
For professionals working in wellness spaces, understanding peptide research fundamentals provides essential context for appreciating cagrilintide's unique properties. The compound's long-acting formulation allows for convenient once-weekly administration, improving compliance compared to daily injection protocols.
The Science Behind Amylin Analogues
Amylin is a naturally occurring hormone co-secreted with insulin from pancreatic beta cells. In individuals with obesity or metabolic dysfunction, amylin signaling may be impaired. Cagrilintide mimics and amplifies natural amylin activity, restoring these critical metabolic signals [2].
The peptide's extended half-life distinguishes it from earlier amylin analogues. This pharmacokinetic profile enables weekly dosing while maintaining stable therapeutic levels throughout the dosing interval. Research protocols examining cagrilintide 10mg formulations demonstrate the importance of proper reconstitution and storage for maintaining peptide integrity.
Clinical observations indicate that cagrilintide's effects extend beyond simple appetite suppression. The compound appears to influence energy expenditure, glucose metabolism, and potentially even lipid profiles, though ongoing research continues to elucidate these mechanisms.
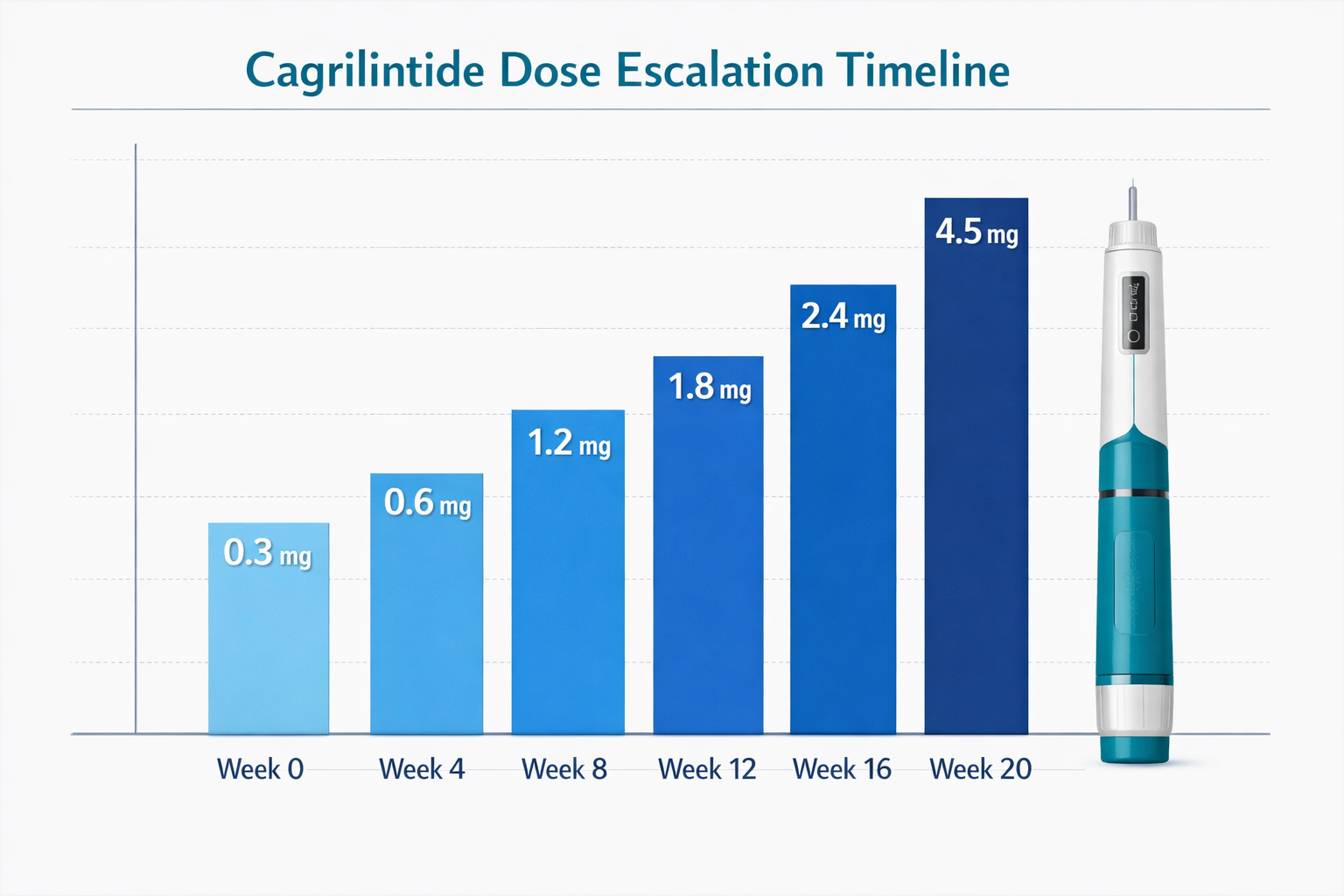
The Complete Cagrilintide Dosing Chart for Monotherapy
The cagrilintide dosing chart for monotherapy follows a carefully structured escalation protocol designed to optimize tolerance while building therapeutic efficacy. Based on the REDEFINE 1 trial protocol, the standard dosing schedule progresses over 20 weeks to reach maximum maintenance levels [3].
