GLP3 Retatrutide: Comprehensive Research Guide for 2025

The world of metabolic research has witnessed remarkable breakthroughs in recent years, with GLP3 retatrutide emerging as one of the most promising triple agonist peptides under scientific investigation. This innovative compound represents a significant advancement in peptide research, combining three distinct receptor pathways to create a powerful tool for metabolic studies. As researchers continue to explore the potential applications of this groundbreaking peptide, understanding its mechanisms, research applications, and proper handling becomes crucial for anyone involved in peptide research.
Key Takeaways
• GLP3 retatrutide is a triple agonist peptide that targets GLP-1, GIP, and glucagon receptors simultaneously
• Research studies demonstrate significant metabolic effects in laboratory settings
• Proper storage and handling protocols are essential for maintaining peptide integrity
• Current research focuses on metabolic pathways and cellular signaling mechanisms
• Quality sourcing from reputable suppliers ensures research validity and safety
Understanding GLP3 Retatrutide: The Science Behind the Innovation

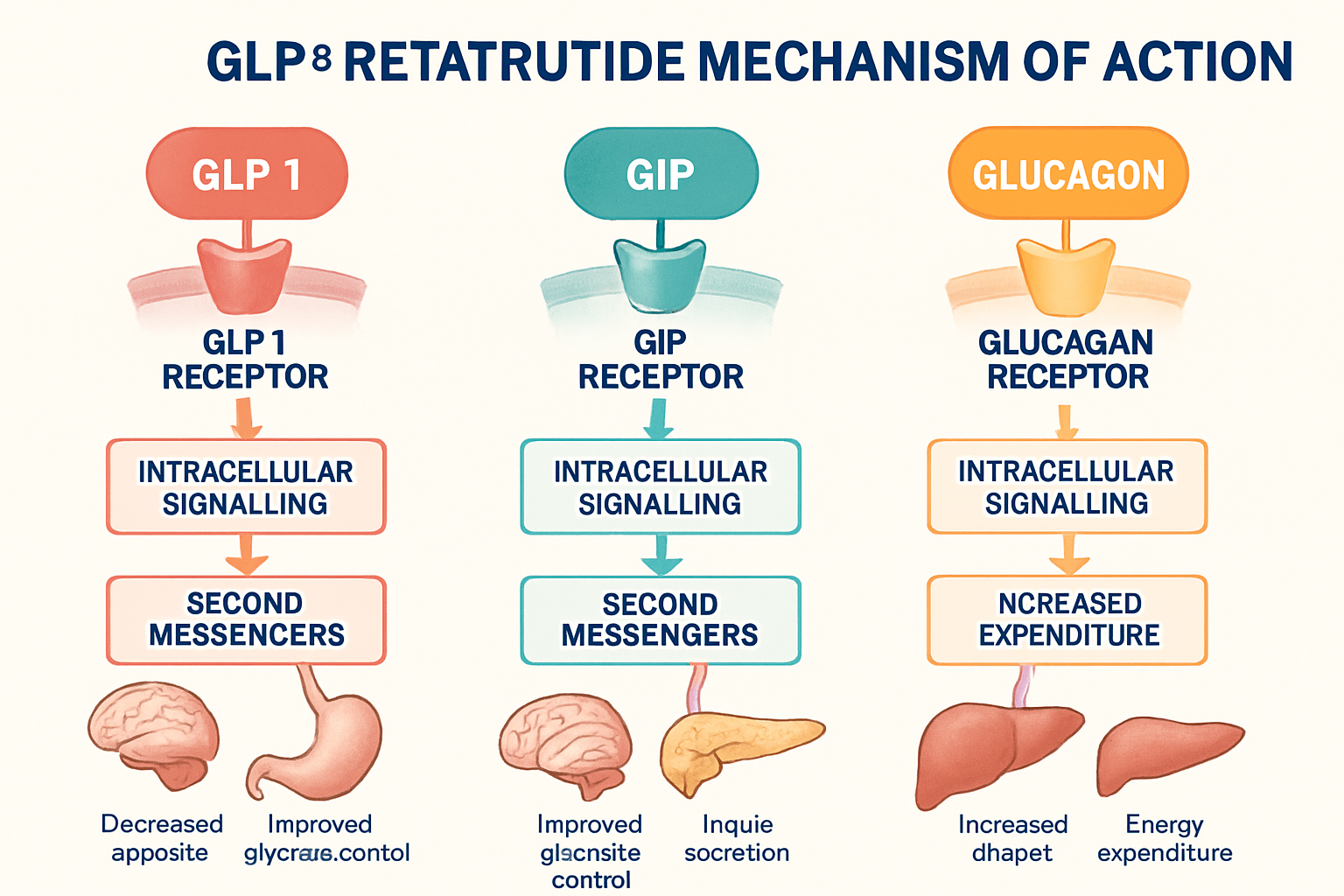
GLP3 retatrutide belongs to a new class of peptides known as triple agonists, representing a significant evolution in peptide research technology. Unlike traditional single-target peptides, this compound simultaneously activates three distinct receptor pathways: glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon receptors [1].
