
Triple Agonist Retatrutide: Revolutionary Breakthrough in Metabolic Health and Weight Management

Imagine a single compound that could revolutionize how we approach weight management, metabolic health, and even cardiovascular wellness. The triple agonist retatrutide represents exactly that breakthrough—a first-in-class therapeutic that's rewriting the rules of what's possible in metabolic medicine. With clinical trial results showing unprecedented weight loss of up to 28.7% and remarkable improvements across multiple health markers, this innovative peptide is capturing the attention of researchers, biohackers, and health optimization enthusiasts worldwide. 🚀
Key Takeaways
• Revolutionary Weight Loss: Triple agonist retatrutide achieved up to 28.7% average weight loss in clinical trials—the highest efficacy seen in obesity treatment to date
• Multi-Target Mechanism: Unlike existing therapies, this compound targets three distinct hormone receptors (GIP, GLP-1, and glucagon) simultaneously for comprehensive metabolic effects
• Cardiovascular Benefits: Beyond weight loss, retatrutide demonstrated significant improvements in blood pressure, cholesterol profiles, and inflammatory markers
• Promising Safety Profile: While side effects exist, discontinuation rates remain manageable, with most adverse events being gastrointestinal in nature
• Future Availability: FDA approval expected by 2027, with commercial availability projected for 2029 and potential sales reaching $15.6 billion by 2031
Understanding Triple Agonist Retatrutide: The Science Behind the Breakthrough
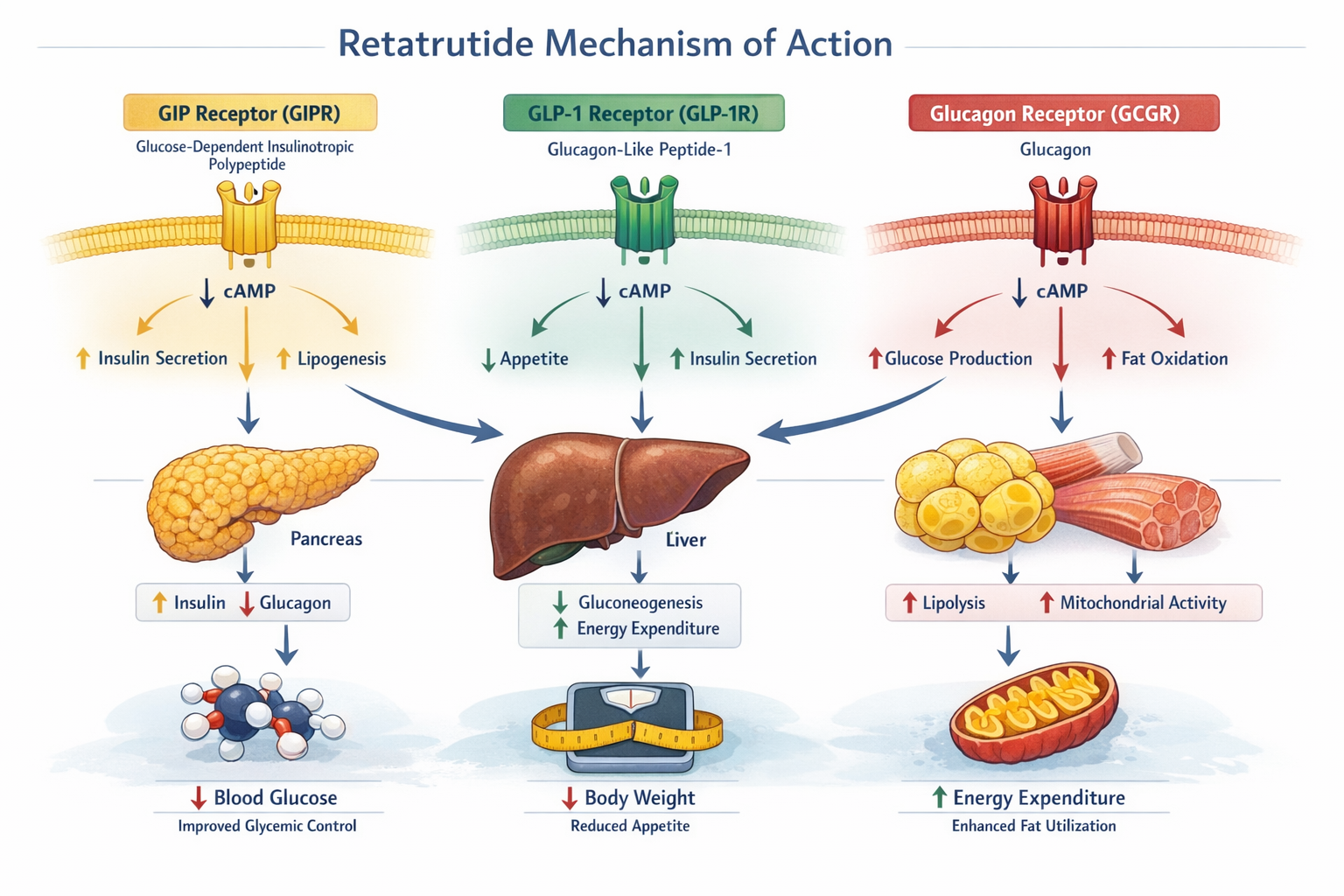
