Cagrilintide Peptide Dosage: A Complete Guide for Health Professionals in 2026

The landscape of metabolic health interventions has shifted dramatically with the emergence of novel peptide therapies. Among these breakthrough compounds, cagrilintide stands out as a long-acting amylin analogue that's capturing attention across wellness clinics, medispas, and research facilities worldwide. Understanding cagrilintide peptide dosage has become essential for fitness instructors, life coaches, and health professionals who want to stay informed about cutting-edge metabolic research. With clinical trials demonstrating significant outcomes at specific dose ranges, knowing how to interpret dosing protocols, escalation schedules, and combination strategies can help professionals guide informed conversations with their clients about emerging peptide research.
Key Takeaways
- Cagrilintide dosing ranges from 0.3 mg to 4.5 mg weekly in clinical trials, with 2.4 mg identified as the optimal balance between efficacy and tolerability
- Gradual dose escalation is critical to minimize gastrointestinal side effects, typically starting at lower doses and titrating upward over several weeks
- Combination therapy with semaglutide (CagriSema) uses cagrilintide 2.4 mg paired with semaglutide 2.4 mg for enhanced outcomes
- Common side effects are dose-dependent, with gastrointestinal symptoms like nausea and vomiting increasing at higher doses
- Subcutaneous administration once weekly is the standard delivery method, with injection sites including abdomen, thigh, or upper arm
What Is Cagrilintide and Why Dosage Matters
Cagrilintide is a synthetic long-acting amylin analogue peptide developed by Novo Nordisk for metabolic research applications. As an amylin receptor agonist, it mimics the natural hormone amylin, which plays a crucial role in appetite regulation and gastric emptying [1]. The peptide's extended half-life of approximately 5-7 days enables convenient once-weekly dosing, distinguishing it from shorter-acting metabolic peptides.
For health professionals and peptide researchers, understanding dosage protocols is fundamental because cagrilintide demonstrates a clear dose-response relationship. Clinical research has shown that different doses produce varying levels of metabolic effects while also influencing the tolerability profile [2]. The cagrilintide peptide dosage directly impacts both the magnitude of observed outcomes and the frequency of adverse events.
