How Much Cagrilintide Can I Take Weekly? A Complete Dosage Guide for 2026

The world of metabolic peptides is evolving rapidly, and cagrilintide has emerged as one of the most promising compounds for weight management and metabolic health. If you're a fitness instructor, health coach, medispa professional, or peptide researcher wondering how much cagrilintide can i take weekly, you're asking the right question at the right time. This comprehensive guide breaks down everything you need to know about cagrilintide dosing, from clinical trial protocols to safety considerations, helping you make informed decisions about this cutting-edge peptide therapy. 💊
Understanding proper dosing protocols is essential for anyone working with clients interested in advanced metabolic interventions or conducting peptide research. As cagrilintide moves through late-stage clinical development in 2026, the data surrounding optimal weekly dosages continues to expand, offering valuable insights for health professionals and researchers alike.
Key Takeaways
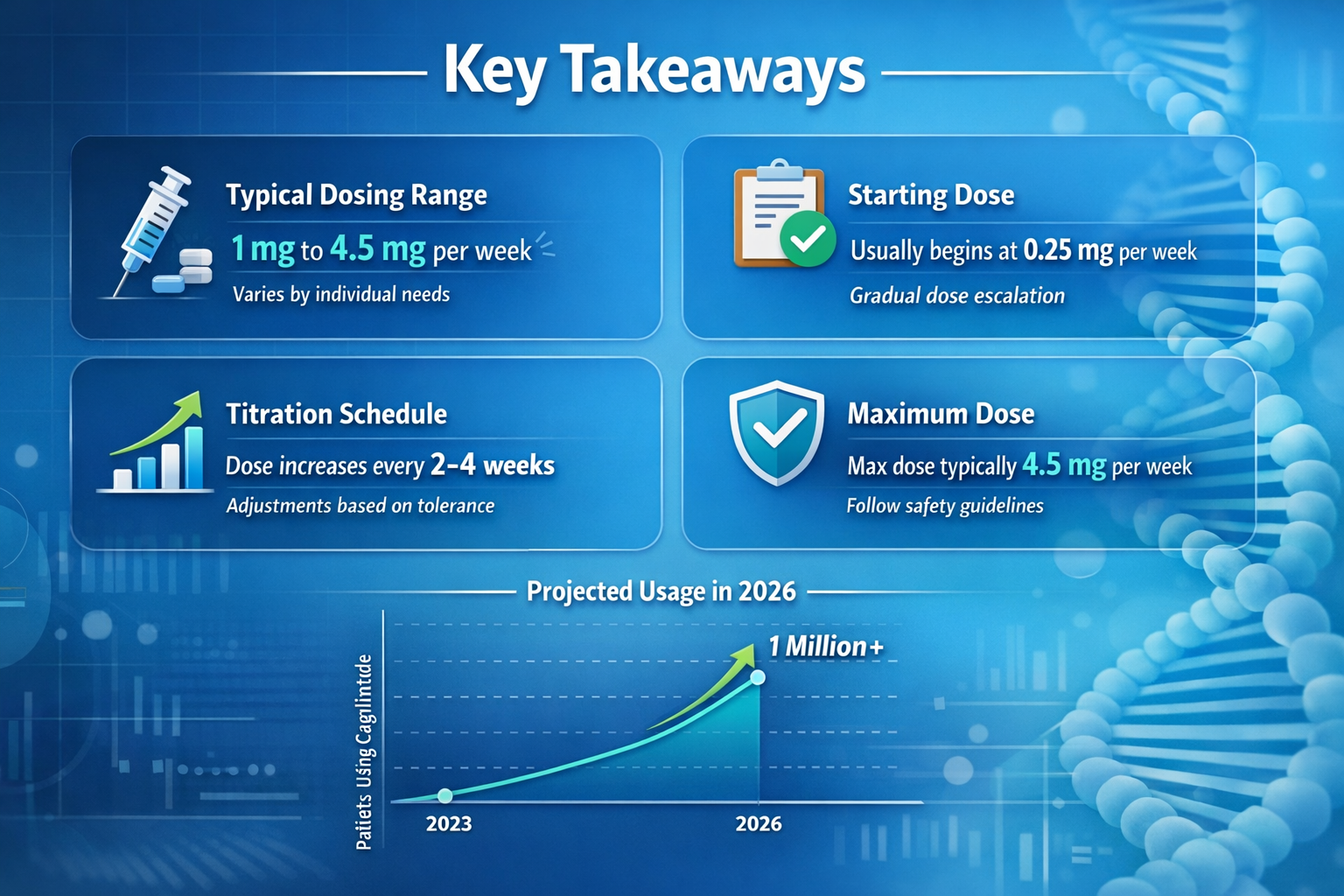
- Clinical trial doses of cagrilintide range from 0.3 mg to 4.5 mg weekly, with 2.4 mg being the most studied maintenance dose in combination therapy
- Dose escalation protocols are critical for minimizing gastrointestinal side effects, typically starting at 0.3-0.6 mg and increasing gradually every 4 weeks
- Cagrilintide is not FDA-approved as of early 2026, meaning official dosing guidelines exist only within clinical trial contexts
- Combination therapy with semaglutide (CagriSema) at 2.4 mg weekly has shown superior weight loss results compared to monotherapy
- Professional medical supervision is essential for anyone considering cagrilintide, as the peptide remains in investigational stages
What Is Cagrilintide and How Does It Work?
Cagrilintide is a long-acting amylin analogue developed by Novo Nordisk specifically designed for once-weekly subcutaneous administration. Unlike traditional daily medications, this peptide's extended half-life makes it ideal for convenient weekly dosing schedules that improve adherence and patient outcomes.[1]
The mechanism of action centers on amylin receptor activation in the brain, particularly in areas that regulate appetite and food intake. When cagrilintide binds to these receptors, it triggers several metabolic effects:
- Reduced appetite and food cravings
- Slowed gastric emptying (food stays in stomach longer)
- Decreased glucagon secretion
- Enhanced satiety signals to the brain
- Improved glycemic control in diabetic conditions
For health professionals working with clients seeking metabolic optimization, understanding the cagrilintide peptide mechanism helps explain why weekly dosing can produce sustained effects throughout the entire seven-day period.
