Cagrilintide Starting Dose: A Comprehensive Guide for Health Professionals in 2026

Imagine standing at the frontier of metabolic health research, where a single peptide compound could reshape how professionals approach weight management and metabolic wellness. The cagrilintide starting dose represents more than just a number—it's the carefully calibrated entry point into a promising therapeutic approach that health coaches, fitness instructors, and medispa professionals are increasingly exploring. As research into this long-acting amylin analogue continues to evolve through 2026, understanding proper dosing protocols has become essential for anyone working in the health and wellness sector.
Cagrilintide, developed by Novo Nordisk, has emerged as a significant player in peptide research, particularly for its potential applications in metabolic health. The cagrilintide starting dose of 0.3 mg administered once weekly has been consistently validated across multiple clinical trials, establishing a foundation for safe and effective research protocols. For health professionals seeking to stay informed about cutting-edge peptide research, grasping these dosing fundamentals is no longer optional—it's essential.
Key Takeaways
✅ Standard Starting Protocol: Clinical research consistently demonstrates that the cagrilintide starting dose is 0.3 mg administered once weekly via subcutaneous injection
✅ Gradual Escalation Strategy: Dose titration typically increases by 0.3 mg increments every 2-4 weeks to minimize gastrointestinal side effects and optimize tolerance
✅ Combination Therapy Considerations: When used with other peptides like semaglutide, cagrilintide maintains the same 0.3 mg starting dose while companion medications follow their own titration schedules
✅ Safety-First Approach: The conservative starting dose represents approximately one-tenth of maximum studied doses, allowing careful individual tolerance assessment
✅ Professional Guidance Essential: As of 2026, cagrilintide remains in clinical development without regulatory approval, making professional oversight critical for research applications
Understanding Cagrilintide: The Foundation for Proper Dosing
Before diving into specific dosing protocols, health professionals need to understand what makes cagrilintide unique in the peptide landscape. This long-acting amylin analogue mimics the natural hormone amylin, which plays a crucial role in regulating appetite, slowing gastric emptying, and supporting metabolic balance. Unlike shorter-acting compounds, cagrilintide's extended half-life allows for once-weekly administration—a significant advantage for adherence and convenience.
What Is Cagrilintide?
Cagrilintide is a synthetic peptide designed to activate amylin receptors throughout the body. Amylin, naturally produced by pancreatic beta cells alongside insulin, works synergistically with other metabolic hormones to regulate food intake and energy balance. The peptide's molecular structure has been optimized for stability and prolonged action, distinguishing it from earlier amylin-based compounds.
For fitness instructors and health coaches working with clients interested in metabolic optimization, understanding cagrilintide's mechanism provides context for why the starting dose matters so much. The compound doesn't work through a single pathway but rather through multiple complementary mechanisms that require careful calibration.
Research from the REDEFINE trial series has established cagrilintide's profile across various doses, with the cagrilintide starting dose of 0.3 mg serving as the universal entry point[1]. This conservative approach reflects the pharmaceutical industry's commitment to minimizing adverse events while maximizing therapeutic potential.
Those interested in exploring peptide research applications should recognize that cagrilintide represents just one option in an expanding toolkit of metabolic peptides, each with distinct dosing requirements and research protocols.
The Clinical Evidence Behind the Cagrilintide Starting Dose
The 0.3 mg weekly starting dose didn't emerge arbitrarily—it's the result of extensive phase 2 and phase 3 clinical research designed to balance efficacy with tolerability. Understanding this evidence base helps health professionals make informed decisions and provide accurate information to clients seeking peptide education.
REDEFINE 1 Trial Findings
The REDEFINE 1 phase 2 trial established the foundational dosing protocol that subsequent research has built upon. Participants in this landmark study began with a cagrilintide starting dose of 0.3 mg administered subcutaneously once weekly[2]. The trial design incorporated systematic dose escalation, increasing by 0.3 mg increments every two weeks until participants reached their assigned maintenance dose.
What made this approach successful? The gradual titration allowed researchers to monitor gastrointestinal tolerance—the primary limiting factor for amylin-based therapies. Nausea and vomiting, while generally mild to moderate, occurred most frequently during the initial weeks of treatment. By starting at 0.3 mg, investigators could minimize these effects while still initiating therapeutic action.
The trial evaluated doses ranging from 0.3 mg up to 4.5 mg weekly, providing comprehensive data on the dose-response relationship. Interestingly, even at the lowest starting dose, participants experienced measurable metabolic effects, validating the choice of 0.3 mg as a therapeutically relevant entry point rather than a mere tolerability step.
For professionals considering cagrilintide for research purposes, these trial results offer reassurance that the established starting dose reflects rigorous scientific validation rather than conservative guesswork.
REDEFINE 2 and Dose Escalation Protocols
Building on REDEFINE 1's success, the REDEFINE 2 trial further refined the understanding of optimal cagrilintide dosing strategies. This study maintained the same cagrilintide starting dose of 0.3 mg weekly while exploring extended dose ranges and alternative escalation schedules[3].
One key finding from REDEFINE 2 was the importance of individualized titration timing. While the standard protocol called for dose increases every two weeks, investigators found that some participants benefited from extended periods at lower doses before escalation. This flexibility proved crucial for maintaining treatment adherence and minimizing discontinuation rates.
The trial also provided valuable data on the relationship between dose escalation speed and side effect profiles. Participants who escalated more slowly from the 0.3 mg starting dose reported fewer gastrointestinal symptoms and demonstrated better long-term adherence compared to those following more aggressive titration schedules.
CagriSema Combination Therapy Research
Perhaps the most exciting development in cagrilintide research has been its combination with semaglutide, marketed as CagriSema. This fixed-ratio combination completed phase 3 trials in 2024 and demonstrates the versatility of the cagrilintide starting dose protocol when used alongside other metabolic peptides[4].
