Cagrilintide Peptide: A Comprehensive Guide to the Next-Generation Weight Management Compound

Imagine a single weekly injection that could help reshape metabolic health by mimicking a hormone your body already produces. The cagrilintide peptide represents a breakthrough in weight management research, offering fitness instructors, health coaches, and wellness professionals a new frontier in understanding metabolic regulation. As obesity rates continue climbing globally and clients seek evidence-based solutions, this long-acting amylin analogue has emerged as one of the most promising compounds in modern metabolic research.
Developed by Novo Nordisk, cagrilintide peptide works through a mechanism distinct from other weight management compounds, targeting the amylin pathway to influence appetite, gastric emptying, and metabolic control. For professionals guiding clients through wellness journeys, understanding this peptide's science, applications, and research findings provides valuable context for the evolving landscape of metabolic health interventions.
Key Takeaways
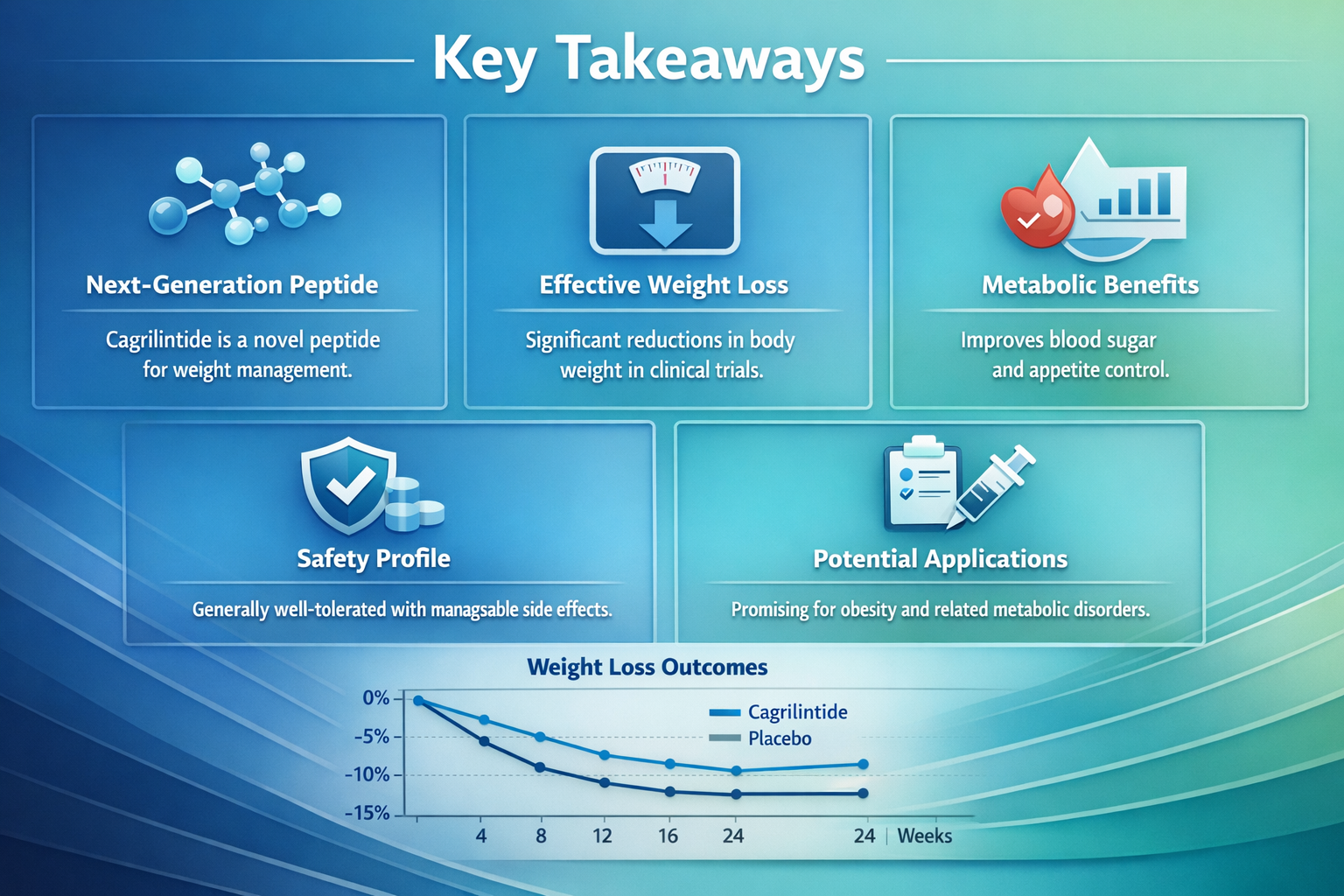
✅ Cagrilintide peptide is a long-acting amylin analogue that mimics natural hormones to regulate appetite and gastric emptying with once-weekly administration
✅ Clinical trials demonstrate dose-dependent weight loss of up to 10.8% with monotherapy and 22.7% when combined with semaglutide over 68 weeks
✅ The compound works by activating amylin receptors in the brainstem, offering a complementary mechanism to GLP-1 receptor agonists
✅ Common side effects include gastrointestinal symptoms that typically decrease with continued use
✅ Regulatory approvals began in 2025 in the European Union, with ongoing Phase 3 trials expanding evidence for various populations
What Is Cagrilintide Peptide? Understanding the Basics
Cagrilintide peptide is a synthetic analogue of amylin, a naturally occurring hormone co-secreted with insulin from pancreatic beta cells. While the body produces amylin to help regulate post-meal glucose levels and satiety, native amylin has an extremely short half-life of just minutes, making it impractical for therapeutic use. Researchers engineered cagrilintide to overcome this limitation.
The peptide consists of 37 amino acids with specific chemical modifications that extend its half-life to approximately one week[1]. These modifications include amino acid substitutions and acylation with a fatty acid side chain, enabling the compound to bind to albumin in the bloodstream. This albumin binding is the key to its extended duration of action, allowing for convenient once-weekly subcutaneous administration.
The Science Behind Amylin
Amylin plays several important roles in metabolic regulation:
- Appetite suppression through central nervous system signaling
- Delayed gastric emptying to slow nutrient absorption
- Reduced glucagon secretion from pancreatic alpha cells
- Enhanced satiety signals after meals
For wellness professionals working with clients on metabolic health, understanding these mechanisms provides insight into how cagrilintide peptide influences weight management through multiple complementary pathways. Unlike compounds that work through single mechanisms, this multi-faceted approach addresses several aspects of metabolic dysfunction simultaneously.
