
Only one growth hormone secretagogue peptide has completed Phase III randomized controlled trials across more than 800 patients — yet the most widely used combination in clinical practice has zero published human trial data. That gap between evidence and popularity defines the current landscape of growth hormone secretagogue peptide research in 2026.
Key Takeaways 🔬
- Tesamorelin holds the strongest clinical evidence among all GH secretagogue peptides
- Ipamorelin is the most clinician-preferred starting compound due to its clean safety profile
- Combining GHRH-class and ghrelin-class peptides produces synergistic GH pulse amplification
- FDA regulatory status varies widely — from full approval to compounding removal
- Most growth hormone secretagogues peptide compounds lack long-term human safety data
What Is a Growth Hormone Secretagogues Peptide?
A growth hormone secretagogues peptide is a compound that stimulates the pituitary gland to release growth hormone (GH) naturally — rather than introducing synthetic GH directly. These peptides work through two primary receptor pathways:
- GHRH pathway — mimics growth hormone-releasing hormone (e.g., Tesamorelin, Sermorelin, CJC-1295)
- Ghrelin/GHS-R pathway — mimics ghrelin, the endogenous GH-stimulating peptide discovered by Kojima et al. in 1999 (e.g., Ipamorelin, GHRP-2, MK-677)
💡 "Ghrelin's discovery in 1999 established the physiological basis for every synthetic secretagogue that followed — fundamentally reshaping GH research."
Metabolic effects extend well beyond GH stimulation itself. Research shows these compounds influence carbohydrate and fat metabolism, muscle tissue growth, and energy utilization simultaneously.
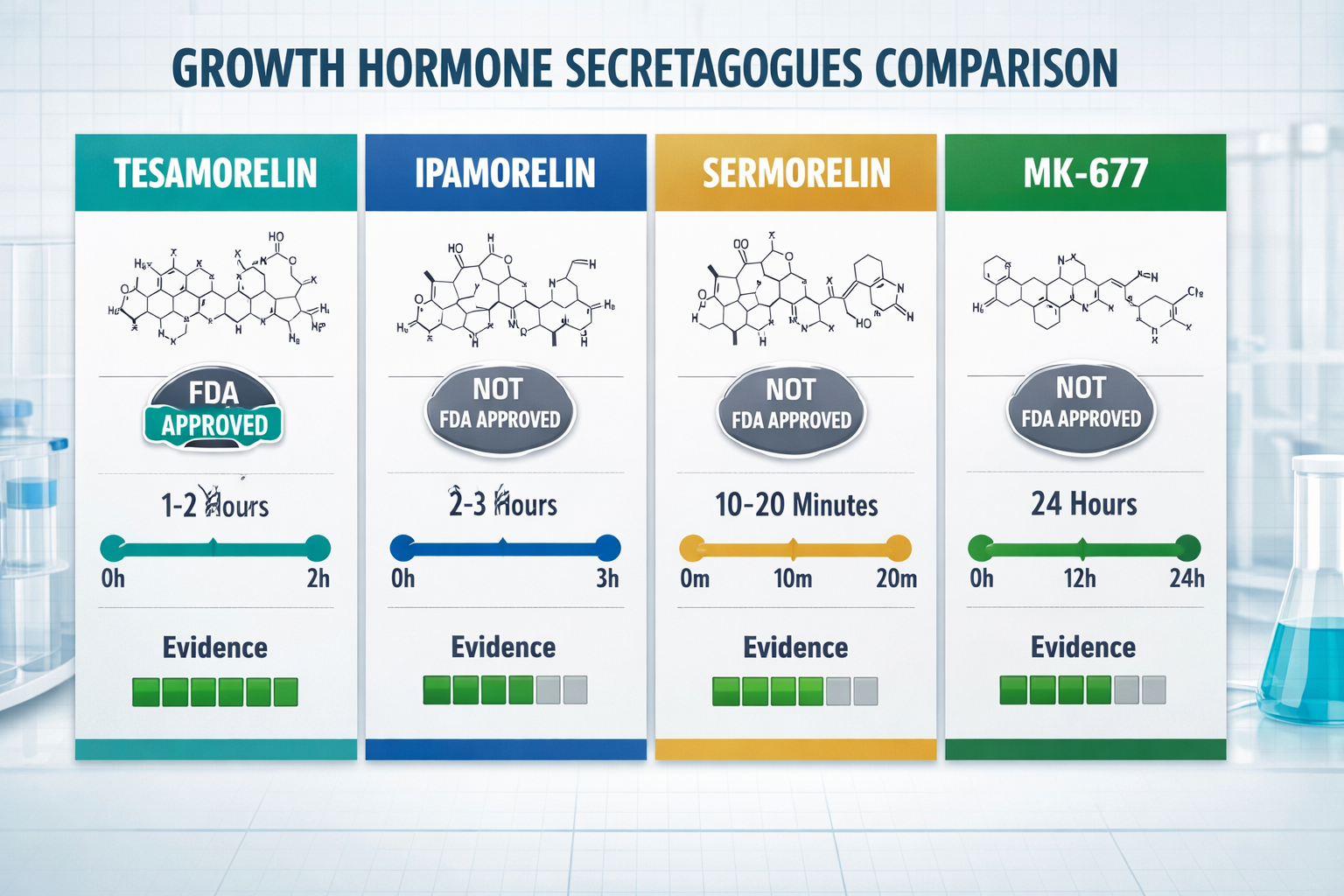
Top Growth Hormone Secretagogues Peptide Compounds Compared
| Peptide | Pathway | FDA Status | Key Evidence |
|---|---|---|---|
| Tesamorelin | GHRH | ✅ Approved (HIV lipodystrophy) | Phase III, 816 patients |
| Ipamorelin | Ghrelin | ❌ Not approved | Preclinical + clinical use |
| MK-677 | Ghrelin (oral) | ❌ Not approved | 2-year RCT, 65 patients |
| Sermorelin | GHRH | ⚠️ Withdrawn 2008 | Prior FDA approval |
| Anamorelin | Ghrelin | ✅ Japan (cachexia) | Phase III ROMANA trials |
| CJC-1295 | GHRH | ❌ Removed from compounding (2023) | No human trial data |
Tesamorelin: The Evidence Leader
Tesamorelin stands alone with Phase III data demonstrating 15–20% visceral fat reduction in HIV-associated lipodystrophy patients. Its FDA approval makes it the most legally defensible growth hormone secretagogues peptide for clinical application. For researchers exploring dosing parameters, the Tesamorelin dosage research overview provides useful context.
Ipamorelin: The Clinician's Starting Point