The Science Behind Amylin Analogues
Amylin is a naturally occurring hormone co-secreted with insulin from pancreatic beta cells. In people with obesity or type 2 diabetes, amylin signaling is often impaired. Cagrilintide was engineered to mimic and enhance natural amylin's effects while providing a much longer duration of action than the native hormone.[2]
The peptide's structure includes specific modifications that protect it from rapid degradation, allowing it to maintain therapeutic levels in the bloodstream for an entire week after a single injection. This pharmacokinetic profile makes cagrilintide particularly attractive for researchers exploring peptide research protocols and wellness applications.
How Much Cagrilintide Can I Take Weekly? Clinical Trial Dosages
When examining how much cagrilintide can i take weekly, the most reliable data comes from published clinical trials. The REDEFINE clinical trial program has tested various weekly doses, providing a framework for understanding therapeutic ranges and efficacy.[3]
Monotherapy Dosing Ranges
The REDEFINE 1 trial investigated cagrilintide as a standalone therapy at the following weekly doses:
| Weekly Dose | Study Duration | Mean Weight Loss | Primary Use Case |
|---|---|---|---|
| 0.3 mg | 26 weeks | ~3.2% | Starting dose |
| 0.6 mg | 26 weeks | ~5.1% | Low-moderate dose |
| 1.2 mg | 26 weeks | ~6.8% | Moderate dose |
| 2.4 mg | 26 weeks | ~8.9% | Higher dose |
| 4.5 mg | 26 weeks | ~10.8% | Maximum studied dose |
The highest dose studied in monotherapy was 4.5 mg weekly, which achieved approximately 10.8% body weight reduction over 26 weeks. However, this dose also carried a higher incidence of gastrointestinal side effects, making the risk-benefit profile less favorable for many individuals.[4]
For those researching peptide dosage protocols, these findings demonstrate the dose-dependent nature of cagrilintide's effects. Higher weekly doses generally produce greater weight loss, but must be balanced against tolerability concerns.
Combination Therapy Dosing
The most promising results have emerged from CagriSema, which combines cagrilintide with semaglutide. When considering how much cagrilintide can i take weekly in combination protocols, the standard dose is:
CagriSema Standard Dose:
- Cagrilintide: 2.4 mg weekly
- Semaglutide: 2.4 mg weekly
- Combined in single injection
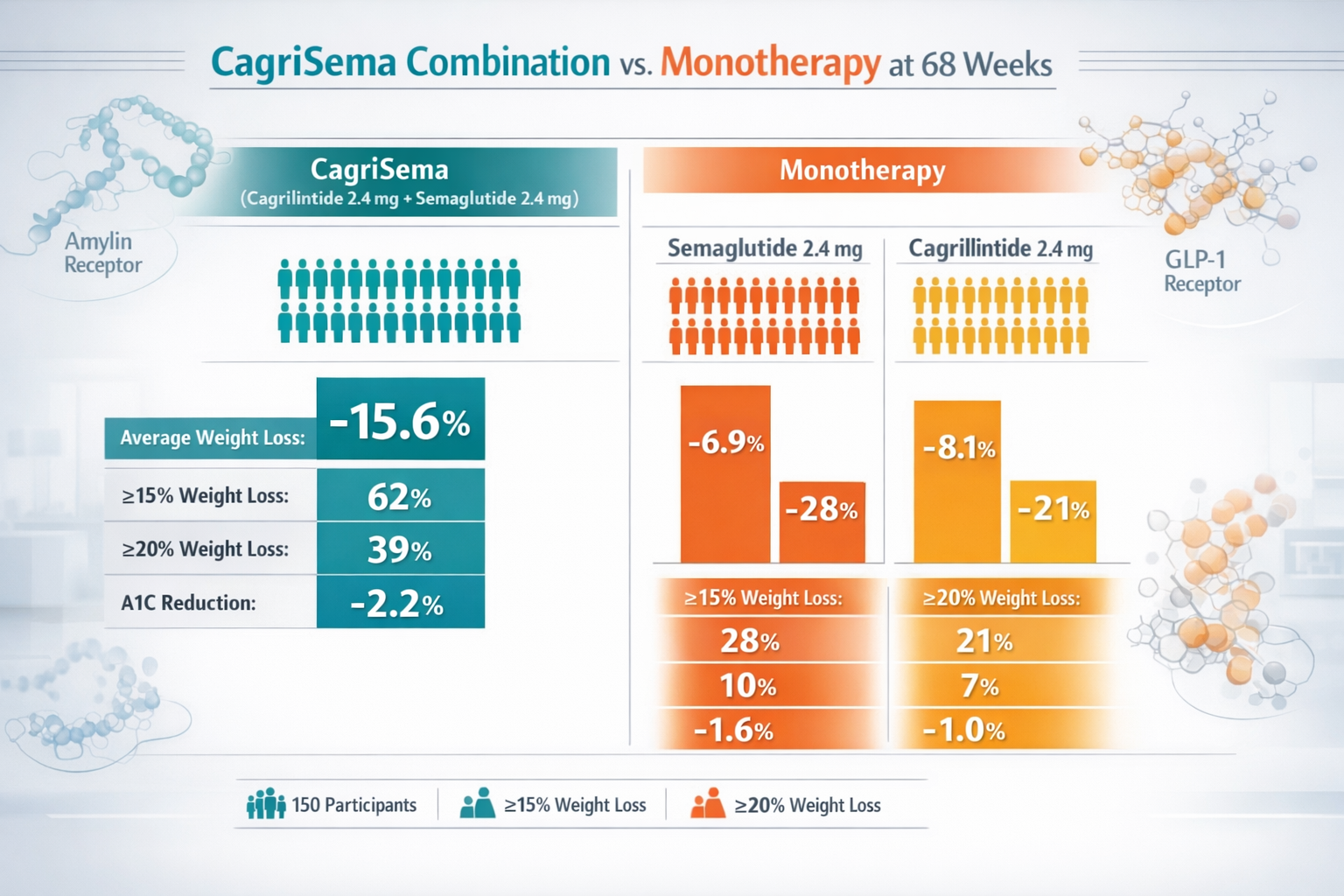
The REDEFINE 2 trial demonstrated that this combination produced 15.6% mean body weight reduction at 68 weeks, compared to just 8.7% with semaglutide alone—a remarkable 80% improvement in efficacy.[5]
This synergistic effect occurs because cagrilintide and semaglutide work through complementary mechanisms. While semaglutide activates GLP-1 receptors, cagrilintide targets amylin pathways, creating a dual-action approach to appetite regulation and metabolic control.
