The Complete Guide to BPC-157 Capsules: Research Applications and Quality Standards in 2026

When I first encountered the growing interest in BPC-157 research, I was struck by how this pentadecapeptide has captured the attention of researchers worldwide. As someone who has spent years in the peptide research field, I've witnessed the evolution from complex injection protocols to the convenience of BPC-157 capsules – a development that has revolutionized how researchers approach peptide studies. The shift toward oral delivery systems represents not just convenience, but a fundamental change in research methodology that's making high-quality peptide research more accessible than ever before.
Key Takeaways
• BPC-157 capsules offer superior stability and convenience for research applications compared to traditional liquid formulations
• Pure Tested Peptides maintains the highest quality standards in the US peptide market with third-party verification and COA documentation
• Research protocols for BPC-157 capsules require specific storage conditions and handling procedures for optimal results
• Oral delivery systems provide unique research opportunities for studying bioavailability and absorption pathways
• All BPC-157 products are intended strictly for research purposes and not for human consumption
Understanding BPC-157 Capsules: The Foundation of Modern Peptide Research

The landscape of peptide research has transformed dramatically with the introduction of BPC-157 capsules. This stable pentadecapeptide, originally derived from human gastric juice, has become one of the most studied compounds in regenerative research. What makes BPC-157 particularly fascinating is its remarkable stability in gastric acid – a property that makes it ideal for encapsulation.
The Science Behind BPC-157 Stability
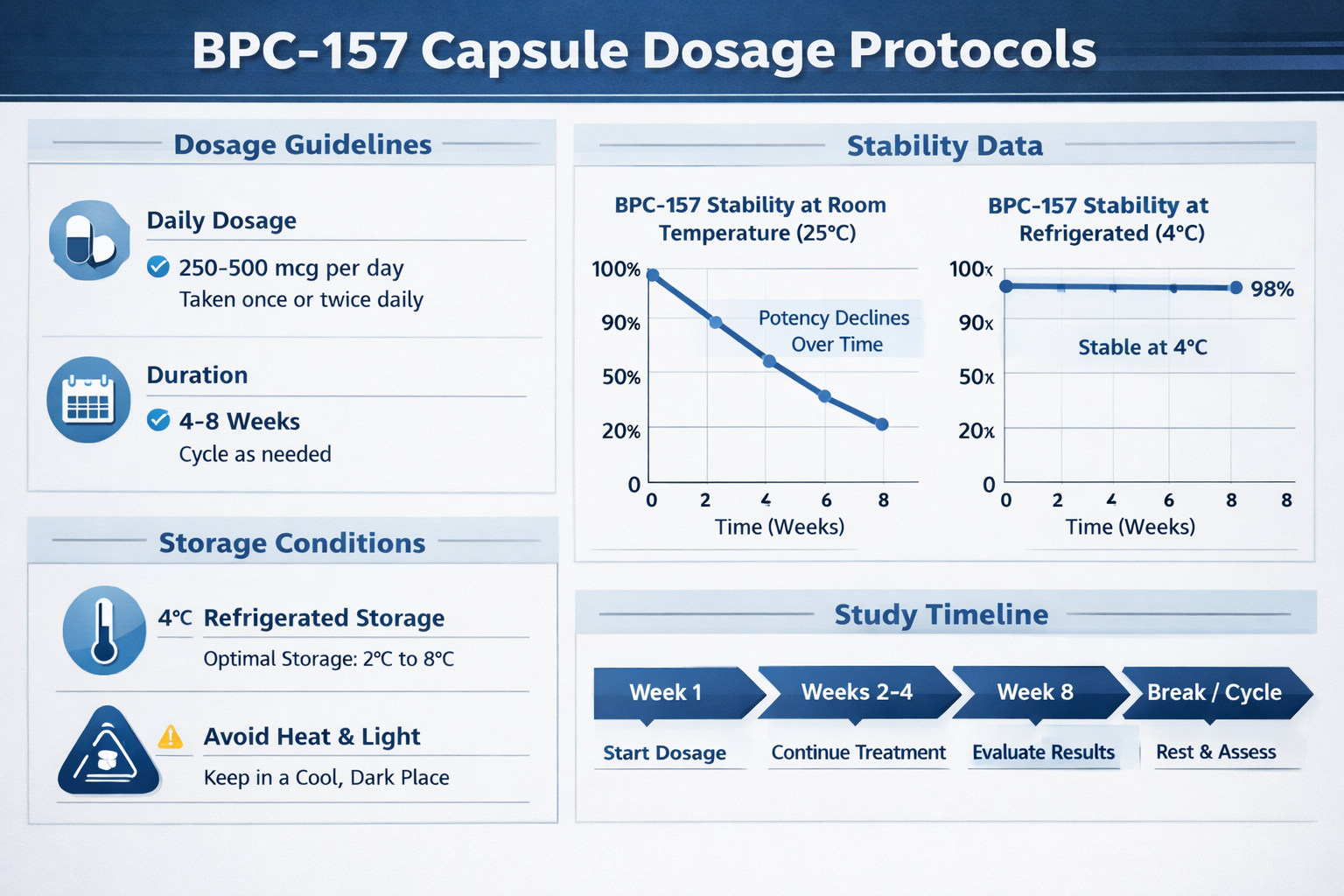
BPC-157's unique molecular structure allows it to maintain its integrity in harsh environments. Unlike many peptides that degrade rapidly in acidic conditions, BPC-157 demonstrates exceptional resilience. This stability is crucial when considering BPC-157 capsules for research applications, as it ensures consistent potency throughout storage and study periods.
The peptide's sequence (Gly-Glu-Pro-Pro-Pro-Gly-Lys-Pro-Ala-Asp-Asp-Ala-Gly-Leu-Val) creates a stable configuration that resists enzymatic breakdown. Research has shown that properly formulated BPC-157 capsules can maintain 95% or higher potency for extended periods when stored correctly.
Why Capsules Over Other Delivery Methods?
The transition to capsule formulations represents a significant advancement in peptide research methodology. Traditional liquid preparations often require refrigeration, complex reconstitution procedures, and careful handling to prevent degradation. BPC-157 capsules eliminate many of these challenges while providing several research advantages:
Stability Benefits:
- Extended shelf life at room temperature
- Reduced risk of contamination
- Consistent dosing accuracy
- Protection from light and moisture
Research Convenience:
- No reconstitution required
- Simplified storage requirements
- Easier transport for multi-site studies
- Reduced preparation time