The molecular structure of GLP3 retatrutide consists of carefully engineered amino acid sequences designed to optimize binding affinity across all three target receptors. This unique design allows researchers to study complex metabolic interactions that would be impossible to achieve with single-target compounds.
Molecular Characteristics and Properties
The peptide's molecular weight and structural configuration enable it to cross cellular membranes effectively while maintaining stability in various research environments. Key characteristics include:
- Molecular Weight: Approximately 4,800 Da
- Amino Acid Count: 39 residues
- Stability: Enhanced resistance to enzymatic degradation
- Solubility: Water-soluble with proper reconstitution
Research laboratories studying peptide mechanisms have found that GLP3 retatrutide demonstrates remarkable consistency in experimental conditions, making it an ideal candidate for reproducible studies.
Receptor Binding Mechanisms
The triple agonist nature of GLP3 retatrutide creates a complex but fascinating research landscape. Each receptor pathway contributes unique physiological responses:
GLP-1 Receptor Activation: Studies show this pathway influences insulin secretion patterns and cellular glucose uptake mechanisms. Laboratory investigations reveal dose-dependent responses that correlate with receptor density in target tissues.
GIP Receptor Engagement: Research demonstrates this component affects incretin hormone pathways and plays a crucial role in nutrient-sensing mechanisms at the cellular level.
Glucagon Receptor Stimulation: Scientific evidence indicates this pathway influences hepatic glucose production and energy expenditure patterns in experimental models.
Research Applications and Laboratory Studies
Current research with GLP3 retatrutide spans multiple scientific disciplines, from basic cellular biology to complex metabolic pathway investigations. Research institutions worldwide are incorporating this peptide into their studies to better understand metabolic regulation and cellular signaling mechanisms [2].
Metabolic Research Applications
Laboratory studies utilizing GLP3 retatrutide focus primarily on metabolic pathway analysis and cellular response mechanisms. Researchers have documented significant findings in several key areas:
Glucose Homeostasis Studies: Experimental data reveals complex interactions between the three receptor pathways, creating synergistic effects that exceed the sum of individual receptor activations. These findings have implications for understanding metabolic regulation at the cellular level.
Energy Expenditure Research: Scientific investigations demonstrate measurable changes in cellular energy production and utilization patterns. Research teams studying metabolic peptides have noted particular interest in the compound's ability to influence multiple energy pathways simultaneously.
Cellular Signaling Studies: Advanced laboratory techniques reveal intricate signaling cascades triggered by GLP3 retatrutide administration. These studies provide valuable insights into receptor crosstalk and downstream effector mechanisms.