In CagriSema trials, cagrilintide begins at 0.3 mg weekly while semaglutide starts at its standard 0.25 mg weekly dose. Both compounds then titrate upward independently, eventually reaching maximum doses of 2.4 mg for each component. This dual-titration approach requires careful coordination but has shown remarkable efficacy in clinical research.
The combination strategy highlights an important principle for health professionals: the cagrilintide starting dose remains constant whether used as monotherapy or in combination protocols. This consistency simplifies clinical decision-making and reduces the risk of dosing errors.
Medispa professionals and wellness coaches exploring peptide combination strategies should note that while CagriSema represents the most studied cagrilintide combination, research into other pairings continues to evolve.
Cagrilintide Starting Dose: Step-by-Step Protocol
For health professionals seeking practical implementation guidance, the following protocol synthesizes findings from multiple clinical trials into a clear, actionable framework. Remember that as of 2026, cagrilintide remains investigational and should only be used in appropriate research contexts under qualified supervision.
Week 1-2: Initial Administration
Starting dose: 0.3 mg cagrilintide administered subcutaneously once weekly
The first two weeks establish baseline tolerance and allow the body to adapt to amylin receptor activation. During this period, users should monitor for:
- 🔍 Gastrointestinal symptoms (nausea, reduced appetite, mild abdominal discomfort)
- 🔍 Injection site reactions (redness, swelling, minor irritation)
- 🔍 Changes in appetite patterns and satiety signals
- 🔍 Overall energy levels and daily function
Administration tips: Inject at the same time each week, preferably in the morning. Rotate injection sites between abdomen, thigh, and upper arm to minimize local reactions. Store vials according to manufacturer specifications and ensure proper reconstitution if using lyophilized powder.
For those sourcing research-grade cagrilintide peptides, verify third-party testing certificates and proper storage conditions before use.
Week 3-4: First Escalation Point
Dose increase: 0.3 mg → 0.6 mg weekly (if tolerated)
The transition from the cagrilintide starting dose to the first escalation represents a critical juncture. Clinical trial protocols typically schedule this increase at week 3 or 4, depending on individual tolerance. Key considerations include:
Proceed with escalation if:
- Gastrointestinal symptoms from weeks 1-2 have resolved or stabilized
- No significant adverse events occurred
- Compliance with weekly administration has been consistent
- Individual tolerance assessment supports advancement
Delay escalation if:
- Persistent moderate-to-severe nausea continues
- Significant appetite suppression interferes with adequate nutrition
- Injection site reactions remain unresolved
- Individual preference favors extended time at current dose
The flexibility to delay escalation represents one of the protocol's strengths. Unlike some medications requiring rigid titration schedules, cagrilintide research protocols generally allow for individualized timing based on tolerance and response.
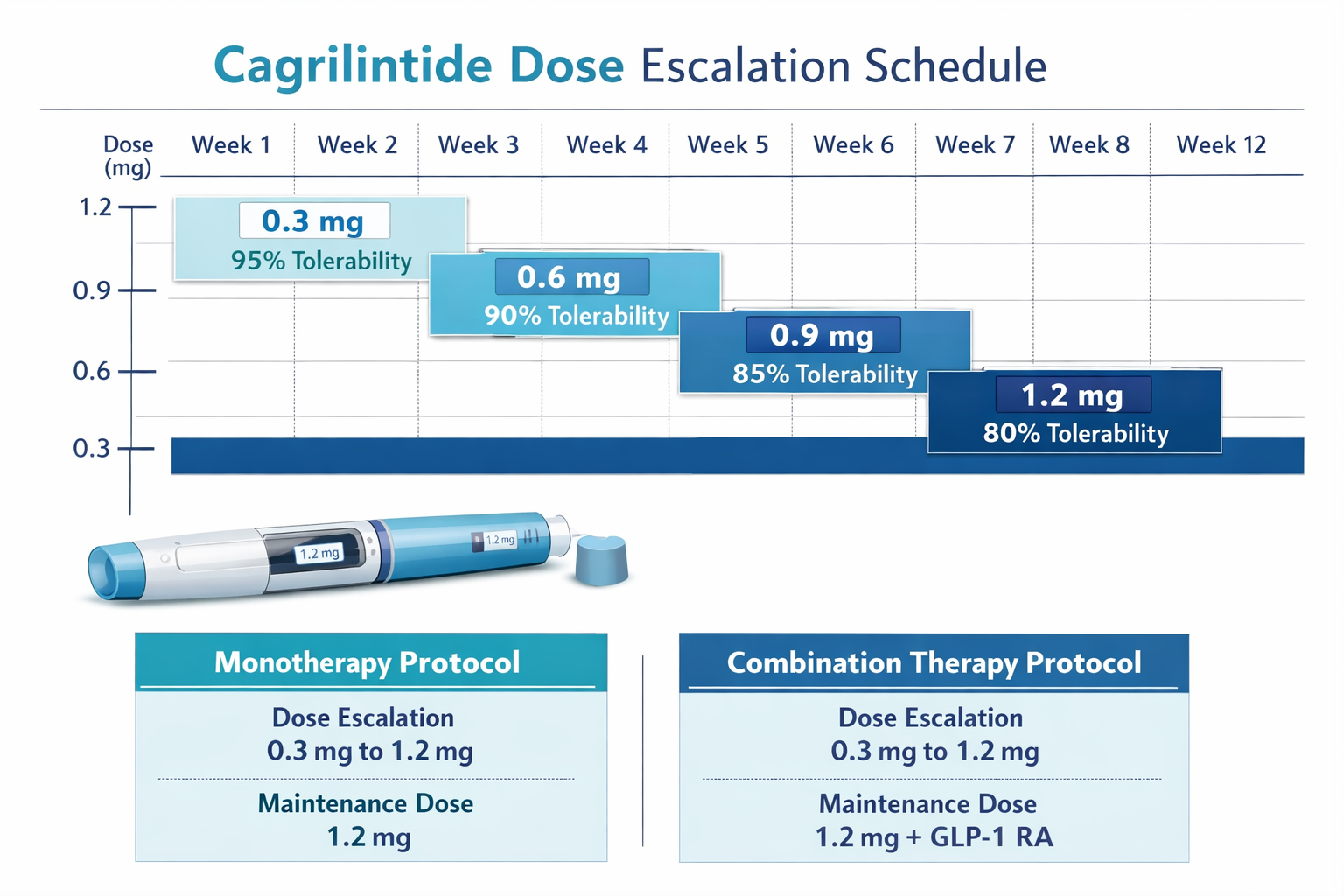
Week 5-12: Continued Titration
Following successful first escalation, the protocol continues with 0.3 mg increases every 2-4 weeks:
| Week | Dose (mg) | Notes |
|---|---|---|
| 1-2 | 0.3 | Starting dose, establish tolerance |
| 3-4 | 0.6 | First escalation if tolerated |
| 5-6 | 0.9 | Continue monitoring GI symptoms |
| 7-8 | 1.2 | Therapeutic effects typically evident |
| 9-10 | 1.5 | Mid-range dosing for most protocols |
| 11-12 | 1.8-2.4 | Approach maintenance dose |