The triple agonist retatrutide represents a quantum leap in peptide-based therapeutics. Unlike traditional single or dual-target approaches, this innovative compound simultaneously activates three critical hormone receptors: glucose-dependent insulinotropic polypeptide (GIP), glucagon-like peptide-1 (GLP-1), and glucagon receptors.
The Molecular Mechanism 🧬
This unique triple-targeting approach creates a synergistic effect that goes far beyond what any single hormone agonist can achieve. The GLP-1 component enhances insulin secretion and slows gastric emptying, while the GIP receptor activation provides additional metabolic benefits. The glucagon receptor engagement adds a powerful fat-burning component that sets retatrutide apart from existing therapies.
Key Receptor Functions:
- GLP-1 Receptor: Controls appetite, slows digestion, enhances insulin sensitivity
- GIP Receptor: Regulates glucose homeostasis, supports insulin function
- Glucagon Receptor: Promotes fat oxidation, increases energy expenditure
The research community has been particularly excited about how these three pathways work together. When you explore incretin research themes, it becomes clear that this multi-target approach addresses the complexity of metabolic dysfunction more comprehensively than previous single-target therapies.
Comparative Advantages Over Existing Therapies
Traditional weight management peptides like semaglutide (Wegovy) and tirzepatide (Zepbound) have shown impressive results, but triple agonist retatrutide takes efficacy to an entirely new level. The addition of glucagon receptor activation provides enhanced lipolysis and energy expenditure that dual agonists simply cannot match.
For biohackers and researchers interested in most popular products for sale, understanding these mechanistic differences is crucial for making informed decisions about research protocols and optimization strategies.