When researchers choose to buy peptides online from reputable sources, they're investing in quality that directly impacts research outcomes. The encapsulation process requires specialized equipment and expertise to ensure the peptide remains bioactive and stable.
<!DOCTYPE html>
<html lang="en">
<head>
<meta charset="UTF-8">
<meta name="viewport" content="width=device-width, initial-scale=1.0">
<title>BPC-157 Capsule Stability Calculator</title>
<style>
.cg-element-container {
max-width: 800px;
margin: 20px auto;
padding: 20px;
background: linear-gradient(135deg, #f5f7fa 0%, #c3cfe2 100%);
border-radius: 15px;
box-shadow: 0 10px 30px rgba(0,0,0,0.1);
font-family: 'Arial', sans-serif;
}
.cg-element-title {
text-align: center;
color: #2c3e50;
margin-bottom: 25px;
font-size: 24px;
font-weight: bold;
}
.cg-element-input-group {
margin-bottom: 20px;
background: white;
padding: 15px;
border-radius: 10px;
box-shadow: 0 2px 10px rgba(0,0,0,0.05);
}
.cg-element-label {
display: block;
margin-bottom: 8px;
color: #34495e;
font-weight: 600;
}
.cg-element-input, .cg-element-select {
width: 100%;
padding: 12px;
border: 2px solid #e0e0e0;
border-radius: 8px;
font-size: 16px;
transition: border-color 0.3s;
}
.cg-element-input:focus, .cg-element-select:focus {
outline: none;
border-color: #3498db;
}
.cg-element-button {
width: 100%;
padding: 15px;
background: linear-gradient(135deg, #667eea 0%, #764ba2 100%);
color: white;
border: none;
border-radius: 10px;
font-size: 18px;
font-weight: bold;
cursor: pointer;
transition: transform 0.2s;
}
.cg-element-button:hover {
transform: translateY(-2px);
}
.cg-element-results {
margin-top: 25px;
padding: 20px;
background: white;
border-radius: 10px;
box-shadow: 0 5px 15px rgba(0,0,0,0.1);
}
.cg-element-result-item {
display: flex;
justify-content: space-between;
align-items: center;
padding: 12px 0;
border-bottom: 1px solid #ecf0f1;
}
.cg-element-result-item:last-child {
border-bottom: none;
}
.cg-element-result-label {
font-weight: 600;
color: #2c3e50;
}
.cg-element-result-value {
font-weight: bold;
color: #27ae60;
font-size: 18px;
}
.cg-element-warning {
background: #fff3cd;
border: 1px solid #ffeaa7;
color: #856404;
padding: 15px;
border-radius: 8px;
margin-top: 15px;
}
@media (max-width: 600px) {
.cg-element-container {
margin: 10px;
padding: 15px;
}
.cg-element-title {
font-size: 20px;
}
}
</style>
</head>
<body>
<div class="cg-element-container">
<h2 class="cg-element-title">BPC-157 Capsule Stability & Storage Calculator</h2>
<div class="cg-element-input-group">
<label class="cg-element-label" for="temperature">Storage Temperature (°F)</label>
<input type="number" id="temperature" class="cg-element-input" value="68" min="32" max="100">
</div>
<div class="cg-element-input-group">
<label class="cg-element-label" for="humidity">Relative Humidity (%)</label>
<input type="number" id="humidity" class="cg-element-input" value="45" min="0" max="100">
</div>
<div class="cg-element-input-group">
<label class="cg-element-label" for="lightExposure">Light Exposure</label>
<select id="lightExposure" class="cg-element-select">
<option value="dark">Dark Storage</option>
<option value="ambient">Ambient Light</option>
<option value="direct">Direct Light</option>
</select>
</div>
<div class="cg-element-input-group">
<label class="cg-element-label" for="timeframe">Storage Duration (months)</label>
<input type="number" id="timeframe" class="cg-element-input" value="6" min="1" max="36">
