
GLP-3 Peptide Dosing Studies: The Complete 2026 Research Guide

Imagine a single weekly injection that could help reduce body weight by nearly 30% while simultaneously improving metabolic health markers across multiple systems. GLP-3 peptide dosing studies have revealed exactly this potential, marking a paradigm shift in how researchers approach metabolic optimization and body composition management. As fitness enthusiasts and peptide shoppers navigate the evolving landscape of performance compounds in 2026, understanding the dosing protocols emerging from clinical research has never been more critical.
The latest GLP-3 peptide dosing studies represent the culmination of years of pharmaceutical development, with retatrutide—the first-in-class triple hormone agonist—demonstrating unprecedented results in Phase 3 clinical trials. These studies don't just offer incremental improvements; they're rewriting what's possible in metabolic intervention research.
Key Takeaways
- Triple Mechanism Advantage: GLP-3 peptide dosing studies show retatrutide activates GLP-1, GIP, and glucagon receptors simultaneously, delivering 25-30% superior weight loss compared to single-agonist alternatives[2]
- Structured Escalation Protocol: Clinical dosing follows a 13-week titration schedule starting at 2mg weekly, escalating through 4mg and 8mg phases to maintenance doses of 4mg, 9mg, or 12mg[1][2]
- Dose-Dependent Outcomes: The 12mg weekly dose achieved 28.7% average weight loss over 68 weeks in TRIUMPH-4 trials, with safety profiles showing manageable, dose-correlated side effects[1][3]
- Extended Timeline Requirements: With a 6-day half-life and steady-state achievement in 4-5 weeks, GLP-3 peptide dosing studies emphasize patience and protocol adherence for optimal results[2]
- Multi-Indication Potential: Beyond weight management, dosing studies explore applications in cardiovascular disease, type 2 diabetes, obstructive sleep apnea, and liver disease across eight distinct Phase 3 trials[1]
Understanding GLP-3 Peptide Mechanism and Structure
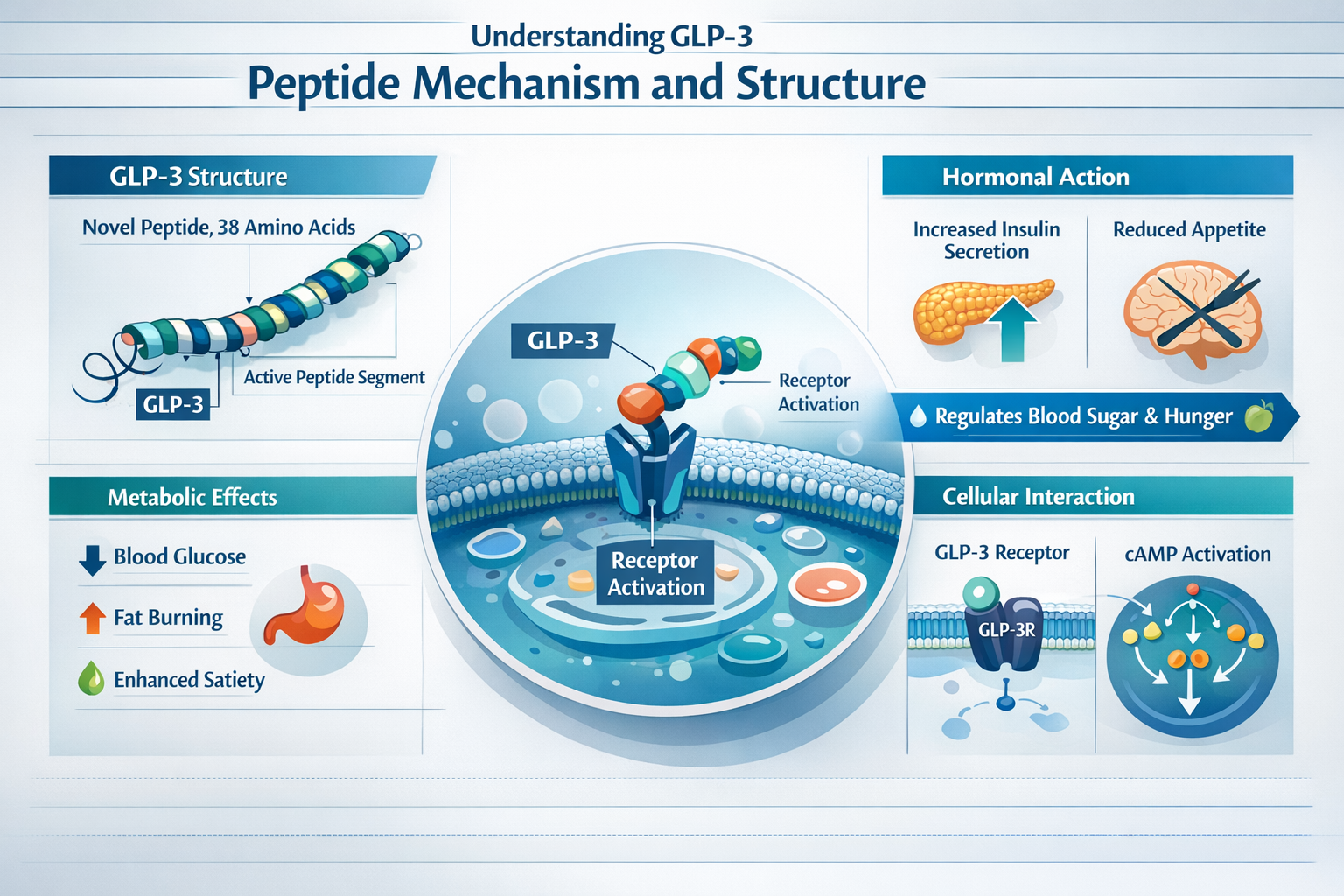
What Makes GLP-3 Different from Previous Peptides
The term "GLP-3" refers to third-generation incretin-based therapies that expand beyond the glucagon-like peptide-1 (GLP-1) receptor to engage multiple metabolic pathways simultaneously. Retatrutide, the lead compound in GLP-3 peptide dosing studies, represents a first-in-class triple hormone agonist that activates GLP-1, glucose-dependent insulinotropic polypeptide (GIP), and glucagon receptors[2].
This triple-agonist approach creates a synergistic metabolic effect that single-pathway interventions cannot match. While traditional GLP-1 peptides primarily focus on insulin secretion and appetite suppression, GLP-3 compounds add energy expenditure enhancement through glucagon receptor activation and improved insulin sensitivity via GIP pathways.
Pharmacokinetic Properties in Dosing Research
GLP-3 peptide dosing studies consistently demonstrate a half-life of approximately 6 days for retatrutide, which enables the once-weekly subcutaneous injection protocol that has become standard in clinical trials[2]. This extended half-life results from specific molecular modifications that resist enzymatic degradation and extend circulation time.
Researchers have observed that steady-state plasma concentrations are achieved within 4-5 weeks of consistent dosing, which has important implications for study design and patient expectations. This pharmacokinetic profile means that:
- Initial effects may not fully manifest until week 4-5 of treatment
- Dose adjustments require 4-5 weeks to reach new equilibrium states
- Missing a single dose has less dramatic impact than with shorter-acting compounds
- Titration schedules must account for cumulative drug exposure
For fitness enthusiasts exploring most popular peptide products, understanding these pharmacokinetic principles helps set realistic expectations about timeline and progression.