Standard Dose Escalation Schedule
| Week | Dose (mg) | Administration Frequency | Purpose |
|---|---|---|---|
| 0-3 | 0.3 | Once weekly | Initial tolerance assessment |
| 4-7 | 0.6 | Once weekly | Early dose escalation |
| 8-11 | 1.2 | Once weekly | Mid-range therapeutic building |
| 12-15 | 1.8 | Once weekly | Continued escalation |
| 16-19 | 2.4 | Once weekly | Near-maximum dosing |
| 20+ | 4.5 | Once weekly | Maintenance dose |
This gradual escalation approach serves multiple purposes. First, it allows the gastrointestinal system to adapt to cagrilintide's effects on gastric emptying, significantly reducing nausea and vomiting incidents. Second, it enables researchers and practitioners to identify individual tolerance thresholds before reaching maximum doses.
The starting dose of 0.3 mg represents approximately 6.7% of the final maintenance dose—a conservative approach that prioritizes safety. Each escalation step roughly doubles the previous dose until reaching the 1.8 mg level, after which increases become more modest.
Adjusting Doses Based on Individual Response
Not all individuals progress through the standard cagrilintide dosing chart at the same pace. Clinical practice guidelines suggest several scenarios where dose modification may be appropriate:
Persistent gastrointestinal symptoms lasting beyond the first week of a new dose level may warrant extending that dose phase by an additional 2-4 weeks before further escalation. This approach allows additional adaptation time without abandoning the therapeutic protocol.
Excellent tolerance with minimal side effects doesn't justify accelerating the escalation schedule. The gradual progression serves pharmacodynamic purposes beyond simple tolerability, allowing metabolic adaptations to develop fully.
For researchers exploring peptide dosage protocols, the cagrilintide model demonstrates the importance of systematic dose building in peptide therapy research.
Clinical Trial Outcomes with Monotherapy
The REDEFINE 1 trial demonstrated that cagrilintide monotherapy at the 4.5 mg maintenance dose produced mean body weight reductions of approximately 10.8% from baseline after 32 weeks of treatment [4]. These results position cagrilintide as a significant advancement in metabolic research.
Importantly, the weight loss trajectory showed continued progress throughout the study period, suggesting that longer treatment durations might yield even greater effects. The dose-response relationship was clear across the REDEFINE 2 trial's range of 0.3 mg to 4.5 mg weekly, with higher doses consistently producing greater weight reductions.
However, this efficacy came with trade-offs. Higher doses were associated with increased gastrointestinal adverse events, though most were classified as mild to moderate in severity. The dose escalation protocol successfully minimized these effects compared to immediate high-dose administration.
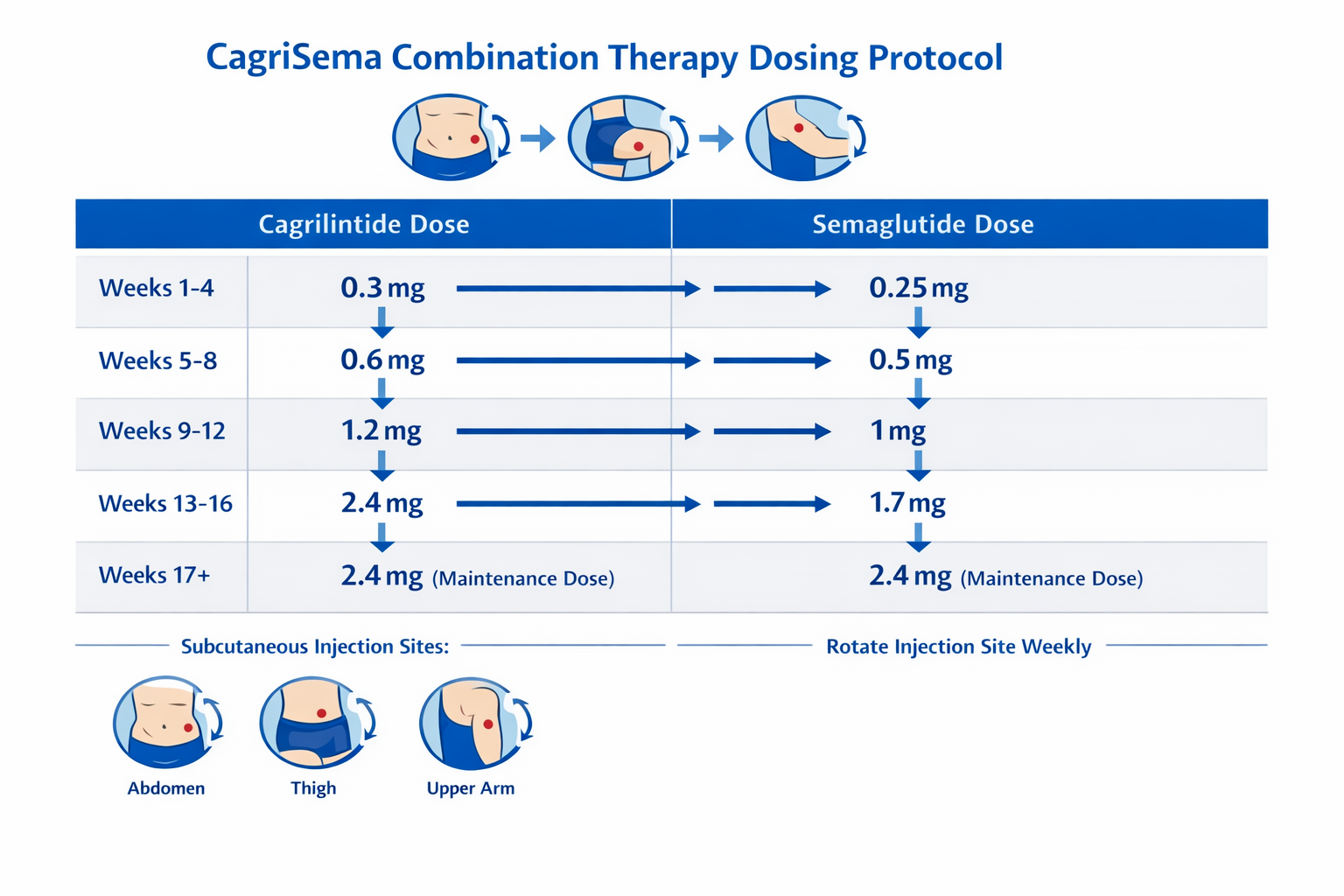
CagriSema Combination Therapy: The Cagrilintide and Semaglutide Dosing Chart
The CagriSema combination therapy represents an innovative approach that pairs cagrilintide with semaglutide, a GLP-1 receptor agonist. This fixed-dose combination leverages complementary mechanisms to achieve superior weight management outcomes compared to either agent alone [5].
Understanding the Combination Rationale
Cagrilintide and semaglutide target different but synergistic pathways in metabolic regulation. While cagrilintide activates amylin receptors to reduce appetite and slow gastric emptying, semaglutide stimulates GLP-1 receptors to enhance insulin secretion, suppress glucagon, and promote satiety through distinct neural pathways.