Clinical Trial Results: Unprecedented Efficacy in Weight Management
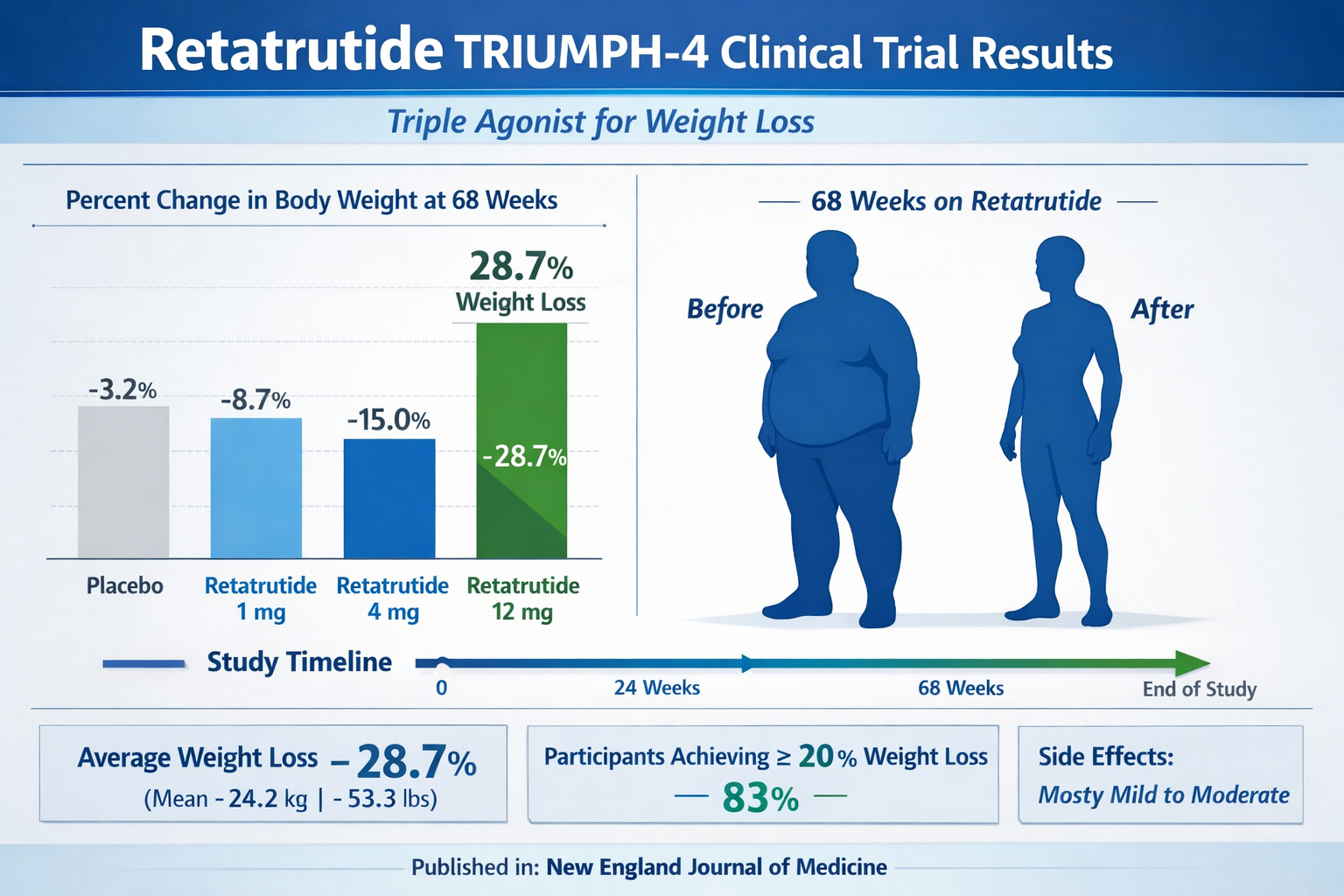
The TRIUMPH-4 clinical trial results have sent shockwaves through the medical and research communities. These findings represent the most significant advancement in obesity treatment efficacy ever recorded in a controlled clinical setting.
TRIUMPH-4 Trial: Breaking New Ground
The phase III TRIUMPH-4 trial enrolled participants with obesity and osteoarthritis of the knee, providing a unique opportunity to assess both metabolic and pain-related outcomes. The results exceeded even the most optimistic projections:
Weight Loss Achievements:
- 9mg dose: 26.4% average weight loss
- 12mg dose: 28.7% average weight loss
- Placebo group: 2.1% weight loss
- Absolute weight loss: Up to 71.2 pounds over 68 weeks [4]
These numbers represent a 26.6% weight loss on a placebo-adjusted basis [2], positioning triple agonist retatrutide as potentially the most effective obesity treatment ever developed.
Beyond Weight Loss: Comprehensive Health Improvements
What makes these results even more remarkable is the broad spectrum of health improvements observed:
Cardiovascular Benefits:
- Systolic blood pressure reduction: 14.0 mmHg at highest dose
- Significant reductions in non-HDL cholesterol
- Decreased triglyceride levels
- Lower high-sensitivity C-reactive protein (inflammation marker) [1]
Pain Management Outcomes:
- 4.5-point reduction in WOMAC pain scores (75.8% improvement)
- 12.0-14.1% of patients became completely pain-free
- Only 4.2% of placebo patients achieved pain-free status [1]
For those researching GLP3 triple agonist applications, these comprehensive benefits suggest potential applications far beyond weight management alone.