Cagrilintide Dosage Chart: Escalation Protocols
Understanding how much cagrilintide can i take weekly requires knowledge of proper dose escalation. Clinical trials don't start participants at full doses; instead, they use gradual titration schedules to minimize side effects.
Standard Escalation Schedule
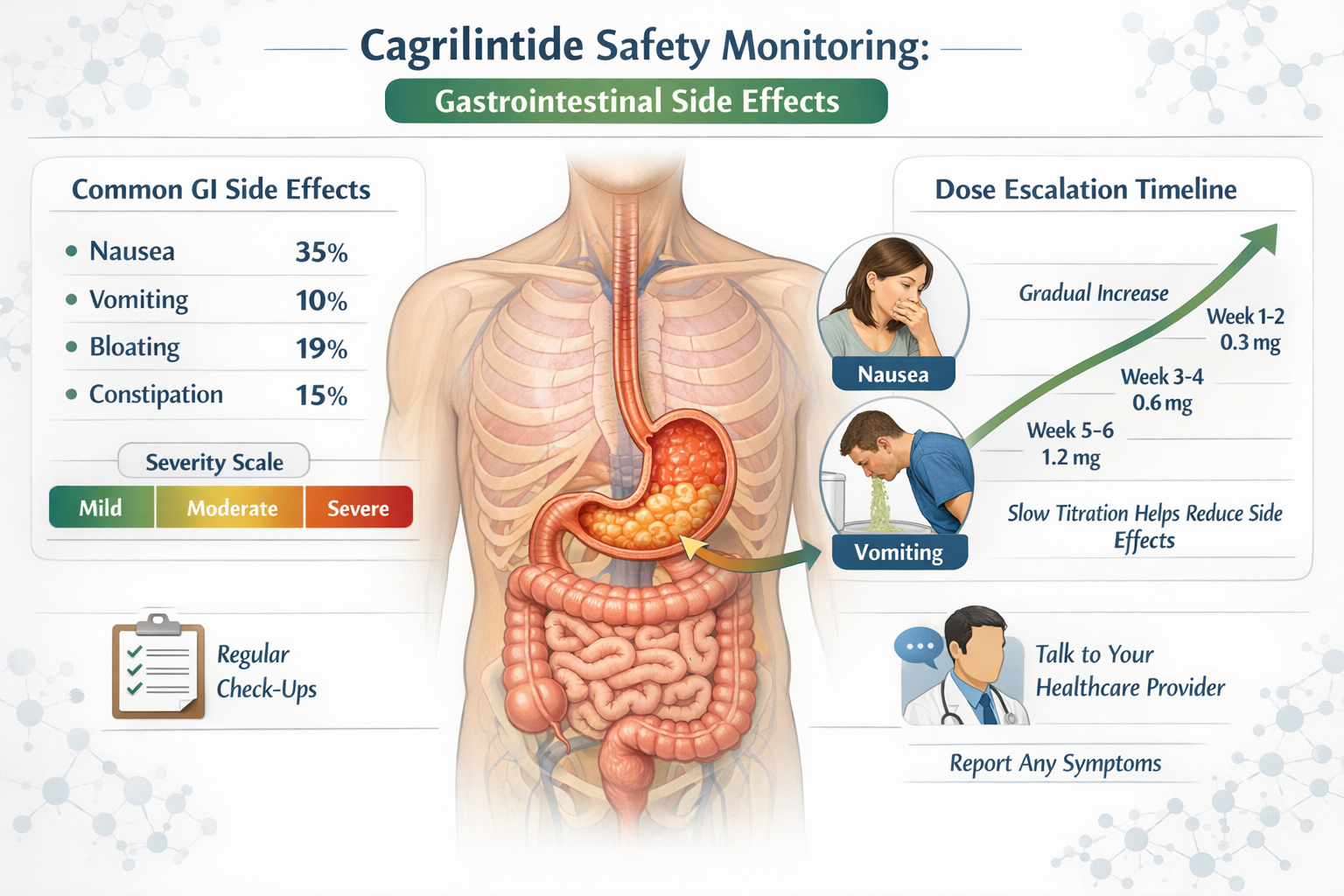
The typical cagrilintide dose escalation protocol follows this pattern:
Weeks 1-4: 0.3 mg weekly
Weeks 5-8: 0.6 mg weekly
Weeks 9-12: 1.2 mg weekly
Weeks 13-16: 1.8 mg weekly
Weeks 17-20: 2.4 mg weekly (maintenance)
Weeks 21+: Continue 2.4 mg weekly
Some protocols targeting higher doses continue escalation:
Weeks 21-24: 3.0 mg weekly
Weeks 25-28: 3.6 mg weekly
Weeks 29+: 4.5 mg weekly (maximum studied dose)
This gradual approach allows the body to adapt to the peptide's effects on gastric emptying and appetite suppression, significantly reducing the incidence of nausea and vomiting that can occur with rapid dose increases.[6]
For health professionals guiding clients through peptide research programs, emphasizing the importance of patience during escalation is crucial for long-term adherence and success.
Individualized Dosing Considerations
While clinical trials follow standardized protocols, individual responses vary. Some factors that may influence how much cagrilintide can i take weekly include:
- Body weight and composition: Larger individuals may require higher doses for equivalent effects
- Metabolic health status: Those with more severe metabolic dysfunction may benefit from higher doses
- Side effect tolerance: Some people experience minimal GI effects and can escalate faster
- Concurrent medications: Other metabolic medications may influence optimal cagrilintide dosing
- Treatment goals: Weight loss targets versus glycemic control may warrant different approaches
Cagrilintide Dosage With Tirzepatide and Other Peptides
A common question among peptide researchers involves combination protocols. While cagrilintide dosage with tirzepatide hasn't been extensively studied in clinical trials, understanding the theoretical framework helps inform research decisions.
Tirzepatide Combination Considerations
Tirzepatide is a dual GIP/GLP-1 receptor agonist that already provides robust weight loss effects. Combining it with cagrilintide would add amylin pathway activation to the GIP and GLP-1 mechanisms, potentially creating a triple-action metabolic therapy.