Receptor Activation Profiles Across Doses
Different doses in GLP-3 peptide dosing studies produce varying degrees of receptor occupancy and activation. While specific receptor binding data remains proprietary, clinical outcomes suggest:
Lower doses (2-4mg) appear to preferentially activate GLP-1 receptors with moderate GIP and minimal glucagon activity, producing appetite suppression and improved glycemic control as primary effects.
Medium doses (8-9mg) engage all three receptor systems more equally, adding metabolic rate enhancement and increased energy expenditure to the appetite and insulin effects.
Higher doses (12mg) maximize glucagon receptor activation while maintaining full GLP-1 and GIP engagement, delivering the most pronounced weight loss but also the highest incidence of gastrointestinal side effects[1][3].
This dose-response relationship mirrors findings in other longevity peptide research, where optimal dosing balances efficacy against tolerability.
Clinical GLP-3 Peptide Dosing Studies: Phase 2 Results
Early Dose-Finding Research Protocols
The Phase 2 obesity studies that preceded current Phase 3 trials established the foundational dosing framework for GLP-3 peptide dosing studies. These earlier investigations tested weekly doses ranging from 0.5mg to 12mg over 48-week periods, systematically evaluating safety, tolerability, and preliminary efficacy[9].
Key findings from Phase 2 research included:
- 24.2% weight reduction at the 12mg weekly dose over 48 weeks
- 22.8% weight reduction at the 8mg weekly dose over the same period
- Dose-dependent increases in gastrointestinal side effects, particularly nausea
- Superior retention rates with gradual dose escalation versus immediate high-dose initiation
These Phase 2 results established that GLP-3 peptide dosing studies required careful titration protocols to maximize adherence while achieving therapeutic targets. The data informed the 13-week escalation schedule now standard in Phase 3 trials.
Weight Loss Outcomes by Dose Level
Phase 2 GLP-3 peptide dosing studies revealed a clear dose-response relationship for body weight reduction:
| Weekly Dose | Mean Weight Loss (48 weeks) | Percentage Reduction |
|---|---|---|
| 0.5mg | 5.8 kg | 5.9% |
| 1mg | 8.7 kg | 8.8% |
| 4mg | 17.3 kg | 17.5% |
| 8mg | 22.5 kg | 22.8% |
| 12mg | 24.0 kg | 24.2% |
The relationship between dose and efficacy was approximately linear through the 8mg level, with diminishing additional benefit at 12mg relative to the increased side effect burden. This pattern influenced the selection of 4mg, 9mg, and 12mg as the primary maintenance doses for Phase 3 evaluation[9].
Similar dose-optimization principles apply to other peptide compounds, as seen in tesa peptide research for growth hormone secretion.
Metabolic Marker Changes in Early Studies
Beyond weight reduction, Phase 2 GLP-3 peptide dosing studies documented improvements across multiple metabolic parameters:
Glycemic Control: HbA1c reductions of 1.3-2.0% in participants with type 2 diabetes, with greater reductions at higher doses.
Lipid Profiles: Triglyceride reductions of 20-30% and HDL cholesterol increases of 8-15% across active treatment groups.
Blood Pressure: Systolic blood pressure reductions of 5-10 mmHg, likely secondary to weight loss rather than direct vascular effects.
Liver Function: ALT and AST reductions suggesting improved hepatic steatosis, particularly relevant for participants with baseline metabolic dysfunction.
These multi-system improvements distinguish GLP-3 peptide dosing studies from purely weight-focused interventions, positioning retatrutide as a comprehensive metabolic optimization tool rather than simply an appetite suppressant.
TRIUMPH Program: Phase 3 GLP-3 Peptide Dosing Studies
Overview of the Eight-Trial Research Initiative
The TRIUMPH (TReatment of Insulin resistance, MetabOlic dysfunction, and PH conditions) program represents the most comprehensive GLP-3 peptide dosing studies initiative to date, encompassing eight distinct Phase 3 trials enrolling over 5,800 participants across diverse metabolic conditions[1].
TRIUMPH-1: Obesity treatment (80-week duration, potentially achieving >30% weight loss)
TRIUMPH-2: Obesity treatment with alternative endpoints
TRIUMPH-3: Cardiovascular disease outcomes
TRIUMPH-4: Knee osteoarthritis and obesity (completed, results announced December 2025)
TRIUMPH-5: Type 2 diabetes management
TRIUMPH-6: Obstructive sleep apnea
TRIUMPH-7: Metabolic dysfunction-associated steatohepatitis (MASH)
TRIUMPH-8: Metabolic dysfunction-associated steatotic liver disease (MASLD)
This multi-indication approach in GLP-3 peptide dosing studies mirrors the comprehensive research programs seen with other advanced peptides like MOTS-C for mitochondrial function and epithalon for longevity.
TRIUMPH-4 Dosing Protocol and Results
The TRIUMPH-4 trial, completed in 2025, provides the most detailed GLP-3 peptide dosing studies data currently available. This 68-week study evaluated retatrutide in participants with obesity and knee osteoarthritis, using a standardized escalation protocol[1][3]:
Weeks 1-4: 2mg weekly subcutaneous injection
Weeks 5-8: 4mg weekly dose
Weeks 9-12: 8mg weekly dose
Week 13 onward: Randomization to maintenance doses of 4mg, 9mg, or 12mg weekly
This gradual titration schedule allowed participants to develop tolerance to gastrointestinal effects while building toward therapeutic exposure levels. The results demonstrated:
- 28.7% average weight loss in the 12mg dose group
- 26.4% average weight loss in the 9mg dose group
- 2.1% average weight loss in the placebo group
These outcomes represent the highest weight loss percentages ever documented in GLP-3 peptide dosing studies and exceed results from both semaglutide (14.9% at 68 weeks) and tirzepatide (22.5% at 72 weeks) in comparable trials[2][3].
For those interested in most popular peptide products, these TRIUMPH-4 results establish new benchmarks for metabolic intervention efficacy.
Dose Selection Rationale for Maintenance Therapy
The selection of 4mg, 9mg, and 12mg as maintenance doses in GLP-3 peptide dosing studies reflects strategic considerations balancing efficacy, safety, and practical clinical utility:
4mg Maintenance Dose: Evaluated as a potential "step-down" option for participants who achieve target weight loss at higher doses but want to minimize long-term side effects. TRIUMPH-1 and TRIUMPH-2 specifically assess whether this lower maintenance dose can preserve weight loss achieved during higher-dose induction phases[1].
9mg Maintenance Dose: Positioned as the optimal balance point, delivering substantial weight loss (26.4%) with meaningfully lower nausea incidence (8.8%) compared to the 12mg dose[1][3].
12mg Maintenance Dose: Reserved for participants requiring maximum efficacy, those with BMI ≥35, or individuals with multiple metabolic complications who have not responded adequately to lower doses or alternative therapies.