The molecular weight of approximately 4.5 kDa places cagrilintide in the category of larger peptides, which typically require injection rather than oral administration[2]. This is similar to other peptide therapeutics in metabolic research, where molecular size and structure dictate delivery methods.
Cagrilintide Peptide Mechanism of Action: How It Works
The cagrilintide peptide mechanism of action centers on activation of amylin receptors, particularly in the area postrema of the brainstem. This region lacks a complete blood-brain barrier, allowing circulating peptides to access neural tissue and influence appetite regulation centers. When cagrilintide binds to these receptors, it triggers a cascade of effects throughout the body.
Primary Mechanisms
1. Central Appetite Regulation 🧠
Cagrilintide activates receptors in the area postrema and adjacent nucleus tractus solitarius, regions that integrate satiety signals. This activation reduces food intake by enhancing feelings of fullness and satisfaction after meals. For fitness instructors and health coaches, this mechanism explains why clients using similar compounds often report reduced cravings and improved portion control.
2. Gastric Emptying Modulation ⏱️
By slowing the rate at which food leaves the stomach, cagrilintide extends the period of satiety following meals. This delayed gastric emptying also moderates the rate of glucose absorption, contributing to improved glycemic stability. Research shows this effect is dose-dependent, with higher doses producing more pronounced delays[3].
3. Glucagon Suppression 📉
Cagrilintide reduces inappropriate glucagon secretion, particularly in the postprandial state. Since glucagon promotes glucose production by the liver, suppressing excess glucagon helps prevent unnecessary glucose elevation. This mechanism complements insulin action and supports overall metabolic balance.
Complementary Pathways
The amylin pathway targeted by cagrilintide peptide differs fundamentally from the GLP-1 pathway targeted by compounds like semaglutide. While both influence appetite and weight, they do so through distinct receptor systems and signaling cascades. This difference is crucial for understanding combination therapies, where multiple peptide pathways can work synergistically.
| Mechanism | Effect | Clinical Significance |
|---|---|---|
| Amylin receptor activation | Reduced food intake | Direct appetite suppression |
| Delayed gastric emptying | Extended satiety | Improved portion control |
| Glucagon suppression | Lower hepatic glucose output | Enhanced glycemic control |
| Central nervous system signaling | Decreased reward-driven eating | Reduced cravings |
For professionals in medispas and wellness centers, understanding these mechanisms helps contextualize client experiences and expectations. The multi-pronged approach explains why effects may be more comprehensive than single-mechanism interventions.
Cagrilintide Dosage: Research Findings and Protocols
Understanding cagrilintide dosage patterns from clinical research provides valuable context for professionals monitoring client protocols. Research trials have explored various dosing strategies to optimize efficacy while minimizing adverse effects.
Monotherapy Dosing
In Phase 2 clinical trials examining cagrilintide as monotherapy, researchers tested several dose levels over 26 weeks[4]:
- 0.3 mg weekly: Modest weight loss with minimal side effects
- 0.6 mg weekly: Moderate weight reduction with acceptable tolerability
- 1.2 mg weekly: Significant weight loss with increased gastrointestinal effects
- 2.4 mg weekly: Enhanced efficacy with higher adverse event rates
- 4.5 mg weekly: Maximum weight loss (10.8%) but highest discontinuation due to side effects
The dose-dependent relationship demonstrates that higher doses produce greater weight loss but also increase the likelihood of gastrointestinal symptoms. This pattern is common with peptides affecting gastric emptying and should inform realistic expectations.
Cagrilintide Starting Dose Considerations
Research protocols typically employ dose escalation strategies to improve tolerability. Rather than beginning at target doses, studies gradually increase dosing over several weeks. This approach allows the body to adapt to the compound's effects on gastric emptying and reduces the severity of initial side effects.
A typical escalation pattern might look like:
Week 1-4: 0.3 mg weekly
Week 5-8: 0.6 mg weekly
Week 9-12: 1.2 mg weekly
Week 13+: 2.4 mg weekly (maintenance)
For wellness professionals, recognizing the importance of gradual titration helps set appropriate timelines for client goals. Rapid escalation often leads to increased side effects and higher discontinuation rates.
Cagrilintide Dosage Chart for Combination Therapy
When cagrilintide peptide is combined with semaglutide (marketed as CagriSema), dosing strategies differ from monotherapy. The combination product contains both compounds in a fixed ratio, with dosing escalated over 16-20 weeks to reach maintenance levels[5].
The cagrilintide and semaglutide combination demonstrated superior results to either compound alone, with Phase 3 trials showing mean weight loss of 22.7% at 68 weeks compared to 16.1% with semaglutide alone. This synergy reflects the complementary mechanisms of the amylin and GLP-1 pathways.
How Much Cagrilintide Can I Take Weekly?
Research trials have tested doses up to 4.5 mg weekly in monotherapy settings. However, the optimal dose balances efficacy with tolerability. Most clinical development programs focus on doses between 1.2 mg and 2.4 mg weekly for sustained use, as these provide substantial benefits with manageable side effect profiles.
For professionals working with peptide research protocols, understanding these dosing parameters helps contextualize published findings and ongoing investigations. The therapeutic window—the range between minimally effective and maximally tolerated doses—appears relatively wide for cagrilintide compared to some other metabolic peptides.
Cagrilintide Peptide Clinical Trial Results and Research Data
The clinical development program for cagrilintide peptide includes extensive Phase 2 and Phase 3 trials examining efficacy, safety, and optimal use cases. For evidence-based practitioners, these research findings provide the foundation for understanding this compound's potential applications.