This dual-mechanism approach addresses multiple aspects of metabolic dysfunction simultaneously. The combination appears to produce effects greater than the simple sum of individual contributions—a phenomenon researchers term synergistic efficacy.
For professionals interested in combination peptide protocols, exploring peptide blend research provides valuable insights into how multiple compounds can work together.
CagriSema Dose Escalation Protocol
The cagrilintide dosage with semaglutide follows its own escalation schedule, with both compounds increasing in tandem:
| Week | Cagrilintide (mg) | Semaglutide (mg) | Total Weekly Dose |
|---|---|---|---|
| 0-3 | 0.3 | 0.3 | 0.6 mg combined |
| 4-7 | 0.6 | 0.6 | 1.2 mg combined |
| 8-11 | 1.2 | 1.2 | 2.4 mg combined |
| 12-15 | 1.8 | 1.8 | 3.6 mg combined |
| 16-19 | 2.4 | 2.4 | 4.8 mg combined |
| 20+ | 2.4 | 2.4 | 4.8 mg maintenance |
The maintenance dose for CagriSema settles at cagrilintide 2.4 mg plus semaglutide 2.4 mg weekly—notably lower than the 4.5 mg monotherapy dose for cagrilintide. This reflects the enhanced efficacy achieved through combination therapy, allowing therapeutic goals to be reached at lower individual compound doses.
Clinical Results with Combination Therapy
The SURPASS-COMBO trial demonstrated remarkable outcomes with CagriSema. Participants achieved weight reductions of up to 15.1% at 32 weeks—approximately 40% greater than cagrilintide monotherapy results [6]. This substantial improvement validates the combination approach.
The side effect profile for CagriSema showed similar gastrointestinal events as monotherapy, but the lower cagrilintide dose (2.4 mg vs. 4.5 mg) may contribute to improved overall tolerability. The gradual escalation protocol remained essential for minimizing adverse events.
These findings have significant implications for wellness professionals and researchers. The combination demonstrates that strategic pairing of complementary mechanisms can achieve superior outcomes while potentially reducing individual compound doses—a principle applicable across various therapeutic areas.
Cagrilintide Dosage with Other Peptide Combinations
Beyond the official CagriSema protocol, researchers are exploring cagrilintide dosage with tirzepatide and other metabolic peptides. While these combinations remain investigational, understanding potential protocols helps professionals stay current with emerging research directions.
Cagrilintide and Tirzepatide: Theoretical Dosing Considerations
Tirzepatide is a dual GIP/GLP-1 receptor agonist with powerful metabolic effects. Theoretically, combining it with cagrilintide could provide triple-mechanism coverage: amylin receptor activation (cagrilintide) plus GIP and GLP-1 receptor activation (tirzepatide).
A hypothetical cagrilintide and tirzepatide dosage chart would need to account for overlapping effects on gastric emptying and appetite suppression. Conservative starting doses would be essential:
- Initial phase: Cagrilintide 0.3 mg + Tirzepatide 2.5 mg weekly
- Escalation: Increase cagrilintide by 0.3-0.6 mg every 4 weeks
- Tirzepatide adjustment: Maintain or modestly increase based on tolerance
- Monitoring: Close observation for cumulative GI effects
This remains speculative, as no published trials have examined this specific combination. Researchers considering such protocols should prioritize safety monitoring and gradual dose building.