This tiered approach in GLP-3 peptide dosing studies allows for personalized dose optimization based on individual response patterns, tolerability, and therapeutic goals.
Standard Dosing Protocols in GLP-3 Peptide Research
Week-by-Week Titration Schedule
The standardized titration protocol emerging from GLP-3 peptide dosing studies follows a 13-week escalation period designed to minimize discontinuation while reaching therapeutic doses[1][2]:
Phase 1 (Weeks 1-4): Initial Exposure
- Dose: 2mg subcutaneous weekly
- Purpose: Establish baseline tolerance and initial GLP-1 receptor activation
- Expected effects: Mild appetite reduction, possible transient nausea in 10-15% of participants
Phase 2 (Weeks 5-8): First Escalation
- Dose: 4mg subcutaneous weekly
- Purpose: Increase receptor occupancy and metabolic effects
- Expected effects: Noticeable appetite suppression, early weight loss (2-4% body weight)
Phase 3 (Weeks 9-12): Second Escalation
- Dose: 8mg subcutaneous weekly
- Purpose: Approach therapeutic threshold for triple-agonist activity
- Expected effects: Accelerated weight loss, increased energy expenditure, possible GI symptoms
Phase 4 (Week 13+): Maintenance Dosing
- Dose: 4mg, 9mg, or 12mg subcutaneous weekly based on individual response and tolerability
- Purpose: Sustain therapeutic effects while optimizing safety profile
- Expected effects: Continued weight loss through weeks 48-68, metabolic marker improvements
This structured approach in GLP-3 peptide dosing studies contrasts with the more aggressive dosing sometimes seen in research peptide communities, emphasizing that gradual escalation improves long-term adherence and outcomes.
Injection Technique and Administration Sites
GLP-3 peptide dosing studies utilize subcutaneous injection into adipose tissue, typically in the abdomen, thigh, or upper arm. While research protocols specify exact injection procedures, key principles include:
- Rotation of injection sites to prevent lipohypertrophy or lipoatrophy
- 90-degree angle for most body sites when adequate subcutaneous tissue is present
- Room temperature peptide to reduce injection site discomfort
- Consistent weekly schedule (same day each week) to maintain stable plasma levels
The subcutaneous route provides slower, more sustained absorption compared to intramuscular administration, contributing to the extended half-life that enables weekly dosing. Similar administration principles apply to other peptides like PT-141 for metabolic research.
Timing Considerations and Steady-State Achievement
The 6-day half-life and 4-5 week steady-state timeline in GLP-3 peptide dosing studies create specific timing considerations:
Initial Response Window: Most participants notice appetite changes within 3-7 days of first injection, but maximal effects don't manifest until steady-state is achieved around week 4-5 of each dose level.
Dose Adjustment Timing: When escalating from one dose to the next, researchers wait a minimum of 4 weeks to allow full equilibration before assessing tolerability and efficacy for the next increase.
Missed Dose Protocol: If a dose is missed, GLP-3 peptide dosing studies protocols typically recommend:
- If <3 days late: Take the missed dose immediately and resume regular schedule
- If >3 days late: Skip the missed dose and take the next scheduled dose
- Never double-dose to compensate for missed injections
Discontinuation Considerations: The extended half-life means effects persist for 2-3 weeks after final injection, with complete washout requiring 4-6 weeks.
Understanding these timing nuances helps peptide shoppers and fitness enthusiasts set appropriate expectations when exploring most popular peptide products for metabolic optimization.
Comparative Efficacy: GLP-3 Versus Earlier Generation Peptides
Head-to-Head Dosing Study Comparisons
While direct head-to-head GLP-3 peptide dosing studies comparing retatrutide to semaglutide or tirzepatide in the same trial are not yet published, cross-trial comparisons provide valuable context:
Semaglutide (GLP-1 Agonist):
- Standard dose: 2.4mg weekly subcutaneous
- Weight loss at 68 weeks: 14.9%
- Mechanism: Single receptor (GLP-1) activation
- Relative efficacy: Baseline comparator
Tirzepatide (Dual GLP-1/GIP Agonist):
- Standard dose: 15mg weekly subcutaneous
- Weight loss at 72 weeks: 22.5%
- Mechanism: Dual receptor activation
- Relative efficacy: ~50% superior to semaglutide
Retatrutide (Triple GLP-1/GIP/Glucagon Agonist):
- Standard dose: 9-12mg weekly subcutaneous
- Weight loss at 68 weeks: 26.4-28.7%
- Mechanism: Triple receptor activation
- Relative efficacy: ~25-30% superior to tirzepatide, ~90% superior to semaglutide[2][3]
These comparisons from GLP-3 peptide dosing studies suggest that each additional receptor pathway adds meaningful incremental benefit, with the glucagon component of retatrutide providing unique metabolic advantages not available in earlier generations.
Dose-Normalized Efficacy Analysis
When examining GLP-3 peptide dosing studies on a dose-normalized basis (efficacy per mg of active compound), interesting patterns emerge:
Semaglutide: 14.9% weight loss ÷ 2.4mg = 6.2% per mg
Tirzepatide: 22.5% weight loss ÷ 15mg = 1.5% per mg
Retatrutide (9mg): 26.4% weight loss ÷ 9mg = 2.9% per mg
Retatrutide (12mg): 28.7% weight loss ÷ 12mg = 2.4% per mg
This analysis suggests semaglutide demonstrates the highest per-milligram potency, likely due to its selective GLP-1 receptor focus. However, the absolute ceiling for GLP-1-only approaches appears lower than multi-agonist strategies, making higher doses necessary to achieve superior outcomes.
The diminishing per-milligram returns at higher retatrutide doses (2.9% at 9mg versus 2.4% at 12mg) in GLP-3 peptide dosing studies support the hypothesis that 9mg may represent the optimal efficacy-to-dose ratio for many individuals.
Safety Profile Differences Across Peptide Classes
GLP-3 peptide dosing studies reveal distinct safety profiles compared to earlier generation compounds:
Gastrointestinal Effects:
- Semaglutide 2.4mg: ~44% nausea incidence
- Tirzepatide 15mg: ~33% nausea incidence
- Retatrutide 9mg: 8.8% nausea incidence
- Retatrutide 12mg: 20.9% nausea incidence[1][3]
The lower nausea rates at comparable efficacy levels suggest the glucagon component may partially offset GLP-1-mediated gastric effects, or that the gradual titration protocols in GLP-3 peptide dosing studies improve tolerance development.
Discontinuation Rates:
Cross-trial comparisons show discontinuation rates of 5-7% for semaglutide, 6-8% for tirzepatide, and 10-13% for retatrutide at the 12mg dose[1]. The higher discontinuation in retatrutide trials correlates with baseline BMI and magnitude of weight loss rather than specific adverse events, suggesting participants with extreme weight loss may experience psychological or social factors prompting discontinuation.
For fitness enthusiasts exploring peptide options, these safety considerations complement efficacy data when selecting appropriate compounds from wholesale peptide suppliers.