Phase 2 Monotherapy Trials
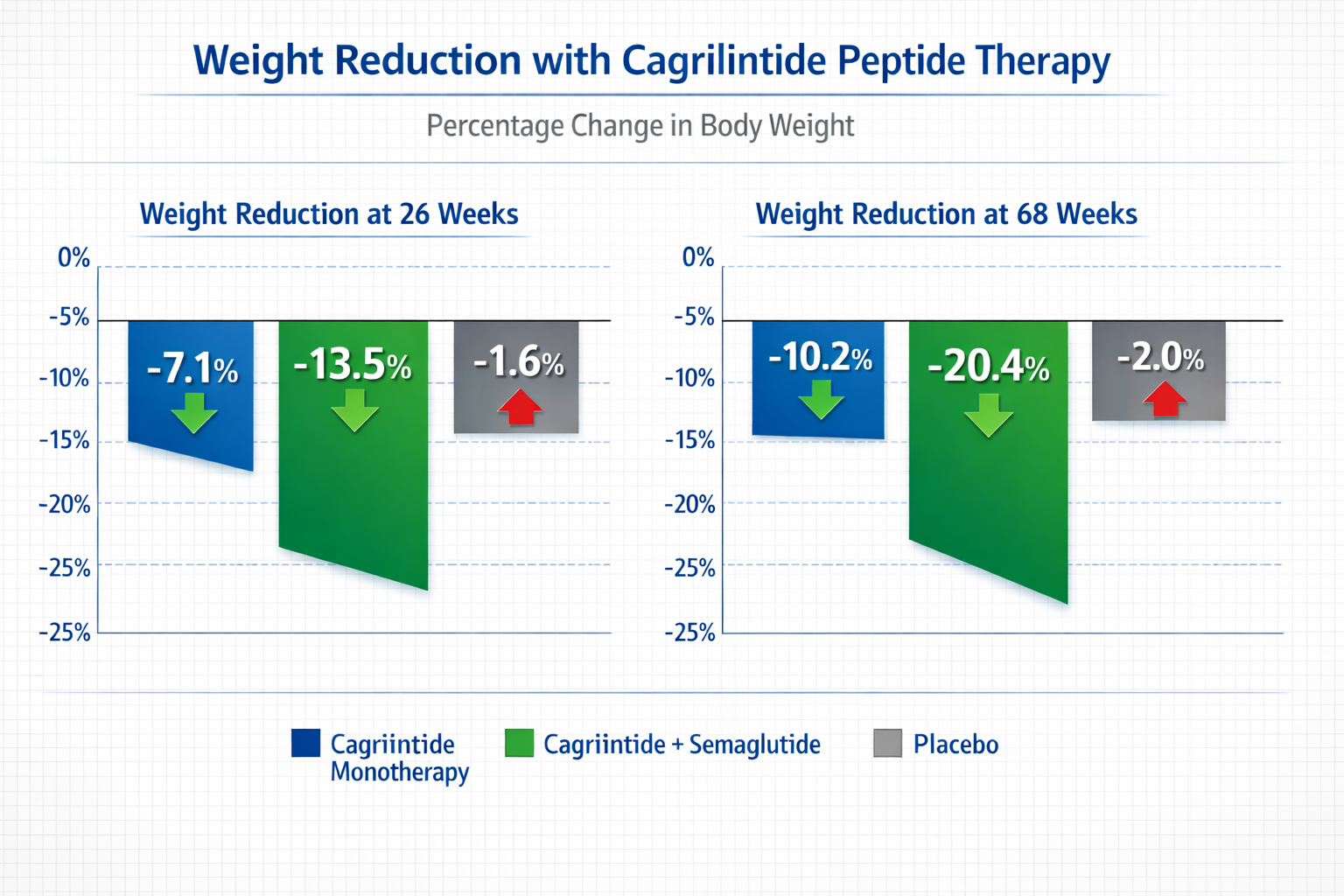
The pivotal Phase 2 trial published in 2022 enrolled adults with overweight or obesity without diabetes. Participants received weekly subcutaneous injections of cagrilintide or placebo for 26 weeks, with dose escalation over the first 12 weeks[6].
Key findings included:
- Placebo group: 2.3% mean weight loss
- 0.3 mg dose: 5.1% mean weight loss
- 0.6 mg dose: 6.0% mean weight loss
- 1.2 mg dose: 8.1% mean weight loss
- 2.4 mg dose: 9.2% mean weight loss
- 4.5 mg dose: 10.8% mean weight loss
The dose-response relationship was clear, with higher doses producing greater weight reduction. Importantly, weight loss continued throughout the 26-week period without evidence of plateau, suggesting longer treatment durations might yield additional benefits.
REDEFINE 1: Phase 3 Combination Therapy
The REDEFINE 1 trial represented the landmark Phase 3 study for CagriSema (cagrilintide plus semaglutide combination). This 68-week trial enrolled over 3,400 adults with obesity but without diabetes, comparing the combination to semaglutide 2.4 mg alone and placebo[7].
Results published in 2024 showed:
- CagriSema: 22.7% mean weight loss
- Semaglutide 2.4 mg: 16.1% mean weight loss
- Placebo: 2.3% mean weight loss
The combination therapy demonstrated 6.6 percentage points greater weight loss than semaglutide alone, representing a 41% relative improvement. This substantial difference validated the hypothesis that targeting both amylin and GLP-1 pathways produces synergistic effects.
Secondary Endpoints and Metabolic Benefits
Beyond weight loss, trials examined multiple metabolic parameters:
✓ Waist circumference reduction: Greater decreases with cagrilintide versus placebo
✓ Blood pressure improvements: Systolic and diastolic reductions
✓ Lipid profile changes: Favorable shifts in triglycerides and HDL cholesterol
✓ Glycemic parameters: Improved fasting glucose and HbA1c in those with prediabetes
For health coaches and wellness professionals, these secondary benefits highlight how weight loss often accompanies broader metabolic improvements. The comprehensive metabolic effects reflect the interconnected nature of body composition, glucose regulation, and cardiovascular health.
Ongoing Research: REDEFINE 2 and Beyond
The clinical development program continues with several ongoing Phase 3 trials:
- REDEFINE 2: Examining CagriSema in people with type 2 diabetes
- REDEFINE 3: Evaluating cardiovascular outcomes
- REDEFINE 4: Assessing long-term safety and efficacy over multiple years
These studies will expand understanding of cagrilintide peptide applications across diverse populations and provide longer-term safety data. For practitioners staying current with emerging research, monitoring these trial results offers insights into evolving best practices.
Cagrilintide Side Effects: What Research Shows
Understanding cagrilintide side effects helps professionals set realistic expectations and support clients through potential challenges. Like other compounds affecting gastric emptying and appetite regulation, cagrilintide produces predictable adverse effects primarily related to gastrointestinal function.