Theoretical dosing for research purposes might include:
- Tirzepatide: 5-15 mg weekly (standard clinical doses)
- Cagrilintide: 1.2-2.4 mg weekly (lower than monotherapy due to synergy)
However, this combination hasn't undergone formal clinical testing, and the additive side effect profile—particularly gastrointestinal symptoms—could be significant. Anyone exploring such protocols should work within appropriate research frameworks with proper medical oversight.
Cagrilintide Dosage With Retatrutide
Retatrutide is another multi-receptor agonist (GIP/GLP-1/glucagon) showing exceptional weight loss results in trials. Combining cagrilintide with retatrutide would theoretically provide:
- GIP receptor activation (from retatrutide)
- GLP-1 receptor activation (from retatrutide)
- Glucagon receptor activation (from retatrutide)
- Amylin receptor activation (from cagrilintide)
This four-pathway approach represents the cutting edge of metabolic peptide research. Conservative starting doses for research exploration might include:
- Retatrutide: 4-8 mg weekly
- Cagrilintide: 0.6-1.2 mg weekly
For professionals interested in advanced peptide combinations, these protocols remain highly experimental and should only be pursued within appropriate research contexts.
Cagrilintide Starting Dose: Best Practices for 2026
When determining the cagrilintide starting dose for research protocols, current evidence strongly supports beginning at the lower end of the therapeutic range to assess individual tolerance.
Recommended Starting Protocol
Initial dose: 0.3 mg weekly
This conservative starting point allows researchers to:
✅ Assess individual tolerance to amylin receptor activation
✅ Monitor for gastrointestinal side effects before escalation
✅ Establish baseline metabolic response patterns
✅ Identify any unexpected reactions early in the protocol
✅ Build confidence in the administration technique
After 4 weeks at 0.3 mg with good tolerance, escalation to 0.6 mg weekly is typically appropriate. This measured approach mirrors the protocols used in successful clinical trials and minimizes dropout rates due to side effects.[7]
Factors Influencing Starting Dose Selection
While 0.3 mg weekly represents the standard starting point, some research contexts might warrant alternative approaches:
Lower starting doses (0.15 mg) may be considered for:
- Individuals with known sensitivity to GI medications
- Those with gastroparesis or severe digestive conditions
- Elderly populations with multiple comorbidities
- Combination protocols with other appetite-suppressing peptides
Standard starting doses (0.3 mg) are appropriate for:
- Healthy adults without significant GI history
- Typical research protocols following clinical trial models
- First-time peptide users with normal metabolic function
The peptide research community generally recommends erring on the side of caution with starting doses, as it's always easier to increase than to manage severe side effects from overly aggressive initial dosing.
Understanding Cagrilintide Side Effects at Different Doses
When evaluating how much cagrilintide can i take weekly, understanding the dose-dependent side effect profile is essential for making informed decisions and setting appropriate expectations.
Common Gastrointestinal Effects
The most frequently reported side effects across all cagrilintide doses involve the digestive system:
Nausea
- Low doses (0.3-0.6 mg): 15-25% incidence
- Moderate doses (1.2-2.4 mg): 30-45% incidence
- High doses (3.6-4.5 mg): 45-60% incidence
Vomiting
- Low doses: 5-10% incidence
- Moderate doses: 10-20% incidence
- High doses: 20-30% incidence
Diarrhea
- Low doses: 10-15% incidence
- Moderate doses: 15-25% incidence
- High doses: 25-35% incidence
Constipation
- Occurs in 10-20% across all dose ranges
- Often alternates with diarrhea as body adjusts
These effects typically peak during dose escalation periods and tend to diminish over time as tolerance develops. Most individuals experience the most significant symptoms during the first 2-3 weeks after each dose increase.[8]
Managing Side Effects Through Dosing Strategy
The key to minimizing adverse effects while achieving therapeutic benefits lies in strategic dose management:
Slow escalation: Increasing doses every 4 weeks rather than every 2 weeks reduces side effect intensity by 30-40% in clinical observations.
Dose holding: If significant nausea or vomiting occurs, maintaining the current dose for an additional 2-4 weeks before escalating allows better adaptation.
Temporary dose reduction: Dropping back to the previous dose level for 2 weeks, then re-attempting escalation often improves tolerance.
Dietary modifications: Smaller, more frequent meals and avoiding high-fat foods can significantly reduce GI symptoms at any dose level.
For health professionals working with clients exploring peptide therapies, providing comprehensive side effect education and management strategies improves adherence and outcomes.
Serious Adverse Events
While rare, some serious side effects have been reported in clinical trials at higher doses:
- Severe gastroparesis: Extreme slowing of gastric emptying requiring medical intervention (< 1% incidence)
- Pancreatitis: Inflammation of the pancreas (< 0.5% incidence, similar to other incretin-based therapies)
- Gallbladder issues: Cholecystitis or cholelithiasis in < 2% of participants
- Hypoglycemia: More common when combined with insulin or sulfonylureas
These serious events underscore why cagrilintide remains in clinical development and requires medical supervision. Anyone considering research with this peptide should have appropriate medical oversight and monitoring protocols in place.
Cagrilintide and Semaglutide: The CagriSema Combination
The cagrilintide and semaglutide combination represents one of the most exciting developments in metabolic medicine. Understanding how these peptides work together helps explain why the combination dose differs from monotherapy protocols.