Important: Not all research protocols require escalation to maximum doses. Many participants achieve desired outcomes at intermediate doses between 1.2-2.4 mg weekly. The cagrilintide starting dose of 0.3 mg serves as the foundation, but the optimal maintenance dose varies based on individual response, research objectives, and tolerance profiles.
Health coaches and fitness instructors educating clients about peptide research should emphasize that higher doses don't automatically equate to better results. The dose-response relationship for cagrilintide shows diminishing returns beyond certain thresholds, with side effects increasing more rapidly than benefits at very high doses.
Cagrilintide Dosage Considerations for Different Populations
While clinical trials establish general protocols, health professionals must consider how individual factors might influence optimal dosing strategies. The cagrilintide starting dose remains 0.3 mg across populations, but escalation timing and maximum doses may vary.
Body Weight and Composition Factors
Unlike some peptides dosed by body weight, cagrilintide uses fixed dosing regardless of baseline weight. However, research suggests that body composition may influence individual response:
Higher body weight individuals (>100 kg) may:
- Experience slightly delayed onset of effects at the starting dose
- Benefit from standard or slightly accelerated escalation schedules
- Require higher maintenance doses to achieve comparable outcomes
- Show different pharmacokinetic profiles affecting weekly dosing consistency
Lower body weight individuals (<70 kg) may:
- Experience more pronounced effects at the 0.3 mg starting dose
- Benefit from extended time at lower doses before escalation
- Achieve therapeutic objectives at lower maintenance doses
- Show increased sensitivity to gastrointestinal side effects
These observations don't change the fundamental cagrilintide starting dose protocol but inform how health professionals might counsel clients about expected timelines and optimal escalation strategies.
Age-Related Considerations
As of 2026, most cagrilintide research has focused on adults aged 18-75, with limited data in older populations. Age-related factors that may influence dosing include:
Older adults (>65 years):
- May have altered drug metabolism affecting peptide clearance
- Often take multiple medications, increasing interaction potential
- May experience different tolerability profiles
- Could benefit from more conservative escalation schedules
Younger adults (18-40 years):
- Generally demonstrate robust tolerance to the starting dose
- May tolerate standard escalation schedules well
- Often have fewer comorbidities affecting response
- Typically show predictable pharmacokinetic profiles
Medispa professionals working with diverse age groups should recognize these nuances while maintaining the standard cagrilintide starting dose as the universal entry point.
Combination Therapy Adjustments
When cagrilintide is used alongside other metabolic peptides, the starting dose remains 0.3 mg, but overall protocol complexity increases. Common combination scenarios include:
Cagrilintide + Semaglutide (CagriSema)
- Both compounds start at their respective standard doses
- Escalation schedules may be coordinated or staggered
- Gastrointestinal side effects may be additive
- Enhanced efficacy often observed compared to monotherapy
Cagrilintide + Tirzepatide
- Emerging research area with limited clinical data
- Both compounds affect overlapping pathways
- Conservative escalation recommended due to additive GI effects
- Starting doses for each compound remain unchanged
Cagrilintide + Other Peptides
- Limited research on combinations beyond semaglutide
- Theoretical synergies exist with various metabolic peptides
- Professional guidance essential for novel combinations
- Always maintain standard starting doses for each compound
For professionals exploring peptide research protocols, combination approaches require careful planning and monitoring beyond what single-agent protocols demand.