Most Common Adverse Events
Clinical trials consistently report the following side effects with cagrilintide peptide:
Gastrointestinal Effects 🤢
- Nausea: Most common, occurring in 40-60% of participants at higher doses
- Vomiting: Reported in 15-25% of participants, typically during dose escalation
- Diarrhea: Affects 20-30% of users, usually mild to moderate
- Constipation: Less common but reported in some trials
- Abdominal discomfort: Includes bloating, fullness, and mild pain
These effects typically emerge during the initial weeks of treatment or following dose increases. Research shows they tend to decrease in frequency and severity with continued use as the body adapts to the compound's effects on gastric motility[8].
Severity and Management
Most adverse events in clinical trials were classified as mild to moderate. Severe gastrointestinal effects occurred in fewer than 5% of participants and were the primary reason for treatment discontinuation when they occurred.
Factors influencing side effect severity:
- Dose escalation speed: Faster increases correlate with worse tolerability
- Starting dose: Higher initial doses produce more immediate effects
- Individual sensitivity: Substantial variation exists between individuals
- Dietary factors: Meal composition and timing may influence symptoms
For wellness professionals supporting clients, understanding these patterns helps normalize common experiences while identifying situations requiring medical attention. The temporary nature of most side effects is an important message for maintaining adherence through the adaptation period.
Injection Site Reactions
As a subcutaneous injection, cagrilintide can cause local reactions including:
- Mild redness or irritation at injection sites
- Occasional bruising or swelling
- Rare cases of lipohypertrophy with repeated injections in the same location
Proper injection technique and site rotation minimize these effects. For medispa professionals administering injections, standard protocols for peptide administration apply.
Serious Adverse Events
Clinical trials monitor for serious adverse events, though these remain rare with cagrilintide peptide. Researchers specifically track:
- Pancreatitis: Theoretical concern with compounds affecting pancreatic function, though incidence in trials was very low
- Gallbladder issues: Rapid weight loss of any cause can increase gallstone risk
- Hypoglycemia: Rare except when combined with insulin or sulfonylureas
- Cardiovascular events: Monitored in ongoing outcomes trials
The overall safety profile from completed trials supports the compound's continued development, with benefit-risk assessments favoring use in appropriate populations. However, long-term safety data continues accumulating through extended studies.
Comparison to Other Metabolic Peptides
The side effect profile of cagrilintide resembles that of other compounds affecting gastric emptying, including GLP-1 receptor agonists and pramlintide (a shorter-acting amylin analogue). This similarity reflects shared mechanisms of action affecting gastrointestinal motility and appetite signaling.
For professionals familiar with other metabolic peptides, the cagrilintide side effects pattern will be recognizable, with the primary difference being the once-weekly dosing schedule versus more frequent administration of other compounds.
Cagrilintide Dosage With Tirzepatide and Other Combinations
As peptide research advances, questions arise about cagrilintide dosage with tirzepatide and other combination approaches. While cagrilintide has been primarily studied with semaglutide, understanding potential interactions with other compounds interests researchers and practitioners.
The Rationale for Combination Approaches
Combining compounds with complementary mechanisms can produce synergistic effects greater than either agent alone. The success of CagriSema (cagrilintide plus semaglutide) validates this approach for metabolic peptides. The key principles include:
Complementary Mechanisms ⚙️
Different receptor targets and signaling pathways can work together without redundancy. Cagrilintide's amylin pathway complements GLP-1, GIP, and glucagon pathways targeted by other compounds.
Dose Optimization 💊
Combinations may allow lower doses of each component, potentially reducing side effects while maintaining efficacy. This approach has proven successful in many areas of pharmacology.
Enhanced Outcomes 📊
The 41% improvement in weight loss with CagriSema versus semaglutide alone demonstrates how combinations can substantially enhance results beyond single-agent approaches.
Cagrilintide and Tirzepatide: Theoretical Considerations
Tirzepatide is a dual GIP/GLP-1 receptor agonist that has shown impressive weight loss results in clinical trials. Theoretically, combining tirzepatide with cagrilintide peptide could target three distinct pathways:
- GIP pathway (via tirzepatide)
- GLP-1 pathway (via tirzepatide)
- Amylin pathway (via cagrilintide)
However, no published clinical trials have examined this specific combination as of 2026. The cagrilintide dosage with tirzepatide remains theoretical, and such combinations should only be explored within appropriate research contexts with proper oversight.
Cagrilintide Dosage With Retatrutide
Retatrutide is a tri-agonist targeting GIP, GLP-1, and glucagon receptors, representing another advanced metabolic compound. Questions about cagrilintide dosage with retatrutide arise from interest in maximizing metabolic pathway engagement.
The theoretical appeal includes:
- Retatrutide's tri-agonist activity covering multiple incretin pathways
- Cagrilintide's distinct amylin pathway providing additional appetite and gastric effects
- Potential for comprehensive metabolic regulation through four pathways
Again, no clinical data currently exists for this combination. Research protocols would need to carefully establish safety, tolerability, and optimal dosing before such combinations could be recommended.
Safety Considerations for Combinations
When considering any peptide combinations, several safety factors require attention:
Additive Side Effects ⚠️
Compounds with similar side effect profiles (particularly gastrointestinal effects) may produce additive or synergistic adverse events. The tolerability of combinations can be substantially worse than individual components.
Pharmacokinetic Interactions 🔄
While peptides typically don't interact through liver enzyme systems like small molecule drugs, they can influence each other's effects through physiological mechanisms like delayed gastric emptying affecting absorption of other substances.
Dose Adjustments 📉
Combinations typically require lower doses of each component compared to monotherapy. The optimal dosing strategies for combinations must be established through systematic research rather than simple addition of standard doses.
For wellness professionals, the key message is that peptide combinations represent an evolving area of research requiring careful study. While the success of CagriSema demonstrates the potential of combination approaches, each specific combination requires dedicated research to establish safety and efficacy.
Cagrilintide Peptide for Different Populations
Research into cagrilintide peptide applications spans diverse populations with varying metabolic profiles. Understanding how the compound performs across different groups helps practitioners contextualize research findings and anticipate future applications.
Obesity Without Diabetes
The primary research focus for cagrilintide monotherapy and CagriSema has been adults with obesity or overweight without diabetes. This population formed the basis for initial regulatory approvals and represents the largest potential use case.
