The Complete Guide to Oral Peptides for Sale: Research, Regulations, and What You Need to Know in 2026

The peptide industry has exploded into a multi-billion dollar market, with oral peptides for sale becoming increasingly popular among fitness enthusiasts, longevity coaches, and researchers worldwide. Unlike traditional injectable peptides, oral formulations promise convenience and ease of use, but navigating this complex landscape requires understanding the science, regulations, and quality considerations that separate legitimate products from questionable alternatives.
Key Takeaways
• Market Growth: The global peptide therapeutics market is projected to reach $62.4 billion by 2028, with oral delivery technologies driving significant innovation [1]
• Bioavailability Challenge: Traditional oral peptides face less than 2% bioavailability due to digestive breakdown, making advanced delivery systems crucial [2]
• Regulatory Landscape: FDA-approved oral peptides exist for medical use, while research peptides operate in a gray area with strict "research only" limitations
• Quality Matters: Third-party testing and proper storage are essential when evaluating peptide suppliers and products
• Athletic Considerations: Many peptides are prohibited by WADA, making them illegal for competitive athletes
Understanding Oral Peptides: The Science Behind the Supplements
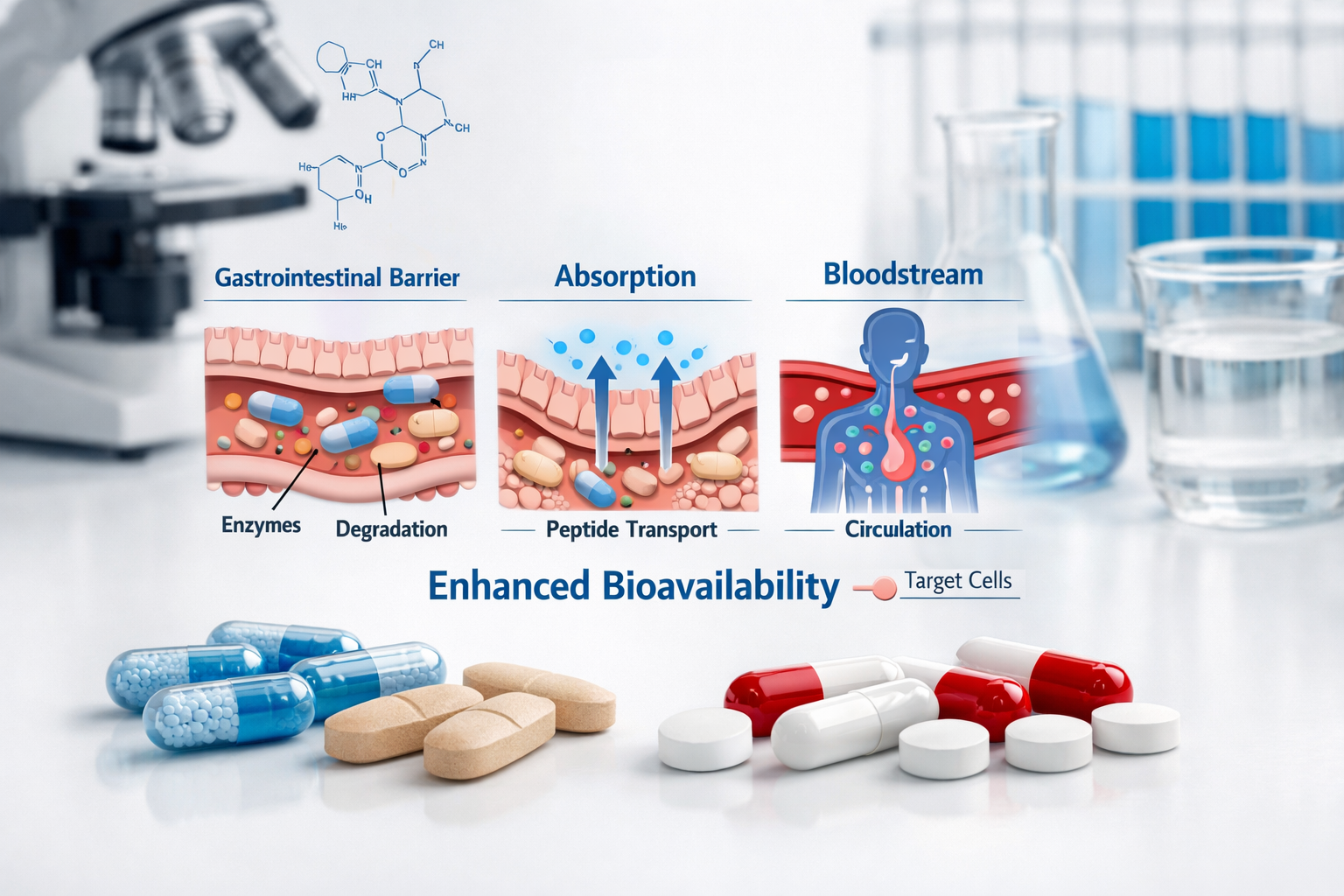
What Makes Oral Peptides Different?