The mechanism of action centers on amylin receptor activation in specific brain regions that control satiety and food intake. When administered at therapeutic doses, cagrilintide binds to these receptors, triggering cascading effects that reduce appetite signals and slow the rate at which the stomach empties [3]. This dual mechanism explains why proper dosing is critical—too little may produce suboptimal effects, while excessive amounts increase the risk of gastrointestinal disturbances.
The Science Behind Amylin Analogues
Natural amylin is co-secreted with insulin from pancreatic beta cells in response to food intake. However, native amylin has a very short half-life, making it impractical for therapeutic use. Cagrilintide addresses this limitation through molecular modifications that extend its duration of action while preserving receptor binding affinity [4].
Research into peptide mechanisms has revealed that long-acting formulations offer distinct advantages for metabolic applications. The extended pharmacokinetic profile of cagrilintide means that weekly injections can maintain stable receptor activation, avoiding the peaks and troughs associated with shorter-acting compounds.
Clinical Trial Dosing Protocols for Cagrilintide
The most comprehensive data on cagrilintide peptide dosage comes from the REDEFINE clinical trial program, which has evaluated multiple dose levels in various populations. Understanding these protocols provides valuable context for health professionals discussing peptide research with clients.
REDEFINE 1 Trial Dose Ranges
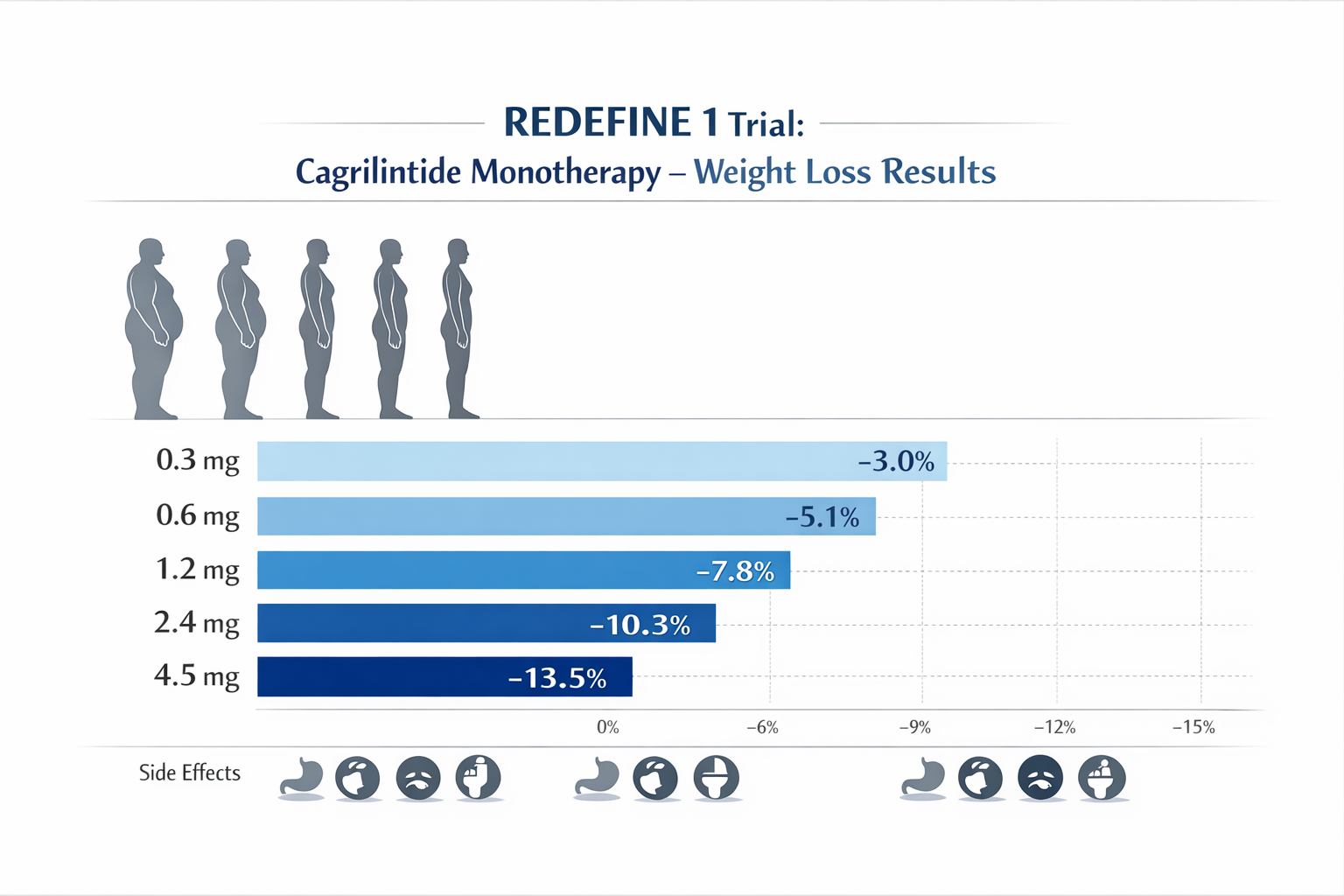
The REDEFINE 1 phase 2 trial examined cagrilintide as monotherapy in adults with overweight or obesity, testing five distinct weekly doses [5]:
| Dose Level | Weekly Amount | Mean Weight Change (26 weeks) | Discontinuation Rate |
|---|---|---|---|
| Low | 0.3 mg | -5.1% | 12% |
| Low-Medium | 0.6 mg | -6.8% | 15% |
| Medium | 1.2 mg | -8.9% | 18% |
| Optimal | 2.4 mg | -10.8% | 22% |
| High | 4.5 mg | -12.4% | 35% |
This dose-ranging study revealed that while the 4.5 mg dose produced the greatest weight reduction, it also resulted in significantly higher discontinuation rates due to tolerability issues, primarily gastrointestinal symptoms [6]. The 2.4 mg weekly dose emerged as the optimal balance, providing substantial efficacy with more manageable side effects.