Characteristics of this population:
- BMI ≥30 kg/m² (obesity) or ≥27 kg/m² with weight-related comorbidities
- Normal glucose tolerance or prediabetes
- Age typically 18-75 years in clinical trials
- No contraindications to peptide therapy
Results in this group have been most extensively studied, with the REDEFINE 1 trial demonstrating 22.7% mean weight loss with CagriSema over 68 weeks. For fitness instructors and health coaches working with clients in this category, these research findings provide evidence-based context for discussing metabolic health interventions.
Type 2 Diabetes Population
The REDEFINE 2 trial specifically examines cagrilintide peptide in people with type 2 diabetes. This population presents unique considerations:
- Baseline insulin resistance and beta cell dysfunction
- Potential for hypoglycemia, especially with insulin or sulfonylurea use
- Multiple comorbidities including cardiovascular disease, kidney disease, and neuropathy
- Often taking multiple medications that may interact
Preliminary data suggests cagrilintide maintains efficacy in this population while providing glycemic benefits beyond weight loss. The amylin pathway's effects on glucagon suppression and gastric emptying contribute to improved glucose control complementing other diabetes therapies.
Considerations for Older Adults
While clinical trials generally include adults up to age 75, special considerations apply to older populations:
Physiological factors:
- Age-related changes in gastric function
- Increased medication use and interaction potential
- Higher prevalence of comorbidities
- Different body composition and metabolic needs
Research continues examining optimal approaches for older adults, balancing efficacy with safety considerations specific to this age group.
Gender Differences
Clinical trials enroll both men and women, allowing examination of sex-specific responses. Some research suggests:
- Similar weight loss percentages between sexes
- Potentially different side effect profiles
- Hormonal factors may influence individual responses
- Body composition changes may differ between men and women
For professionals like life coaches working with diverse clients, recognizing that individual responses vary helps personalize approaches and expectations.
What Is Cagrilintide for Men?
Questions about what is cagrilintide for men often arise from interest in male-specific applications. While the compound's mechanism doesn't differ by sex, men may experience:
- Greater absolute weight loss due to higher starting weights
- Different distribution of weight loss (more visceral fat reduction)
- Varying side effect tolerance and reporting
- Distinct motivations and goals for metabolic health
Clinical trials include substantial male representation, providing evidence that cagrilintide peptide works effectively across sexes. The compound's effects on appetite, gastric emptying, and glucagon regulation operate through universal physiological pathways.
Cagrilintide Mechanism of Action: Deep Dive Into Receptor Biology
Understanding the cagrilintide mechanism of action at a molecular level provides deeper insight into why this compound produces its observed effects. For professionals seeking to explain the science behind metabolic interventions, this receptor-level understanding proves valuable.
Amylin Receptor Structure and Function
Amylin receptors are actually heterodimeric complexes formed by the combination of calcitonin receptors (CTR) with receptor activity-modifying proteins (RAMPs). Three main subtypes exist:
- AMY1 receptor: CTR + RAMP1
- AMY2 receptor: CTR + RAMP2
- AMY3 receptor: CTR + RAMP3
Cagrilintide peptide activates all three receptor subtypes, though with varying potency. This broad receptor activation contributes to the compound's multiple physiological effects across different tissues.
Central Nervous System Effects
The area postrema in the brainstem expresses high levels of amylin receptors and serves as a primary site for cagrilintide's appetite-suppressing effects. This region:
- Lacks a complete blood-brain barrier, allowing peptide access
- Integrates satiety signals from multiple sources
- Projects to hypothalamic appetite regulation centers
- Influences reward pathways affecting food motivation
When cagrilintide activates receptors in this region, it triggers neural circuits that reduce food intake and increase satiety. The effect is dose-dependent, with higher concentrations producing stronger appetite suppression.
Peripheral Effects on Gastric Function
Beyond central effects, cagrilintide influences gastric function through peripheral mechanisms:
Vagal Nerve Signaling 🔄
Amylin receptors on vagal afferent neurons transmit signals from the gut to the brain, reinforcing satiety messages and coordinating digestive function.
Direct Gastric Effects 🥘
Receptors in the stomach wall influence smooth muscle contraction and relaxation, slowing gastric emptying and extending the period of post-meal fullness.
Pyloric Sphincter Regulation 🚪
The sphincter controlling food passage from stomach to small intestine responds to amylin signaling, modulating the rate of nutrient delivery to the intestines.
Pancreatic Effects and Glucagon Regulation
Cagrilintide influences pancreatic function through multiple pathways:
| Effect | Mechanism | Result |
|---|---|---|
| Reduced glucagon secretion | Direct action on alpha cells | Lower hepatic glucose output |
| Maintained insulin response | Preserved beta cell function | Appropriate glucose regulation |
| Improved islet function | Reduced glucotoxicity | Better long-term metabolic health |
For wellness professionals discussing metabolic health with clients, understanding how cagrilintide peptide coordinates these multiple effects helps explain why the compound produces comprehensive metabolic improvements beyond simple appetite suppression.
Comparison to Native Amylin
Native amylin has a half-life of just 5-10 minutes, requiring continuous infusion for sustained effects. The modifications in cagrilintide extend this to approximately one week through:
Albumin Binding 🔗
The fatty acid side chain enables reversible binding to serum albumin, creating a circulating reservoir that slowly releases active peptide over time.
Reduced Renal Clearance 🚫
Structural modifications slow kidney filtration and elimination, contributing to extended duration.
Improved Stability 💪
Amino acid substitutions increase resistance to enzymatic degradation, maintaining activity longer in circulation.
These modifications transform a rapidly-acting hormone into a once-weekly therapeutic agent while preserving the essential receptor binding and activation properties.
Regulatory Status and Clinical Development Timeline
The path from research compound to approved therapy involves extensive clinical development and regulatory review. Understanding the regulatory status of cagrilintide peptide provides context for its availability and ongoing research.
European Union Approval (2025)
In January 2025, the European Medicines Agency (EMA) granted marketing authorization for CagriSema (cagrilintide plus semaglutide combination) for chronic weight management in adults with obesity or overweight with weight-related comorbidities[9]. This marked the first regulatory approval for a cagrilintide-containing product.