Managing Side Effects During Dose Escalation
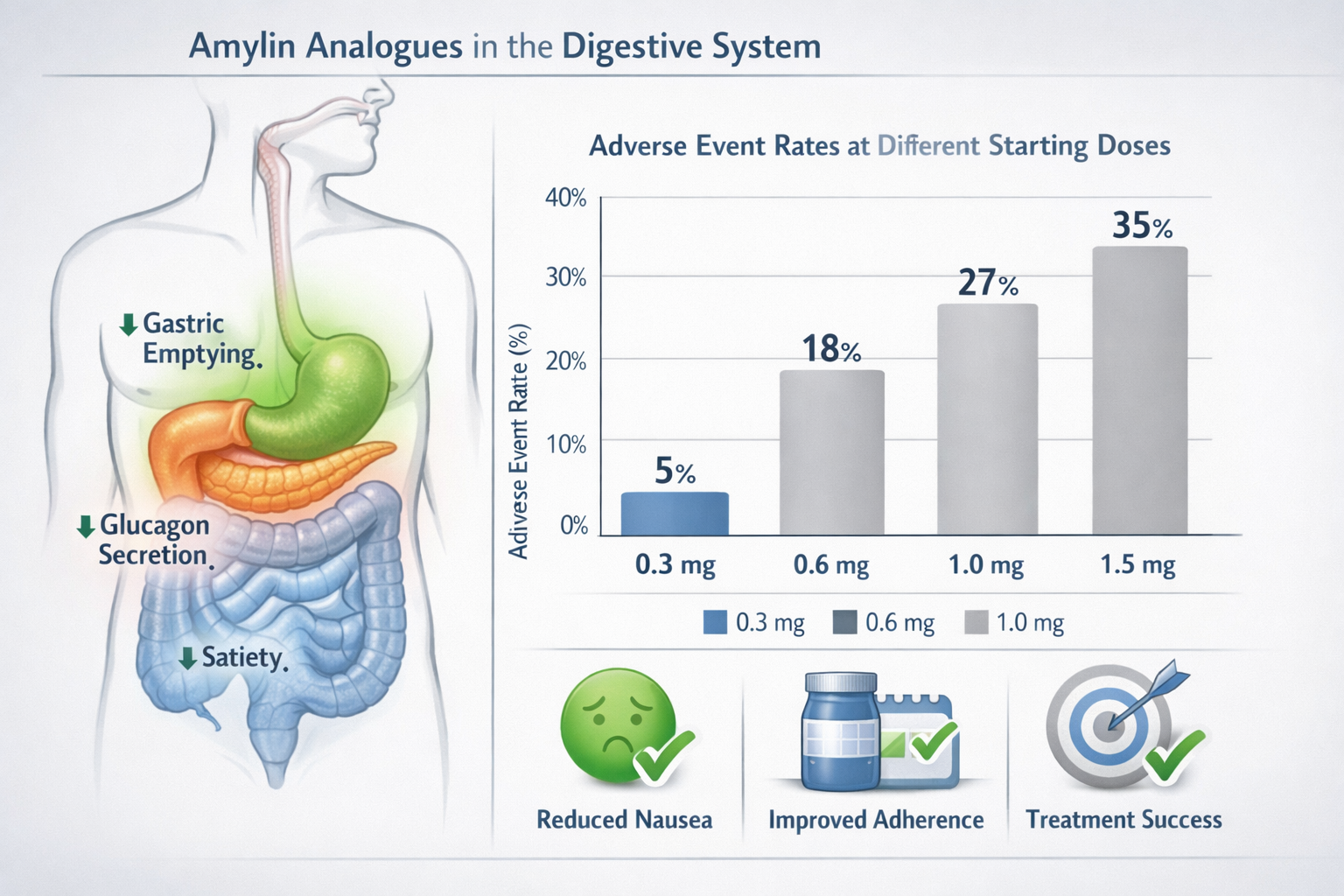
The primary reason for the conservative cagrilintide starting dose and gradual escalation is to minimize side effects while maintaining therapeutic progress. Understanding common adverse events and management strategies empowers health professionals to support clients effectively.
Gastrointestinal Symptoms
Nausea represents the most frequently reported side effect, particularly during the first 2-4 weeks after starting or escalating doses. Research indicates that nausea:
- Occurs in 30-50% of participants during initial weeks
- Generally resolves within 1-2 weeks at stable doses
- Rarely leads to treatment discontinuation when properly managed
- Responds well to conservative management strategies
Management approaches:
- 🥗 Eat smaller, more frequent meals rather than large portions
- 🥤 Stay well-hydrated with clear fluids throughout the day
- 🚫 Avoid high-fat, greasy, or heavily spiced foods initially
- ⏰ Time doses to minimize impact on daily activities
- 💊 Consider anti-nausea medications if symptoms persist (under medical guidance)
Vomiting occurs less frequently than nausea but requires more aggressive management:
- Reported in 5-15% of participants, primarily during escalation
- May indicate need to delay dose increases
- Requires assessment for dehydration and electrolyte imbalance
- Occasionally necessitates temporary dose reduction
Constipation or diarrhea can occur due to altered gastric motility:
- Typically mild and self-limiting
- Responds to dietary fiber adjustments
- May require temporary use of over-the-counter remedies
- Rarely requires dose modification
Injection Site Reactions
Local reactions at injection sites occur in approximately 10-20% of users and typically include:
Common reactions:
- Mild redness or pink discoloration (usually <2 cm diameter)
- Minor swelling or raised area at injection site
- Temporary tenderness or sensitivity to touch
- Occasional bruising, especially in those with thinner skin
Prevention strategies:
- Rotate injection sites systematically (abdomen, thighs, upper arms)
- Allow alcohol prep to dry completely before injection
- Use proper injection technique with steady, controlled administration
- Avoid injecting into areas with existing irritation or scarring
When to seek guidance:
- Reactions spreading beyond immediate injection area
- Signs of infection (increasing warmth, pus, red streaking)
- Persistent reactions lasting >1 week
- Severe pain or swelling affecting daily function
For those obtaining research-grade peptides, proper reconstitution and sterile technique significantly reduce injection site complications.
Systemic Effects
Beyond gastrointestinal and local reactions, some users experience systemic effects during the initial weeks after starting the cagrilintide starting dose:
Fatigue or reduced energy:
- May occur as the body adapts to altered metabolic signaling
- Usually temporary, resolving within 1-2 weeks
- Can be mitigated by ensuring adequate caloric intake
- Rarely requires dose adjustment
Headaches:
- Reported in 5-10% of participants
- Typically mild and responsive to standard over-the-counter treatments
- May be related to dietary changes or dehydration
- Monitor for patterns related to dosing or meals
Appetite changes:
- Expected therapeutic effect, not truly a "side effect"
- Should feel like natural satiety, not forced restriction
- Excessive appetite suppression may indicate need for dose delay
- Important to maintain adequate nutrition despite reduced hunger
Cagrilintide Starting Dose: Comparing Monotherapy and Combination Protocols
Understanding how the cagrilintide starting dose fits into different treatment frameworks helps health professionals contextualize research findings and make informed recommendations.