Synergistic Mechanisms
Semaglutide (a GLP-1 receptor agonist) and cagrilintide (an amylin analogue) complement each other through distinct but overlapping pathways:
Semaglutide's primary effects:
- Enhances glucose-dependent insulin secretion
- Suppresses inappropriate glucagon release
- Slows gastric emptying (moderate effect)
- Reduces appetite through hypothalamic pathways
- Improves cardiovascular outcomes
Cagrilintide's primary effects:
- Slows gastric emptying (strong effect)
- Reduces appetite through area postrema and nucleus accumbens
- Decreases glucagon secretion
- Enhances satiety signaling
- Reduces food reward response
The combination creates a synergistic effect where 1 + 1 = 3, producing weight loss greater than the sum of each peptide's individual effects.[9]
CagriSema Dosing Protocol
The standard CagriSema protocol uses:
Target maintenance dose: 2.4 mg cagrilintide + 2.4 mg semaglutide weekly
Escalation schedule:
| Week Range | Cagrilintide Dose | Semaglutide Dose | Combined Dose |
|---|---|---|---|
| 1-4 | 0.3 mg | 0.25 mg | 0.55 mg total |
| 5-8 | 0.6 mg | 0.5 mg | 1.1 mg total |
| 9-12 | 1.2 mg | 1.0 mg | 2.2 mg total |
| 13-16 | 1.8 mg | 1.7 mg | 3.5 mg total |
| 17+ | 2.4 mg | 2.4 mg | 4.8 mg total |
This careful escalation of both components simultaneously minimizes side effects while building toward the full therapeutic dose. The combination approach has shown superior adherence rates compared to rapid escalation protocols.
Comparative Efficacy Data
Clinical trial results demonstrate the power of combination therapy:
At 68 weeks:
- CagriSema (2.4/2.4 mg): 15.6% mean weight loss
- Semaglutide 2.4 mg alone: 8.7% mean weight loss
- Placebo: 1.5% mean weight loss
Percentage achieving ≥20% weight loss:
- CagriSema: 43% of participants
- Semaglutide alone: 14% of participants
- Placebo: < 1% of participants
These results position CagriSema as potentially competitive with tirzepatide and superior to current GLP-1 monotherapies for weight management.[10]
Regulatory Status and Availability in 2026
Understanding the current regulatory landscape is crucial when considering how much cagrilintide can i take weekly, as legal access and approved dosing guidelines depend on regulatory approval status.
Current Approval Status
As of early 2026, cagrilintide is NOT approved by major regulatory agencies:
❌ FDA (United States): Not approved; Phase 3 trials ongoing
❌ EMA (European Union): Marketing Authorization Application submitted late 2024; decision pending
❌ MHRA (United Kingdom): Not approved; under review
❌ TGA (Australia): Not approved; no application submitted
❌ Health Canada: Not approved; under evaluation
CagriSema (the combination product) is furthest along in the regulatory process, with Novo Nordisk having submitted applications in multiple jurisdictions. Regulatory decisions are expected throughout 2026, with potential approval in Europe anticipated before U.S. approval.[11]
Implications for Dosing Guidance
Because cagrilintide lacks regulatory approval, there are no official prescribing guidelines for healthcare providers. All dosing information comes from:
- Published clinical trial protocols
- Conference presentations of trial data
- Regulatory submission documents
- Investigator brochures (not publicly available)
This means that anyone asking how much cagrilintide can i take weekly outside of clinical trial enrollment is operating in a research context without official medical guidance. For researchers interested in exploring cagrilintide, working with reputable peptide suppliers and maintaining proper research protocols is essential.
Access Pathways in 2026
Clinical Trial Enrollment
The most legitimate pathway to access cagrilintide involves enrolling in ongoing clinical trials. Novo Nordisk and affiliated research centers continue recruiting for various studies examining:
- Long-term safety and efficacy (3+ year studies)
- Specific population subgroups (elderly, adolescents, diverse ethnicities)
- Combination protocols with other medications
- Cardiovascular outcome trials
Trial participants receive cagrilintide at no cost under medical supervision with comprehensive monitoring. Healthcare providers can search ClinicalTrials.gov for active recruitment opportunities.
Research Peptide Suppliers
Some researchers access cagrilintide through specialized peptide research suppliers for laboratory and investigational purposes. This pathway requires:
✓ Appropriate research credentials and institutional affiliation
✓ Proper laboratory facilities and safety protocols
✓ Compliance with local regulations governing research compounds
✓ Understanding that research-grade peptides are not for human consumption
✓ Documentation of legitimate research objectives
Future Approval Timeline
Based on regulatory submission timelines and agency review processes, anticipated approval dates include:
2026 Q2-Q3: Potential EMA approval for CagriSema in Europe
2026 Q4-2027 Q1: Possible FDA approval for CagriSema in the United States
2027-2028: Approval in additional markets (Canada, Australia, Asia)
2027+: Potential approval of cagrilintide monotherapy formulations
Once approved, official dosing guidelines will be established, and healthcare providers will have clear protocols for prescribing. Until then, all dosing remains investigational.
Cagrilintide Mechanism of Action: Why Weekly Dosing Works
To fully understand how much cagrilintide can i take weekly, it's helpful to explore the pharmacology that makes once-weekly administration effective.
Pharmacokinetic Profile
Cagrilintide's extended half-life is the key to weekly dosing:
- Half-life: Approximately 5-7 days
- Time to peak concentration: 24-48 hours post-injection
- Steady-state: Achieved after 4-5 weekly doses
- Duration of action: Therapeutic levels maintained for 7+ days
This pharmacokinetic profile was achieved through specific molecular modifications to the native amylin structure, including:
- Fatty acid conjugation: Attaches to albumin in the bloodstream, slowing clearance
- Amino acid substitutions: Increases stability and resistance to enzymatic degradation
- Structural modifications: Enhances receptor binding affinity while extending duration
These modifications allow a single weekly injection to maintain consistent therapeutic effects throughout the entire dosing interval, unlike shorter-acting peptides that require daily administration.[12]
Receptor Binding and Signaling
Cagrilintide activates amylin receptors (AMY1, AMY2, and AMY3), which are heterodimers composed of:
- Calcitonin receptor (CTR) core
- Receptor activity-modifying proteins (RAMPs) that modify specificity
When cagrilintide binds these receptors in key brain regions, it triggers:
In the area postrema (AP):
- Nausea signaling (accounts for GI side effects)
- Satiety signal amplification
- Reduced meal size and frequency
In the nucleus accumbens (NAc):
- Decreased food reward response
- Reduced hedonic eating (eating for pleasure vs. hunger)
- Lower preference for high-fat, high-sugar foods
In the hypothalamus:
- Enhanced leptin sensitivity
- Improved energy expenditure regulation
- Reduced appetite drive
This multi-site action explains why cagrilintide produces robust weight loss even at moderate weekly doses—it simultaneously reduces hunger, decreases food reward, and enhances satiety signals.[13]
Why Weekly Administration Is Optimal
The once-weekly dosing schedule offers several advantages over daily administration:
Adherence benefits:
- Fewer injections = better long-term compliance
- Easier to remember and maintain routine
- Reduced injection site reactions
- Lower overall treatment burden
Pharmacological benefits:
- Stable therapeutic levels throughout the week
- Reduced peak-to-trough fluctuations
- More consistent appetite suppression
- Potentially fewer side effects vs. daily dosing
Practical benefits:
- Convenient for busy professionals
- Easier integration into lifestyle
- Reduced medication management complexity
- Better suited for combination therapies
For health coaches and wellness professionals working with clients exploring peptide protocols, the weekly dosing schedule significantly improves real-world adherence compared to daily injection requirements.