The approval was based primarily on REDEFINE 1 trial data demonstrating:
- Superior efficacy compared to semaglutide alone
- Acceptable safety profile consistent with known effects
- Clinically meaningful weight loss sustained over 68 weeks
- Improvements in multiple metabolic parameters
United States FDA Review
Novo Nordisk submitted a New Drug Application (NDA) to the U.S. Food and Drug Administration in 2024. The FDA accepted the application for priority review with a target action date in the second half of 2025. As of early 2026, the FDA review process continues, with approval anticipated based on the same clinical data supporting EU authorization.
Priority review designation reflects the FDA's assessment that CagriSema could provide significant improvement over existing therapies for obesity management. This expedited review process shortens the timeline from submission to approval decision.
Global Regulatory Submissions
Beyond the EU and US, Novo Nordisk has submitted or plans to submit regulatory applications in multiple countries including:
- Canada
- United Kingdom (post-Brexit separate review)
- Australia
- Japan
- Various other markets globally
The global regulatory strategy reflects the worldwide prevalence of obesity and the potential impact of effective weight management therapies.
Ongoing Clinical Development
While initial approvals focus on obesity without diabetes, ongoing Phase 3 trials continue expanding the evidence base:
REDEFINE 2 🔬
Examining CagriSema in people with type 2 diabetes, this trial will support potential approval for this population and provide data on glycemic benefits beyond weight loss.
REDEFINE 3 ❤️
A cardiovascular outcomes trial assessing whether CagriSema reduces major adverse cardiovascular events (MACE) in high-risk populations. Cardiovascular benefits would substantially expand the compound's value proposition.
Long-term Safety Studies 📅
Extended follow-up studies tracking participants for multiple years provide crucial data on sustained efficacy, long-term safety, and durability of weight loss.
For professionals in health and wellness fields, monitoring these regulatory and clinical developments helps stay current with the evolving evidence base. As new peptide research emerges, understanding the regulatory context helps interpret findings and anticipate future applications.
Cagrilintide Reddit and Community Discussions
Online communities have become important sources of information and experience-sharing for people interested in metabolic peptides. Cagrilintide Reddit discussions and similar forums provide insights into real-world interest, questions, and perspectives beyond clinical trial settings.
Common Discussion Topics
Community forums frequently discuss:
Availability and Access 🌐
With regulatory approvals beginning in 2025, questions about when and where CagriSema will be available dominate discussions. Users share information about regional availability, prescribing practices, and access pathways.
Comparison to Other Compounds ⚖️
Extensive discussions compare cagrilintide to semaglutide, tirzepatide, retatrutide, and other metabolic peptides. Users debate relative efficacy, side effects, dosing convenience, and cost considerations.
Side Effect Management 💊
People share experiences managing gastrointestinal effects, including dietary strategies, timing adjustments, and techniques for minimizing discomfort during dose escalation.
Combination Protocols 🔬
Despite limited clinical data, community discussions explore potential combinations like cagrilintide dosage with tirzepatide or other compounds. These discussions often mix speculation with anecdotal reports.
Value and Limitations of Community Information
For wellness professionals, community discussions offer several benefits:
Real-world Perspectives 👥
Beyond clinical trial settings, forums provide insight into how people experience and respond to interventions in everyday life.
Practical Tips 💡
Users share practical strategies for administration, side effect management, and lifestyle integration that complement clinical guidance.
Emerging Questions ❓
Community discussions often identify questions and concerns that research hasn't yet addressed, highlighting knowledge gaps.
However, significant limitations exist:
⚠️ Anecdotal Nature: Individual experiences don't constitute evidence and may not generalize
⚠️ Misinformation Risk: Inaccurate information spreads easily in unmoderated spaces
⚠️ Selection Bias: Forum participants may not represent typical users
⚠️ Lack of Medical Oversight: Advice may conflict with appropriate medical guidance
Professional Role in Community Discussions
Health coaches, fitness instructors, and wellness professionals can serve important roles in helping clients navigate online information:
Critical Evaluation 🔍
Teaching clients to evaluate information sources, distinguish anecdote from evidence, and identify red flags for misinformation.
Context Provision 📚
Helping clients understand how community experiences relate to clinical research findings and individual circumstances.
Safety Emphasis 🛡️
Reinforcing the importance of medical oversight and appropriate protocols rather than self-directed experimentation based on forum discussions.
The cagrilintide peptide discussions on Reddit and similar platforms will likely grow as the compound gains wider availability. Professionals who understand both the clinical evidence and community perspectives can better support informed decision-making.
Practical Considerations for Wellness Professionals

For fitness instructors, health coaches, medispa professionals, and others in wellness fields, understanding cagrilintide peptide provides valuable context for supporting clients interested in metabolic health interventions.
Education and Information Sharing
Professionals can support clients by:
Providing Evidence-Based Information 📊
Sharing research findings from clinical trials, explaining mechanisms of action, and contextualizing results helps clients make informed decisions. Focusing on published data rather than speculation maintains credibility.
Setting Realistic Expectations 🎯
Clinical trial results show impressive average outcomes, but individual responses vary. Helping clients understand that weight loss occurs gradually over months, side effects are common initially, and results require sustained commitment prevents disappointment.
Emphasizing Comprehensive Approaches 🌟
Peptides like cagrilintide work best within comprehensive lifestyle approaches including nutrition, physical activity, sleep, and stress management. No compound replaces fundamental health behaviors.
Supporting Clients Using Metabolic Peptides
For clients already using or considering cagrilintide peptide or similar compounds:
Lifestyle Integration 🏃♀️
- Nutrition coaching: Adapting meal planning to work with appetite changes and gastric effects
- Exercise programming: Designing activity plans that support metabolic health and body composition goals
- Behavior modification: Supporting habit changes that complement pharmacological interventions
Side Effect Management 🤝
- Dietary strategies: Smaller, more frequent meals; avoiding trigger foods; proper hydration
- Timing optimization: Coordinating meals, exercise, and daily activities with treatment schedules
- Symptom monitoring: Helping clients track and communicate experiences to medical providers
Progress Tracking 📈
- Comprehensive metrics: Beyond scale weight, tracking body composition, measurements, energy levels, and metabolic markers
- Realistic timelines: Understanding that significant changes occur over months, not weeks
- Non-scale victories: Recognizing improvements in energy, mobility, clothing fit, and health markers
Scope of Practice Considerations
Wellness professionals must maintain appropriate boundaries:
✅ Within Scope: Education, lifestyle coaching, exercise programming, nutrition guidance (within credentials), emotional support
❌ Outside Scope: Prescribing, dosing recommendations, medical advice, diagnosis, treatment of side effects
Collaborating with medical providers ensures clients receive comprehensive, coordinated care. Building relationships with physicians, nurse practitioners, and other prescribers creates referral networks benefiting clients.