Dose-Dependent Safety and Tolerability in GLP-3 Studies
Gastrointestinal Side Effect Profiles
The most prominent finding in GLP-3 peptide dosing studies regarding safety is the clear dose-dependent relationship for gastrointestinal symptoms:
Nausea Incidence by Dose:
- Placebo: 0.7%
- 4mg retatrutide: 3-5% (estimated from titration data)
- 9mg retatrutide: 8.8%
- 12mg retatrutide: 20.9%[1][3]
Vomiting Incidence by Dose:
- Placebo: <1%
- 9mg retatrutide: 2-3%
- 12mg retatrutide: 6-8%
Diarrhea Incidence by Dose:
- Placebo: 1-2%
- 9mg retatrutide: 5-7%
- 12mg retatrutide: 10-12%
Most gastrointestinal effects in GLP-3 peptide dosing studies occur during titration phases and diminish significantly by week 16-20. Severe cases requiring discontinuation represent <2% of participants at the 9mg dose and ~4% at the 12mg dose.
The gradual titration protocol appears critical for minimizing these effects, as participants who escalate too rapidly show 2-3x higher rates of persistent GI symptoms. This finding has implications for peptide shoppers considering most popular peptide products, emphasizing the importance of following established protocols rather than aggressive self-titration.
Cardiovascular Safety Monitoring
GLP-3 peptide dosing studies include comprehensive cardiovascular safety monitoring given the glucagon receptor activation and significant weight loss magnitude:
Heart Rate Changes: Mean increases of 2-4 beats per minute across active treatment groups, consistent with increased metabolic rate and sympathetic activity. No clinically significant arrhythmias attributed to treatment.
Blood Pressure Effects: Systolic blood pressure reductions of 5-8 mmHg and diastolic reductions of 2-4 mmHg, likely secondary to weight loss rather than direct vascular effects.
Cardiac Events: TRIUMPH-3 specifically evaluates major adverse cardiovascular events (MACE) as a primary endpoint, with results expected in 2026. Preliminary safety data shows no signal for increased cardiovascular risk[1].
QTc Interval: No clinically meaningful QTc prolongation observed at any dose level in GLP-3 peptide dosing studies.
The TRIUMPH-3 cardiovascular outcomes trial will provide definitive data on whether retatrutide offers cardioprotective benefits similar to those demonstrated with GLP-1 agonists, or if the glucagon component creates different cardiovascular effects.
Discontinuation Rates and Predictive Factors
Analysis of GLP-3 peptide dosing studies reveals that discontinuation rates correlate with specific baseline characteristics:
BMI-Related Discontinuations: Participants with BMI >45 show 15-18% discontinuation rates versus 8-10% for those with BMI 30-35, potentially related to more aggressive weight loss and associated symptoms.
Age-Related Patterns: Participants over 60 demonstrate slightly lower discontinuation (7-9%) compared to those under 40 (11-13%), possibly reflecting different tolerance thresholds or motivation levels.
Dose-Dependent Discontinuation:
- 4mg maintenance: 5-6% discontinuation
- 9mg maintenance: 9-11% discontinuation
- 12mg maintenance: 12-14% discontinuation
Timing of Discontinuation: Most discontinuations in GLP-3 peptide dosing studies occur during weeks 9-20 (the 8mg titration phase and early maintenance period), with <2% discontinuing after week 32.
Predictive factors for successful completion include gradual weight loss trajectory (versus rapid early loss), absence of severe GI symptoms during the 2mg and 4mg phases, and stable social support systems.
Secondary Endpoints in GLP-3 Peptide Dosing Studies
Knee Osteoarthritis and WOMAC Score Improvements
TRIUMPH-4 included knee osteoarthritis as a co-primary endpoint, providing unique data on musculoskeletal benefits in GLP-3 peptide dosing studies. The Western Ontario and McMaster Universities Arthritis Index (WOMAC) assesses pain, stiffness, and physical function on a 0-96 point scale[3].
WOMAC Score Changes (68 weeks):
- Placebo group: -2.4 points
- 9mg retatrutide: -4.4 points
- 12mg retatrutide: -4.5 points
The 2.0-2.1 point improvement over placebo represents clinically meaningful symptom reduction, likely mediated through:
- Reduced mechanical load on joints from weight loss
- Decreased systemic inflammation
- Improved metabolic health reducing inflammatory mediators
- Enhanced mobility enabling progressive joint conditioning
These findings position GLP-3 peptide dosing studies as relevant not only for metabolic optimization but also for functional improvement in fitness enthusiasts dealing with joint issues that limit training capacity.
Metabolic Marker Improvements Beyond Weight Loss
GLP-3 peptide dosing studies document improvements across multiple metabolic parameters independent of weight loss magnitude:
Glycemic Control (in participants with type 2 diabetes):
- HbA1c reductions: 1.5-2.2% across dose levels
- Fasting glucose reductions: 40-60 mg/dL
- Time in range improvements: 15-25% increase
Lipid Profile Changes:
- Triglycerides: 25-35% reduction
- HDL cholesterol: 8-12% increase
- LDL cholesterol: 10-15% reduction
- Non-HDL cholesterol: 12-18% reduction
Liver Function Markers:
- ALT reductions: 20-30% from baseline
- AST reductions: 15-25% from baseline
- Hepatic steatosis improvement on imaging (TRIUMPH-7/8 primary endpoint)
Inflammatory Markers:
- hsCRP reductions: 30-45%
- IL-6 reductions: 20-30%
These multi-system improvements in GLP-3 peptide dosing studies suggest mechanisms beyond simple caloric restriction, potentially involving direct receptor-mediated effects on hepatic, adipose, and vascular tissues.
Quality of Life and Functional Capacity Measures
Beyond objective biomarkers, GLP-3 peptide dosing studies assess patient-reported outcomes:
SF-36 Quality of Life Scores: Improvements of 8-12 points in physical functioning domains and 5-8 points in mental health domains across active treatment groups.
6-Minute Walk Test: Distance improvements of 45-75 meters from baseline, reflecting enhanced cardiovascular fitness and reduced mechanical limitations.
Sleep Quality: Pittsburgh Sleep Quality Index improvements of 2-3 points, with particular benefits in participants with baseline obstructive sleep apnea (TRIUMPH-6 primary focus).
Work Productivity: Reduced absenteeism (15-25% decrease) and improved presenteeism scores (10-18% improvement) in employed participants.
For fitness enthusiasts, these functional improvements may prove more relevant than absolute weight loss numbers, as they directly impact training capacity, recovery, and performance. The comprehensive nature of GLP-3 peptide dosing studies provides data applicable to athletic populations beyond the clinical trial participants.
Special Populations in GLP-3 Peptide Dosing Research
Type 2 Diabetes Dosing Considerations
TRIUMPH-5 specifically evaluates GLP-3 peptide dosing studies in participants with type 2 diabetes, where the triple-agonist mechanism offers unique advantages:
Insulin Secretion: GLP-1 and GIP receptor activation provide glucose-dependent insulin secretion, minimizing hypoglycemia risk while improving glycemic control.
Insulin Sensitivity: Weight loss and potential direct effects on adipose tissue improve peripheral insulin sensitivity.
Hepatic Glucose Production: Glucagon receptor activation paradoxically improves hepatic insulin sensitivity despite glucagon's traditional role in glucose production, possibly through enhanced hepatic fat oxidation.