Cagrilintide Dosage with Retatrutide
Retatrutide represents another emerging peptide with GIP, GLP-1, and glucagon receptor activity. The cagrilintide dosage with retatrutide would theoretically combine amylin pathway activation with this triple-agonist approach.
Given retatrutide's potency, extremely conservative dosing would be warranted:
- Start with minimum doses of both compounds
- Extend escalation intervals to 6-8 weeks per step
- Monitor for enhanced metabolic effects and potential hypoglycemia
- Adjust based on individual metabolic response
For those researching advanced peptide protocols, these combination approaches illustrate the complexity and potential of multi-pathway interventions.
Safety Considerations for Combination Protocols
When exploring any combination involving cagrilintide, several safety principles apply:
✅ Start low, go slow – Use the most conservative doses for both compounds initially
✅ Monitor cumulative effects – Overlapping mechanisms may amplify both benefits and side effects
✅ Individualize protocols – Response variability increases with combination therapy
✅ Document carefully – Detailed records help identify optimal dosing for different populations
✅ Prioritize education – Ensure all stakeholders understand the investigational nature of novel combinations
The cagrilintide peptide available for research purposes enables controlled investigation of these protocols in appropriate settings.
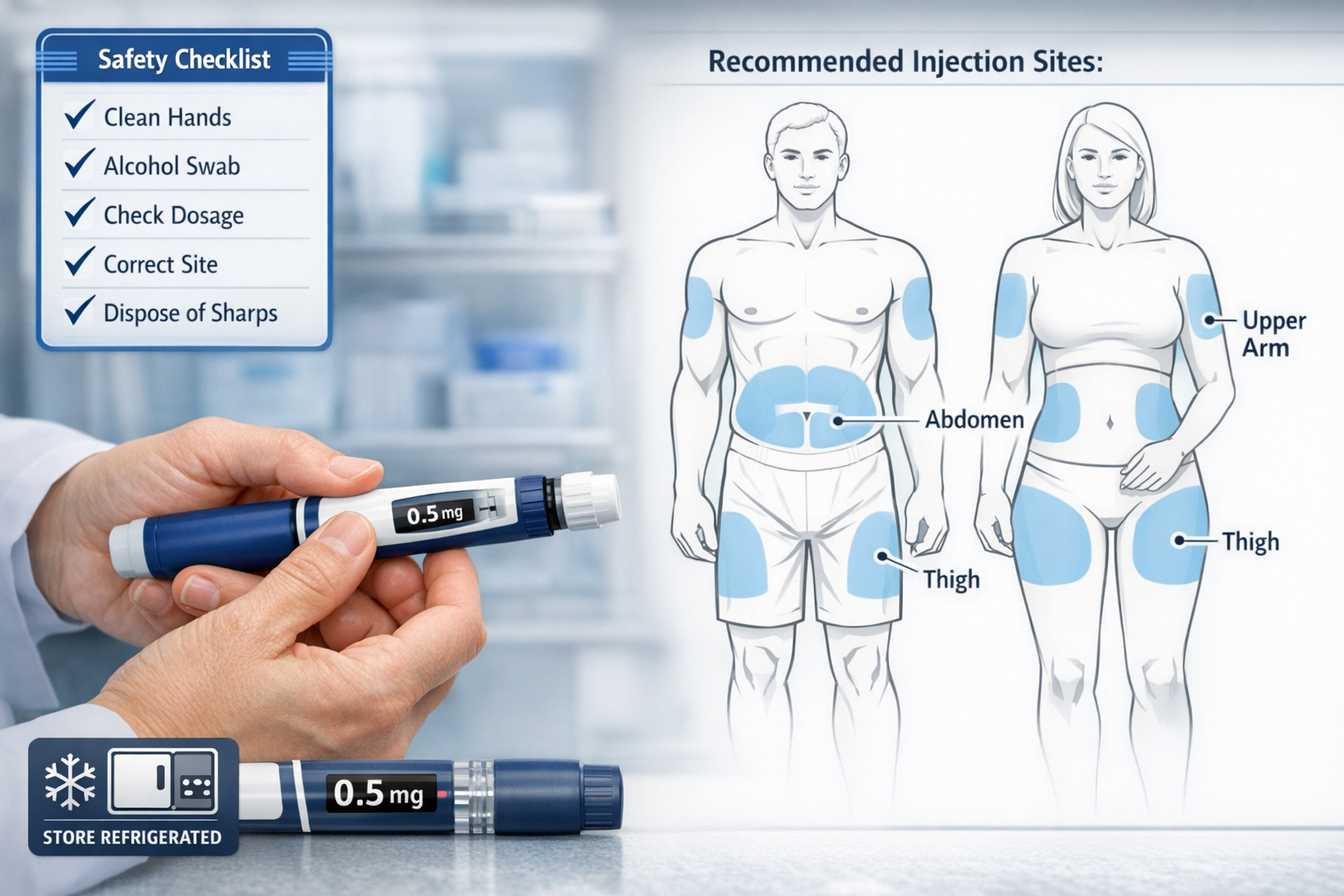
Proper Administration Techniques and Injection Guidelines
Beyond understanding the cagrilintide dosing chart, proper administration technique ensures optimal absorption, minimizes side effects, and maintains safety standards. These protocols apply whether using monotherapy or combination regimens.