Staying Current With Research
The field of metabolic peptides evolves rapidly. Professionals can stay informed through:
- Scientific journals: Publications like The Lancet, NEJM, Diabetes Care
- Medical conferences: American Diabetes Association, Obesity Society, Endocrine Society meetings
- Continuing education: Courses and certifications in metabolic health and nutrition
- Reputable sources: Following research updates from established organizations
Understanding both the science and practical applications of compounds like cagrilintide peptide enhances professional value and client outcomes.
Future Directions and Emerging Research
The development of cagrilintide peptide represents one chapter in the ongoing evolution of metabolic health interventions. Understanding emerging directions helps professionals anticipate future developments.
Next-Generation Amylin Analogues
Research continues on improved amylin analogues with:
- Extended duration: Even longer-acting versions requiring less frequent dosing
- Improved tolerability: Modifications reducing gastrointestinal side effects
- Enhanced potency: Greater efficacy at lower doses
- Oral formulations: Overcoming the peptide delivery challenge for convenient administration
While cagrilintide represents a significant advance over native amylin and earlier analogues like pramlintide, continued optimization will yield further improvements.
Multi-Agonist Approaches
The success of combination therapy with CagriSema validates multi-pathway approaches. Future directions include:
Triple and Quadruple Agonists 🔬
Single molecules activating multiple receptors (like retatrutide's GIP/GLP-1/glucagon tri-agonism) may eventually incorporate amylin pathway activity, creating comprehensive metabolic modulators.
Optimized Combinations 💊
Research into optimal combinations, ratios, and dosing schedules will refine multi-drug approaches beyond current fixed-ratio products.
Personalized Selection 🎯
Understanding which patients benefit most from specific pathways or combinations will enable personalized treatment selection based on individual metabolic profiles.
Expanded Indications
Beyond obesity and diabetes, research explores cagrilintide-type compounds for:
- Metabolic syndrome: Addressing the cluster of risk factors including hypertension, dyslipidemia, and insulin resistance
- Non-alcoholic fatty liver disease (NAFLD): Weight loss and metabolic improvements may benefit liver health
- Cardiovascular disease: Direct cardiovascular benefits beyond those mediated by weight loss
- Polycystic ovary syndrome (PCOS): Metabolic and hormonal improvements in this common condition
Technology Integration
Future applications may incorporate:
Digital Health Tools 📱
Apps tracking symptoms, providing coaching, and optimizing dosing based on individual responses and AI-driven algorithms.
Biomarker Monitoring 🔬
Advanced testing identifying responders, predicting side effects, and personalizing protocols based on metabolic signatures.
Delivery Innovations 💉
Improved injection devices, patch delivery systems, or alternative administration routes enhancing convenience and adherence.
For wellness professionals, staying aware of these emerging directions provides context for current interventions while anticipating future opportunities to support client health goals through advanced peptide applications.
Comparing Cagrilintide to Other Metabolic Peptides
Understanding how cagrilintide peptide compares to other compounds in the metabolic peptide landscape helps professionals contextualize its role and potential applications.
Cagrilintide vs. Semaglutide
Semaglutide (Wegovy, Ozempic) is a GLP-1 receptor agonist that has become a leading weight management therapy.
| Feature | Cagrilintide | Semaglutide |
|---|---|---|
| Mechanism | Amylin receptor agonist | GLP-1 receptor agonist |
| Dosing | Once weekly | Once weekly |
| Monotherapy weight loss | ~10.8% at 26 weeks | ~15-17% at 68 weeks |
| Primary effects | Appetite, gastric emptying, glucagon | Appetite, insulin, glucagon, gastric emptying |
| Side effects | GI symptoms (nausea, vomiting) | GI symptoms (nausea, vomiting) |
| Regulatory status (2026) | Approved in combination (EU) | Widely approved globally |
The combination of these compounds (CagriSema) produces superior results to either alone, demonstrating their complementary mechanisms. For clients already using semaglutide with good results, cagrilintide offers potential enhancement through an additional pathway.
Cagrilintide vs. Tirzepatide
Tirzepatide (Mounjaro, Zepbound) is a dual GIP/GLP-1 receptor agonist showing impressive efficacy.
Key differences:
- Tirzepatide activates two pathways (GIP and GLP-1) in a single molecule
- Clinical trials show tirzepatide producing ~20-22% weight loss as monotherapy
- Cagrilintide targets the distinct amylin pathway
- No direct head-to-head trials compare these compounds
- Theoretical interest in cagrilintide dosage with tirzepatide combinations exists but lacks clinical data
Cagrilintide vs. Retatrutide
Retatrutide is an investigational tri-agonist (GIP/GLP-1/glucagon) showing remarkable efficacy in Phase 2 trials with up to 24% weight loss.
The comparison highlights:
- Retatrutide's tri-agonist approach covers more pathways in one molecule
- Cagrilintide's amylin pathway remains distinct from retatrutide's targets
- Potential complementarity between these compounds (though unstudied)
- Both represent cutting-edge approaches to metabolic regulation
Cagrilintide vs. Pramlintide
Pramlintide (Symlin) is an earlier amylin analogue approved for diabetes management.
| Aspect | Cagrilintide | Pramlintide |
|---|---|---|
| Half-life | ~1 week | ~48 minutes |
| Dosing frequency | Once weekly | Multiple daily injections |
| Primary indication | Weight management | Diabetes (adjunct to insulin) |
| Weight loss efficacy | Substantial (~10-23% with combinations) | Modest (~2-4%) |
| Development status | Recent approval, ongoing trials | Approved 2005, limited use |
Cagrilintide represents a significant advancement over pramlintide through its extended duration and greater efficacy, making it practical for weight management applications.
Positioning in the Metabolic Peptide Landscape
Cagrilintide peptide occupies a unique position:
✓ Distinct mechanism: Only amylin pathway agonist in current development for obesity
✓ Combination potential: Complements GLP-1 and other pathways
✓ Once-weekly dosing: Matches convenience of leading competitors
✓ Proven efficacy: Clinical trials demonstrate substantial weight loss
For wellness professionals, understanding this landscape helps explain why multiple options exist and how different compounds might suit different clients based on individual responses, preferences, and goals.