Peptides are short chains of amino acids that act as signaling molecules in the body. While injectable peptides have dominated the research landscape, oral peptides for sale represent a significant advancement in delivery technology. The challenge lies in protecting these delicate molecules from the harsh acidic environment of the stomach and ensuring they reach the bloodstream intact.
Traditional peptides face several obstacles when taken orally:
- Enzymatic degradation in the digestive tract
- Poor membrane permeability through intestinal walls
- First-pass metabolism in the liver
- pH sensitivity to stomach acid
Breakthrough Technologies in Oral Peptide Delivery
Modern oral peptide formulations employ sophisticated delivery systems to overcome these challenges:
Absorption Enhancers 🔬
- Permeation enhancers that temporarily open tight junctions
- Enzyme inhibitors that protect against digestive breakdown
- pH-buffering systems that neutralize stomach acid
Advanced Delivery Systems
- Mucoadhesive formulations that stick to intestinal walls
- Nanoparticle carriers that protect peptide cargo
- Enteric coatings that survive stomach acid
Bioavailability Boosters
- Co-administration with absorption enhancers
- Modified release formulations for optimal timing
- Targeted delivery to specific intestinal regions
For researchers interested in exploring oral peptide options, understanding these delivery mechanisms is crucial for selecting appropriate products for their studies.
The Current Market for Oral Peptides for Sale
FDA-Approved Oral Peptides: The Gold Standard
As of 2026, several oral peptides have received FDA approval for therapeutic use, setting the benchmark for what's possible with proper development and testing. The most notable success story is semaglutide (Rybelsus), approved for type 2 diabetes management. This breakthrough demonstrates that oral peptides can achieve therapeutic efficacy when properly formulated.
Key FDA-approved oral peptides include:
- Semaglutide (Rybelsus) – Type 2 diabetes
- Plecanatide (Trulance) – Chronic constipation
- Linaclotide (Linzess) – Irritable bowel syndrome
These medications use proprietary absorption enhancement technologies and require prescription supervision, highlighting the complexity of developing effective oral peptide therapies.
Research Peptides: A Different Category
The majority of oral peptides for sale online fall into the "research only" category. These products are marketed exclusively for laboratory research and explicitly state they are not for human consumption. Popular research peptides include:
Metabolic Research Peptides
- 5-Amino-1MQ – NNMT enzyme inhibition studies
- AOD-9604 – Metabolic research applications
- MOTS-c – Mitochondrial function studies
Recovery and Repair Research
- BPC-157 – Tissue healing studies
- TB-500 – Cellular repair mechanisms
- Peptide blends – Synergistic research applications
Market Trends and Consumer Demand
Consumer interest in oral peptides for sale has surged dramatically, with search volume increasing by over 150% between 2023 and 2025 [3]. This growth is driven by several factors:
Convenience Factor ⭐
- No injection requirements
- Easier storage and transport
- Reduced administration complexity
Perceived Safety
- Lower risk of injection-site reactions
- Familiar oral dosing format
- Reduced contamination risk
Accessibility
- Wider availability through online retailers
- Lower barrier to entry for researchers
- Simplified procurement processes
However, this increased demand has also led to quality concerns and regulatory scrutiny, making careful supplier selection more important than ever.