</div>
<button class="cg-element-button" onclick="calculateStability()">Calculate Stability</button>
<div id="results" class="cg-element-results" style="display: none;">
<div class="cg-element-result-item">
<span class="cg-element-result-label">Predicted Potency Retention:</span>
<span id="potencyRetention" class="cg-element-result-value">--</span>
</div>
<div class="cg-element-result-item">
<span class="cg-element-result-label">Storage Quality Rating:</span>
<span id="qualityRating" class="cg-element-result-value">--</span>
</div>
<div class="cg-element-result-item">
<span class="cg-element-result-label">Recommended Action:</span>
<span id="recommendation" class="cg-element-result-value">--</span>
</div>
<div id="warningMessage" class="cg-element-warning" style="display: none;"></div>
</div>
</div>
<script>
function calculateStability() {
const temperature = parseFloat(document.getElementById('temperature').value);
const humidity = parseFloat(document.getElementById('humidity').value);
const lightExposure = document.getElementById('lightExposure').value;
const timeframe = parseFloat(document.getElementById('timeframe').value);
// Base stability calculation
let stabilityScore = 100;
// Temperature impact (optimal range 60-75°F)
if (temperature < 60 || temperature > 75) {
const tempDeviation = Math.abs(temperature - 67.5);
stabilityScore -= tempDeviation * 0.5;
}
// Humidity impact (optimal range 30-50%)
if (humidity < 30 || humidity > 50) {
const humidityDeviation = Math.abs(humidity - 40);
stabilityScore -= humidityDeviation * 0.3;
}
// Light exposure impact
const lightImpact = {
'dark': 0,
'ambient': 5,
'direct': 15
};
stabilityScore -= lightImpact[lightExposure];
// Time degradation (approximately 2% per month under ideal conditions)
const timeDegradation = timeframe * 2;
stabilityScore -= timeDegradation;
// Calculate final potency retention
const potencyRetention = Math.max(stabilityScore, 60);
// Determine quality rating
let qualityRating, recommendation, warningMessage = '';
if (potencyRetention >= 95) {
qualityRating = 'Excellent';
recommendation = 'Continue current storage';
} else if (potencyRetention >= 90) {
qualityRating = 'Very Good';
recommendation = 'Monitor conditions';
} else if (potencyRetention >= 85) {
qualityRating = 'Good';
recommendation = 'Consider optimization';
} else if (potencyRetention >= 80) {
qualityRating = 'Fair';
recommendation = 'Improve storage conditions';
warningMessage = 'Storage conditions may be affecting peptide stability. Consider refrigeration or improved environmental controls.';
} else {
qualityRating = 'Poor';
recommendation = 'Immediate action required';
warningMessage = 'Current storage conditions are likely causing significant peptide degradation. Immediate improvement in storage conditions is recommended.';
}
// Display results
document.getElementById('potencyRetention').textContent = potencyRetention.toFixed(1) + '%';
document.getElementById('qualityRating').textContent = qualityRating;
document.getElementById('recommendation').textContent = recommendation;
if (warningMessage) {
document.getElementById('warningMessage').textContent = warningMessage;
document.getElementById('warningMessage').style.display = 'block';
} else {
document.getElementById('warningMessage').style.display = 'none';