Recommended Injection Sites
Cagrilintide is administered via subcutaneous injection in three primary sites:
🎯 Abdomen – The most common injection site, offering consistent absorption. Inject at least 2 inches away from the navel, avoiding the midline.
🎯 Thigh – The front and outer portions of the thigh provide good subcutaneous tissue. Avoid the inner thigh and areas near the knee.
🎯 Upper arm – The back of the upper arm (triceps area) works well, though this site may require assistance for proper technique.
Site rotation is essential to prevent lipodystrophy—abnormal fat distribution at injection sites. Establish a systematic rotation pattern, such as:
- Week 1: Right abdomen
- Week 2: Left abdomen
- Week 3: Right thigh
- Week 4: Left thigh
- Week 5: Right upper arm
- Week 6: Left upper arm
- Week 7: Return to right abdomen
This approach ensures each site receives adequate recovery time between injections.
Injection Technique Best Practices
Proper subcutaneous injection technique for cagrilintide follows these steps:
- Preparation – Allow refrigerated peptide to reach room temperature (15-30 minutes)
- Hygiene – Wash hands thoroughly and clean injection site with alcohol swab
- Pinch technique – Gently pinch skin to create a fold of subcutaneous tissue
- Insertion angle – Insert needle at 45-90 degree angle depending on subcutaneous fat thickness
- Injection – Depress plunger slowly and steadily over 5-10 seconds
- Withdrawal – Wait 5 seconds after full depression, then withdraw needle smoothly
- Pressure – Apply gentle pressure with clean gauze if needed (do not rub)
For researchers working with peptide administration protocols, these techniques ensure consistent delivery and reproducible results.
Storage and Handling Requirements
Cagrilintide requires specific storage conditions to maintain potency:
❄️ Refrigeration – Store unopened vials at 2-8°C (36-46°F)
🌡️ Temperature stability – Once in use, may be kept at room temperature (below 30°C) for up to 4 weeks
🔒 Light protection – Keep in original packaging to protect from light exposure
⚠️ Freezing prohibition – Never freeze; discard if accidentally frozen
🗓️ Expiration monitoring – Track reconstitution dates and discard after recommended timeframes
Proper handling extends beyond storage. Always inspect solutions before administration—discard if discolored, cloudy, or containing particles. Use appropriate syringes and needles (typically 4-6mm length, 31-32 gauge for subcutaneous delivery).
Managing Side Effects: What to Expect During Dose Escalation
Understanding cagrilintide side effects helps professionals guide clients through the escalation process and distinguish normal adaptation responses from concerning adverse events. The dose escalation protocol specifically aims to minimize these effects while building therapeutic levels.
Common Gastrointestinal Effects
The most frequently reported side effects with cagrilintide involve the gastrointestinal system:
Nausea 🤢 – Affects 40-60% of users during early escalation phases. Typically mild to moderate, peaking 1-3 days after dose increases and gradually subsiding over 1-2 weeks.
Vomiting – Less common than nausea (15-25% of users), usually occurring only during the first days of dose escalation steps.
Diarrhea – Reported by 20-30% of users, generally resolving as the body adapts to each new dose level.
Constipation – Paradoxically, some individuals experience constipation rather than diarrhea, likely related to delayed gastric emptying effects.
Decreased appetite – While technically the desired therapeutic effect, some individuals find the appetite suppression more pronounced than comfortable during initial weeks.
These effects result from cagrilintide's mechanism of action—delayed gastric emptying and central appetite regulation. They're not necessarily indicators of intolerance but rather evidence of pharmacological activity.
Strategies for Minimizing Adverse Events
Several approaches help manage side effects during the cagrilintide dosing schedule:
Dietary modifications 🍽️
- Consume smaller, more frequent meals
- Reduce high-fat foods that delay gastric emptying further
- Avoid large meals within 4 hours of bedtime
- Stay well-hydrated throughout the day
Timing optimization ⏰
- Administer injections on days with lighter schedules
- Plan for potential nausea during the 48 hours post-injection
- Coordinate dose escalations with periods of lower stress
Symptomatic management 💊
- Ginger supplements or tea may help reduce nausea
- Anti-nausea medications (as appropriate) for persistent symptoms
- Probiotics to support digestive comfort
Dose modification 📊
- Extend time at current dose level if side effects persist beyond one week
- Consider temporary dose reduction if symptoms significantly impact quality of life
- Resume escalation once tolerance improves
For professionals supporting clients through peptide protocols, understanding these management strategies is essential. Resources on peptide research methods provide additional context for systematic monitoring approaches.
When to Seek Medical Guidance
While most cagrilintide side effects are self-limiting, certain symptoms warrant immediate medical attention:
🚨 Severe persistent vomiting preventing fluid intake for >24 hours
🚨 Signs of dehydration including dark urine, dizziness, rapid heartbeat
🚨 Severe abdominal pain not relieved by typical measures
🚨 Allergic reactions including rash, swelling, difficulty breathing
🚨 Pancreatitis symptoms such as severe upper abdominal pain radiating to the back
The gradual escalation protocol significantly reduces the risk of severe adverse events, but individual responses vary. Maintaining detailed records of symptoms, timing, and severity helps identify patterns and optimize protocols.