Safety Profile and Tolerability
While the efficacy results are groundbreaking, understanding the safety profile is equally important for researchers and biohackers considering this compound:
Common Side Effects:
- Nausea (most frequent)
- Diarrhea
- Constipation
- Decreased appetite
- Gastrointestinal discomfort
Discontinuation Rates:
- 12.2% discontinued on 9mg dose due to adverse events
- 18.2% discontinued on 12mg dose due to adverse events
- 4% discontinued on placebo [1]
Analysts note that discontinuation rates correlate with baseline BMI and the magnitude of weight loss achieved, suggesting that proper dosing strategies and patient selection may optimize tolerability.
Research Applications and Biohacking Potential
For the fitness and biohacking community, triple agonist retatrutide opens up fascinating research possibilities that extend far beyond traditional weight management applications.
Metabolic Optimization Protocols
The unique mechanism of action makes this compound particularly interesting for metabolic optimization research. Unlike traditional approaches that focus on single pathways, the triple agonist approach may provide insights into:
Enhanced Fat Oxidation Research:
- Glucagon receptor activation promotes increased lipolysis
- Potential for improved body composition even at maintenance calories
- Enhanced metabolic flexibility through multiple pathway activation
Insulin Sensitivity Studies:
- Dual incretin receptor activation may provide superior glucose control
- Potential applications in metabolic syndrome research
- Investigation of long-term metabolic health improvements
Performance and Recovery Applications
While weight loss dominates the headlines, researchers are exploring broader applications. The compound's effects on inflammation (demonstrated by C-reactive protein reductions) suggest potential benefits for recovery and performance optimization.
Research Areas of Interest:
- Post-exercise recovery enhancement
- Inflammatory response modulation
- Metabolic efficiency during training phases
- Body recomposition during cutting cycles
For those interested in comprehensive most popular products for sale, combining retatrutide research with other peptides like growth hormone releasing compounds could provide synergistic effects worth investigating.
Longevity and Healthspan Research
The cardiovascular and metabolic improvements observed in clinical trials align with key biomarkers of healthy aging. Researchers interested in longevity applications are particularly excited about:
Cardiovascular Health Markers:
- Blood pressure optimization
- Improved lipid profiles
- Reduced systemic inflammation
- Enhanced metabolic efficiency
Metabolic Health Indicators:
- Improved insulin sensitivity
- Better glucose control
- Enhanced fat metabolism
- Optimized hormone signaling
Future Outlook: What's Next for Triple Agonist Retatrutide
The future of triple agonist retatrutide looks incredibly promising, with multiple phase III trials currently underway and commercial availability on the horizon.
Upcoming Clinical Trials
Seven additional phase III trials are expected to report results throughout 2026, testing various dosing strategies and patient populations:
TRIUMPH-1 Trial Expectations:
- 80-week treatment duration (longer than TRIUMPH-4)
- Potential for over 30% weight loss achievement
- Testing of maintenance doses (4mg) alongside higher doses
- Broader patient population inclusion [1]
Additional Research Areas:
- Cardiovascular outcome studies
- Long-term safety assessments
- Combination therapy investigations
- Pediatric population studies
Regulatory Timeline and Availability
The regulatory pathway for triple agonist retatrutide is progressing rapidly:
Key Milestones:
- 2026: Additional phase III trial results
- 2027: Expected FDA approval [1]
- 2029: Projected commercial availability [1]
- 2031: Estimated $15.6 billion in global sales [1]
Market Impact and Accessibility
The commercial success projections reflect the revolutionary potential of this therapy. However, for researchers and biohackers, the timeline to availability means continued reliance on research-grade compounds and careful protocol development.
Market Considerations:
- High demand expected upon approval
- Potential supply constraints initially
- Research applications likely to continue growing
- Integration with existing peptide protocols
For those planning research protocols, exploring current most popular products for sale can help establish baseline approaches while awaiting broader availability.
Integration with Existing Research
The success of triple agonist retatrutide is likely to accelerate research into other multi-target approaches. Researchers are already investigating:
Combination Protocols:
- Integration with growth hormone peptides
- Synergistic effects with other metabolic compounds
- Optimized dosing schedules for different research goals
- Personalized approaches based on individual response patterns
Practical Considerations for Researchers
For biohackers and researchers interested in exploring triple agonist retatrutide, several practical considerations are essential for successful research protocols.