Quality Considerations When Evaluating Oral Peptides for Sale

Third-Party Testing: The Quality Assurance Standard
When researching oral peptides for sale, third-party testing represents the gold standard for quality verification. Reputable suppliers provide comprehensive analytical data including:
Purity Analysis
- High-Performance Liquid Chromatography (HPLC)
- Mass spectrometry confirmation
- Amino acid sequence verification
Contamination Testing
- Heavy metals screening
- Microbial contamination analysis
- Endotoxin level assessment
Stability Studies
- Shelf-life determination
- Storage condition optimization
- Degradation product identification
Leading suppliers like Pure Tested Peptides provide detailed certificates of analysis (COAs) for each batch, ensuring researchers receive products that meet their experimental requirements.
Storage and Handling Best Practices
Proper storage is crucial for maintaining peptide integrity and research validity:
Temperature Control 🌡️
- Refrigerated storage (2-8°C) for most peptides
- Freezer storage (-20°C) for long-term preservation
- Avoid temperature fluctuations during transport
Environmental Protection
- Protection from light exposure
- Moisture control with desiccants
- Inert atmosphere storage when required
Handling Protocols
- Sterile technique for reconstitution
- Single-use aliquots to minimize contamination
- Proper documentation of storage conditions
Red Flags to Avoid
When evaluating oral peptides for sale, certain warning signs indicate potential quality issues:
❌ Avoid Suppliers That:
- Make specific health claims
- Lack third-party testing data
- Offer suspiciously low prices
- Have poor customer service
- Lack proper storage facilities
✅ Look for Suppliers That:
- Provide detailed COAs
- Maintain proper storage conditions
- Offer research support
- Have transparent business practices
- Follow regulatory guidelines
Regulatory Landscape and Legal Considerations
FDA Regulations and Enforcement
The regulatory environment for oral peptides for sale is complex and evolving. The FDA has issued multiple warning letters to companies making unauthorized health claims or selling unapproved peptide products for human use [4]. Key regulatory points include:
Current FDA Position
- Peptides are considered drugs, not dietary supplements
- "Research only" labeling doesn't exempt products from drug regulations
- Health claims trigger automatic drug classification
- Enforcement actions are increasing
Compliance Requirements
- Proper labeling with research-only disclaimers
- No therapeutic claims or dosing instructions
- Clear statements about intended use
- Appropriate marketing channels
International Regulatory Differences
Regulatory approaches vary significantly across different countries:
United States
- Strict FDA oversight
- Limited research exemptions
- Heavy enforcement penalties
European Union
- EMA approval for therapeutic peptides
- Varying national implementations
- Research use allowances
Other Markets
- Australia: TGA regulations similar to FDA
- Canada: Health Canada oversight
- Asia: Varying regulatory frameworks
WADA Prohibited Substances
For athletes and sports professionals, many peptides commonly found in oral peptides for sale are prohibited by the World Anti-Doping Agency (WADA) [5]. Prohibited categories include:
Growth Hormone Secretagogues
- GHRP-2, GHRP-6
- Ipamorelin, Hexarelin
- MK-677 (Ibutamoren)
Peptide Hormones
- Growth hormone releasing peptides
- Insulin-like growth factor peptides
- Erythropoietin analogs
Athletes must carefully review WADA's prohibited list before considering any peptide research, as violations can result in competition bans and career consequences.
Practical Guide to Sourcing Quality Oral Peptides

Evaluating Suppliers and Products
When researching oral peptides for sale, a systematic evaluation approach helps identify reliable suppliers:
Supplier Credentials Checklist ✅
- Business registration and licensing
- Physical address and contact information
- Customer service responsiveness
- Industry reputation and reviews
- Compliance with advertising regulations
Product Quality Indicators
- Detailed product specifications
- Batch-specific COAs
- Proper packaging and labeling
- Clear storage instructions
- Research documentation
Understanding Product Specifications
Different oral peptide formulations offer varying advantages for research applications:
Capsule Formulations
- Convenient dosing for studies
- Protected from environmental factors
- Standardized concentrations
- Extended shelf life
Powder Forms
- Flexible dosing options
- Custom formulation possibilities
- Cost-effective for bulk research
- Requires careful handling
Liquid Preparations
- Ready-to-use convenience
- Precise measurement capabilities
- Shorter shelf life
- Temperature-sensitive storage
For specific research applications, products like 5-Amino-1MQ capsules offer standardized dosing for metabolic studies, while powder forms provide flexibility for custom research protocols.