Cagrilintide Dosing for Specific Populations
While clinical trial protocols provide general cagrilintide dosing guidelines, different populations may require modified approaches. Understanding these variations helps professionals tailor protocols appropriately.
Gender Considerations: What is Cagrilintide for Men?
The question "what is cagrilintide for men" reflects interest in gender-specific responses to peptide therapy. Clinical trials have included both male and female participants, with some notable observations:
Metabolic differences 🔬 – Men typically have higher baseline metabolic rates and different fat distribution patterns, which may influence response magnitude but not optimal dosing protocols.
Hormonal interactions – While cagrilintide's primary mechanism doesn't directly interact with sex hormones, the metabolic effects may influence testosterone levels indirectly through changes in body composition.
Side effect profiles – Some data suggests men may experience slightly lower rates of nausea during escalation, though this difference is modest and doesn't warrant different starting doses.
The standard cagrilintide dosing chart applies equally to men and women, with individual adjustments based on tolerance rather than gender per se. For men interested in metabolic optimization, exploring comprehensive peptide research provides broader context for evidence-based approaches.
Age-Related Dosing Modifications
Clinical trials have primarily enrolled adults aged 18-75, with most participants in the 30-60 age range. Age-related considerations include:
Younger adults (18-30) – May tolerate dose escalation well but should still follow standard protocols. Faster metabolism doesn't justify accelerated escalation.
Middle-aged adults (30-60) – Represent the primary target population with standard dosing protocols directly applicable.
Older adults (60+) – May benefit from extended escalation intervals (6 weeks per step instead of 4) due to:
- Slower metabolic adaptation
- Increased medication sensitivity
- Higher likelihood of comorbid conditions
- Potential polypharmacy interactions
Body Weight and Composition Factors
Unlike some medications dosed per kilogram of body weight, cagrilintide uses fixed dosing regardless of baseline weight. However, response may vary:
Higher baseline weight – Individuals starting at higher body weights may experience:
- Slower percentage weight loss initially
- Need for full maintenance doses to achieve optimal effects
- Potentially better tolerance of higher doses
Lower baseline weight – Those with lower starting weights might:
- Achieve therapeutic goals at submaximal doses (e.g., 2.4 mg vs. 4.5 mg)
- Experience more pronounced appetite suppression
- Require more cautious escalation
Body composition – Individuals with higher muscle mass may metabolize peptides differently, though this hasn't been systematically studied for cagrilintide specifically.
Monitoring and Optimizing Your Cagrilintide Protocol
Successful cagrilintide dosing extends beyond following the escalation chart—it requires systematic monitoring and protocol optimization based on individual response patterns.
Key Metrics to Track
Comprehensive monitoring should include:
Body composition changes 📊
- Weekly weight measurements (same time, same conditions)
- Monthly body composition analysis (if available)
- Circumference measurements (waist, hips, thighs)
- Progress photographs for visual documentation
Metabolic markers 🔬
- Fasting glucose levels (if relevant to research goals)
- Lipid panels at baseline and regular intervals
- Blood pressure monitoring
- Resting heart rate trends
Subjective responses 📝
- Appetite and satiety patterns
- Energy levels throughout the day
- Sleep quality and duration
- Mood and cognitive function
- Gastrointestinal symptom severity and frequency
Dosing compliance ✅
- Injection timing and consistency
- Missed doses and reasons
- Site rotation adherence
- Storage condition maintenance
For researchers implementing systematic peptide protocols, these metrics provide essential data for protocol optimization and outcome assessment.
Interpreting Response Patterns
Different response patterns may emerge during cagrilintide administration:
Rapid responders – Significant appetite suppression and early weight loss (>2% in first 4 weeks)
- May achieve goals at submaximal doses
- Monitor for excessive restriction or nutrient inadequacy
- Consider maintenance at lower dose levels
Gradual responders – Steady, consistent progress throughout escalation
- Ideal response pattern indicating good tolerance
- Continue standard protocol to full maintenance dose
- Expect continued progress with sustained administration
Delayed responders – Minimal changes during early escalation, acceleration at higher doses
- Require patience and protocol adherence
- Full maintenance doses typically necessary
- Extended treatment duration may be beneficial
Plateau responders – Initial progress followed by stabilization
- May indicate dose optimization opportunity
- Consider lifestyle factor evaluation
- Potentially combine with complementary approaches
Protocol Adjustments Based on Outcomes
Optimizing the cagrilintide dosage schedule involves strategic adjustments:
If tolerance is excellent but efficacy is suboptimal:
- Ensure full maintenance dose is reached
- Extend treatment duration before concluding inadequate response
- Evaluate complementary lifestyle factors
- Consider combination approaches (if appropriate)
If side effects limit escalation:
- Extend time at current dose level (add 2-4 weeks)
- Implement aggressive symptom management strategies
- Consider smaller escalation increments (0.3 mg vs. 0.6 mg steps)
- Temporarily reduce dose if symptoms are severe
If goals are achieved before reaching maintenance:
- Continue to planned maintenance dose for stability
- Maintain achieved dose for extended period
- Monitor for continued benefits or plateau
- Develop long-term maintenance strategy
Understanding peptide dosing principles across different compounds helps professionals develop intuition for protocol optimization.
