The Complete Guide to Peptides in Canada: Regulations, Research, and Legal Considerations for 2026

The Canadian peptide landscape is experiencing unprecedented growth, with the market projected to reach new heights as research institutions and biotech companies drive innovation across the nation. Whether you're a fitness enthusiast exploring performance enhancement options, a longevity coach seeking evidence-based solutions, or an MMA athlete looking to optimize recovery, understanding the complex regulatory environment surrounding peptides in Canada is crucial for making informed decisions in 2026.
Key Takeaways
• Peptides in Canada are strictly regulated under the Food and Drugs Act, requiring Health Canada authorization for therapeutic use
• Research peptides are available for laboratory purposes only and must be clearly labeled as "not for human consumption"
• The Canadian peptide market is experiencing significant growth, driven by university research and biotech innovation
• Purchasing unapproved peptides for personal use carries legal risks and safety concerns
• Legitimate peptide access requires proper prescriptions from licensed healthcare providers or authorized research credentials
Understanding Peptide Regulations in Canada
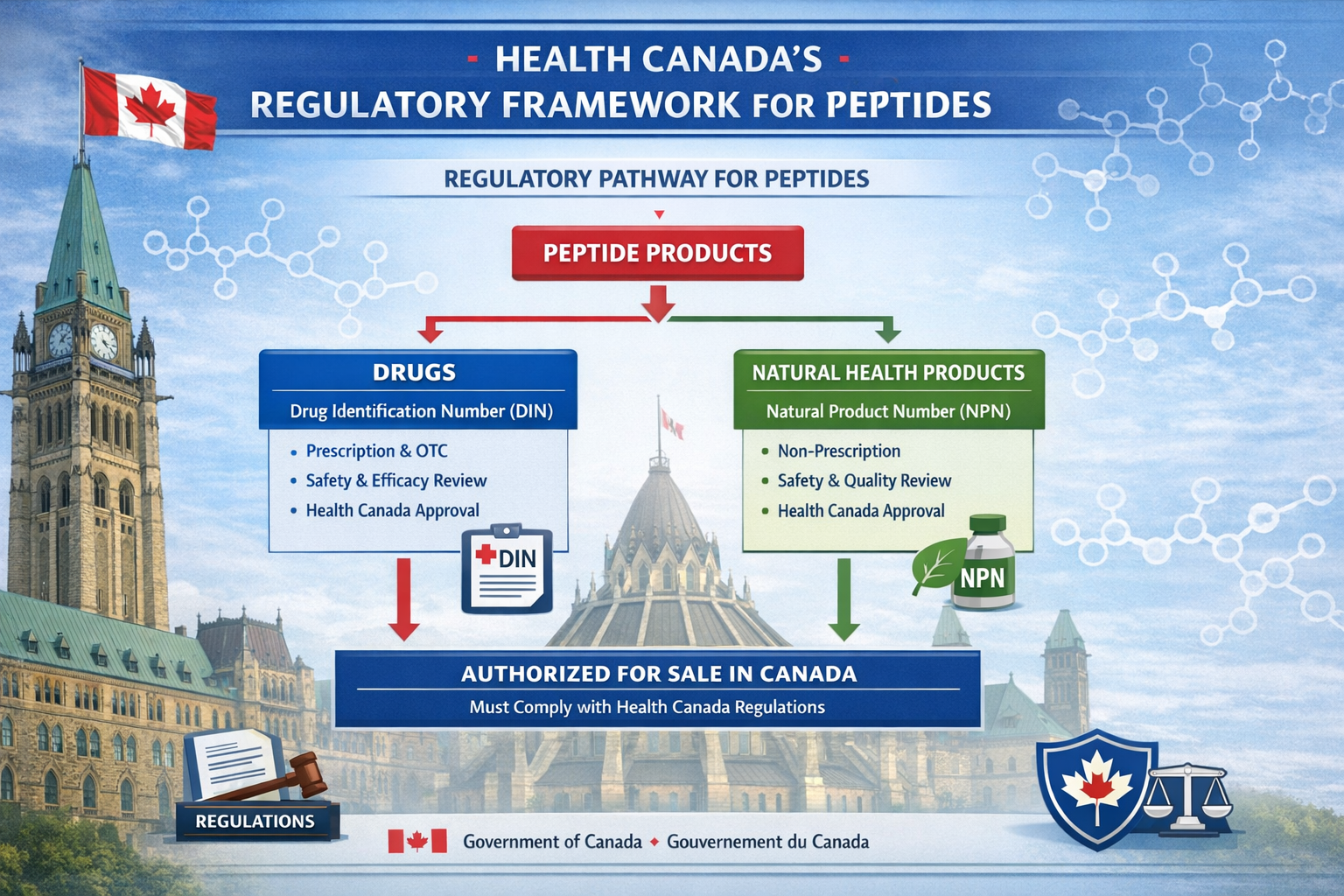
Health Canada's Regulatory Framework
Peptides in Canada fall under strict federal oversight through Health Canada's comprehensive regulatory system. The Food and Drugs Act governs all peptide products, classifying them based on their intended use, structure, and therapeutic claims [1]. This regulatory framework ensures that any peptide marketed for human consumption meets rigorous safety and efficacy standards.