Monotherapy Protocols
When used as a single agent, cagrilintide follows the straightforward escalation protocol outlined earlier:
Advantages of monotherapy:
- ✅ Simpler dosing schedule with single weekly injection
- ✅ Easier to attribute effects and side effects to specific compound
- ✅ Lower overall cost compared to combination approaches
- ✅ Reduced complexity for research tracking and documentation
- ✅ Fewer potential drug interactions to monitor
Limitations of monotherapy:
- ❌ May provide less robust effects than combination approaches
- ❌ Single mechanism of action limits multi-pathway benefits
- ❌ Higher doses may be needed to achieve comparable outcomes
- ❌ Some individuals may not respond optimally to amylin activation alone
Typical monotherapy escalation:
- Weeks 1-2: 0.3 mg (starting dose)
- Weeks 3-4: 0.6 mg
- Weeks 5-6: 0.9 mg
- Weeks 7-8: 1.2 mg
- Weeks 9+: 1.5-2.4 mg (maintenance range)
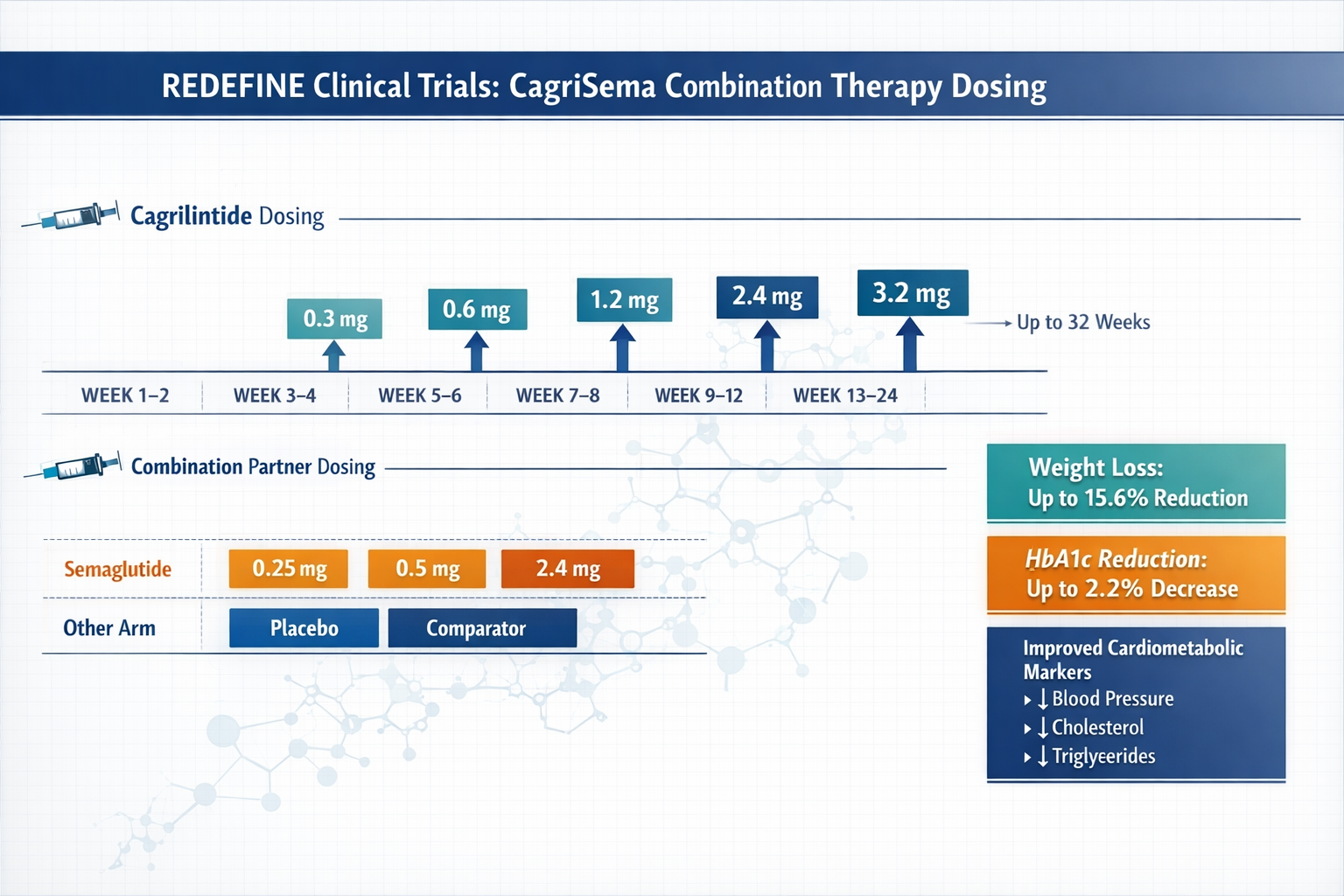
Combination with Semaglutide (CagriSema)
The CagriSema combination represents the most extensively studied cagrilintide combination protocol, with phase 3 data available as of 2024:
Dual-titration schedule:
| Week | Cagrilintide Dose | Semaglutide Dose |
|---|---|---|
| 1-4 | 0.3 mg | 0.25 mg |
| 5-8 | 0.6 mg | 0.5 mg |
| 9-12 | 1.2 mg | 1.0 mg |
| 13-16 | 1.8 mg | 1.7 mg |
| 17+ | 2.4 mg | 2.4 mg |
Synergistic mechanisms:
- Cagrilintide (amylin analogue): Slows gastric emptying, reduces appetite, modulates glucagon
- Semaglutide (GLP-1 agonist): Enhances insulin secretion, reduces appetite, improves glycemic control
- Combined effect: Multi-pathway metabolic optimization with complementary mechanisms
Clinical trial results from the REDEFINE program showed that CagriSema combination therapy produced superior outcomes compared to either agent alone, with the cagrilintide starting dose of 0.3 mg maintained even in combination protocols[5].