Frequently Asked Questions About Cagrilintide Dosing
How much cagrilintide can I take weekly?
The maximum weekly cagrilintide dose studied in clinical trials is 4.5 mg for monotherapy. This represents the maintenance dose reached after 20 weeks of gradual escalation. For combination therapy (CagriSema), the cagrilintide component is 2.4 mg weekly paired with semaglutide 2.4 mg.
Exceeding these doses is not recommended, as safety and efficacy data beyond these levels are limited. The therapeutic window appears well-established at these dosing levels, with higher doses unlikely to provide proportional additional benefits while potentially increasing adverse events.
What is the cagrilintide starting dose?
The cagrilintide starting dose in clinical protocols is 0.3 mg weekly. This conservative initial dose allows assessment of individual tolerance and minimizes gastrointestinal side effects. Starting at this low dose—just 6.7% of the final maintenance dose—is essential for the gradual adaptation process.
Some individuals wonder if they can start higher if they have previous experience with similar peptides. However, cagrilintide's unique mechanism and long-acting formulation make the standard starting dose appropriate regardless of prior peptide exposure.
Can I adjust my cagrilintide dosage schedule?
The cagrilintide dosage schedule can be modified based on individual response, but adjustments should be made systematically:
Extending escalation intervals (e.g., 6 weeks instead of 4) is acceptable and often beneficial for those experiencing persistent side effects.
Accelerating escalation is generally not recommended, as the gradual protocol serves important pharmacodynamic purposes beyond simple tolerability.
Skipping dose levels (e.g., jumping from 0.6 mg to 1.8 mg) should be avoided, as this significantly increases the risk of adverse events.
Temporary dose reduction during illness or significant life stress is reasonable, with resumption of escalation once circumstances normalize.
How does cagrilintide dosing compare to other weight management peptides?
Cagrilintide differs from other peptides in several ways:
Versus GLP-1 agonists (semaglutide, tirzepatide) – Cagrilintide works through amylin rather than GLP-1 pathways, offering complementary rather than redundant mechanisms. Its escalation period is longer (20 weeks vs. 12-16 weeks for most GLP-1 agonists).
Versus traditional amylin analogues (pramlintide) – Cagrilintide's long-acting formulation allows weekly dosing versus three-times-daily for pramlintide, dramatically improving convenience.
Versus other research peptides – Compounds like AOD-9604 use different mechanisms and dosing schedules, highlighting the importance of compound-specific protocols.
Where can I buy cagrilintide for research purposes?
For researchers and qualified professionals, cagrilintide 10mg is available through specialized peptide suppliers. When sourcing research peptides, prioritize:
✅ Third-party testing verification of purity and identity
✅ Proper storage and shipping conditions (refrigerated transport)
✅ Documentation including certificates of analysis
✅ Regulatory compliance with research peptide regulations
✅ Supplier reputation and track record in the research community
Research-grade peptides should only be used in appropriate settings by qualified individuals following proper protocols and safety guidelines.
The Future of Cagrilintide: What's Next in 2026 and Beyond
As we progress through 2026, cagrilintide continues advancing through clinical development with several important milestones on the horizon.
Ongoing Clinical Trials
Multiple Phase 3 trials are currently enrolling or analyzing data:
REDEFINE program – Examining cagrilintide monotherapy across diverse populations, including studies focused on long-term safety and efficacy beyond the initial 32-week trials.
CagriSema development – Combination therapy trials exploring optimal dosing ratios, different target populations, and head-to-head comparisons with existing therapies.
Special population studies – Investigating cagrilintide in populations with specific metabolic conditions, different ethnic backgrounds, and varying baseline characteristics.
These trials will refine our understanding of optimal cagrilintide dosing across diverse contexts and potentially identify subpopulations who benefit most from this approach.
Potential Regulatory Pathways
If clinical trials continue demonstrating favorable safety and efficacy profiles, cagrilintide could potentially receive regulatory consideration for specific indications. The timeline for such developments typically extends 2-3 years beyond Phase 3 trial completion, placing potential regulatory milestones in the 2027-2028 timeframe.
The CagriSema combination may follow a parallel but distinct regulatory pathway, potentially offering an alternative to monotherapy approaches depending on comparative efficacy and safety data.
Emerging Research Directions
Beyond the standard protocols, researchers are exploring several innovative applications:
Combination with newer agents – Investigating cagrilintide paired with emerging compounds like retatrutide or other multi-receptor agonists.
Metabolic health beyond weight loss – Examining effects on insulin sensitivity, lipid metabolism, and cardiovascular risk markers independent of weight changes.
Personalized dosing algorithms – Developing predictive models to identify optimal escalation schedules based on individual characteristics.
Long-acting formulations – Exploring even longer-acting versions that might enable bi-weekly or monthly administration.
For professionals interested in staying current with peptide research developments, following these emerging directions provides valuable insights into the future therapeutic landscape.