The Natural and Non-prescription Health Products Directorate (NNHPD) oversees peptides intended for health purposes, while more complex therapeutic peptides require full drug approval processes. Understanding these distinctions is essential for anyone considering peptide research or therapeutic applications.
Key regulatory classifications include:
- Natural Product Number (NPN): For peptides with health claims but lower risk profiles
- Drug Identification Number (DIN): For therapeutic peptides requiring prescription access
- Research-only designation: For laboratory and scientific investigation purposes
Classification Systems and Requirements
Health Canada employs a sophisticated classification system that determines how different peptides are regulated and accessed. Synthetic peptides used for therapeutic purposes typically require extensive documentation and approval before legal sale in Canada.
The classification process considers several factors:
- Molecular structure and complexity
- Intended therapeutic applications
- Safety profile and potential risks
- Manufacturing standards and quality control
For fitness enthusiasts and athletes interested in performance enhancement, it's crucial to understand that most peptides marketed for these purposes fall under prescription drug regulations or are considered unapproved therapeutic products.
The Canadian Peptide Research Landscape
Leading Institutions and Innovation Centers
Canada's peptide research sector represents a cornerstone of the nation's biotechnology advancement, with several world-renowned institutions driving innovation. The University of Toronto and McGill University lead groundbreaking research in antimicrobial peptides, cancer therapeutics, and metabolic disorders as of 2026 [2].
These institutions benefit from substantial federal funding through the Canadian Institutes of Health Research (CIHR) and the Natural Sciences and Engineering Research Council (NSERC), supporting cutting-edge peptide-based therapeutic development.
Major research focus areas include:
🔬 Antimicrobial peptides for infection control
🎯 Cancer-targeting peptides for precision oncology
⚡ Metabolic peptides for diabetes and obesity treatment
🧬 Neuropeptides for neurological disorders
Research Peptide Availability and Compliance
For legitimate researchers and institutions, research peptides are available through authorized suppliers who must verify purchaser credentials and maintain strict compliance protocols. These suppliers are required to clearly label all products as "not for human consumption" and maintain detailed records of sales to qualified research entities.
The research peptide market in Canada operates under specific guidelines that ensure products are used exclusively for scientific investigation. This includes popular research compounds like 5-amino-1MQ and AOD 9604, which are available only to qualified researchers with appropriate institutional backing.
Legal Considerations and Safety Concerns for Peptides in Canada

Import Regulations and Customs Enforcement
Canadian customs and border services maintain active monitoring of peptide imports, with strict enforcement protocols for unauthorized products. Individuals attempting to import peptides in Canada for personal use without proper authorization face significant legal consequences under the Food and Drugs Act [3].
The Canada Border Services Agency (CBSA) has implemented sophisticated detection systems that identify peptide shipments, particularly those marketed for bodybuilding, anti-aging, or performance enhancement without Health Canada approval.
Common enforcement scenarios include:
- Package seizure at border crossings
- Legal notifications and warnings
- Potential criminal charges for large-scale importation
- Forfeiture of products and associated costs
Risks of Unauthorized Peptide Use
Health Canada has issued comprehensive warnings about the dangers of purchasing unapproved peptides online, highlighting serious concerns about product purity, contamination, incorrect dosing, and lack of quality control [4]. These risks are particularly relevant for fitness enthusiasts and athletes who may be tempted by unregulated online sources.
"The purchase and use of unauthorized peptides poses significant health risks, including unknown contaminants, incorrect concentrations, and potentially dangerous interactions with other substances." – Health Canada Advisory
Primary safety concerns include:
- Product contamination with harmful substances
- Incorrect dosing leading to adverse effects
- Unknown purity levels affecting efficacy and safety
- Lack of sterility in manufacturing processes
- Absence of quality control testing
For those interested in specific peptides like AOD 9604 for metabolic research, working with legitimate research institutions or obtaining proper medical supervision is essential.