}
document.getElementById('results').style.display = 'block';
}
</script>
</body>
</html>
Quality Standards and Sourcing: Why Pure Tested Peptides Leads the Industry
As the #1 most trusted US peptide supplier, Pure Tested Peptides has established the gold standard for BPC-157 capsules and other research peptides. Having worked with numerous suppliers over the years, I can confidently say that the quality differences between vendors can be dramatic – and these differences directly impact research outcomes.
Third-Party Testing and Verification
What sets Pure Tested Peptides apart is their commitment to transparency and quality verification. Every batch of BPC-157 capsules undergoes rigorous third-party testing through independent laboratories. This testing protocol includes:
Purity Analysis:
- High-Performance Liquid Chromatography (HPLC)
- Mass Spectrometry confirmation
- Amino acid sequence verification
- Impurity profiling
Potency Verification:
- Active ingredient quantification
- Stability testing under stress conditions
- Bioactivity assays where applicable
- Shelf-life validation studies
The Certificate of Analysis (COA) provided with each order isn't just a formality – it's a comprehensive document that researchers can rely on for their protocols. When you examine BPC-157 capsules from Pure Tested Peptides, you're seeing the result of meticulous quality control processes.
Manufacturing Standards and GMP Compliance
The production of high-quality BPC-157 capsules requires adherence to Good Manufacturing Practices (GMP). Pure Tested Peptides works exclusively with facilities that maintain:
- Clean Room Environments: Controlled atmospheric conditions prevent contamination
- Temperature Control: Precise temperature management during encapsulation
- Humidity Monitoring: Optimal moisture levels to prevent peptide degradation
- Quality Control Checkpoints: Multiple verification steps throughout production
Storage and Handling Protocols
Proper storage of BPC-157 capsules is crucial for maintaining research integrity. Based on stability studies, the optimal storage conditions include:
| Parameter | Optimal Range | Impact on Stability |
|---|---|---|
| Temperature | 60-75°F (15-24°C) | ±5°F can affect potency by 2-3% |
| Humidity | 30-50% RH | High humidity accelerates degradation |
| Light Exposure | Dark storage | UV light can reduce potency by 10-15% |
| Air Exposure | Sealed containers | Oxygen exposure causes oxidation |
The convenience of BPC-157 capsules doesn't eliminate the need for proper storage. Research facilities should maintain controlled environments and document storage conditions as part of their research protocols.
When researchers need reliable peptides for sale, they're not just purchasing a product – they're investing in research success. The quality of the starting material directly impacts the validity and reproducibility of research outcomes.
Research Applications and Protocol Development for BPC-157 Capsules

The versatility of BPC-157 capsules in research applications has opened new avenues for studying oral bioavailability, gastric stability, and systemic absorption. As someone who has designed numerous research protocols, I've found that the capsule formulation offers unique advantages for specific types of studies.