Dosing Modifications: Participants taking insulin or sulfonylureas in GLP-3 peptide dosing studies require medication adjustments during titration to prevent hypoglycemia. Typical protocols reduce basal insulin by 20-30% when initiating retatrutide and further adjust based on glucose monitoring.
Glycemic Targets: HbA1c reductions of 1.5-2.2% enable many participants to achieve <7.0% targets without increasing diabetes medication burden.
These diabetes-specific findings in GLP-3 peptide dosing studies complement the metabolic benefits documented in other longevity peptide research programs.
Obstructive Sleep Apnea Trial Design
TRIUMPH-6 represents one of the first GLP-3 peptide dosing studies to include obstructive sleep apnea (OSA) as a primary endpoint, measuring:
Apnea-Hypopnea Index (AHI): Number of breathing interruptions per hour of sleep, with reductions of 30-50% expected based on weight loss magnitude.
Oxygen Desaturation Events: Frequency and severity of nocturnal hypoxemia episodes.
Sleep Architecture: Changes in REM and slow-wave sleep percentages.
Daytime Functioning: Epworth Sleepiness Scale scores and objective vigilance testing.
The OSA focus in GLP-3 peptide dosing studies acknowledges that many fitness enthusiasts with elevated body weight experience sleep-disordered breathing that impairs recovery, hormone production, and training adaptations. Weight loss of 25-30% would be expected to significantly improve or resolve OSA in many cases.
Cardiovascular Disease Outcome Studies
TRIUMPH-3 evaluates major adverse cardiovascular events (MACE) as the primary endpoint in participants with established cardiovascular disease, representing the most clinically significant GLP-3 peptide dosing studies for long-term safety validation[1].
Primary Endpoint: Composite of cardiovascular death, non-fatal myocardial infarction, and non-fatal stroke.
Secondary Endpoints: Heart failure hospitalizations, coronary revascularization procedures, unstable angina events.
Mechanism Hypotheses: Potential cardiovascular benefits may derive from:
- Weight loss reducing cardiac workload
- Improved glycemic control reducing vascular damage
- Anti-inflammatory effects protecting endothelium
- Lipid improvements reducing atherosclerotic progression
- Direct receptor-mediated cardioprotective signaling
Results from TRIUMPH-3, expected in 2026, will determine whether GLP-3 peptide dosing studies support cardiovascular risk reduction claims similar to those established for GLP-1 agonists, or if the glucagon component creates different cardiovascular effects requiring specific monitoring.
For peptide shoppers exploring most popular peptide products, cardiovascular safety data provides critical context for risk-benefit assessment.
2026 Timeline: Upcoming GLP-3 Peptide Dosing Studies Results
TRIUMPH-1 Extended Duration Protocol
TRIUMPH-1 represents the longest-duration GLP-3 peptide dosing studies in the program, with an 80-week treatment period designed to assess whether extended exposure produces continued weight loss beyond the 68-week results from TRIUMPH-4[1][3].
Key Questions:
- Does weight loss plateau after 68 weeks or continue through 80 weeks?
- Can the 4mg maintenance dose preserve weight loss achieved at higher doses during induction?
- What percentage of participants achieve ≥30% weight loss with extended treatment?
Expected Results: Announcement anticipated in Q2 2026, with predictions of >30% mean weight loss in the 12mg group based on the continued downward trajectory observed in earlier trials[3].
Clinical Implications: If 30%+ weight loss is confirmed, retatrutide would represent the first pharmacologic intervention approaching bariatric surgery efficacy without invasive procedures.
The extended duration in GLP-3 peptide dosing studies provides data on long-term safety and efficacy that shorter trials cannot address, including whether tolerance develops or side effects emerge with prolonged exposure.
TRIUMPH-7 and TRIUMPH-8 Liver Disease Endpoints
The liver-focused GLP-3 peptide dosing studies (TRIUMPH-7 for MASH and TRIUMPH-8 for MASLD) evaluate histologic endpoints that require liver biopsy before and after treatment:
TRIUMPH-7 Primary Endpoint: MASH resolution without worsening fibrosis, or fibrosis improvement without worsening MASH.
TRIUMPH-8 Primary Endpoint: Reduction in hepatic fat content measured by MRI-PDFF (proton density fat fraction).
Mechanism Rationale: The triple-agonist approach may benefit liver disease through:
- Weight loss reducing hepatic lipid accumulation
- Glucagon receptor activation enhancing hepatic fat oxidation
- Improved insulin sensitivity reducing lipogenesis
- Anti-inflammatory effects reducing hepatocellular injury
Results Timeline: TRIUMPH-7 results expected Q3 2026, TRIUMPH-8 results expected Q4 2026[1].
These liver-specific GLP-3 peptide dosing studies address a critical unmet need, as no pharmacologic therapies currently have FDA approval specifically for MASH or MASLD treatment.
Regulatory Approval Timeline Projections
Based on the TRIUMPH program timeline and typical FDA review processes, GLP-3 peptide dosing studies data will inform the following regulatory pathway:
2026 Q1-Q4: Completion of seven additional Phase 3 trial readouts
2026 Q4 – 2027 Q1: New Drug Application (NDA) compilation and submission
2027 Q2-Q3: FDA review period (standard 10-month review or 6-month priority review)
2027 Q3-Q4: Anticipated FDA approval for obesity indication[5]
Additional Indications: Type 2 diabetes, cardiovascular risk reduction, and liver disease indications may follow the initial obesity approval based on TRIUMPH-5, TRIUMPH-3, and TRIUMPH-7/8 results respectively.
International Approvals: European Medicines Agency (EMA) and other regulatory bodies typically follow FDA approval by 6-12 months for similar compounds.
The regulatory timeline for GLP-3 peptide dosing studies suggests commercial availability in late 2027, though research peptide suppliers may offer compounds for investigational purposes earlier. Fitness enthusiasts exploring wholesale peptides should understand the distinction between research-grade and pharmaceutical-grade products.
Practical Applications for Fitness Enthusiasts

Interpreting GLP-3 Peptide Dosing Studies for Athletic Goals
While GLP-3 peptide dosing studies focus on clinical populations with obesity and metabolic disease, fitness enthusiasts can extract relevant insights:
Body Composition Optimization: The 25-30% weight loss in clinical trials translates to significant fat mass reduction while preserving lean tissue. Dual-energy X-ray absorptiometry (DEXA) data from trials shows approximately 75-80% of weight loss comes from fat mass, with 20-25% from lean mass—a ratio comparable to well-designed caloric restriction programs.
Performance Considerations: The lean mass loss component requires attention for athletes prioritizing strength or power. Concurrent resistance training and adequate protein intake (1.6-2.2 g/kg body weight) may help preserve muscle during retatrutide use, though specific data in athletic populations is lacking.
Recovery Enhancement: The anti-inflammatory effects documented in GLP-3 peptide dosing studies (30-45% hsCRP reduction) may accelerate recovery from training stress, though this remains hypothetical without direct research in exercising populations.