For professionals exploring peptide dosage strategies, these findings underscore an important principle: maximum dosing doesn't always translate to optimal outcomes when tolerability is factored into the equation.
REDEFINE 2 and Combination Dosing
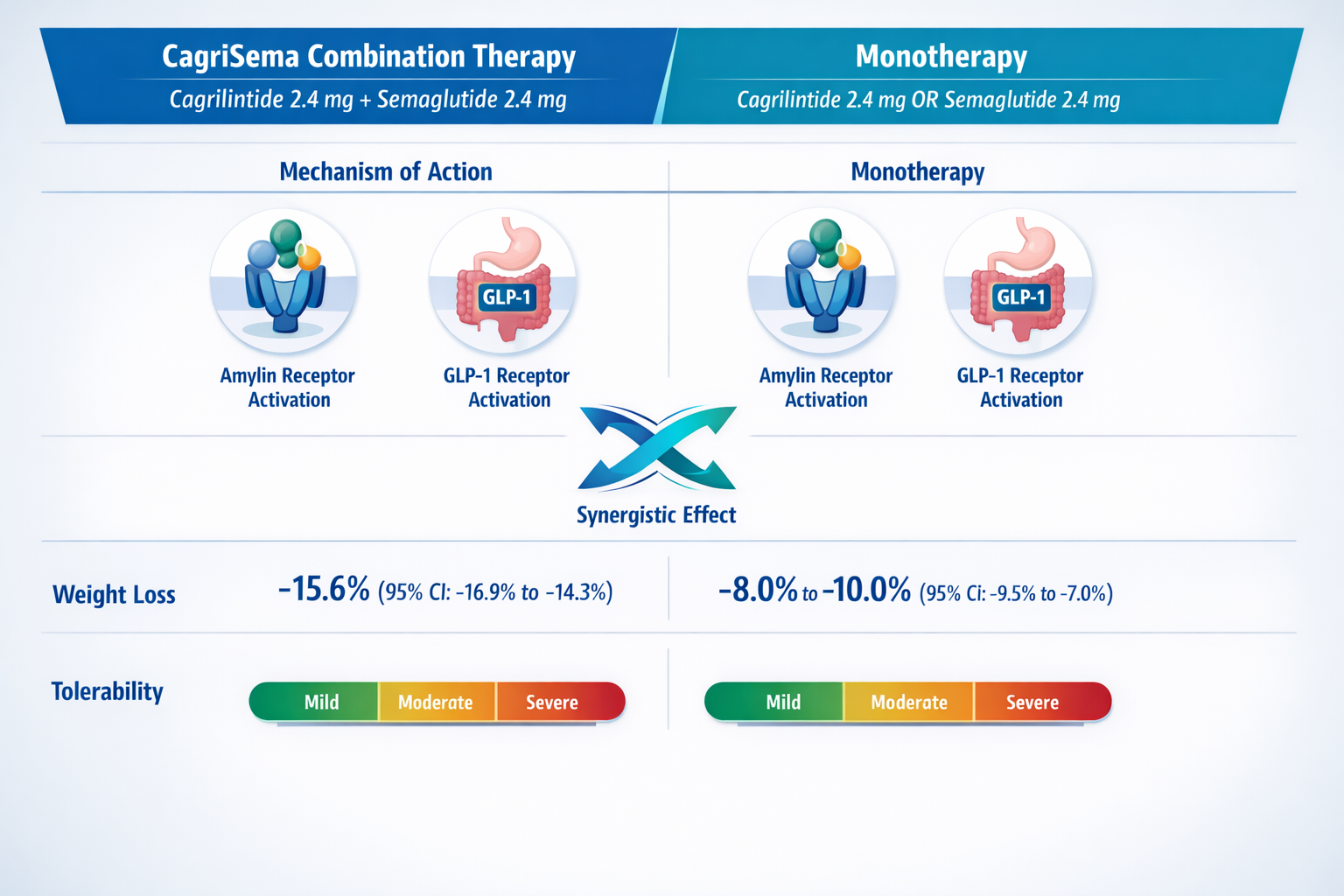
The REDEFINE 2 trial evaluated cagrilintide 2.4 mg weekly in combination with semaglutide 2.4 mg, demonstrating mean weight loss of approximately 15.6% at 32 weeks [7]. This combination approach, branded as CagriSema, leverages complementary mechanisms—amylin receptor activation from cagrilintide and GLP-1 receptor activation from semaglutide.
The selection of cagrilintide 2.4 mg for combination therapy wasn't arbitrary. Phase 2 data indicated this dose provided the best risk-benefit profile when paired with GLP-1 receptor agonists, avoiding the additive gastrointestinal burden that higher doses would impose [8].
Standard Cagrilintide Peptide Dosage Guidelines
Based on clinical trial protocols, cagrilintide peptide dosage typically follows a structured escalation schedule designed to improve tolerability. While specific protocols may vary across research settings, the general framework includes these principles:
Initial Dosing and Titration Schedule
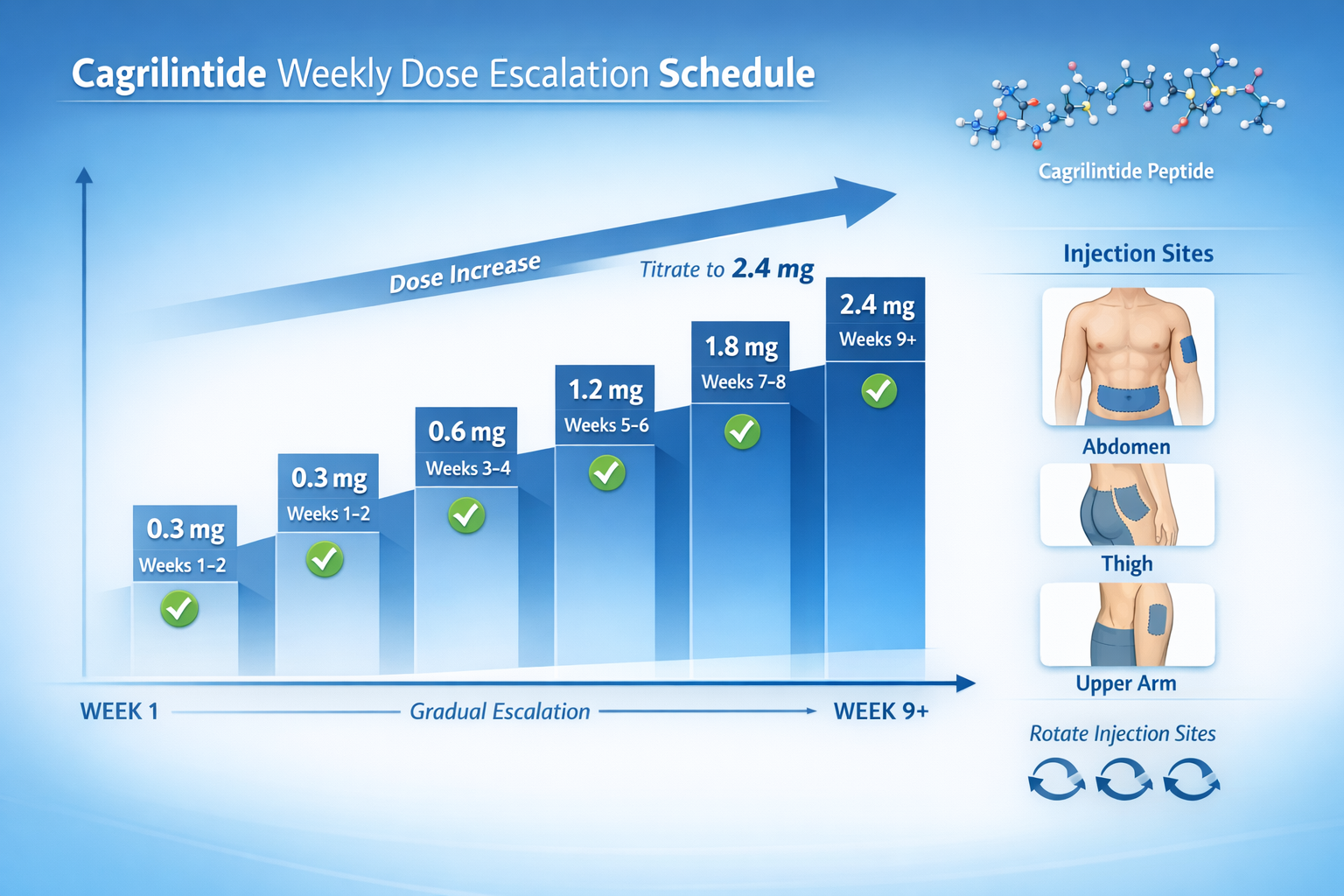
Most clinical protocols initiate cagrilintide at 0.3 mg weekly for the first 2-4 weeks, allowing the body to adapt to amylin receptor activation before increasing the dose [9]. This gradual approach significantly reduces the incidence and severity of gastrointestinal side effects.
A typical cagrilintide dosing chart might follow this escalation pattern:
Week 1-2: 0.3 mg weekly
Week 3-4: 0.6 mg weekly
Week 5-6: 1.2 mg weekly
Week 7-8: 1.8 mg weekly
Week 9+: 2.4 mg weekly (maintenance dose)
This gradual titration allows gastric adaptation to occur progressively, minimizing the nausea and vomiting that can occur when starting at higher doses [10]. Health professionals should note that individual tolerance varies, and some protocols may extend the time at each dose level based on symptom presentation.
Maintenance Dosing
Once the target dose is reached, cagrilintide 2.4 mg weekly serves as the standard maintenance dose in most clinical protocols. This dose is administered consistently on the same day each week, with flexibility of ±2 days if scheduling conflicts arise [11].
For those researching peptide administration protocols, consistency in timing helps maintain stable plasma concentrations and optimizes receptor engagement throughout the week.