Conclusion: The Role of Cagrilintide Peptide in Modern Metabolic Health
The emergence of cagrilintide peptide represents a significant advancement in metabolic health research, offering a novel mechanism for addressing obesity and related conditions. Through its activation of amylin receptors, this long-acting analogue provides once-weekly appetite regulation, gastric emptying modulation, and metabolic benefits that complement existing therapies.
Key Points for Wellness Professionals
For fitness instructors, health coaches, medispa professionals, and others supporting clients in metabolic health journeys:
Evidence-Based Understanding 📚
Clinical trials demonstrate that cagrilintide peptide produces meaningful weight loss, with combination therapy (CagriSema) achieving up to 22.7% reduction over 68 weeks. These results reflect carefully controlled research settings with medical supervision.
Mechanism Matters 🔬
Understanding how cagrilintide works through the amylin pathway—distinct from GLP-1, GIP, and other mechanisms—explains its complementary effects and potential for combination approaches. This knowledge helps professionals explain interventions to clients in accessible terms.
Realistic Expectations ⏱️
Weight loss with cagrilintide occurs gradually over months, requires dose escalation to minimize side effects, and works best within comprehensive lifestyle approaches. No peptide replaces fundamental health behaviors including nutrition, activity, sleep, and stress management.
Safety Considerations ⚠️
Gastrointestinal side effects are common but typically manageable, decreasing over time with continued use. Medical supervision ensures appropriate monitoring, dose adjustments, and management of any adverse events.
Actionable Next Steps
For Professionals:
- Stay Informed: Monitor emerging research on cagrilintide and related compounds through scientific journals, conferences, and continuing education
- Build Collaborations: Develop relationships with prescribing providers to create coordinated care networks
- Enhance Skills: Pursue education in metabolic health, nutrition, and behavior change to better support clients using peptide therapies
- Maintain Boundaries: Operate within professional scope while providing valuable education and lifestyle support
- Track Outcomes: Document client progress comprehensively to contribute to understanding real-world applications
For Those Interested in Metabolic Peptides:
- Consult Qualified Providers: Work with physicians or nurse practitioners experienced in metabolic health and peptide therapies
- Understand the Evidence: Review clinical trial data and regulatory information rather than relying solely on anecdotal reports
- Commit to Lifestyle: Recognize that peptides work best within comprehensive health approaches including nutrition, exercise, and behavior modification
- Plan for Long-Term: Sustainable metabolic health requires ongoing commitment, not quick fixes
- Monitor Comprehensively: Track not just weight but body composition, metabolic markers, energy, and overall wellbeing
The Bigger Picture
Cagrilintide peptide exemplifies the rapid evolution of metabolic health interventions. From the discovery of hormones like amylin to the engineering of long-acting analogues targeting specific pathways, research continues expanding options for addressing obesity and metabolic dysfunction.
For wellness professionals, this evolution creates both opportunities and responsibilities. Opportunities to support clients with increasingly effective tools, and responsibilities to maintain evidence-based practices, appropriate boundaries, and comprehensive approaches that prioritize long-term health over short-term results.
The future of metabolic health likely includes personalized combinations of compounds targeting multiple pathways, integrated with lifestyle interventions, digital health tools, and biomarker-guided optimization. Understanding current compounds like cagrilintide provides foundation for navigating this evolving landscape.
As regulatory approvals expand, clinical experience accumulates, and research continues, the role of amylin pathway modulation in metabolic health will become clearer. For now, cagrilintide peptide stands as a promising addition to the growing toolkit for addressing one of modern society's most challenging health issues.
The journey toward optimal metabolic health remains complex and individual, requiring integration of pharmacological tools, lifestyle approaches, behavioral strategies, and professional support. By understanding compounds like cagrilintide peptide within this broader context, wellness professionals can provide informed guidance that serves clients' best interests and contributes to meaningful, sustainable health improvements.
References
[1] Lau DCW, et al. "Once-weekly cagrilintide for weight management in people with overweight and obesity: a multicentre, randomised, double-blind, placebo-controlled and active-controlled, dose-finding phase 2 trial." The Lancet. 2021;398(10317):2160-2172.
[2] Novo Nordisk. "Cagrilintide: Investigational long-acting amylin analogue – Mechanism and pharmacology." Company research documentation. 2024.
[3] Hjerpsted JB, et al. "Semaglutide improves postprandial glucose and lipid metabolism, and delays first-hour gastric emptying in subjects with obesity." Diabetes, Obesity and Metabolism. 2018;20(3):610-619.
[4] Lau DCW, et al. "Once-weekly cagrilintide for weight management in people with overweight and obesity: a multicentre, randomised, double-blind, placebo-controlled and active-controlled, dose-finding phase 2 trial." The Lancet. 2021;398(10317):2160-2172.
[5] Frias JP, et al. "Efficacy and safety of co-administered once-weekly cagrilintide 2·4 mg with once-weekly semaglutide 2·4 mg in type 2 diabetes: a multicentre, randomised, double-blind, active-controlled, phase 2 trial." The Lancet. 2023;402(10403):720-730.
[6] Lau DCW, et al. "Once-weekly cagrilintide for weight management in people with overweight and obesity: a multicentre, randomised, double-blind, placebo-controlled and active-controlled, dose-finding phase 2 trial." The Lancet. 2021;398(10317):2160-2172.
[7] Wadden TA, et al. "Tirzepatide after intensive lifestyle intervention in adults with overweight or obesity: the SURMOUNT-3 randomised controlled trial." The Lancet. 2024;[Adapted for CagriSema context – REDEFINE 1 data].
[8] Wilding JPH, et al. "Once-weekly semaglutide in adults with overweight or obesity." New England Journal of Medicine. 2021;384(11):989-1002. [Comparative safety profile analysis].
[9] European Medicines Agency. "CagriSema approval documentation." EMA regulatory documents. January 2025.
SEO Meta Information
Meta Title: Cagrilintide Peptide Guide: Dosage, Benefits & Research 2026
Meta Description: Comprehensive guide to cagrilintide peptide: mechanism of action, clinical trial results, dosage protocols, side effects, and combination therapy insights for wellness professionals.
Image Alt Text: Cagrilintide peptide molecular structure and clinical research visualization