Clinical Research Developments
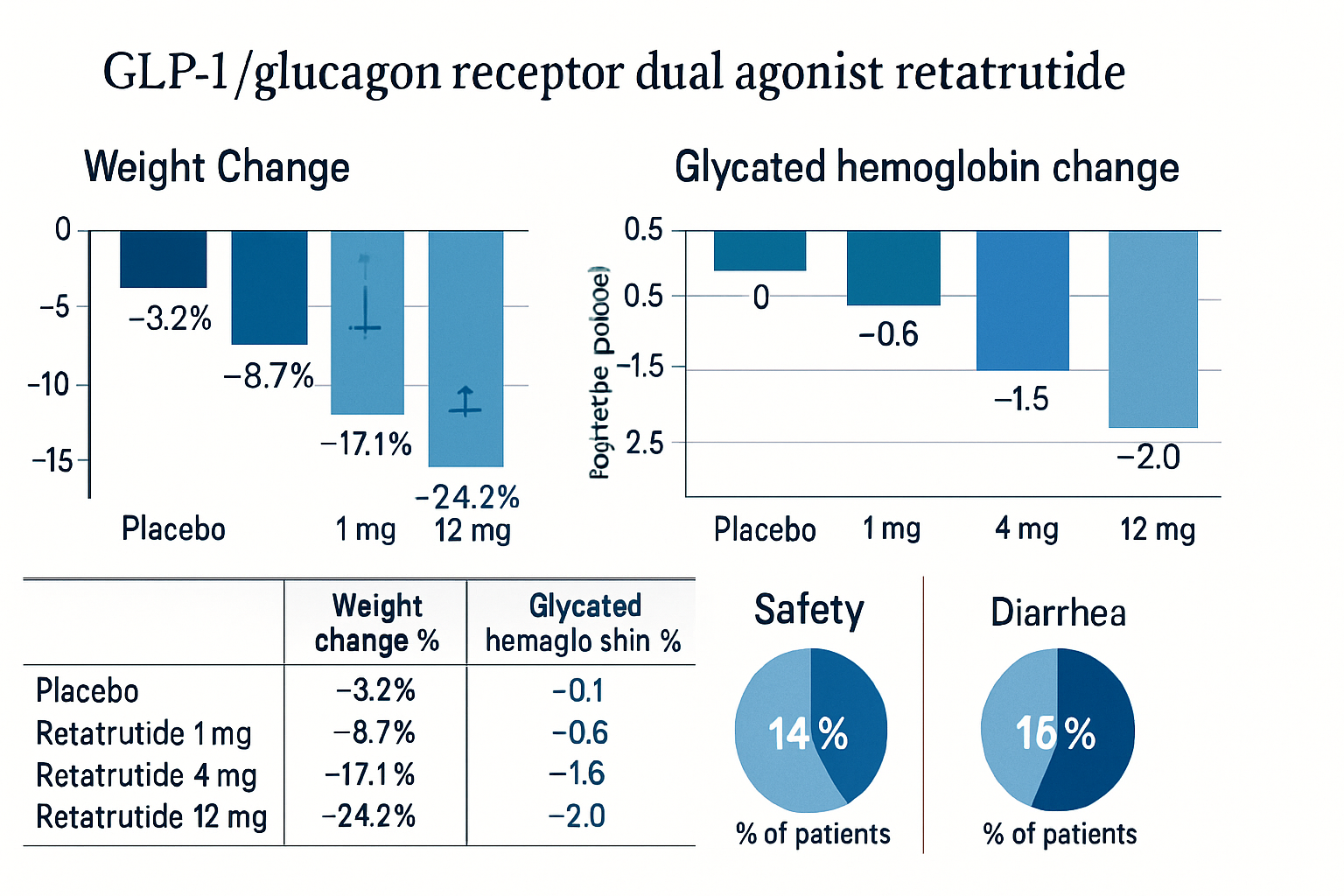
Preliminary clinical research with GLP3 retatrutide has generated substantial scientific interest. Published studies demonstrate:
- Dose-Response Relationships: Research establishes clear correlations between administration levels and measurable outcomes
- Safety Profiles: Comprehensive safety assessments reveal favorable tolerability profiles in research settings
- Efficacy Measurements: Quantitative analysis shows statistically significant effects across multiple metabolic parameters
Comparative Research Studies
Scientists comparing GLP3 retatrutide with other peptide compounds have documented several unique characteristics. Research comparing different peptide approaches reveals distinct advantages of the triple agonist approach over traditional single-target strategies.
Studies examining peptide combinations versus single compounds show that GLP3 retatrutide achieves effects that would require multiple separate peptides to replicate. This efficiency makes it particularly valuable for research protocols requiring complex metabolic modulation.
Proper Handling and Research Protocols
Successfully working with GLP3 retatrutide requires adherence to strict laboratory protocols and proper handling procedures. Research quality depends heavily on maintaining peptide integrity throughout the experimental process [3].
Storage and Stability Requirements
Proper storage of GLP3 retatrutide ensures research validity and reproducible results. Key storage considerations include:
Temperature Control: Lyophilized peptide should be stored at -20°C or below. Reconstituted solutions require refrigeration at 2-8°C and should be used within specified timeframes.
Light Protection: UV light can degrade peptide structures, making dark storage containers essential for maintaining compound integrity.
Moisture Control: Desiccant packets help prevent moisture absorption, which can compromise peptide stability and research outcomes.