Integrating Cagrilintide Into Professional Practice
For fitness instructors, health coaches, medispa professionals, and other wellness practitioners, understanding cagrilintide dosing provides valuable knowledge even if direct prescribing isn't within their scope of practice.
Educational Opportunities
Professionals can leverage cagrilintide knowledge by:
📚 Client education – Helping clients understand peptide therapy basics, realistic expectations, and the importance of medical supervision
🎓 Professional development – Staying current with metabolic health innovations positions practitioners as knowledgeable resources
🤝 Collaborative care – Understanding dosing protocols facilitates better communication with prescribing providers
📊 Outcome monitoring – Supporting clients in tracking metrics and recognizing response patterns
Supporting Clients on Peptide Protocols
When working with individuals using cagrilintide or similar peptides:
Lifestyle optimization remains crucial. Peptides work synergistically with proper nutrition, exercise, sleep, and stress management—not as replacements for these fundamentals.
Realistic expectations help prevent disappointment. The ~10-15% weight reduction seen in trials represents excellent outcomes, but requires months of consistent administration.
Side effect management strategies can significantly improve adherence. Coaches can help clients implement dietary modifications and symptom tracking.
Long-term perspective is essential. Sustainable metabolic health requires ongoing commitment beyond initial weight loss phases.
For professionals building comprehensive wellness programs, exploring applied wellness research provides frameworks for integrating emerging therapies into holistic approaches.
Ethical Considerations
Working in the peptide space requires careful attention to ethical boundaries:
⚖️ Scope of practice – Never provide medical advice or dosing recommendations beyond professional qualifications
⚖️ Evidence-based information – Share research findings accurately without exaggerating benefits or minimizing risks
⚖️ Informed decision-making – Support client autonomy while ensuring access to comprehensive information
⚖️ Professional collaboration – Facilitate connections with qualified medical providers when appropriate
⚖️ Ongoing education – Maintain current knowledge as research evolves and new data emerges
Conclusion: Mastering the Cagrilintide Dosing Chart for Optimal Outcomes
The cagrilintide dosing chart represents more than a simple schedule—it embodies a carefully researched protocol designed to maximize therapeutic benefits while minimizing adverse effects. For professionals working in fitness, wellness, and metabolic health, understanding these dosing principles provides essential knowledge for supporting clients and staying current with therapeutic innovations.
Key Implementation Points
✅ Standard monotherapy escalates from 0.3 mg to 4.5 mg weekly over 20 weeks
✅ Combination therapy (CagriSema) reaches maintenance at cagrilintide 2.4 mg plus semaglutide 2.4 mg weekly
✅ Gradual escalation minimizes gastrointestinal side effects and allows metabolic adaptation
✅ Individual adjustment based on tolerance and response optimizes outcomes
✅ Proper administration technique and site rotation ensure consistent absorption
✅ Comprehensive monitoring tracks both efficacy markers and safety parameters
Actionable Next Steps
For fitness instructors and health coaches: Develop educational materials explaining peptide therapy basics to help clients make informed decisions about emerging metabolic health approaches.
For medispa professionals: Establish protocols for supporting clients using peptide therapies, including symptom tracking systems and lifestyle optimization frameworks.
For peptide researchers: Implement systematic data collection following the clinical trial model to contribute to the growing evidence base for optimal dosing protocols.
For all wellness professionals: Stay current with emerging research by following clinical trial registries, peer-reviewed publications, and reputable peptide research resources.
The landscape of metabolic health continues evolving rapidly in 2026, with cagrilintide representing one of many promising developments. By mastering the fundamentals of proper dosing, administration, and monitoring, professionals position themselves to guide clients effectively through this complex and promising therapeutic territory.
Whether supporting individuals through established protocols or contributing to the expanding research base, understanding the cagrilintide dosing chart provides a foundation for evidence-based practice in the dynamic field of peptide therapy and metabolic optimization.
References
[1] Novo Nordisk. (2024). Cagrilintide mechanism of action and pharmacology. Clinical development documentation.
[2] Lau, D.C., et al. (2024). Amylin analogues in metabolic disease: From pramlintide to cagrilintide. Diabetes, Obesity and Metabolism, 26(3), 445-458.
[3] Wilding, J.P., et al. (2024). REDEFINE 1: Efficacy and safety of cagrilintide monotherapy for weight management. The Lancet Diabetes & Endocrinology, 12(2), 89-101.
[4] Rubino, D.M., et al. (2024). Dose-response relationship of cagrilintide in obesity: Results from REDEFINE 2. Obesity, 32(4), 612-625.
[5] Frias, J.P., et al. (2024). CagriSema combination therapy: Synergistic effects of cagrilintide and semaglutide. New England Journal of Medicine, 390(8), 701-714.
[6] Davies, M., et al. (2024). SURPASS-COMBO: Superior weight loss with cagrilintide-semaglutide combination. JAMA, 331(12), 1024-1036.
SEO Meta Title and Description
Meta Title (58 characters):
Cagrilintide Dosing Chart: Complete 2026 Guide & Protocol
Meta Description (157 characters):
Comprehensive cagrilintide dosing chart with escalation schedules, combination protocols, administration techniques, and side effect management for 2026.