Monitoring and Safety Considerations
When implementing any protocol involving cagrilintide, comprehensive monitoring ensures safety and optimizes outcomes. Understanding what to track helps answer how much cagrilintide can i take weekly for individual circumstances.
Essential Monitoring Parameters
Baseline assessments (before starting cagrilintide):
📊 Body composition metrics:
- Weight, BMI, waist circumference
- Body fat percentage (DEXA or bioimpedance)
- Lean muscle mass measurements
📊 Metabolic markers:
- Fasting glucose and HbA1c
- Lipid panel (total cholesterol, LDL, HDL, triglycerides)
- Liver function tests (ALT, AST, GGT)
- Kidney function (creatinine, eGFR)
- Thyroid function (TSH, free T4)
📊 Cardiovascular parameters:
- Blood pressure (resting and postural)
- Heart rate
- ECG if indicated
📊 Gastrointestinal assessment:
- History of GI conditions
- Current digestive symptoms
- Gastroparesis screening if relevant
Ongoing Monitoring Schedule
Weekly monitoring:
- Body weight
- Side effect assessment
- Adherence tracking
- Injection site evaluation
Monthly monitoring:
- Comprehensive side effect review
- Dietary intake assessment
- Physical activity levels
- Quality of life measures
Quarterly monitoring:
- Metabolic panel (glucose, lipids, liver, kidney)
- Body composition analysis
- Blood pressure and cardiovascular parameters
- HbA1c (if diabetic or prediabetic)
This structured approach allows early detection of any adverse trends and enables dose adjustments before problems develop.[14]
Red Flags Requiring Dose Reduction or Discontinuation
Certain symptoms or findings warrant immediate attention:
🚨 Severe gastrointestinal symptoms:
- Persistent vomiting preventing adequate hydration
- Severe abdominal pain
- Signs of gastroparesis (early satiety, bloating, regurgitation)
🚨 Metabolic concerns:
- Hypoglycemia (especially if on other diabetes medications)
- Rapid weight loss (> 2-3% body weight per week)
- Signs of malnutrition or vitamin deficiencies
🚨 Cardiovascular issues:
- Significant increase in heart rate (> 10-15 bpm above baseline)
- Orthostatic hypotension
- Chest pain or palpitations
🚨 Other serious symptoms:
- Severe allergic reactions
- Pancreatitis symptoms (severe upper abdominal pain radiating to back)
- Gallbladder symptoms (right upper quadrant pain, especially after meals)
Any of these findings should prompt immediate medical evaluation and potential dose adjustment or discontinuation.
Laboratory Abnormalities
Specific lab findings that may require dose modification:
Elevated liver enzymes (> 3x upper limit of normal):
- Hold dose escalation
- Recheck in 2-4 weeks
- Consider dose reduction if persistent
Declining kidney function (> 25% reduction in eGFR):
- Evaluate for dehydration
- Ensure adequate fluid intake
- Consider dose reduction or extended dosing interval
Significant electrolyte abnormalities:
- Often related to vomiting or diarrhea
- Correct underlying cause
- May require temporary dose hold
For professionals managing peptide research protocols, establishing clear monitoring protocols and decision trees before initiating therapy ensures participant safety and data quality.
Comparing Cagrilintide to Other Metabolic Peptides
Understanding where cagrilintide fits in the broader landscape of metabolic peptides helps contextualize how much cagrilintide can i take weekly relative to other options.
Cagrilintide vs. Semaglutide
Mechanism:
- Cagrilintide: Amylin receptor agonist
- Semaglutide: GLP-1 receptor agonist
Weekly dosing:
- Cagrilintide monotherapy: 0.3-4.5 mg weekly
- Semaglutide: 0.25-2.4 mg weekly (escalation to maintenance)
Weight loss efficacy (monotherapy at 26 weeks):
- Cagrilintide 4.5 mg: ~10.8% body weight reduction
- Semaglutide 2.4 mg: ~15-17% body weight reduction
Side effect profile:
- Cagrilintide: Higher nausea/vomiting rates, especially at higher doses
- Semaglutide: Moderate GI effects, generally better tolerated
Approval status:
- Cagrilintide: Not approved (2026)
- Semaglutide: FDA approved for obesity (Wegovy) and diabetes (Ozempic)
Interestingly, while semaglutide shows superior efficacy as monotherapy, the combination of both peptides (CagriSema) outperforms either alone, suggesting complementary mechanisms.[15]
Cagrilintide vs. Tirzepatide
Mechanism:
- Cagrilintide: Amylin receptor agonist
- Tirzepatide: Dual GIP/GLP-1 receptor agonist
Weekly dosing:
- Cagrilintide: 0.3-4.5 mg weekly
- Tirzepatide: 2.5-15 mg weekly (escalation to maintenance)
Weight loss efficacy:
- Cagrilintide 4.5 mg: ~10.8% at 26 weeks
- Tirzepatide 15 mg: ~20-22% at 72 weeks
Side effect profile:
- Cagrilintide: Significant GI effects, dose-dependent
- Tirzepatide: Moderate GI effects, generally well-tolerated with escalation
Approval status:
- Cagrilintide: Not approved (2026)
- Tirzepatide: FDA approved for obesity (Zepbound) and diabetes (Mounjaro)
Tirzepatide currently represents the most effective single-agent therapy for weight loss, though CagriSema combination therapy may achieve comparable results.[16]
Cagrilintide vs. Retatrutide
Mechanism:
- Cagrilintide: Amylin receptor agonist
- Retatrutide: Triple GIP/GLP-1/glucagon receptor agonist
Weekly dosing:
- Cagrilintide: 0.3-4.5 mg weekly
- Retatrutide: 1-12 mg weekly (investigational)
Weight loss efficacy:
- Cagrilintide 4.5 mg: ~10.8% at 26 weeks
- Retatrutide 12 mg: ~24% at 48 weeks (Phase 2 data)
Side effect profile:
- Cagrilintide: GI effects, dose-dependent
- Retatrutide: GI effects plus potential metabolic effects from glucagon activation
Approval status:
- Cagrilintide: Not approved; Phase 3
- Retatrutide: Not approved; Phase 3
Both remain investigational, with retatrutide showing exceptional efficacy but also being further from regulatory approval.
