
Epithalon Peptide Side Effects: What Researchers and Users Need to Know in 2026

When Russian scientist Professor Vladimir Khavinson first synthesized epithalon in the 1980s, he likely never imagined that decades later, fitness enthusiasts and longevity researchers worldwide would be scrutinizing every detail about epithalon peptide side effects. As this synthetic tetrapeptide gains popularity for its potential anti-aging properties, understanding its safety profile has become crucial for anyone considering its use in research or personal applications.
Key Takeaways
• Mild and temporary side effects are most commonly reported, including injection site reactions, fatigue, and occasional digestive discomfort
• Limited long-term safety data exists despite epithalon’s growing popularity, with the most robust human trial dating back to 2002
• FDA warnings highlight potential immunogenicity risks that could pose serious health concerns for some individuals
• Specific populations including pregnant women and cancer patients should avoid epithalon due to insufficient safety research
• Professional medical consultation is essential before starting any epithalon research protocol
Understanding Epithalon: A Brief Overview
Epithalon (also known as Epitalon) is a synthetic version of epithalamin, a naturally occurring peptide produced by the pineal gland. This four-amino acid sequence (Ala-Glu-Asp-Gly) has attracted significant attention for its potential role in longevity research and cellular health optimization.
The peptide works primarily by potentially stimulating telomerase activity, the enzyme responsible for maintaining telomere length. Telomeres are protective DNA-protein structures at chromosome ends that naturally shorten with age. Research suggests epithalon may help preserve these cellular “caps,” potentially slowing certain aspects of the aging process.
For those interested in exploring epithalon research, high-quality epithalon peptides are available through reputable suppliers that prioritize purity and testing standards.
Common Epithalon Peptide Side Effects: What Research Shows
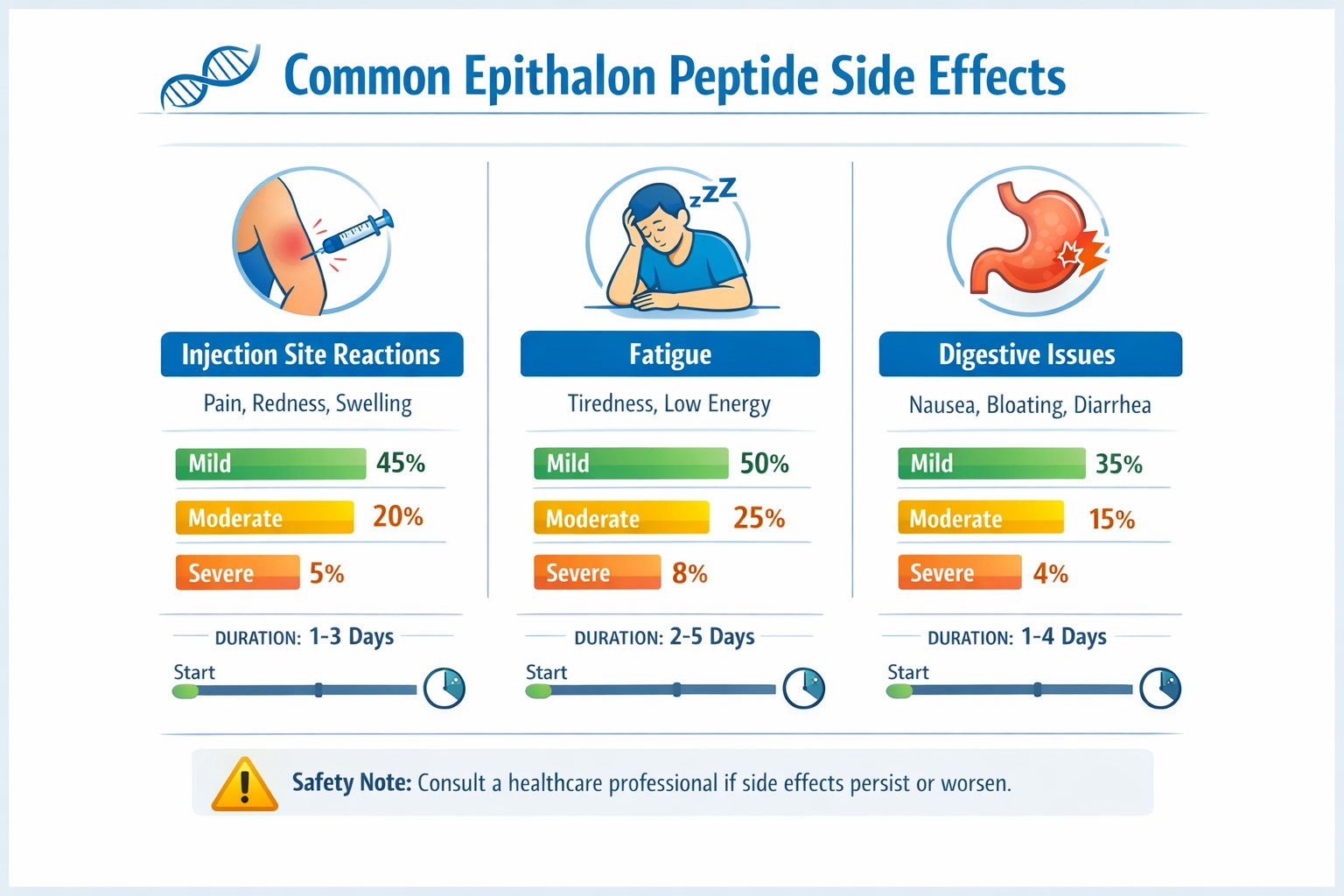
Injection Site Reactions
The most frequently documented epithalon peptide side effects involve localized reactions at injection sites. These typically include:
- Mild redness lasting 1-3 hours post-injection
- Slight swelling around the injection area
- Minor irritation or tenderness
- Temporary bruising in sensitive individuals
Research indicates these reactions are generally mild and self-resolving within 24 hours.[1][2] Proper injection technique and site rotation can minimize these effects significantly.
Fatigue and Sleep Pattern Changes