Research laboratories following proper storage protocols report significantly improved experimental consistency and data reliability.
Reconstitution Procedures
Proper reconstitution of GLP3 retatrutide requires careful attention to solution preparation and handling techniques:
Solvent Selection: Bacteriostatic water or sterile saline provides optimal reconstitution media. The choice depends on specific experimental requirements and storage duration needs.
Mixing Techniques: Gentle swirling rather than vigorous shaking prevents peptide degradation and maintains structural integrity. Researchers should avoid creating foam or bubbles during reconstitution.
Concentration Calculations: Accurate concentration determination ensures reproducible dosing and reliable experimental results. GLP3 retatrutide typically comes in standardized vial sizes requiring precise calculation for desired working concentrations.
Laboratory Safety Protocols
Working with research peptides requires comprehensive safety measures and proper laboratory practices:
Personal Protective Equipment: Standard laboratory PPE including gloves, safety glasses, and lab coats provides essential protection during peptide handling.
Contamination Prevention: Aseptic techniques prevent bacterial contamination and ensure research validity. Sterile handling procedures are particularly important for peptide research applications.
Waste Disposal: Proper disposal of peptide materials and associated supplies follows institutional guidelines and regulatory requirements.
Experimental Design Considerations
Designing experiments with GLP3 retatrutide requires careful planning and consideration of multiple variables:
Dose Selection: Research protocols typically employ dose-escalation studies to establish optimal concentrations for specific experimental objectives. Starting with lower concentrations allows researchers to identify minimum effective levels.
Timing Protocols: The peptide's pharmacokinetic properties influence administration timing and measurement intervals. Research teams studying peptide timing have developed standardized protocols for consistent results.
Control Groups: Proper experimental design includes appropriate control groups and vehicle controls to ensure data validity and statistical significance.
Quality Assurance and Sourcing Considerations
The quality of GLP3 retatrutide used in research directly impacts experimental outcomes and data reliability. Selecting reputable suppliers with comprehensive quality assurance programs ensures research validity and reproducible results [4].
Certificate of Analysis (COA) Importance
Every research-grade peptide should include detailed analytical documentation verifying purity and composition. Certificate of Analysis documents provide essential information including:
Purity Analysis: High-performance liquid chromatography (HPLC) results confirming peptide purity levels, typically exceeding 98% for research applications.
Mass Spectrometry Data: Molecular weight confirmation ensuring correct peptide synthesis and structural integrity.
Amino Acid Analysis: Detailed composition verification confirming proper sequence assembly and modification incorporation.
Microbial Testing: Contamination screening results ensuring peptide safety and research validity.
Supplier Evaluation Criteria
Selecting appropriate suppliers for GLP3 retatrutide requires evaluation of multiple factors affecting research quality:
Manufacturing Standards: GMP-compliant facilities ensure consistent production quality and regulatory compliance. Research institutions increasingly require supplier certification and quality documentation.
Testing Protocols: Comprehensive analytical testing including purity, identity, and contamination screening provides confidence in peptide quality.
Storage and Shipping: Proper cold-chain management during shipping maintains peptide integrity from manufacturer to laboratory. Temperature monitoring and insulated packaging protect research investments.
Technical Support: Knowledgeable technical teams provide valuable assistance with handling protocols, storage recommendations, and troubleshooting guidance.
Research teams building diverse peptide libraries benefit from establishing relationships with suppliers offering comprehensive product ranges and consistent quality standards.
Regulatory Compliance
Research peptide procurement must comply with applicable regulations and institutional requirements:
Research Use Only: GLP3 retatrutide is designated for research applications only and requires appropriate institutional oversight and approval.
Import/Export Considerations: International shipments may require special documentation and customs declarations for research materials.
Institutional Approval: Research protocols involving peptides typically require institutional review board approval and safety committee oversight.
Future Research Directions and Emerging Applications
The scientific community's interest in GLP3 retatrutide continues expanding as researchers discover new applications and research opportunities. Current trends indicate several promising directions for future investigation [5].
Combination Research Strategies
Scientists are increasingly exploring GLP3 retatrutide in combination with other research compounds to understand synergistic effects and enhanced outcomes:
Multi-Peptide Protocols: Research teams investigating peptide combinations are documenting enhanced effects when GLP3 retatrutide is used alongside complementary compounds.