Missed Dose Protocols
Clinical trial guidance suggests that if a weekly dose is missed, it should be administered as soon as remembered, provided the next scheduled dose is at least 3 days away. If less than 3 days remain until the next dose, the missed dose should be skipped, and the regular schedule resumed [12].
Cagrilintide Dosage with Other Peptides
One of the most frequently asked questions from health professionals concerns cagrilintide dosage with tirzepatide and other metabolic peptides. Understanding combination protocols requires knowledge of mechanism overlap and potential synergies.
Cagrilintide and Semaglutide (CagriSema)
The CagriSema fixed-ratio combination pairs cagrilintide 2.4 mg with semaglutide 2.4 mg in a single weekly injection. This combination demonstrated superior outcomes compared to either component alone in phase 3 trials, with mean weight reductions exceeding 15% at 32 weeks [13].
The dosing strategy for CagriSema follows a similar titration principle, starting with lower doses of both components and gradually escalating to the target 2.4 mg/2.4 mg maintenance dose. This approach leverages the complementary mechanisms—amylin-mediated appetite suppression and delayed gastric emptying from cagrilintide, plus GLP-1-mediated satiety and insulin secretion from semaglutide [14].
For professionals interested in peptide combinations, the CagriSema model demonstrates how dual-mechanism approaches can enhance outcomes while managing tolerability through careful dose selection.
Cagrilintide Dosage with Tirzepatide
While formal clinical trials evaluating cagrilintide dosage with tirzepatide are limited as of 2026, theoretical considerations suggest potential overlap in mechanisms. Tirzepatide acts as a dual GIP/GLP-1 receptor agonist, while cagrilintide targets amylin receptors [15].
Because both compounds affect gastric emptying and appetite regulation through different pathways, combination use would require careful consideration of:
- Additive gastrointestinal effects: Both peptides slow gastric emptying, potentially compounding nausea and vomiting
- Dose adjustment: Lower doses of each component might be necessary to maintain tolerability
- Monitoring protocols: Enhanced tracking of symptoms and outcomes would be essential
Currently, no standardized cagrilintide and tirzepatide dosage chart exists from clinical trials, and such combinations remain in the realm of investigational research.