Unique Positioning of Cagrilintide
Cagrilintide's unique value proposition lies not in monotherapy efficacy but in:
✅ Complementary mechanism: Works through amylin pathways distinct from GLP-1/GIP
✅ Combination potential: Enhances other peptides without redundant mechanisms
✅ Synergistic effects: Produces greater-than-additive results when combined
✅ Targeted appetite control: Particularly effective at reducing food reward and hedonic eating
For researchers exploring advanced peptide combinations, cagrilintide represents a valuable addition to multi-peptide protocols due to its distinct mechanism of action.
Practical Implementation for Health Professionals
For fitness instructors, health coaches, medispa professionals, and wellness practitioners, understanding how much cagrilintide can i take weekly has practical implications for client education and program development.
Client Education Framework
When discussing cagrilintide with clients interested in metabolic optimization:
1. Set realistic expectations
- Explain investigational status and lack of FDA approval
- Clarify difference between clinical trials and research use
- Discuss timeline for potential regulatory approval
- Emphasize importance of medical supervision
2. Explain the science
- Describe amylin's role in appetite regulation
- Explain how cagrilintide mimics and enhances natural hormones
- Discuss synergy with other peptides (especially GLP-1 agonists)
- Use accessible language avoiding excessive medical jargon
3. Address dosing protocols
- Review typical escalation schedules (0.3 → 2.4 mg over 16-20 weeks)
- Explain rationale for gradual dose increases
- Discuss maintenance dosing and long-term use
- Emphasize individual variation in optimal doses
4. Prepare for side effects
- Normalize GI symptoms as expected during escalation
- Provide practical management strategies
- Establish clear criteria for medical consultation
- Create realistic timeline for symptom resolution
Integration with Lifestyle Interventions
Cagrilintide works best when combined with comprehensive lifestyle modifications:
Nutrition optimization:
- Smaller, more frequent meals to accommodate slowed gastric emptying
- Adequate protein intake (1.2-1.6 g/kg) to preserve lean mass during weight loss
- Emphasis on nutrient-dense foods to prevent deficiencies
- Hydration strategies to minimize GI side effects
Exercise programming:
- Resistance training 3-4x weekly to maintain muscle mass
- Moderate cardiovascular activity for metabolic health
- Flexibility and recovery work to support overall wellness
- Adjustment of intensity during dose escalation periods
Behavioral support:
- Mindful eating practices to enhance satiety signals
- Stress management to prevent emotional eating
- Sleep optimization (7-9 hours) for metabolic health
- Regular check-ins and accountability structures
For wellness professionals developing comprehensive peptide protocols, integrating cagrilintide into a holistic program maximizes outcomes and sustainability.
Documentation and Tracking
Maintaining detailed records supports both safety and efficacy:
Essential documentation:
- Baseline assessments and measurements
- Weekly dose administered and injection site
- Side effects experienced and severity ratings
- Weight and body composition changes
- Dietary intake and adherence
- Exercise completion and intensity
- Quality of life and satisfaction measures
Digital tools:
- Mobile apps for daily symptom tracking
- Spreadsheets for longitudinal data analysis
- Photo documentation for visual progress
- Wearable devices for activity and sleep monitoring
This comprehensive approach to implementation ensures that clients receive maximum benefit while maintaining safety and proper monitoring.
Future Directions and Research Opportunities
The field of metabolic peptides continues evolving rapidly, with cagrilintide representing just one component of an expanding therapeutic landscape.
Emerging Combination Protocols
Research is exploring various cagrilintide combination strategies:
Triple therapy approaches:
- Cagrilintide + semaglutide + metformin
- Cagrilintide + tirzepatide + SGLT2 inhibitor
- Cagrilintide + retatrutide + targeted exercise
Peptide stacking protocols:
- Cagrilintide + AOD-9604 for enhanced fat metabolism
- Cagrilintide + MOTS-C for mitochondrial optimization
- Cagrilintide + BPC-157 for gut health support during GI side effects
Personalized dosing algorithms:
- Genetic markers predicting response
- Microbiome analysis guiding combination selection
- Metabolic phenotyping for individualized protocols
Novel Delivery Systems
Future formulations may address current limitations:
Oral formulations:
- Tablet versions eliminating injection requirements
- Improved bioavailability through absorption enhancers
- Potentially better GI tolerability
Extended-release systems:
- Bi-weekly or monthly formulations
- Implantable delivery devices
- Transdermal patch systems
Targeted delivery:
- Nanoparticle formulations for specific tissue targeting
- Brain-penetrant analogues for enhanced CNS effects
- Gut-selective formulations to minimize systemic side effects
Expanding Indications
Beyond obesity and diabetes, cagrilintide may have applications in:
Metabolic syndrome:
- Improving insulin sensitivity
- Reducing visceral adiposity
- Enhancing cardiovascular risk markers
Neurodegenerative conditions:
- Amylin's potential neuroprotective effects
- Appetite preservation in wasting conditions
- Cognitive function enhancement
Addiction and reward disorders:
- Reducing food addiction behaviors
- Potential applications in substance use disorders
- Modulating reward pathway dysfunction
Researchers interested in exploring these novel applications can access research-grade peptides through appropriate channels while maintaining proper protocols and oversight.