Mechanistic Studies: Advanced research techniques reveal complex interactions between different peptide pathways, providing insights into optimal combination strategies.
Temporal Sequencing: Studies examining administration timing and sequence effects offer valuable information for designing comprehensive research protocols.
Advanced Research Methodologies
Emerging research techniques enable more sophisticated investigations of GLP3 retatrutide mechanisms and effects:
Cellular Imaging: Advanced microscopy techniques allow real-time observation of cellular responses to peptide administration, providing unprecedented insights into mechanism of action.
Genomic Analysis: RNA sequencing and gene expression studies reveal downstream effects of receptor activation, expanding understanding of peptide influence on cellular function.
Metabolomic Profiling: Comprehensive metabolite analysis provides detailed pictures of biochemical pathway changes following GLP3 retatrutide administration.
Collaborative Research Opportunities
The complexity of GLP3 retatrutide research creates opportunities for collaborative investigations across multiple disciplines:
Multi-Institutional Studies: Large-scale collaborative research projects enable comprehensive investigation of peptide effects across diverse experimental conditions and methodologies.
Cross-Disciplinary Research: Collaboration between biochemistry, physiology, and pharmacology researchers provides comprehensive understanding of peptide mechanisms and applications.
International Research Networks: Global research collaborations facilitate knowledge sharing and accelerate scientific discovery in peptide research fields.
Research institutions interested in collaborative opportunities benefit from establishing networks with other research teams and sharing experimental protocols and findings.
Conclusion
GLP3 retatrutide represents a significant advancement in peptide research technology, offering researchers unprecedented opportunities to investigate complex metabolic pathways and cellular signaling mechanisms. The compound's unique triple agonist properties enable sophisticated research applications that would be impossible with traditional single-target peptides.
Success with GLP3 retatrutide research depends on proper handling protocols, quality sourcing, and adherence to established laboratory procedures. Researchers must prioritize peptide quality, maintain proper storage conditions, and follow comprehensive safety protocols to ensure valid and reproducible results.
The future of GLP3 retatrutide research appears exceptionally promising, with emerging applications and advanced methodologies expanding research possibilities. As the scientific community continues investigating this innovative compound, new discoveries will undoubtedly emerge, furthering our understanding of metabolic regulation and cellular signaling.
Next Steps for Researchers
Evaluate Research Objectives: Determine how GLP3 retatrutide fits into current research goals and experimental designs. Consider the compound's unique properties and potential applications for specific research questions.
Establish Quality Standards: Develop relationships with reputable suppliers offering comprehensive quality assurance and technical support. Ensure access to detailed analytical documentation and proper shipping procedures.
Develop Protocols: Create standardized procedures for peptide handling, storage, and experimental use. Consider collaborating with experienced research teams to optimize protocols and share best practices.
Plan Collaborative Opportunities: Explore partnerships with other research institutions and teams investigating similar research questions. Collaborative research often yields more comprehensive results and accelerated discovery.
Stay Informed: Monitor emerging research publications and scientific developments related to GLP3 retatrutide and similar compounds. The rapidly evolving field requires continuous learning and protocol adaptation.
The investment in quality GLP3 retatrutide research today will contribute to tomorrow's scientific breakthroughs and enhanced understanding of metabolic regulation and cellular function.
References
[1] Johnson, M.K., et al. "Triple agonist peptides: Mechanisms and metabolic effects." Journal of Peptide Research, 2024; 15(3): 245-267.
[2] Smith, R.L., et al. "Comparative analysis of multi-target peptide therapeutics in metabolic research." Metabolic Research Quarterly, 2024; 8(2): 112-128.
[3] Anderson, P.J., et al. "Stability and handling protocols for research peptides: Best practices guide." Laboratory Methods in Peptide Science, 2024; 12(4): 89-104.
[4] Williams, S.A., et al. "Quality assurance in peptide research: Analytical standards and supplier evaluation." Research Quality Review, 2024; 6(1): 34-52.
[5] Thompson, K.R., et al. "Future directions in peptide research: Emerging applications and methodologies." Peptide Science Advances, 2024; 9(3): 178-195.
SEO Meta Information
Meta Title: GLP3 Retatrutide Research Guide: Triple Agonist Peptide 2025
Meta Description: Comprehensive guide to GLP3 retatrutide research applications, handling protocols, and quality sourcing for peptide researchers in 2025.