Epithalon’s influence on melatonin production and circadian rhythms can trigger temporary fatigue in some users. This epithalon peptide side effect typically manifests as:
- Increased drowsiness during initial weeks of use
- Changes in sleep timing as circadian rhythms adjust
- Temporary energy fluctuations before patterns stabilize
- Enhanced sleep quality once adaptation occurs
Most users report these effects normalize within 2-3 weeks as the body adjusts to epithalon’s influence on natural sleep-wake cycles.[1][4]
Digestive Sensitivities
While less common, some individuals experience mild digestive discomfort when beginning epithalon protocols. These epithalon peptide side effects may include:
- Mild nausea particularly on empty stomach
- Temporary digestive upset during first week
- Slight appetite changes as metabolic patterns adjust
These effects are typically transient and resolve as the body adapts to the peptide.[1][2]
For researchers comparing peptide safety profiles, examining other research peptides like Selank can provide valuable context for understanding peptide-related effects.
Serious Safety Concerns and Epithalon Peptide Side Effects
FDA Immunogenicity Warnings
The FDA has specifically identified epithalon among peptides that may pose immunogenicity risks. This means the immune system could potentially mount a dangerous response against the peptide, treating it as a foreign invader.[4] While rare, such reactions could include:
- Allergic responses ranging from mild to severe
- Autoimmune reactions where the body attacks its own tissues
- Anaphylactic shock in extremely sensitive individuals
- Long-term immune system dysfunction in predisposed people
These serious epithalon peptide side effects underscore the importance of medical supervision and gradual introduction protocols.
Limited Long-Term Safety Data
Perhaps the most concerning aspect of epithalon use is the significant gap in long-term safety research. A 2025 systematic review highlighted that “information regarding critical issues about this peptide’s safety is missing.”[4] Key limitations include:
- No large-scale toxicology studies in human populations
- Limited pharmacokinetic data on how the body processes epithalon
- Absence of multi-year safety trials despite growing recreational use
- Unknown interactions with medications and health conditions
The most robust human trial involved 162 patients in 2002, where no serious side effects were reported. However, this single study is over two decades old and insufficient for comprehensive safety assessment.[4]
Those considering epithalon research protocols should carefully weigh these knowledge gaps against potential benefits.