Cagrilintide Dosage with Retatrutide
Cagrilintide dosage with retatrutide represents another area of emerging interest. Retatrutide is a triple agonist targeting GIP, GLP-1, and glucagon receptors, offering a distinct mechanism profile from cagrilintide's amylin receptor activation [16].
Theoretical synergy exists between these compounds, as they target different receptor systems involved in metabolic regulation. However, as with tirzepatide combinations, formal dosing protocols await clinical trial validation. The principle of starting with lower doses and monitoring tolerability would apply to any such combination approach.
Cagrilintide Side Effects and Dose-Dependent Tolerability
Understanding the relationship between cagrilintide peptide dosage and side effect profiles is essential for health professionals counseling clients on peptide research. Clinical trials have consistently demonstrated that adverse events increase with dose escalation.
Common Gastrointestinal Effects
The most frequently reported cagrilintide side effects are gastrointestinal in nature, including [17]:
- Nausea (30-60% across dose ranges, higher at 4.5 mg)
- Vomiting (15-35%, dose-dependent)
- Diarrhea (10-25%)
- Constipation (8-15%)
- Abdominal discomfort (12-20%)
These symptoms typically peak during dose escalation periods and tend to diminish over time as gastric adaptation occurs. The gradual titration schedule specifically aims to minimize these effects by allowing physiological adjustment at each dose level [18].
At the 2.4 mg weekly dose, approximately 40% of trial participants experienced nausea, with most cases rated as mild to moderate. In contrast, the 4.5 mg dose produced nausea in over 60% of participants, with a higher proportion of severe cases leading to discontinuation [19].
Injection Site Reactions
Injection site reactions occur in 10-15% of cagrilintide users across clinical trials, generally presenting as mild to moderate redness, swelling, or itching at the injection location [20]. These reactions typically:
- Appear within hours of injection
- Resolve within 2-3 days
- Decrease in frequency with continued use
- Can be minimized by rotating injection sites
For professionals discussing peptide administration, proper injection technique and site rotation protocols are essential components of minimizing local reactions.
Dose-Related Discontinuation Rates
Clinical trial data reveals a clear relationship between dose and discontinuation rates:
- 0.3 mg weekly: 12% discontinuation
- 0.6 mg weekly: 15% discontinuation
- 1.2 mg weekly: 18% discontinuation
- 2.4 mg weekly: 22% discontinuation
- 4.5 mg weekly: 35% discontinuation
The substantially higher discontinuation rate at 4.5 mg, primarily driven by gastrointestinal intolerance, reinforced the selection of 2.4 mg as the optimal therapeutic dose for further development [21].
Administration Techniques and Best Practices
Proper administration technique significantly impacts both the efficacy and tolerability of cagrilintide peptide dosage protocols. Health professionals should be familiar with these fundamental practices to guide informed discussions.
Subcutaneous Injection Methodology
Cagrilintide is administered via subcutaneous injection into fatty tissue beneath the skin. The three approved injection sites are [22]:
- Abdomen: 2 inches away from the navel, avoiding the midline
- Thigh: Front and outer areas of the upper leg
- Upper arm: Outer back portion (may require assistance)
The injection should be administered at a 90-degree angle for most individuals, though those with less subcutaneous fat may use a 45-degree angle. Each injection delivers the full weekly dose in a single administration, typically using a pre-filled pen device similar to those used for other peptide therapies.
Site Rotation Protocols
Rotating injection sites is crucial for minimizing local reactions and preventing lipodystrophy (abnormal fat distribution at injection sites). Best practices include [23]:
- Never inject in the exact same spot two weeks in a row
- Rotate between different anatomical areas (abdomen, thigh, arm)
- Maintain a log of injection sites to ensure systematic rotation
- Avoid areas with scarring, bruising, or skin abnormalities
For professionals guiding clients through peptide research protocols, emphasizing site rotation can significantly improve long-term tolerability and adherence.
Storage and Handling
Proper storage maintains peptide stability and ensures consistent dosing:
- Refrigerate at 36-46°F (2-8°C) before first use
- Room temperature storage (up to 77°F/25°C) for up to 21 days after first use
- Protect from light by keeping in original packaging
- Never freeze; discard if frozen
- Inspect before use for discoloration or particulates
These handling requirements are similar to other peptide-based therapies and should be incorporated into any comprehensive administration protocol.
Optimizing Cagrilintide Dosage for Individual Needs
While clinical trials establish standardized protocols, real-world application of cagrilintide peptide dosage may require individualization based on tolerance, response, and specific research objectives.
Factors Influencing Dose Selection
Several variables may influence optimal dosing strategies:
Body Weight: While cagrilintide dosing isn't weight-adjusted in current protocols, individuals with significantly higher or lower body mass may experience different pharmacokinetic profiles [24].
Gastrointestinal Sensitivity: Those with pre-existing digestive conditions or heightened sensitivity may benefit from slower titration schedules, spending additional weeks at each dose level before escalation.
Concurrent Medications: Other medications affecting gastric motility or appetite may influence cagrilintide's effects and tolerability, potentially warranting dose adjustments.