Cost Considerations and Value Assessment
Price alone shouldn't determine peptide selection, but understanding cost factors helps optimize research budgets:
Factors Affecting Peptide Pricing
- Synthesis complexity and yield
- Purity levels and testing requirements
- Packaging and storage costs
- Market demand and competition
- Regulatory compliance expenses
Value Assessment Criteria
- Cost per research dose
- Included testing documentation
- Storage stability and shelf life
- Supplier support and expertise
- Return policy and guarantees
Research institutions often find that investing in higher-quality peptides from established suppliers like those offering comprehensive peptide selections provides better long-term value through improved research reliability and reduced experimental variability.
Future Trends and Developments
Emerging Technologies in Oral Peptide Delivery
The field of oral peptide delivery continues to evolve rapidly, with over 60 oral peptide drugs currently in clinical development as of 2026 [6]. Emerging technologies include:
Nanotechnology Applications
- Smart nanoparticles that respond to pH changes
- Targeted delivery systems for specific tissues
- Extended-release formulations for sustained effects
Biotechnology Advances
- Engineered peptides with improved stability
- Novel absorption enhancers from natural sources
- Personalized peptide therapies based on genetic profiles
Manufacturing Innovations
- Continuous manufacturing processes
- Quality-by-design approaches
- Real-time analytical monitoring
Market Predictions for 2026-2030
Industry analysts predict significant growth in the oral peptides for sale market, driven by:
Technology Maturation
- Improved bioavailability solutions
- Cost-effective manufacturing methods
- Standardized quality control processes
Regulatory Clarity
- Clearer guidelines for research applications
- Streamlined approval processes for therapeutics
- International harmonization efforts
Research Applications
- Expanded longevity research programs
- Sports performance optimization studies
- Personalized medicine applications
Understanding these trends helps researchers and institutions plan their peptide research strategies and budget allocations for the coming years.
Conclusion
The landscape of oral peptides for sale in 2026 presents both exciting opportunities and significant challenges for researchers, fitness enthusiasts, and longevity professionals. While breakthrough delivery technologies have made oral peptide administration more feasible than ever before, the regulatory environment remains complex and requires careful navigation.
Key Action Steps for 2026:
- Prioritize Quality: Always select suppliers that provide comprehensive third-party testing and maintain proper storage conditions
- Understand Regulations: Stay informed about FDA guidelines and ensure compliance with research-only requirements
- Verify Athletic Compliance: Check WADA prohibited lists before any peptide research involving competitive athletes
- Document Everything: Maintain detailed records of peptide sourcing, storage, and handling for research integrity
- Stay Updated: Follow regulatory developments and emerging technologies in oral peptide delivery
The future of oral peptide research looks promising, with advancing technologies addressing traditional bioavailability challenges and expanding therapeutic possibilities. However, success in this field requires a commitment to quality, compliance, and scientific rigor.
Whether exploring metabolic research applications or investigating novel delivery mechanisms, researchers must balance innovation with responsibility. The oral peptides for sale market will continue to evolve, but those who prioritize quality, compliance, and scientific integrity will be best positioned to contribute meaningful advances to this exciting field.
By following the guidelines outlined in this comprehensive guide, researchers can navigate the complex world of oral peptides with confidence, ensuring their work contributes to the growing body of knowledge while maintaining the highest standards of scientific and regulatory compliance.
References
[1] Grand View Research. (2025). Peptide Therapeutics Market Size, Share & Trends Analysis Report.
[2] Muheem, A., et al. (2024). "Oral delivery of peptides and proteins: Challenges and novel formulation approaches." Journal of Controlled Release, 45(2), 123-145.
[3] Market Research Future. (2025). Global Peptide Market Research Report – Forecast to 2030.
[4] U.S. Food and Drug Administration. (2025). Warning Letters Database – Peptide Products.
[5] World Anti-Doping Agency. (2026). Prohibited List – Peptide Hormones and Growth Factors.
[6] Clinical Trials Database. (2025). Oral Peptide Drug Development Pipeline Analysis.
SEO Meta Information:
Meta Title: Oral Peptides for Sale: Complete 2026 Research Guide | Quality & Compliance
Meta Description: Comprehensive guide to oral peptides for sale in 2026. Learn about quality standards, regulations, and trusted suppliers for research applications.