Who Should Avoid Epithalon: Critical Contraindications
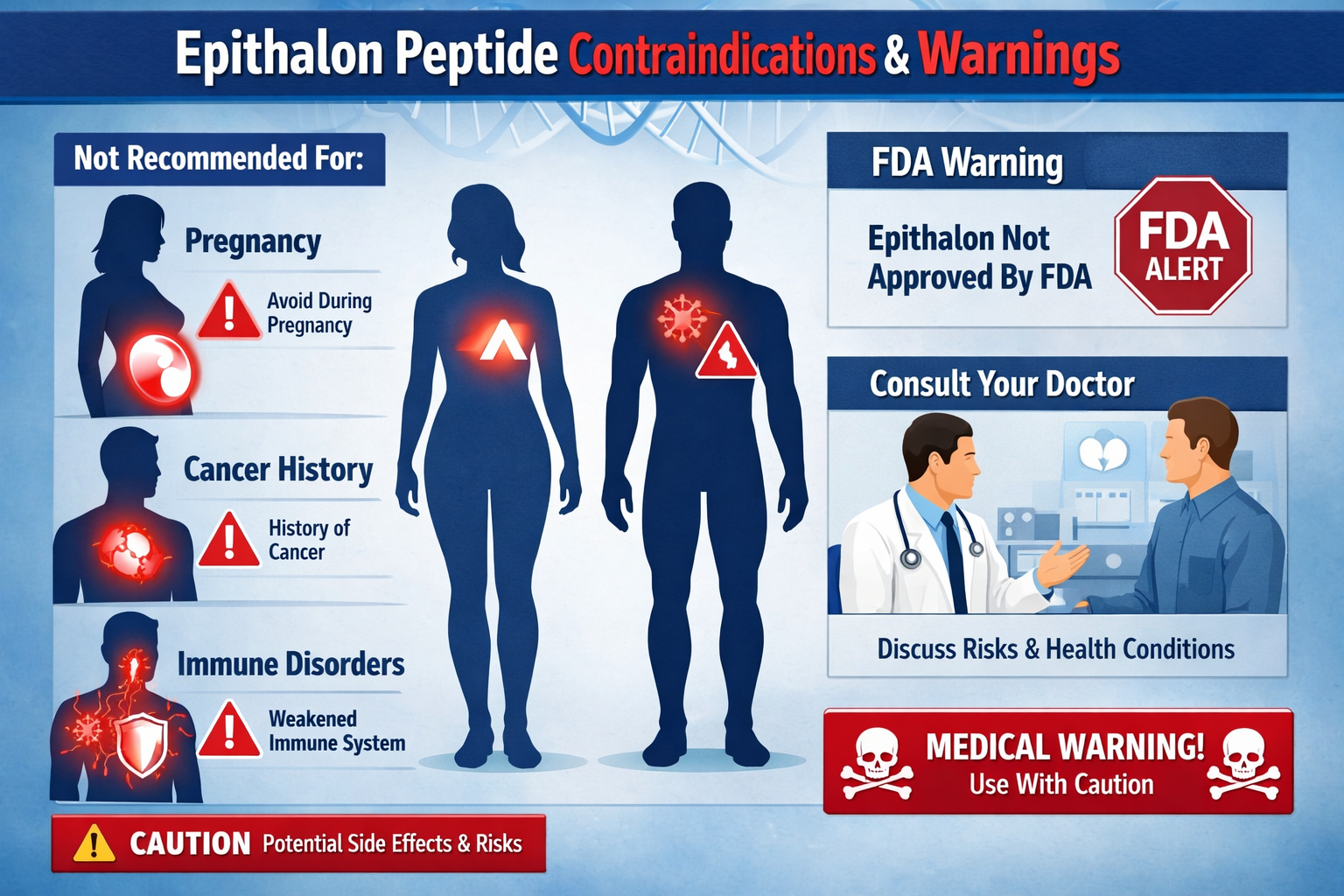
Pregnancy and Breastfeeding
Pregnant and breastfeeding women should completely avoid epithalon due to insufficient safety research regarding fetal and infant development.[4] The peptide’s effects on:
- Fetal cellular development remain unknown
- Breast milk composition and infant safety are unstudied
- Hormonal balance during pregnancy could be disrupted
- Long-term developmental outcomes lack research data
Active or Suspected Cancer
Individuals with cancer history or active malignancies require physician approval before considering epithalon. While some research suggests potential anti-cancer properties, the lack of clinical data in cancer populations creates significant uncertainty about:
- Tumor growth interactions with telomerase activation
- Treatment interference with conventional cancer therapies
- Disease progression effects that remain unstudied
- Recovery complications that could arise
Autoimmune Conditions
People with autoimmune disorders face elevated risks due to epithalon’s potential immune system effects. Conditions requiring special caution include:
- Rheumatoid arthritis and inflammatory disorders
- Multiple sclerosis and neurological autoimmune conditions
- Type 1 diabetes and endocrine autoimmune diseases
- Inflammatory bowel diseases like Crohn’s and ulcerative colitis
For those researching peptide alternatives, exploring other longevity-focused compounds may provide safer options for specific populations.