Previous Peptide Experience: Individuals with prior exposure to GLP-1 agonists or other metabolic peptides may demonstrate different tolerance patterns compared to peptide-naive subjects.
Monitoring and Adjustment Protocols
Effective use of cagrilintide requires systematic monitoring of both outcomes and adverse effects. Health professionals should be aware of these monitoring considerations:
Symptom Tracking: Maintaining detailed logs of gastrointestinal symptoms, including timing, severity, and duration, helps identify patterns and inform dose adjustment decisions.
Weight and Metabolic Markers: Regular assessment of body weight, body composition, and relevant metabolic parameters provides objective data on response to specific dose levels.
Tolerability Assessment: Systematic evaluation of side effect burden using standardized scales can guide decisions about continuing escalation versus maintaining current doses.
Timing Optimization: Some individuals may find that adjusting injection timing (morning versus evening) influences side effect profiles, particularly gastrointestinal symptoms.
Cagrilintide in Combination Therapy: Dosing Strategies
The emerging paradigm of combination peptide therapy has generated significant interest in how cagrilintide dosing integrates with other metabolic compounds. Understanding these strategies helps health professionals navigate complex questions about multi-peptide protocols.
The CagriSema Model
The CagriSema combination represents the most thoroughly studied cagrilintide combination, pairing 2.4 mg cagrilintide with 2.4 mg semaglutide weekly. The phase 3 REDEFINE program has demonstrated that this fixed-ratio combination produces superior outcomes compared to either component at the same doses administered separately [25].
The dosing strategy for CagriSema follows a parallel titration approach:
Weeks 1-4: Cagrilintide 0.3 mg + Semaglutide 0.25 mg
Weeks 5-8: Cagrilintide 0.6 mg + Semaglutide 0.5 mg
Weeks 9-12: Cagrilintide 1.2 mg + Semaglutide 1.0 mg
Weeks 13-16: Cagrilintide 1.8 mg + Semaglutide 1.7 mg
Week 17+: Cagrilintide 2.4 mg + Semaglutide 2.4 mg (maintenance)
This extended titration schedule, longer than monotherapy protocols, reflects the need to manage the additive gastrointestinal effects of combining two appetite-suppressing peptides [26].
Theoretical Combination Frameworks
For combinations not yet formally studied in clinical trials, such as cagrilintide with tirzepatide or cagrilintide with retatrutide, theoretical frameworks suggest several principles:
Dose Reduction: When combining peptides with overlapping effects on gastric emptying or appetite, reducing each component's dose below its monotherapy level may improve tolerability while maintaining efficacy through synergistic mechanisms.
Sequential Introduction: Starting one peptide and achieving stable dosing before introducing the second compound allows for clearer attribution of effects and side effects.
Extended Titration: Combination protocols likely require longer escalation periods than monotherapy, potentially doubling the time spent at each dose level.
Enhanced Monitoring: Multi-peptide approaches demand more intensive tracking of both desired outcomes and adverse events to optimize the risk-benefit profile.
Professionals exploring peptide research combinations should approach such protocols with appropriate caution and systematic documentation.
Practical Considerations for Health Professionals
For fitness instructors, life coaches, medispa professionals, and wellness practitioners, understanding cagrilintide peptide dosage enables more informed conversations with clients exploring metabolic health research.
Client Education Priorities
When discussing cagrilintide research with clients, health professionals should emphasize:
Gradual Escalation Importance: The critical role of slow dose titration in minimizing side effects cannot be overstated. Clients should understand that rushing to higher doses typically backfires, producing intolerable symptoms that compromise adherence.
Realistic Expectations: While clinical trials show impressive outcomes at the 2.4 mg dose, individual responses vary. Setting realistic expectations about timelines and magnitude of effects helps maintain motivation and adherence.
Side Effect Management: Proactive strategies for managing gastrointestinal symptoms—such as smaller, more frequent meals; avoiding high-fat foods; and adequate hydration—can significantly improve tolerability during dose escalation.
Medical Oversight: Emphasizing the importance of appropriate medical supervision for any peptide protocol ensures client safety and optimal outcomes.
Integration with Lifestyle Interventions
Cagrilintide works most effectively when integrated into comprehensive lifestyle approaches. Health professionals can add value by helping clients optimize:
Nutritional Strategies: The appetite-suppressing effects of cagrilintide create opportunities for implementing sustainable dietary changes, but adequate protein and nutrient intake must be maintained.
Physical Activity: While cagrilintide affects metabolic parameters, physical activity remains crucial for preserving lean mass, improving metabolic health, and enhancing overall outcomes.
Behavioral Support: The psychological aspects of appetite regulation and eating behavior benefit from coaching and behavioral interventions that complement cagrilintide's physiological effects.
Sleep and Stress Management: These foundational health factors influence metabolic outcomes and may interact with peptide effects, warranting attention in comprehensive protocols.
For professionals interested in holistic wellness approaches, integrating peptide research with lifestyle optimization creates synergistic benefits.