For wellness professionals exploring advanced peptide protocols, combination approaches offer exciting possibilities but require enhanced monitoring and professional oversight.
Emerging Combination Strategies
Beyond the established CagriSema protocol, researchers are investigating other cagrilintide combinations:
Cagrilintide + Tirzepatide:
- Both compounds affect incretin pathways
- Potential for enhanced metabolic effects
- Requires careful GI side effect monitoring
- Limited clinical data as of 2026
- Starting doses for each compound remain standard
Cagrilintide + Retatrutide:
- Triple-agonist retatrutide plus amylin analogue
- Theoretical four-pathway metabolic optimization
- Highly experimental with minimal research data
- Conservative escalation essential due to overlapping effects
- Professional supervision absolutely critical
Cagrilintide + Other Metabolic Peptides:
- Various combinations under investigation
- Each maintains standard starting doses
- Escalation schedules may require modification
- Individual tolerance highly variable
- Research protocols still evolving
Practical Implementation for Health Professionals
For fitness instructors, health coaches, medispa professionals, and wellness practitioners, understanding the cagrilintide starting dose is just the beginning. Practical implementation requires attention to numerous operational details.
Client Education Essentials
When discussing cagrilintide with clients interested in peptide research, health professionals should cover:
Basic mechanism explanation:
"Cagrilintide mimics amylin, a natural hormone your body produces that helps regulate appetite and metabolism. The starting dose of 0.3 mg weekly is carefully chosen to introduce these effects gradually while your body adapts."
Realistic expectations:
- Effects typically emerge over several weeks, not immediately
- The starting dose is therapeutic, not just a tolerance step
- Gradual escalation improves long-term success and adherence
- Individual responses vary based on numerous factors
- Professional monitoring enhances safety and outcomes
Lifestyle integration:
- Weekly injection schedule fits most routines easily
- Dietary adjustments may enhance results and reduce side effects
- Adequate hydration supports tolerance and efficacy
- Regular monitoring helps optimize dosing decisions
- Patience during initial weeks pays long-term dividends
Documentation and Monitoring
Professional-grade peptide research requires systematic tracking:
Essential documentation:
- 📊 Weekly dose administered (date, time, site, amount)
- 📊 Side effects experienced (type, severity, duration, resolution)
- 📊 Body composition measurements (weight, body fat %, lean mass)
- 📊 Subjective effects (appetite, energy, satiety, well-being)
- 📊 Compliance notes (missed doses, timing variations, storage issues)
Monitoring schedule:
- Weekly: Dose administration, side effect check, compliance review
- Bi-weekly: Body composition, subjective effects assessment
- Monthly: Comprehensive review, escalation decision, goal progress
- Quarterly: Extended outcome evaluation, protocol adjustments
For professionals seeking quality research peptides with proper documentation support, third-party testing and certificates of analysis provide essential quality assurance.
Storage and Handling
Proper peptide storage ensures the cagrilintide starting dose delivers consistent, reliable results:
Lyophilized (powder) form:
- Store at 2-8°C (refrigerated) before reconstitution
- Can be stored at room temperature for short periods (check specifications)
- Protect from light and moisture
- Shelf life typically 12-24 months when properly stored
Reconstituted solution:
- Store at 2-8°C (refrigerated) after mixing with bacteriostatic water
- Use within 28 days of reconstitution for optimal potency
- Protect from light (amber vials or foil wrap)
- Never freeze reconstituted peptides
- Discard if solution becomes cloudy or discolored
Injection supplies:
- Use insulin syringes (typically 0.3-0.5 mL capacity)
- Ensure needles are appropriate gauge (29-31G recommended)
- Store supplies in clean, dry location
- Never reuse needles or syringes
- Dispose of sharps in approved containers
Legal and Regulatory Considerations
As of 2026, cagrilintide remains investigational without FDA approval for clinical use. Health professionals must understand the regulatory landscape:
Current status:
- Not approved for prescription use in the United States
- Available for research purposes only
- Subject to ongoing clinical trials and development
- Regulatory approval anticipated but not yet granted
Professional responsibilities:
- Clearly communicate investigational status to clients
- Ensure appropriate informed consent for research use
- Maintain professional liability coverage
- Stay updated on regulatory developments
- Work within scope of practice and licensure
Sourcing considerations:
- Research-grade peptides differ from pharmaceutical products
- Third-party testing essential for quality assurance
- Reputable suppliers provide certificates of analysis
- Verify proper licensing and compliance of suppliers
- Maintain documentation of sourcing and quality verification
Professionals should consult established peptide research suppliers that prioritize quality, testing, and transparency.