Ipamorelin is widely selected as a first-choice growth hormone secretagogues peptide because it releases GH without triggering cortisol, prolactin, or appetite changes — even at supra-physiologic doses. This selective action distinguishes it from older GHRPs like GHRP-2 and GHRP-6.
MK-677: The Only Oral Option
MK-677 offers a ~24-hour half-life and oral administration — unique among major secretagogues. A 2-year randomized controlled trial with 65 participants supports its GH-stimulating effects, making it one of the better-studied non-injectable options.
The Synergy Principle: Why Combinations Work
Research by Veldhuis and Bowers quantified a striking finding:
- GHRP-2 alone → 47-fold increase in pulsatile GH secretion
- GHRH alone → 20-fold increase
- Combined → 54-fold increase
This synergistic effect explains why the Sermorelin + Ipamorelin + CJC-1295 combination dominates clinical practice — even though published human trial data for this specific stack remains absent. Researchers interested in longevity-focused peptide applications should note this evidence gap when evaluating protocols.
[product_slider]
Regulatory Status: What Researchers Must Know ⚠️
Regulatory standing for growth hormone secretagogues peptide compounds varies significantly:
- Tesamorelin — FDA-approved for HIV lipodystrophy
- Macimorelin — FDA-approved for GH deficiency diagnosis only
- Anamorelin — Approved in Japan for cancer cachexia
- CJC-1295 — Removed from FDA compounding approval in 2023
- Sermorelin — Withdrawn from market in 2008 due to manufacturing issues
Always verify current regulatory status before sourcing any research compound. Reviewing quality testing protocols is an essential step for responsible research procurement.
Conclusion: Actionable Next Steps for Researchers
The growth hormone secretagogues peptide category offers some of the most compelling — and most misunderstood — compounds in peptide science. Here's what to prioritize in 2026:
- ✅ Start with evidence — Tesamorelin and Ipamorelin have the strongest documented profiles
- ✅ Understand synergy — Combining GHRH and ghrelin-pathway peptides amplifies GH pulse output
- ✅ Verify regulatory status — Compounding rules changed in 2023; confirm current standing
- ✅ Acknowledge safety gaps — Most compounds lack long-term human safety characterization
- ✅ Source responsibly — Use verified, tested suppliers with documented quality controls
Staying current with new developments in peptide research ensures research decisions are grounded in the latest available science.
Tags: growth hormone secretagogues peptide, GH secretagogue, tesa, ipamorelin, peptide therapy, GHRH peptide, ghrelin mimetic, MK-677, sermorelin, peptide research 2026, GH releasing peptide, longevity peptides