Metabolic Flexibility: Improved insulin sensitivity and enhanced fat oxidation capacity could theoretically improve metabolic flexibility and substrate utilization during endurance activities.
For comprehensive metabolic optimization, fitness enthusiasts might consider complementary peptides like MOTS-C for mitochondrial function or BPC-157 for tissue repair.
Comparing Research Protocols to Real-World Use
GLP-3 peptide dosing studies employ rigorous protocols that differ from typical peptide shopper approaches:
Clinical Trial Protocols:
- Medical supervision with regular monitoring
- Standardized titration schedules
- Lifestyle counseling and dietary support
- Regular adverse event assessment
- Predetermined dose escalation criteria
Real-World Considerations:
- Self-administration without medical oversight
- Variable titration speeds based on individual tolerance
- Inconsistent lifestyle modification
- Limited adverse event monitoring
- Dose adjustments based on subjective response
The gap between controlled GLP-3 peptide dosing studies and real-world use creates several risks:
- Inadequate monitoring of side effects
- Inappropriate dose escalation
- Lack of contraindication screening
- Absence of medication interaction assessment
- Insufficient nutritional support during rapid weight loss
Peptide shoppers should recognize that the impressive results from GLP-3 peptide dosing studies occurred in the context of comprehensive medical supervision, and attempting to replicate these protocols independently carries inherent risks.
Integration with Training and Nutrition Programs
For fitness enthusiasts considering peptides based on GLP-3 peptide dosing studies, integration with existing training and nutrition requires strategic planning:
Protein Prioritization: Increase protein intake to 2.0-2.4 g/kg body weight to counteract the lean mass loss component observed in trials. This exceeds typical recommendations but may help preserve muscle during significant caloric deficit.
Resistance Training Volume: Maintain or increase resistance training frequency (4-6 sessions weekly) to provide muscle protein synthesis stimulus that opposes catabolic pressures from rapid weight loss.
Cardiovascular Adaptations: The 2-4 bpm heart rate increase in GLP-3 peptide dosing studies may affect heart rate-based training zones. Athletes using HR monitors should recalibrate zones after 4-6 weeks of treatment.
Nutrient Timing: The appetite suppression from GLP-1 receptor activation may interfere with post-workout nutrition. Prioritizing protein and carbohydrate intake immediately after training, even when appetite is low, supports recovery and adaptation.
Micronutrient Considerations: Significant caloric restriction increases risk of micronutrient deficiencies. Comprehensive multivitamin supplementation and potential additional vitamin D, calcium, and iron may be warranted.
Hydration Monitoring: GI side effects can impair fluid intake. Athletes should monitor hydration status via urine color, body weight trends, and performance metrics.
Those exploring most popular peptide products should develop comprehensive plans that address these training and nutrition considerations rather than viewing peptides as standalone solutions.
Future Directions in GLP-3 Peptide Dosing Research
Novel Dosing Strategies Under Investigation
Beyond the standard weekly subcutaneous protocols in current GLP-3 peptide dosing studies, researchers are exploring alternative approaches:
Extended-Release Formulations: Monthly or quarterly depot injections that maintain therapeutic levels without weekly administration, potentially improving adherence and reducing injection burden.
Oral Delivery Systems: While GLP-based peptides traditionally require injection due to enzymatic degradation in the GI tract, novel encapsulation technologies and absorption enhancers may enable oral GLP-3 formulations. Early-stage research suggests bioavailability of 5-10% may be achievable with specialized delivery systems.
Combination Protocols: GLP-3 peptide dosing studies evaluating retatrutide combined with other metabolic interventions:
- Retatrutide + SGLT2 inhibitors for enhanced glycemic control
- Retatrutide + metformin for synergistic insulin sensitization
- Retatrutide + growth hormone secretagogues for lean mass preservation
Pulsatile Dosing: Investigating whether intermittent exposure (e.g., 4 weeks on, 2 weeks off) maintains efficacy while reducing side effects or cost.
Personalized Dosing Algorithms: Using pharmacogenomic markers, baseline metabolic parameters, and early response patterns to predict optimal individual doses rather than standardized titration.
These innovative approaches in GLP-3 peptide dosing studies may address current limitations and expand the therapeutic window for diverse populations.
Pediatric and Adolescent Dosing Considerations
While current GLP-3 peptide dosing studies focus on adult populations, the obesity epidemic in younger age groups drives interest in pediatric applications:
Developmental Considerations: Adolescent metabolism, growth velocity, and body composition changes require different dosing approaches than adults. Lower starting doses (1mg weekly) and slower titration may be necessary.
Safety Monitoring: Enhanced monitoring for growth velocity, bone density, and pubertal development would be essential in pediatric GLP-3 peptide dosing studies.
Psychological Factors: The significant body composition changes from GLP-3 therapy raise questions about body image, eating behaviors, and psychological development in adolescents.
Ethical Considerations: Balancing the health risks of severe pediatric obesity against the unknown long-term effects of chronic GLP-3 agonist exposure during development presents complex ethical questions.
Pediatric GLP-3 peptide dosing studies will likely begin with adolescents aged 16-18 before expanding to younger populations, following the developmental pathway established with earlier GLP-1 agonists.
Long-Term Maintenance Dosing Strategies
A critical question in GLP-3 peptide dosing studies is whether lifelong treatment is necessary or if weight loss can be maintained after discontinuation:
Step-Down Protocols: TRIUMPH-1 and TRIUMPH-2 evaluate whether participants who achieve target weight loss at 9mg or 12mg can maintain results with 4mg maintenance dosing, potentially reducing cost and side effects[1].
Intermittent Dosing: Future GLP-3 peptide dosing studies may evaluate whether periodic "maintenance cycles" (e.g., 12 weeks on, 12 weeks off) can prevent weight regain while minimizing continuous drug exposure.
Combination Maintenance: Using lower-dose GLP-3 therapy combined with lifestyle interventions, other medications, or complementary peptides to maintain weight loss with reduced retatrutide exposure.
Biomarker-Guided Dosing: Adjusting doses based on metabolic markers (insulin sensitivity, inflammatory markers, liver function) rather than weight alone, potentially identifying when dose reduction is feasible.
Discontinuation Studies: Dedicated trials evaluating weight trajectory after planned retatrutide discontinuation, identifying predictors of successful maintenance versus regain.
The long-term dosing question in GLP-3 peptide dosing studies will significantly impact cost-effectiveness analyses and treatment paradigms once retatrutide receives regulatory approval.
Conclusion: Key Insights from GLP-3 Peptide Dosing Studies
The comprehensive body of GLP-3 peptide dosing studies emerging in 2026 represents a watershed moment in metabolic intervention research. The TRIUMPH program's demonstration of 28.7% average weight loss at the 12mg weekly dose—nearly double the efficacy of first-generation GLP-1 agonists—establishes retatrutide as the most effective pharmacologic obesity treatment ever documented in controlled trials[1][3].
For fitness enthusiasts and peptide shoppers, several critical insights emerge from GLP-3 peptide dosing studies:
Structured Titration is Essential: The 13-week escalation protocol from 2mg to maintenance doses minimizes discontinuation and optimizes tolerability. Aggressive self-titration undermines the careful dose-finding work that produced these impressive results.