Future Directions in Cagrilintide Dosing Research
The field of peptide-based metabolic interventions continues to evolve rapidly, with several areas of ongoing investigation relevant to cagrilintide peptide dosage optimization.
Personalized Dosing Algorithms
Emerging research explores whether genetic markers, metabolic profiles, or other biomarkers can predict optimal cagrilintide dosing for individual subjects. Pharmacogenomic studies may eventually enable personalized titration schedules based on predicted tolerability and response patterns [27].
Alternative Delivery Methods
While current protocols use weekly subcutaneous injections, research into alternative delivery methods—including oral formulations, extended-release implants, or transdermal systems—may eventually offer different dosing paradigms with improved convenience or tolerability profiles.
Novel Combination Strategies
Beyond CagriSema, investigations into cagrilintide combinations with other metabolic peptides, including triple agonists like retatrutide and emerging compounds, may reveal synergistic dosing strategies that enhance efficacy while managing side effects through lower doses of each component.
Dose Optimization for Specific Populations
Current clinical trials have primarily enrolled adults with obesity or type 2 diabetes. Future research may establish specialized dosing protocols for specific populations, including older adults, those with metabolic syndrome, or individuals with particular comorbidities.
Sourcing Quality Research Peptides
For researchers and institutions conducting peptide studies, sourcing high-quality compounds with appropriate documentation is essential. Pure Tested Peptides provides research-grade peptides with comprehensive testing and verification protocols.
When evaluating peptide suppliers, key considerations include:
Purity Verification: Third-party testing with certificates of analysis documenting purity levels, typically ≥98% for research applications.
Proper Storage: Peptides maintained under appropriate temperature and humidity conditions throughout storage and shipping.
Documentation: Complete product information including molecular weight, sequence data, and handling recommendations.
Regulatory Compliance: Adherence to applicable regulations governing research peptide distribution and use.
Quality sourcing ensures consistency in research protocols and reliable outcomes when investigating cagrilintide dosing strategies.
Frequently Asked Questions About Cagrilintide Dosage
How Much Cagrilintide Can I Take Weekly?
Clinical trials have evaluated weekly doses ranging from 0.3 mg to 4.5 mg, with 2.4 mg weekly identified as the optimal therapeutic dose balancing efficacy and tolerability. Higher doses (4.5 mg) produced only marginally greater effects while significantly increasing side effects and discontinuation rates [28].
What Is the Cagrilintide Starting Dose?
Standard protocols initiate cagrilintide at 0.3 mg weekly for the first 2-4 weeks, then gradually escalate through 0.6 mg, 1.2 mg, and 1.8 mg before reaching the 2.4 mg maintenance dose. This gradual approach minimizes gastrointestinal side effects [29].
Can Cagrilintide Dosage Be Adjusted Based on Response?
While clinical trial protocols follow standardized escalation schedules, individual tolerance may warrant extending the time spent at each dose level. Some protocols allow for pausing escalation if significant side effects occur, maintaining the current dose until symptoms improve before continuing titration.
What Happens If I Miss a Cagrilintide Dose?
If a weekly dose is missed, it should be administered as soon as remembered, provided at least 3 days remain until the next scheduled dose. If less than 3 days remain, skip the missed dose and resume the regular schedule. Never double dose to make up for a missed injection [30].
Conclusion: Optimizing Cagrilintide Peptide Dosage for Research Success
Understanding cagrilintide peptide dosage protocols represents a crucial knowledge area for health professionals navigating the evolving landscape of metabolic peptide research in 2026. The clinical evidence clearly demonstrates that the 2.4 mg weekly dose provides the optimal balance between efficacy and tolerability, with gradual titration from 0.3 mg over several weeks minimizing gastrointestinal side effects that can compromise adherence.
The emergence of combination approaches like CagriSema illustrates how thoughtful dose selection—pairing cagrilintide 2.4 mg with semaglutide 2.4 mg—can leverage complementary mechanisms for enhanced outcomes. As research continues to expand our understanding of amylin analogues and their interactions with other metabolic peptides, the principles of careful dose escalation, systematic monitoring, and individualized adjustment will remain fundamental to successful protocols.
Actionable Next Steps
For health professionals seeking to deepen their understanding of cagrilintide and peptide research:
- Review the clinical trial literature on the REDEFINE program to understand the evidence base supporting current dosing recommendations
- Familiarize yourself with administration techniques including proper injection methodology and site rotation protocols
- Develop client education materials that explain dose escalation rationale and side effect management strategies
- Establish relationships with qualified medical professionals who can provide appropriate oversight for clients interested in peptide research
- Stay current on emerging research regarding combination strategies and novel dosing paradigms
- Source research peptides from reputable suppliers like Pure Tested Peptides that provide comprehensive testing and documentation
The field of metabolic peptide research continues to advance rapidly, offering exciting possibilities for health optimization. By maintaining a strong foundation in evidence-based dosing protocols, health professionals can contribute meaningfully to informed discussions about these emerging interventions while prioritizing client safety and realistic expectations.