Market Trends and Future Outlook for Peptides in Canada
Economic Growth and Market Projections
The Canadian peptide therapeutics market represents a significant portion of the global market, which is projected to reach USD 96.7 billion by 2032 [5]. This growth is driven by increasing research investments, biotech company expansion, and growing recognition of peptide therapeutic potential.
Key market drivers include:
- 📈 Increased research funding from government and private sources
- 🏢 Biotech company expansion in major Canadian cities
- 🎓 University-industry partnerships fostering innovation
- 💊 Growing therapeutic applications across multiple medical fields
Emerging Applications and Research Directions
Canadian researchers are exploring innovative peptide applications that extend far beyond traditional therapeutic uses. This includes advanced research into peptide blends and combination therapies that may offer enhanced benefits for various health conditions.
The longevity and wellness sectors are particularly interested in peptide research, with studies examining compounds like MOTS-C combinations for metabolic optimization and aging intervention.
Emerging research areas:
- Personalized peptide therapy based on genetic profiles
- Combination peptide treatments for complex conditions
- Oral peptide delivery systems for improved patient compliance
- Targeted peptide therapies for rare diseases
Regulatory Evolution and Future Changes
As the peptide industry continues to evolve, Health Canada is adapting its regulatory framework to accommodate new therapeutic applications while maintaining safety standards. This includes potential streamlined approval processes for certain peptide categories and enhanced oversight of research peptide distribution.
The regulatory landscape for peptides in Canada is expected to become more sophisticated, with clearer guidelines for different peptide categories and improved pathways for legitimate research and therapeutic access.
Legitimate Access Pathways and Professional Guidance

Prescription Peptides and Medical Supervision
For individuals seeking legitimate peptide therapy, several prescription options are available through licensed healthcare providers in Canada. These include approved medications like semaglutide (Ozempic, Wegovy) for diabetes and weight management, which require valid prescriptions and medical supervision [6].
Healthcare providers specializing in peptide therapy can assess individual needs and recommend appropriate treatments within the legal framework. This approach ensures safety, efficacy, and compliance with Canadian regulations.
Compounding Pharmacy Options
Compounding pharmacies in Canada may prepare peptide formulations under specific circumstances with valid prescriptions, but must comply with provincial pharmacy regulations and Health Canada's guidelines for compounded preparations [7]. This option provides access to customized peptide treatments while maintaining regulatory compliance.
Requirements for compounded peptides:
- Valid prescription from licensed physician
- Compliance with provincial pharmacy regulations
- Adherence to Health Canada compounding guidelines
- Quality assurance and documentation protocols
For researchers interested in specific compounds like 5-amino-1MQ research, working with accredited institutions and following proper protocols ensures legal compliance and research integrity.
Conclusion
The landscape of peptides in Canada in 2026 presents both exciting opportunities and significant regulatory challenges. While the Canadian peptide market continues to grow and evolve, strict federal oversight ensures that safety and efficacy remain paramount considerations.
For fitness enthusiasts, longevity coaches, and athletes, the key to navigating this complex environment lies in understanding the legal framework, working with qualified healthcare providers, and avoiding the temptations of unregulated online sources. The risks associated with unauthorized peptide use far outweigh any perceived benefits, making professional guidance essential.
Actionable next steps:
- Consult healthcare professionals for legitimate peptide therapy options
- Research institutional partnerships for scientific peptide investigation
- Stay informed about regulatory updates and approved treatments
- Avoid unauthorized sources that may pose legal and safety risks
- Consider approved alternatives that meet therapeutic goals safely
As Canada's peptide research sector continues to advance, new therapeutic options will likely become available through proper regulatory channels. By maintaining patience and working within the legal framework, individuals can access the benefits of peptide therapy while ensuring safety and compliance with Canadian law.
References

[1] Health Canada. (2025). Food and Drugs Act: Regulatory Framework for Therapeutic Products.
[2] Canadian Institutes of Health Research. (2025). Peptide Research Funding and Innovation Report.
[3] Canada Border Services Agency. (2025). Import Regulations for Pharmaceutical and Research Products.
[4] Health Canada. (2025). Advisory on Unauthorized Peptide Products and Associated Risks.
[5] Global Peptide Therapeutics Market Analysis. (2025). Market Research Report on Canadian Sector Growth.
[6] Health Canada. (2025). Approved Prescription Peptide Medications Database.
[7] National Association of Pharmacy Regulatory Authorities. (2025). Compounding Guidelines for Peptide Preparations.
SEO Meta Information
Meta Title: Peptides in Canada 2026: Complete Regulatory Guide & Legal Access
Meta Description: Comprehensive guide to peptides in Canada covering regulations, research, legal access, and safety. Essential information for fitness enthusiasts and researchers in 2026.