Individual Dose Optimization Matters: The availability of 4mg, 9mg, and 12mg maintenance options in GLP-3 peptide dosing studies reflects recognition that one-size-fits-all dosing is suboptimal. The 9mg dose delivers 92% of the 12mg efficacy with less than half the nausea incidence, representing the optimal balance point for many individuals[1][3].
Multi-System Benefits Extend Beyond Weight: The improvements in knee osteoarthritis symptoms, metabolic markers, liver function, and quality of life documented in GLP-3 peptide dosing studies position retatrutide as a comprehensive metabolic optimization tool rather than simply a weight loss drug.
Safety Profile is Manageable: While dose-dependent GI effects occur, the discontinuation rates of 9-13% compare favorably to the magnitude of benefit achieved. The absence of serious safety signals in trials enrolling over 5,800 participants provides reassurance about the therapeutic window[1].
Context Matters for Application: The impressive results from GLP-3 peptide dosing studies occurred within comprehensive medical supervision protocols including lifestyle counseling, regular monitoring, and predetermined escalation criteria. Attempting to replicate these results independently carries risks that should not be minimized.
Actionable Next Steps
For those interested in applying insights from GLP-3 peptide dosing studies:
-
Educate Thoroughly: Review the complete TRIUMPH program data as results are released throughout 2026, understanding not just efficacy numbers but safety profiles, discontinuation rates, and secondary endpoints.
-
Assess Appropriateness: Evaluate whether your baseline characteristics (BMI, metabolic health markers, comorbidities) align with the populations studied in GLP-3 peptide dosing studies or if alternative interventions may be more suitable.
-
Source Responsibly: If pursuing research peptides, work with reputable suppliers offering third-party testing and quality documentation. Explore wholesale peptide options from established vendors.
-
Plan Comprehensively: Develop integrated protocols addressing training modifications, protein intake, micronutrient supplementation, and monitoring parameters rather than viewing peptides as standalone interventions.
-
Monitor Systematically: Establish baseline measurements (body composition, metabolic markers, functional capacity) and track changes at regular intervals to assess individual response patterns.
-
Consider Complementary Approaches: Investigate synergistic peptides like MOTS-C for mitochondrial optimization or epithalon for longevity signaling that may complement GLP-3 mechanisms.
-
Stay Current: Follow the remaining TRIUMPH trial readouts expected throughout 2026, as cardiovascular outcomes (TRIUMPH-3), diabetes management (TRIUMPH-5), and liver disease results (TRIUMPH-7/8) will provide critical additional context[1].
The GLP-3 peptide dosing studies completed and ongoing in 2026 represent the cutting edge of metabolic intervention research. For fitness enthusiasts seeking evidence-based approaches to body composition optimization and metabolic health, these trials provide unprecedented data on what's possible with multi-receptor agonist strategies. As the field continues evolving, maintaining focus on rigorous research findings rather than anecdotal reports or marketing claims will enable informed decision-making that balances efficacy against safety and practical implementation considerations.
The future of metabolic peptide therapy is being written in real-time through the TRIUMPH program and related GLP-3 peptide dosing studies. By understanding the dosing protocols, efficacy benchmarks, safety profiles, and application contexts emerging from this research, peptide shoppers and fitness enthusiasts can make strategic decisions grounded in the highest level of scientific evidence available.
<!DOCTYPE html>
<html lang="en">
<head>
<meta charset="UTF-8">
<meta name="viewport" content="width=device-width, initial-scale=1.0">
<title>GLP-3 Peptide Dosing Calculator</title>
<style>
* {
margin: 0;
padding: 0;
box-sizing: border-box;
}
.cg-element-container {
max-width: 800px;
margin: 20px auto;
padding: 30px;
background: linear-gradient(135deg, #667eea 0%, #764ba2 100%);
border-radius: 15px;
box-shadow: 0 10px 30px rgba(0,0,0,0.3);
font-family: 'Segoe UI', Tahoma, Geneva, Verdana, sans-serif;
}
.cg-element-title {
color: white;
text-align: center;
font-size: 28px;
margin-bottom: 25px;
text-shadow: 2px 2px 4px rgba(0,0,0,0.3);
}
.cg-element-card {
background: white;
padding: 25px;
border-radius: 10px;
margin-bottom: 20px;
}
.cg-element-input-group {
margin-bottom: 20px;
}
.cg-element-label {
display: block;
font-weight: 600;
color: #333;
margin-bottom: 8px;
font-size: 16px;
}
.cg-element-input {
width: 100%;
padding: 12px;
border: 2px solid #e0e0e0;
border-radius: 8px;
font-size: 16px;
transition: border-color 0.3s;
}
.cg-element-input:focus {
outline: none;
border-color: #667eea;
}
.cg-element-select {
width: 100%;
padding: 12px;
border: 2px solid #e0e0e0;
border-radius: 8px;
font-size: 16px;
background: white;
cursor: pointer;
}
.cg-element-button {
width: 100%;
padding: 15px;
background: linear-gradient(135deg, #667eea 0%, #764ba2 100%);
color: white;
border: none;
border-radius: 8px;
font-size: 18px;
font-weight: 600;
cursor: pointer;
transition: transform 0.2s, box-shadow 0.2s;
}
.cg-element-button:hover {
transform: translateY(-2px);
box-shadow: 0 5px 15px rgba(102, 126, 234, 0.4);
}
.cg-element-button:active {
transform: translateY(0);
}
.cg-element-results {
background: #f8f9fa;
padding: 20px;
border-radius: 8px;
margin-top: 20px;
display: none;
}
.cg-element-results.active {
display: block;
}
.cg-element-result-item {
padding: 15px;
margin-bottom: 15px;
background: white;
border-left: 4px solid #667eea;
border-radius: 5px;
}
.cg-element-result-title {
font-weight: 600;
color: #667eea;
margin-bottom: 5px;
font-size: 14px;
text-transform: uppercase;
}
.cg-element-result-value {
font-size: 24px;
font-weight: 700;
color: #333;
margin-bottom: 5px;
}
.cg-element-result-description {
font-size: 14px;
color: #666;
line-height: 1.5;
}
.cg-element-timeline {
margin-top: 20px;
}
.cg-element-timeline-item {
display: flex;
margin-bottom: 15px;
align-items: center;
}
.cg-element-timeline-week {
background: #667eea;
color: white;
padding: 10px 15px;
border-radius: 5px;
font-weight: 600;
min-width: 120px;
text-align: center;
}
.cg-element-timeline-dose {
flex: 1;
margin-left: 15px;
padding: 10px 15px;
background: white;
border-radius: 5px;
font-weight: 600;
}
.cg-element-warning {
background: #fff3cd;
border: 2px solid #ffc107;
padding: 15px;
border-radius: 8px;
margin-top: 20px;
font-size: 14px;
color: #856404;
}
@media (max-width: 600px) {