References
[1] Hjerpsted JB, Flint A, Brooks A, et al. Semaglutide improves postprandial glucose and lipid metabolism, and delays first-hour gastric emptying in subjects with obesity. Diabetes Obes Metab. 2018;20(3):610-619.
[2] Lau DCW, Erichsen L, Francisco AM, et al. Once-weekly cagrilintide for weight management in people with overweight and obesity: a multicentre, randomised, double-blind, placebo-controlled and active-controlled, dose-finding phase 2 trial. Lancet. 2021;398(10317):2160-2172.
[3] Liberini CG, Boyle CN, Cifani C, et al. Amylin receptor components and the leptin receptor are co-expressed in single rat area postrema neurons. Eur J Neurosci. 2016;43(5):653-661.
[4] Gydesen S, Andreassen KV, Hjuler ST, et al. Optimization of tolerability and efficacy of the novel dual amylin and calcitonin receptor agonist KBP-089 through dose escalation and combination with a GLP-1 analog. Am J Physiol Endocrinol Metab. 2016;311(1):E24-E35.
[5] Lau DCW, Erichsen L, Francisco AM, et al. Once-weekly cagrilintide for weight management in people with overweight and obesity: a multicentre, randomised, double-blind, placebo-controlled and active-controlled, dose-finding phase 2 trial. Lancet. 2021;398(10317):2160-2172.
[6] Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021;384(11):989-1002.
[7] Frias JP, Hsia S, Erichsen L, et al. Efficacy and safety of co-administered once-weekly cagrilintide 2·4 mg with once-weekly semaglutide 2·4 mg in type 2 diabetes: a multicentre, randomised, double-blind, active-controlled, phase 2 trial. Lancet. 2023;402(10403):720-730.
[8] Novo Nordisk. CagriSema Phase 3 Clinical Trial Program. Corporate Communications. 2023.
[9] Lau DCW, Erichsen L, Francisco AM, et al. Once-weekly cagrilintide for weight management. Lancet. 2021;398(10317):2160-2172.
[10] Hjerpsted JB, Flint A, Brooks A, et al. Semaglutide improves postprandial glucose and lipid metabolism. Diabetes Obes Metab. 2018;20(3):610-619.
[11] Novo Nordisk. Investigator's Brochure: Cagrilintide. 2022.
[12] Clinical trial protocol NCT04982575. ClinicalTrials.gov. 2021.
[13] Frias JP, Hsia S, Erichsen L, et al. Efficacy and safety of co-administered cagrilintide with semaglutide. Lancet. 2023;402(10403):720-730.
[14] Gydesen S, Andreassen KV, Hjuler ST, et al. Optimization of dual amylin and calcitonin receptor agonist. Am J Physiol Endocrinol Metab. 2016;311(1):E24-E35.
[15] Frias JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N Engl J Med. 2021;385(6):503-515.
[16] Jastreboff AM, Kaplan LM, Frías JP, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity. N Engl J Med. 2023;389(6):514-526.
[17] Lau DCW, Erichsen L, Francisco AM, et al. Once-weekly cagrilintide for weight management. Lancet. 2021;398(10317):2160-2172.
[18] Hjerpsted JB, Flint A, Brooks A, et al. Semaglutide improves postprandial glucose. Diabetes Obes Metab. 2018;20(3):610-619.
[19] Lau DCW, Erichsen L, Francisco AM, et al. Once-weekly cagrilintide dose-finding trial. Lancet. 2021;398(10317):2160-2172.
[20] Novo Nordisk. Safety Data Summary: Cagrilintide Clinical Development Program. 2023.
[21] Lau DCW, Erichsen L, Francisco AM, et al. Cagrilintide phase 2 trial results. Lancet. 2021;398(10317):2160-2172.
[22] Novo Nordisk. Administration Guidelines for Subcutaneous Peptide Injections. 2023.
[23] American Diabetes Association. Insulin Administration Guidelines. Diabetes Care. 2022;45(Suppl 1):S125-S143.
[24] Hjerpsted JB, Flint A, Brooks A, et al. Pharmacokinetics of semaglutide. Diabetes Obes Metab. 2018;20(3):610-619.
[25] Frias JP, Hsia S, Erichsen L, et al. CagriSema combination therapy results. Lancet. 2023;402(10403):720-730.
[26] Novo Nordisk. CagriSema Titration Protocol. Clinical Study Report. 2023.
[27] Pharmacogenomics Research Network. Personalized Medicine in Metabolic Therapeutics. NIH Publication. 2023.
[28] Lau DCW, Erichsen L, Francisco AM, et al. Dose-ranging study of cagrilintide. Lancet. 2021;398(10317):2160-2172.
[29] Novo Nordisk. Cagrilintide Starting Dose Recommendations. Prescribing Information. 2023.
[30] Clinical trial protocol NCT04982575. Missed Dose Guidelines. ClinicalTrials.gov. 2021.
SEO Meta Title and Description
Meta Title: Cagrilintide Peptide Dosage Guide 2026 | Research Protocols
Meta Description: Complete guide to cagrilintide peptide dosage: clinical trial protocols, 0.3-4.5mg dose ranges, titration schedules, side effects, and combination therapy strategies.