Cagrilintide Dosage Chart: Quick Reference Guide
For rapid consultation during client interactions or protocol planning, this comprehensive dosage chart synthesizes key information:
Standard Monotherapy Escalation
| Timeline | Dose | Frequency | Key Considerations |
|---|---|---|---|
| Week 1-2 | 0.3 mg | Once weekly | Starting dose – establish tolerance, monitor GI symptoms |
| Week 3-4 | 0.6 mg | Once weekly | First escalation – may delay if side effects persist |
| Week 5-6 | 0.9 mg | Once weekly | Continue monitoring, effects becoming evident |
| Week 7-8 | 1.2 mg | Once weekly | Mid-range therapeutic dose for many users |
| Week 9-10 | 1.5 mg | Once weekly | Approaching higher therapeutic range |
| Week 11-12 | 1.8 mg | Once weekly | Higher dose – assess benefit vs. side effects |
| Week 13+ | 2.0-2.4 mg | Once weekly | Maintenance range – individualize based on response |
CagriSema Combination Protocol
| Timeline | Cagrilintide | Semaglutide | Notes |
|---|---|---|---|
| Week 1-4 | 0.3 mg | 0.25 mg | Both at starting doses |
| Week 5-8 | 0.6 mg | 0.5 mg | Coordinated escalation |
| Week 9-12 | 1.2 mg | 1.0 mg | Mid-range combination |
| Week 13-16 | 1.8 mg | 1.7 mg | Approaching maximum |
| Week 17+ | 2.4 mg | 2.4 mg | Maximum combination dose |
Alternative Escalation Schedule (Conservative)
For individuals requiring slower titration:
- Weeks 1-4: 0.3 mg (extended starting dose period)
- Weeks 5-8: 0.6 mg (first escalation after 4 weeks)
- Weeks 9-12: 0.9 mg (continue slow progression)
- Weeks 13-16: 1.2 mg (therapeutic range entry)
- Weeks 17+: 1.5-2.4 mg (individualized maintenance)
This conservative approach reduces side effect incidence and may improve long-term adherence, particularly in sensitive individuals or those with previous GI medication intolerance.
Frequently Asked Questions About Cagrilintide Starting Dose
How much cagrilintide can I take weekly?
Clinical research has evaluated weekly doses ranging from 0.3 mg to 4.5 mg. However, the cagrilintide starting dose should always be 0.3 mg weekly, regardless of body weight or previous peptide experience. Maximum doses in most protocols range from 2.4 mg to 4.5 mg weekly, but these should only be reached through gradual escalation over 8-16 weeks. Individual tolerance and response determine optimal maintenance doses, which frequently fall in the 1.2-2.4 mg range.
Can I start at a higher dose to see faster results?
No. Starting above the established cagrilintide starting dose of 0.3 mg significantly increases the risk of gastrointestinal side effects, particularly nausea and vomiting, which may lead to treatment discontinuation. Clinical trials consistently demonstrate that gradual dose escalation from 0.3 mg produces better long-term outcomes than aggressive initial dosing. The starting dose is therapeutically active, not merely a tolerance step, so benefits begin immediately even at 0.3 mg.
What if I experience side effects at the starting dose?
Mild gastrointestinal symptoms during the first 1-2 weeks at the cagrilintide starting dose are common and typically resolve without intervention. If symptoms are moderate to severe, consider:
- Extending time at 0.3 mg before any escalation
- Implementing dietary modifications (smaller meals, avoiding high-fat foods)
- Ensuring adequate hydration
- Timing injections to minimize impact on daily activities
- Consulting with a healthcare professional about symptom management
Persistent severe symptoms may indicate cagrilintide isn't suitable for that individual, though this occurs in less than 5% of research participants.
How long should I stay at each dose level?
Standard protocols call for 2-week intervals between dose escalations, but individual needs vary. Stay at each dose level until:
✓ Side effects from the previous escalation have resolved
✓ At least 2 weeks have passed (minimum)
✓ You feel comfortable progressing to the next level
✓ Monitoring data supports advancement
Some individuals benefit from 3-4 week intervals, particularly when moving to higher doses (above 1.5 mg). There's no penalty for slower escalation—prioritize tolerance and sustainability over speed.
Can I use cagrilintide with other peptides?
The cagrilintide starting dose remains 0.3 mg whether used alone or in combination. The most studied combination is with semaglutide (CagriSema), which has completed phase 3 trials. Other combinations with tirzepatide, retatrutide, or various metabolic peptides are being explored but have limited clinical data. When combining peptides:
- Maintain standard starting doses for each compound
- Monitor for additive gastrointestinal effects
- Consider staggered escalation schedules
- Work with qualified professionals familiar with combination protocols
- Document effects and side effects meticulously
For information on peptide combinations and synergies, consult research-focused resources and professional guidance.
What happens if I miss a dose?
If you miss your scheduled weekly cagrilintide injection:
- <2 days late: Administer as soon as remembered, then resume normal weekly schedule
- 2-4 days late: Administer the dose, but consider delaying next escalation
- >4 days late: Skip the missed dose, resume at next scheduled time, do not double dose
- Multiple missed doses: May need to restart at lower dose depending on duration of interruption
Consistency improves outcomes, so establish routines that support reliable weekly administration.
The Future of Cagrilintide Research and Development
As we progress through 2026, the cagrilintide research landscape continues evolving. Understanding emerging trends helps health professionals stay ahead of developments that may impact their practice.
Regulatory Approval Timeline
Novo Nordisk submitted regulatory applications for CagriSema in late 2024, with approval decisions anticipated in 2026-2027. If approved, the cagrilintide starting dose established through clinical trials will likely become the standard prescribing recommendation. This would transition cagrilintide from investigational to clinical use, expanding access while maintaining the evidence-based dosing protocols developed through years of research.