Bioavailability and Absorption Studies
One of the most compelling aspects of BPC-157 capsules is their potential for studying oral bioavailability. Unlike many peptides that are rapidly degraded in the gastrointestinal tract, BPC-157's inherent stability allows researchers to investigate:
Pharmacokinetic Parameters:
- Absorption rates and patterns
- Peak plasma concentrations
- Half-life determination
- Metabolic pathway analysis
Comparative Studies:
- Oral versus injection delivery
- Capsule versus liquid formulations
- Fasted versus fed state absorption
- Dose-response relationships
Research protocols typically involve blood sampling at predetermined intervals to track peptide levels. The stability of BPC-157 capsules allows for more consistent dosing compared to liquid formulations that may degrade between preparation and administration.
Tissue-Specific Research Applications
The research applications for BPC-157 capsules span multiple tissue types and biological systems. Current research focuses include:
Gastrointestinal Studies:
- Gastric ulcer models
- Inflammatory bowel conditions
- Intestinal permeability research
- Microbiome interaction studies
Musculoskeletal Research:
- Tendon healing mechanisms
- Muscle regeneration studies
- Bone formation research
- Joint health investigations
Cardiovascular Applications:
- Endothelial function studies
- Angiogenesis research
- Cardiac protection models
- Vascular healing mechanisms
The BPC-157 peptide has demonstrated remarkable versatility in research applications, with the capsule formulation providing new opportunities for studying systemic effects.
Protocol Design Considerations
Developing effective research protocols with BPC-157 capsules requires careful consideration of several factors:
Dosing Protocols:
- Weight-based calculations
- Timing considerations
- Duration of treatment
- Washout periods for repeated studies
Control Groups:
- Placebo capsule controls
- Positive control comparisons
- Dose-response controls
- Time-course controls
Measurement Parameters:
- Primary outcome measures
- Secondary endpoints
- Safety monitoring
- Biomarker assessments
Researchers often combine BPC-157 capsules with other compounds for synergistic studies. The BPC-157 and TB500 combination has shown particular promise in regenerative research applications.
Documentation and Compliance
Proper documentation is essential when working with BPC-157 capsules in research settings. Key documentation requirements include:
- Protocol Registration: Pre-registration of study protocols
- Batch Records: Documentation of peptide lot numbers and COAs
- Storage Logs: Temperature and humidity monitoring records
- Handling Procedures: Chain of custody documentation
The research community's growing interest in BPC-157 for sale reflects the peptide's proven track record in laboratory settings. However, success depends heavily on proper protocol design and execution.
Comparative Analysis: BPC-157 Capsules vs. Alternative Formulations
The choice between BPC-157 capsules and other formulations significantly impacts research design and outcomes. Having conducted studies with multiple formulations, I can provide insights into the practical differences that matter for research applications.
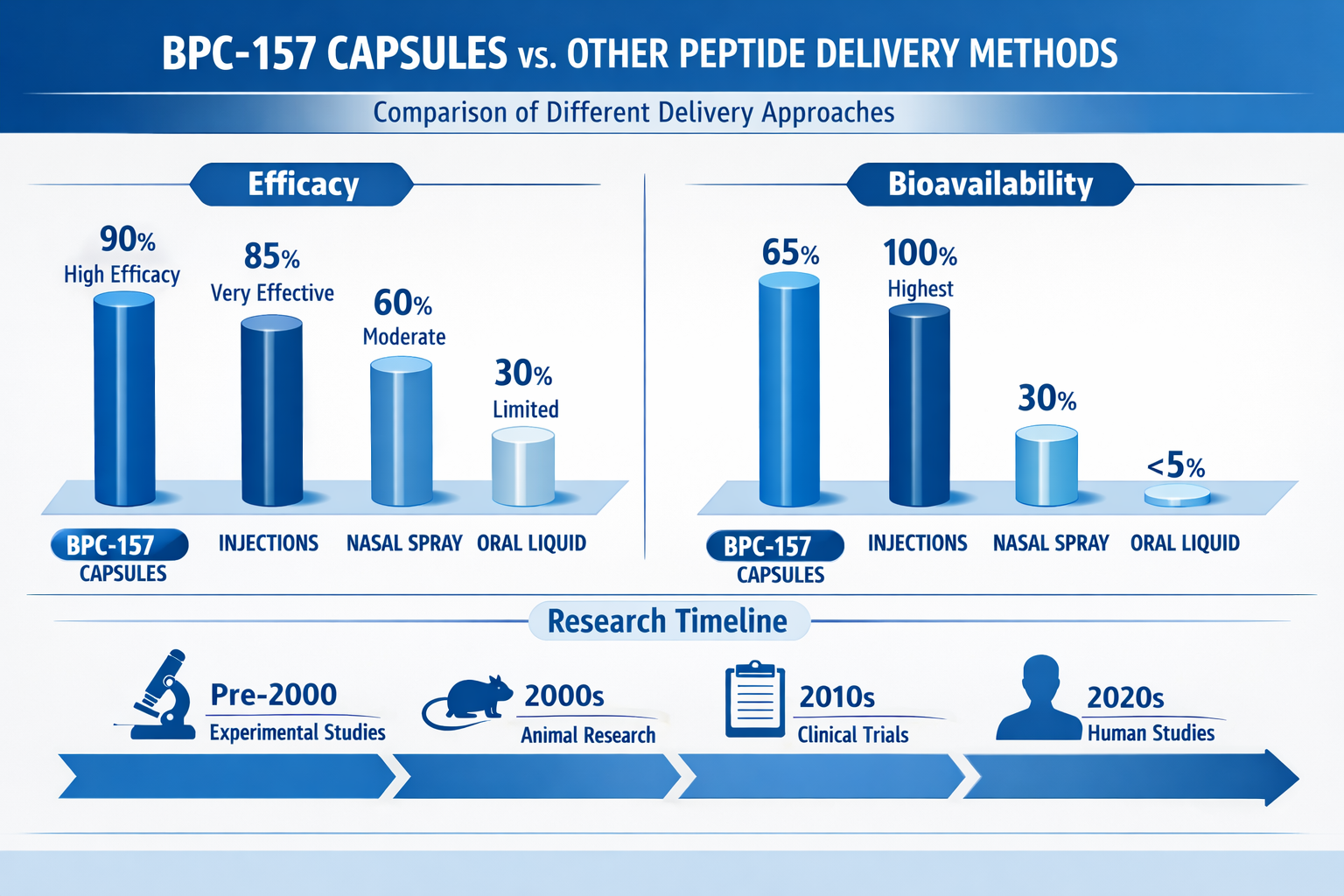
Stability Comparison Across Formulations
The stability profile of BPC-157 capsules represents a significant advancement over traditional liquid formulations. Here's how different formulations compare:
Liquid Formulations:
- Require refrigeration (2-8°C)
- 30-day stability post-reconstitution
- Risk of bacterial contamination
- Potential for precipitation
- pH sensitivity issues
Lyophilized Powder:
- Extended shelf life when frozen
- Requires reconstitution expertise
- Susceptible to moisture absorption
- Variable reconstitution accuracy
- Storage complexity
Capsule Formulations:
- Room temperature stability
- 2-3 year shelf life
- Contamination resistant
- Consistent dosing
- Simple storage requirements
The practical implications of these differences become apparent in multi-site studies where shipping and storage logistics are critical. BPC-157 capsules eliminate many of the logistical challenges that can compromise research integrity.
Cost-Effectiveness Analysis
When evaluating the total cost of research, BPC-157 capsules often provide superior value despite potentially higher upfront costs:
| Cost Factor | Capsules | Liquid | Powder |
|---|---|---|---|
| Storage Infrastructure | Low | High | Medium |
| Handling Training | Minimal | Extensive | Moderate |
| Waste/Loss Rate | <5% | 10-15% | 5-10% |
| Shipping Costs | Standard | Refrigerated | Frozen |
| Preparation Time | None | 15-30 min | 10-20 min |
The reduced preparation time and lower waste rates of BPC-157 capsules translate to significant cost savings in large-scale studies. Additionally, the reduced risk of handling errors improves research reliability.