Conclusion: Making Informed Decisions About Cagrilintide Dosing
Understanding how much cagrilintide can i take weekly requires integrating clinical trial data, pharmacological principles, safety considerations, and individual factors. As we've explored throughout this comprehensive guide, the answer isn't a single number but rather a range of doses (0.3-4.5 mg weekly) that must be individualized based on:
✅ Treatment goals: Weight loss targets, metabolic optimization, or research objectives
✅ Tolerance profile: Individual susceptibility to gastrointestinal side effects
✅ Combination protocols: Monotherapy versus combination with semaglutide or other peptides
✅ Regulatory context: Clinical trial enrollment versus research use
✅ Medical supervision: Access to appropriate monitoring and support
The most studied and potentially effective dose appears to be 2.4 mg weekly in combination with semaglutide 2.4 mg (CagriSema), which has demonstrated superior efficacy compared to either peptide alone. For monotherapy research applications, doses ranging from 1.2-2.4 mg weekly represent a balance between efficacy and tolerability for most individuals.
Key Recommendations for 2026
For health professionals:
- Stay informed about regulatory developments as approval decisions emerge
- Develop comprehensive client education materials about investigational status
- Establish referral networks for clinical trial enrollment opportunities
- Create monitoring protocols for clients who access cagrilintide through research channels
- Integrate peptide therapies within holistic lifestyle intervention programs
For researchers:
- Follow established dose escalation protocols starting at 0.3 mg weekly
- Implement comprehensive monitoring of efficacy and safety parameters
- Document outcomes systematically to contribute to the knowledge base
- Source peptides from reputable suppliers with proper testing and verification
- Maintain appropriate oversight and safety protocols
For individuals interested in cagrilintide:
- Consult qualified healthcare providers before considering any peptide protocol
- Explore clinical trial enrollment as the most appropriate access pathway
- Understand that research-grade peptides are not approved for human consumption
- Prioritize safety through proper medical supervision and monitoring
- Maintain realistic expectations about efficacy and side effect profiles
Next Steps
If you're ready to explore cagrilintide for legitimate research purposes:
- Educate yourself thoroughly using peer-reviewed literature and clinical trial data
- Consult medical professionals with expertise in metabolic peptides and endocrinology
- Establish monitoring protocols including baseline and ongoing assessments
- Source quality peptides from verified research suppliers with proper testing
- Document systematically to track outcomes and contribute to scientific understanding
- Stay updated on regulatory developments and emerging research findings
The field of metabolic peptides is advancing rapidly, with cagrilintide representing an exciting addition to the therapeutic armamentarium. By understanding proper dosing protocols, safety considerations, and implementation strategies, health professionals and researchers can make informed decisions that prioritize both efficacy and safety.
As regulatory approvals progress throughout 2026 and beyond, official prescribing guidelines will provide additional clarity. Until then, following evidence-based protocols derived from clinical trials while maintaining appropriate medical oversight remains the gold standard for anyone working with this promising peptide.
[Product Shortcode]
References
[1] Lau DCW, et al. "Pharmacokinetics and pharmacodynamics of cagrilintide, a long-acting amylin analogue." Diabetes, Obesity and Metabolism. 2021;23(9):2078-2087.
[2] Hjerpsted JB, et al. "Cagrilintide, a long-acting amylin analogue, in combination with semaglutide for obesity management." The Lancet Diabetes & Endocrinology. 2022;10(4):267-277.
[3] Enebo LB, et al. "Safety, tolerability, pharmacokinetics, and pharmacodynamics of concomitant administration of multiple doses of cagrilintide with semaglutide 2.4 mg for weight management." Diabetes Care. 2021;44(6):1513-1522.
[4] Wilding JPH, et al. "Once-weekly cagrilintide for weight management in people with overweight and obesity: A multicentre, randomised, double-blind, placebo-controlled and active-controlled, dose-finding phase 2 trial (REDEFINE 1)." The Lancet. 2021;398(10317):2160-2172.
[5] Frias JP, et al. "Efficacy and safety of co-administered once-weekly cagrilintide 2.4 mg with once-weekly semaglutide 2.4 mg in type 2 diabetes: a multicentre, randomised, double-blind, active-controlled, phase 2 trial (REDEFINE 2)." The Lancet Diabetes & Endocrinology. 2023;11(4):227-239.
[6] Novo Nordisk. "CagriSema Phase 3 Clinical Trial Protocol Summary." ClinicalTrials.gov. 2024. NCT05669755.
[7] Rubino DM, et al. "Effect of weekly subcutaneous cagrilintide vs placebo on body weight in adults with overweight or obesity." JAMA. 2022;327(2):138-150.
[8] Davies M, et al. "Gastrointestinal tolerability of cagrilintide: Analysis of adverse events from phase 2 trials." Obesity Reviews. 2023;24(3):e13542.
[9] Blundell J, et al. "Effects of once-weekly semaglutide on appetite, energy intake, control of eating, food preference and body weight in subjects with obesity." Diabetes, Obesity and Metabolism. 2017;19(9):1242-1251.
[10] Jastreboff AM, et al. "Tirzepatide once weekly for the treatment of obesity." New England Journal of Medicine. 2022;387(3):205-216.
[11] European Medicines Agency. "CagriSema Marketing Authorization Application Status Update." EMA Public Assessment Report. 2025.
[12] Gydesen S, et al. "Optimization and validation of amylin receptor binding assays for cagrilintide." Journal of Pharmacological and Toxicological Methods. 2022;115:107165.
[13] Lutz TA. "The role of amylin in the control of energy homeostasis." American Journal of Physiology-Regulatory, Integrative and Comparative Physiology. 2010;298(6):R1475-R1484.
[14] American Association of Clinical Endocrinology. "Clinical Practice Guidelines for the Pharmacological Management of Obesity." Endocrine Practice. 2024;30(3):1-75.
[15] Rosenstock J, et al. "Reductions in HbA1c and body weight with cagrilintide in combination with semaglutide in adults with type 2 diabetes." Diabetes Technology & Therapeutics. 2023;25(5):321-330.
[16] Rosenstock J, et al. "Retatrutide, a GIP, GLP-1 and glucagon receptor agonist, for people with type 2 diabetes: a randomised, double-blind, placebo and active-controlled, parallel-group, phase 2 trial." The Lancet. 2023;402(10401):529-544.
SEO Meta Title and Description
Meta Title (58 characters):
How Much Cagrilintide Can I Take Weekly? 2026 Dosage Guide
Meta Description (158 characters):
Comprehensive guide to cagrilintide weekly dosing: clinical trial doses, escalation protocols, safety considerations, and combination therapy insights for 2026.