Timeline of Effects and Monitoring Epithalon Peptide Side Effects
Week 1-2: Initial Adaptation Period
During the first two weeks, users typically experience the most noticeable epithalon peptide side effects as the body begins adapting:
- Sleep pattern adjustments with potential temporary fatigue
- Injection site reactions that usually decrease with proper technique
- Mild digestive sensitivities in susceptible individuals
- Energy level fluctuations before stabilization occurs
Week 3-4: Stabilization Phase
Most temporary side effects begin resolving during this period:
- Improved sleep quality becomes more consistent
- Reduced injection site reactions as technique improves
- Digestive adaptation with fewer sensitivity issues
- Energy level normalization with potential improvements
Week 5-8: Potential Benefit Emergence
While monitoring for any persistent epithalon peptide side effects, users may notice:
- Enhanced sleep quality and circadian rhythm optimization
- Improved skin health and reduced oxidative stress markers[4]
- Cognitive function improvements in some individuals
- Overall vitality enhancement reported by some users
Regular monitoring throughout this timeline helps identify any concerning patterns that warrant medical attention.
Those interested in comprehensive peptide research might explore synergistic approaches with other compounds for potentially enhanced benefits.
Minimizing Epithalon Peptide Side Effects: Best Practices
Proper Administration Techniques
Injection technique significantly impacts the severity of localized side effects:
- Subcutaneous injection in fatty tissue areas (abdomen, thigh)
- Site rotation to prevent tissue irritation and scarring
- Sterile technique using alcohol swabs and clean needles
- Slow injection speed to minimize tissue trauma
- Proper needle gauge (typically 29-31G) for comfort
Dosage Considerations
Starting with conservative dosages helps minimize initial side effects:
- Begin with lower doses (5-10mg) to assess individual tolerance
- Gradual increase over 1-2 weeks if well-tolerated
- Cycle protocols with breaks to prevent adaptation issues
- Medical supervision for dosage optimization and safety monitoring
Timing and Lifestyle Factors
Strategic timing can reduce certain epithalon peptide side effects:
- Evening administration to work with natural circadian rhythms
- Empty stomach injection 2-3 hours after meals
- Consistent timing to support circadian rhythm optimization
- Adequate hydration to support peptide metabolism
For researchers seeking professionally tested peptides, quality sourcing remains crucial for minimizing contamination-related side effects.
Research Gaps and Future Safety Studies

Current Research Limitations
The epithalon peptide side effects profile remains incomplete due to several research gaps:
- Small study populations in most published research
- Short-term follow-up periods inadequate for long-term safety assessment
- Limited demographic diversity in research participants
- Absence of dose-response studies for optimal safety protocols
Needed Research Areas
Future studies should address critical safety questions:
- Long-term toxicology studies spanning multiple years
- Drug interaction research with common medications
- Population-specific studies in elderly, diseased, and healthy cohorts
- Optimal dosing protocols for safety and efficacy balance
Regulatory Considerations
As epithalon gains popularity, regulatory oversight may evolve:
- FDA guidance updates on peptide safety requirements
- International regulatory harmonization for peptide research
- Quality control standards for peptide manufacturing
- Clinical trial requirements for therapeutic applications
Researchers should stay informed about regulatory developments affecting peptide research to ensure compliance and safety.
SEO Meta Title: Epithalon Peptide Side Effects: Complete Safety Guide 2026
Meta Description: Discover epithalon peptide side effects, safety concerns, and contraindications. Expert guide covers FDA warnings, research gaps, and best practices for 2026.
{“@context”:”https://schema.org”,”@type”:”Article”,”headline”:”epithalon peptide side effects”,”description”:”Discover epithalon peptide side effects, safety concerns, and contraindications. Expert guide covers FDA warnings, research gaps, and best practices for 2026.”,”image”:”https://zsxkvszxbhpwnvzxdydv.supabase.co/storage/v1/object/public/generated-images/kie/a7c9526a-74ce-4e73-9412-b9183f437cab/slot-0-1770080115844.png”,”datePublished”:”2026-02-03T00:53:05.394007+00:00″,”dateModified”:”2026-02-03T00:56:08.132Z”,”author”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″},”publisher”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″}}