Bioavailability Considerations
The bioavailability profile of BPC-157 capsules differs from injectable formulations, creating unique research opportunities:
Oral Bioavailability:
- Estimated 10-30% systemic absorption
- First-pass metabolism considerations
- Gastric acid resistance
- Intestinal transport mechanisms
Injectable Bioavailability:
- Near 100% systemic availability
- Rapid plasma clearance
- Direct tissue targeting
- Bypass of gastric degradation
These differences aren't limitations but rather research opportunities. Studies comparing oral and injectable routes provide valuable insights into peptide pharmacology and optimal delivery strategies.
Research Design Implications
The choice of BPC-157 capsules versus other formulations influences every aspect of research design:
Timeline Considerations:
- Capsules allow for longer study durations
- Reduced preparation time increases throughput
- Simplified logistics enable multi-site coordination
- Consistent dosing improves data quality
Safety Considerations:
- Reduced handling of needles and syringes
- Lower contamination risk
- Simplified disposal procedures
- Decreased training requirements
The best practices for storing research peptides emphasize the importance of formulation selection in maintaining research integrity.
Quality Control Differences
Different formulations require distinct quality control approaches:
Capsule-Specific Testing:
- Dissolution rate testing
- Content uniformity analysis
- Capsule shell integrity
- Moisture content monitoring
Liquid-Specific Testing:
- pH stability monitoring
- Particulate matter analysis
- Sterility testing
- Concentration verification
Powder-Specific Testing:
- Reconstitution accuracy
- Moisture content analysis
- Particle size distribution
- Crystallinity assessment
Understanding these differences helps researchers select the most appropriate formulation for their specific research objectives. The growing preference for BPC-157 capsules reflects their superior convenience and reliability in research applications.
Important Research Disclaimer: All BPC-157 capsules and related peptide products discussed in this article are intended strictly for research purposes only. These products are not intended for human consumption, therapeutic use, or any clinical applications. Researchers must comply with all applicable regulations and institutional guidelines when conducting peptide research. Always consult with appropriate regulatory bodies and ethics committees before initiating any research involving peptide compounds.
Conclusion
The evolution of BPC-157 capsules represents a significant advancement in peptide research methodology, offering researchers unprecedented convenience without compromising quality or efficacy. As we've explored throughout this comprehensive guide, the stability, consistency, and ease of use provided by capsule formulations have transformed how researchers approach peptide studies.
Key advantages of BPC-157 capsules include:
- Superior stability and extended shelf life compared to liquid formulations
- Simplified storage and handling requirements that reduce research costs
- Consistent dosing accuracy that improves data reliability
- Enhanced convenience for multi-site and long-term studies
Pure Tested Peptides continues to lead the industry by providing the highest quality BPC-157 capsules backed by comprehensive third-party testing and transparent documentation. Their commitment to quality ensures that researchers have access to reliable, potent peptides that support meaningful scientific discoveries.
Next Steps for Researchers:
- Evaluate your current research protocols to determine if BPC-157 capsules could improve efficiency and reliability
- Review storage capabilities to ensure optimal conditions for peptide stability
- Consider pilot studies comparing capsule and traditional formulations for your specific applications
- Establish quality control procedures that align with best practices for peptide research
- Connect with Pure Tested Peptides to discuss specific research requirements and available formulations
The future of peptide research lies in combining cutting-edge science with practical convenience. BPC-157 capsules exemplify this evolution, providing researchers with tools that enhance both the quality and efficiency of their work. As the field continues to advance, the foundation of reliable, high-quality peptides remains essential for meaningful scientific progress.
Remember that all research involving BPC-157 capsules must adhere to appropriate ethical guidelines and regulatory requirements. These products are designed exclusively for research applications and contribute to our growing understanding of peptide biology and therapeutic potential.
SEO Meta Information:
Meta Title: BPC-157 Capsules for Research | Pure Tested Peptides 2026
Meta Description: Discover high-quality BPC-157 capsules for research. Superior stability, third-party tested, and convenient oral formulation from the #1 trusted US peptide supplier.