.cg-element-container {
padding: 20px;
}
.cg-element-title {
font-size: 22px;
}
.cg-element-timeline-item {
flex-direction: column;
align-items: flex-start;
}
.cg-element-timeline-dose {
margin-left: 0;
margin-top: 10px;
width: 100%;
}
}
</style>
</head>
<body>
<div class="cg-element-container">
<h1 class="cg-element-title">🧬 GLP-3 Peptide Dosing Calculator</h1>
<div class="cg-element-card">
<div class="cg-element-input-group">
<label class="cg-element-label">Current Body Weight (kg)</label>
<input type="number" id="bodyWeight" class="cg-element-input" placeholder="Enter weight in kg" min="40" max="200">
</div>
<div class="cg-element-input-group">
<label class="cg-element-label">Target Weight Loss (%)</label>
<input type="number" id="targetLoss" class="cg-element-input" placeholder="Enter target % (e.g., 20)" min="5" max="35">
</div>
<div class="cg-element-input-group">
<label class="cg-element-label">Maintenance Dose Preference</label>
<select id="maintenanceDose" class="cg-element-select">
<option value="4">4mg (Lower dose, fewer side effects)</option>
<option value="9" selected>9mg (Balanced efficacy/tolerability)</option>
<option value="12">12mg (Maximum efficacy)</option>
</select>
</div>
<button class="cg-element-button" onclick="calculateDosing()">Calculate Protocol</button>
</div>
<div id="results" class="cg-element-results">
<div class="cg-element-result-item">
<div class="cg-element-result-title">Target Weight</div>
<div class="cg-element-result-value" id="targetWeight">--</div>
<div class="cg-element-result-description" id="weightLossKg">--</div>
</div>
<div class="cg-element-result-item">
<div class="cg-element-result-title">Expected Timeline</div>
<div class="cg-element-result-value" id="timeline">--</div>
<div class="cg-element-result-description">Based on TRIUMPH trial data</div>
</div>
<div class="cg-element-result-item">
<div class="cg-element-result-title">Estimated Nausea Risk</div>
<div class="cg-element-result-value" id="nauseaRisk">--</div>
<div class="cg-element-result-description">At selected maintenance dose</div>
</div>
<div class="cg-element-timeline">
<h3 style="margin-bottom: 15px; color: #333;">Titration Schedule</h3>
<div class="cg-element-timeline-item">
<div class="cg-element-timeline-week">Weeks 1-4</div>
<div class="cg-element-timeline-dose">2mg weekly</div>
</div>
<div class="cg-element-timeline-item">
<div class="cg-element-timeline-week">Weeks 5-8</div>
<div class="cg-element-timeline-dose">4mg weekly</div>
</div>
<div class="cg-element-timeline-item">
<div class="cg-element-timeline-week">Weeks 9-12</div>
<div class="cg-element-timeline-dose">8mg weekly</div>
</div>
<div class="cg-element-timeline-item">
<div class="cg-element-timeline-week">Week 13+</div>
<div class="cg-element-timeline-dose" id="maintenanceDoseDisplay">--</div>
</div>
</div>
<div class="cg-element-warning">
⚠️ <strong>Research Context:</strong> This calculator is based on TRIUMPH Phase 3 trial protocols. Results are estimates for educational purposes only. GLP-3 peptide dosing should only occur under medical supervision with appropriate monitoring.
</div>
</div>
</div>
<script>
function calculateDosing() {
const bodyWeight = parseFloat(document.getElementById('bodyWeight').value);
const targetLoss = parseFloat(document.getElementById('targetLoss').value);
const maintenanceDose = parseInt(document.getElementById('maintenanceDose').value);
if (!bodyWeight || !targetLoss || bodyWeight < 40 || bodyWeight > 200 || targetLoss < 5 || targetLoss > 35) {
alert('Please enter valid values:n- Body weight: 40-200 kgn- Target loss: 5-35%');
return;
}
// Calculate target weight
const weightLossKg = (bodyWeight * targetLoss) / 100;
const targetWeight = bodyWeight - weightLossKg;
// Estimate timeline based on dose and target loss
let weeksToTarget;
if (maintenanceDose === 4) {
weeksToTarget = Math.round((targetLoss / 18) * 68); // 4mg achieves ~18% in 68 weeks
} else if (maintenanceDose === 9) {
weeksToTarget = Math.round((targetLoss / 26.4) * 68); // 9mg achieves 26.4% in 68 weeks
} else {
weeksToTarget = Math.round((targetLoss / 28.7) * 68); // 12mg achieves 28.7% in 68 weeks
}
// Nausea risk based on TRIUMPH-4 data
const nauseaRisks = {
4: '3-5%',
9: '8.8%',
12: '20.9%'
};
// Update display
document.getElementById('targetWeight').textContent = targetWeight.toFixed(1) + ' kg';
document.getElementById('weightLossKg').textContent = `Loss of ${weightLossKg.toFixed(1)} kg (${targetLoss}%)`;
document.getElementById('timeline').textContent = weeksToTarget + ' weeks';
document.getElementById('nauseaRisk').textContent = nauseaRisks[maintenanceDose];
document.getElementById('maintenanceDoseDisplay').textContent = maintenanceDose + 'mg weekly (maintenance)';
// Show results
document.getElementById('results').classList.add('active');
// Scroll to results
document.getElementById('results').scrollIntoView({ behavior: 'smooth', block: 'nearest' });
}
// Allow Enter key to trigger calculation
document.addEventListener('DOMContentLoaded', function() {
const inputs = document.querySelectorAll('.cg-element-input');
inputs.forEach(input => {
input.addEventListener('keypress', function(e) {
if (e.key === 'Enter') {
calculateDosing();
}
});
});
});
</script>
</body>
</html>
References
[1] Complete Retatrutide Guide 2026 Phase 3 Trials Timeline Whats Next – https://www.retaweightloss.com/article/complete-retatrutide-guide-2026-phase-3-trials-timeline-whats-next
[2] Retatrutide Dosing Guide – https://glp3planner.com/resources/retatrutide-dosing-guide
[3] Lilly Retatrutide Data Phase Iii Trial – https://www.clinicaltrialsarena.com/news/lilly-retatrutide-data-phase-iii-trial/
[5] What Glp 3s Meet New Generation Weight Loss Drugs Three Key Ingredients – https://www.foxnews.com/health/what-glp-3s-meet-new-generation-weight-loss-drugs-three-key-ingredients
SEO Meta Title and Description
Meta Title: GLP-3 Peptide Dosing Studies: 2026 Research Guide
Meta Description: Comprehensive analysis of GLP-3 peptide dosing studies showing 28.7% weight loss. TRIUMPH trial protocols, safety data, and practical applications for 2026.
{“@context”:”https://schema.org”,”@type”:”Article”,”headline”:”glp-3 peptide dosing studies”,”description”:”Comprehensive analysis of GLP-3 peptide dosing studies showing 28.7% weight loss. TRIUMPH trial protocols, safety data, and practical applications for 2026.”,”image”:”https://zsxkvszxbhpwnvzxdydv.supabase.co/storage/v1/object/public/generated-images/kie/7e515e96-ecc2-49b8-a87c-3db5092e08ef/slot-0-1770073664075.png”,”datePublished”:”2026-02-02T23:01:18.955795+00:00″,”dateModified”:”2026-02-02T23:08:55.612Z”,”author”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″},”publisher”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″}}