Research Protocol Development
Dosing Considerations:
- Clinical trials used escalating dose protocols
- 9mg and 12mg showed optimal efficacy
- Lower maintenance doses (4mg) under investigation
- Individual response variation requires careful monitoring
Timeline Planning:
- Clinical benefits observed over 68+ weeks
- Significant effects typically seen by 12-16 weeks
- Long-term protocols may be necessary for full assessment
- Regular monitoring essential throughout research period
Quality and Sourcing Considerations
Given the complexity of this triple agonist compound, research quality becomes paramount. The sophisticated mechanism requires precise manufacturing and handling:
Key Quality Factors:
- Proper peptide synthesis and purification
- Appropriate storage and handling protocols
- Third-party testing verification
- Consistent potency across research batches
For researchers prioritizing quality, exploring most popular products for sale from established suppliers ensures access to properly manufactured compounds.
Monitoring and Assessment
Essential Tracking Metrics:
- Body composition changes (not just weight)
- Metabolic markers (glucose, insulin, lipids)
- Cardiovascular parameters (blood pressure, heart rate)
- Subjective measures (appetite, energy, well-being)
- Potential side effects and tolerability
Conclusion

The emergence of triple agonist retatrutide represents a paradigm shift in metabolic health and weight management research. With unprecedented clinical trial results showing up to 28.7% weight loss, significant cardiovascular improvements, and a manageable safety profile, this compound is poised to revolutionize how we approach metabolic optimization.
For the biohacking and research community, retatrutide offers exciting possibilities that extend far beyond traditional weight management. Its unique triple-target mechanism provides a comprehensive approach to metabolic health that aligns perfectly with the goals of optimization-focused researchers.
Next Steps for Researchers
Immediate Actions:
- Stay Informed: Monitor upcoming TRIUMPH trial results throughout 2026
- Protocol Planning: Develop comprehensive research protocols incorporating proper monitoring
- Quality Sourcing: Identify reliable suppliers for research-grade compounds
- Community Engagement: Connect with other researchers exploring similar protocols
Long-term Considerations:
- Regulatory Monitoring: Track FDA approval progress and commercial availability
- Combination Research: Explore synergistic effects with other optimization compounds
- Personalization: Develop individualized approaches based on emerging research
- Safety Protocols: Maintain rigorous safety monitoring throughout research
The future of metabolic optimization has arrived with triple agonist retatrutide. For researchers ready to explore this groundbreaking compound, the opportunity to contribute to our understanding of advanced metabolic therapeutics has never been more exciting. As we await broader availability, the research community continues to push the boundaries of what's possible in human optimization and metabolic health.
References
[1] Lilly Retatrutide Data Phase Iii Trial – https://www.clinicaltrialsarena.com/news/lilly-retatrutide-data-phase-iii-trial/
[2] Roches 22 Weight Loss Benefit Seals Glp 1s Phase Iii Plans Boosts Zealand Combo – https://www.biospace.com/drug-development/roches-22-weight-loss-benefit-seals-glp-1s-phase-iii-plans-boosts-zealand-combo
[3] Retatrutide Approval Status – https://www.peptidestack.io/blog/retatrutide-approval-status
[4] New Weight Loss Drugs – https://www.goodrx.com/conditions/weight-loss/new-weight-loss-drugs
SEO Meta Information
Meta Title: Triple Agonist Retatrutide: Revolutionary Weight Loss Breakthrough 2026
Meta Description: Discover triple agonist retatrutide's groundbreaking 28.7% weight loss results, unique mechanism targeting GIP, GLP-1, and glucagon receptors. Research insights included.
{“@context”:”https://schema.org”,”@type”:”Article”,”headline”:”triple agonist retatrutide”,”description”:”Discover triple agonist retatrutide’s groundbreaking 28.7% weight loss results, unique mechanism targeting GIP, GLP-1, and glucagon receptors. Research insights”,”image”:”https://zsxkvszxbhpwnvzxdydv.supabase.co/storage/v1/object/public/generated-images/kie/d87f8482-47ae-4f9e-a061-8f443051258a/slot-0-1770266885694.png”,”datePublished”:”2026-02-05T04:45:40.541764+00:00″,”dateModified”:”2026-02-05T04:48:50.487Z”,”author”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″},”publisher”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″}}