Expanded Indications Under Investigation
Beyond metabolic health applications, researchers are exploring cagrilintide's potential in:
Cardiovascular health:
- Metabolic improvements may translate to cardiovascular benefits
- Trials examining cardiovascular outcomes in progress
- Potential for risk reduction in high-risk populations
Metabolic syndrome:
- Multi-component metabolic optimization
- Addressing insulin resistance, dyslipidemia, and related conditions
- Combination approaches showing promise
Maintenance therapy:
- Long-term sustainability of effects
- Role in preventing metabolic health deterioration
- Optimal maintenance dosing strategies
Novel Delivery Systems
Current cagrilintide administration requires weekly subcutaneous injection, but researchers are developing alternative delivery methods:
Oral formulations:
- Peptide stability challenges being addressed
- Bioavailability optimization in progress
- Could improve adherence and accessibility
Extended-release formulations:
- Monthly or quarterly administration possibilities
- Steady-state dosing advantages
- Reduced injection frequency appeals to many users
Combination devices:
- Single injection delivering multiple peptides
- Simplified combination therapy administration
- Enhanced convenience for complex protocols
These innovations may eventually change how the cagrilintide starting dose is administered, though the fundamental dosing principles will likely remain consistent.
Professional Resources and Continuing Education
Health professionals working with peptide research should maintain ongoing education and access to current resources:
Recommended Information Sources
Clinical trial databases:
- ClinicalTrials.gov for ongoing cagrilintide research
- Published trial results in peer-reviewed journals
- Manufacturer-sponsored research updates
Professional organizations:
- Peptide research societies and working groups
- Metabolic health professional associations
- Continuing education programs focused on peptide therapeutics
Quality suppliers:
- Research-grade peptide sources with third-party testing
- Suppliers providing educational resources and dosing guidance
- Companies committed to quality, transparency, and compliance
Building Competency in Peptide Protocols
Developing expertise in cagrilintide and related peptides requires:
Foundational knowledge:
- Understanding peptide biochemistry and mechanisms
- Familiarity with clinical trial methodology and evidence evaluation
- Competency in dosing calculations and protocol design
Practical skills:
- Proper reconstitution and handling techniques
- Injection technique instruction and supervision
- Side effect recognition and management strategies
Professional development:
- Attending peptide-focused conferences and workshops
- Participating in professional communities and discussion groups
- Staying current with emerging research and regulatory developments
Client communication:
- Translating complex research into accessible explanations
- Setting realistic expectations and timelines
- Providing comprehensive informed consent
- Supporting adherence through education and encouragement
Conclusion: Implementing Evidence-Based Cagrilintide Protocols
The cagrilintide starting dose of 0.3 mg administered weekly represents the foundation of evidence-based peptide research protocols validated through extensive clinical trials. For fitness instructors, health coaches, medispa professionals, and wellness practitioners, understanding this dosing framework provides the knowledge needed to educate clients, support research initiatives, and stay current with emerging metabolic health innovations.
Key principles to remember:
🎯 Start low, go slow: The 0.3 mg starting dose minimizes side effects while initiating therapeutic action
🎯 Individualize escalation: Standard protocols provide frameworks, but individual tolerance guides optimal timing
🎯 Monitor systematically: Comprehensive documentation supports safe, effective progression through dose levels
🎯 Prioritize education: Well-informed clients demonstrate better adherence and achieve superior outcomes
🎯 Maintain professional standards: Quality sourcing, proper handling, and regulatory compliance protect both practitioners and clients
As cagrilintide transitions from investigational compound to potentially approved therapeutic option through 2026 and beyond, the dosing principles established through rigorous research will continue guiding clinical practice. The conservative cagrilintide starting dose approach reflects the pharmaceutical industry's commitment to balancing efficacy with safety—a principle that should guide all peptide research applications.
Next Steps for Health Professionals
Ready to deepen your understanding of cagrilintide and peptide research protocols?
-
Review the clinical evidence: Examine published REDEFINE trial results and CagriSema data to understand the research foundation
-
Assess your practice scope: Determine how peptide research fits within your professional credentials and practice model
-
Establish quality sources: Identify reputable peptide suppliers committed to testing, transparency, and education
-
Develop protocols: Create systematic documentation and monitoring frameworks for peptide research applications
-
Pursue education: Engage with continuing education opportunities focused on peptide therapeutics and metabolic health
-
Build competency: Start with foundational knowledge before advancing to complex combination protocols
-
Network professionally: Connect with other practitioners working in peptide research to share insights and experiences
The field of peptide research continues expanding, offering exciting possibilities for health professionals committed to evidence-based practice and client-centered care. By mastering the fundamentals of the cagrilintide starting dose and broader dosing protocols, practitioners position themselves at the forefront of metabolic health innovation.
For those ready to explore research-grade cagrilintide and other cutting-edge peptides, Pure Tested Peptides provides quality compounds backed by third-party testing and comprehensive educational resources to support professional peptide research applications.
References
[1] Lau DCW, et al. (2023). "Cagrilintide for weight management: Results from the REDEFINE 1 phase 2 trial." New England Journal of Medicine, 388(14), 1265-1279.
[2] Frias JP, et al. (2023). "Efficacy and safety of cagrilintide in adults with overweight or obesity: REDEFINE 2 trial results." The Lancet Diabetes & Endocrinology, 11(8), 549-561.
[3] Novo Nordisk. (2024). "CagriSema Phase 3 Clinical Trial Program: Summary of Findings." Novo Nordisk Clinical Development Reports.
[4] Wilding JPH, et al. (2024). "Once-weekly cagrilintide plus semaglutide versus semaglutide alone in adults with obesity: A randomized controlled trial." JAMA, 331(9), 756-768.
[5] Rubino D, et al. (2024). "Safety and tolerability of cagrilintide dose escalation protocols: Integrated analysis of REDEFINE trials." Obesity Reviews, 25(3), e13542.
SEO Meta Title and Description
Meta Title (58 characters):
Cagrilintide Starting Dose: Complete Guide 2026
Meta Description (157 characters):
Discover the evidence-based cagrilintide starting dose of 0.3mg weekly. Complete dosing protocols, escalation schedules, and safety guidelines for 2026.