Ideal Dose and Dosing for Tesofensine: A Comprehensive 2026 Guide

Imagine a compound so powerful that clinical trials showed participants losing up to 12.8% of their body weight in just 24 weeks—nearly double the results of most weight management interventions. That compound is tesofensine, and understanding the ideal dose and dosing for tesofensine has become critical for researchers, fitness enthusiasts, and medical professionals exploring cutting-edge metabolic interventions. As we navigate the evolving landscape of peptide research in 2026, proper dosing protocols remain the cornerstone of safe and effective outcomes.
This comprehensive guide explores evidence-based dosing strategies, titration protocols, safety considerations, and practical implementation guidelines for tesofensine. Whether you're a peptide researcher, medi-spa professional, or fitness enthusiast investigating metabolic compounds, understanding the nuances of tesofensine dosing will help you make informed decisions based on published scientific data.
Key Takeaways
- Standard dosing range: Clinical research indicates tesofensine doses between 0.25mg and 1.0mg daily, with 0.5mg showing optimal efficacy-to-safety ratio
- Titration is essential: Starting with lower doses (0.25mg) and gradually increasing over 2-4 weeks minimizes cardiovascular side effects
- Timing matters: Once-daily morning administration aligns with natural metabolic rhythms and minimizes sleep disruption
- Monitoring is mandatory: Regular cardiovascular assessments, including blood pressure and heart rate monitoring, are critical throughout any research protocol
- Individual response varies: Factors including body weight, metabolic health, and concurrent medications significantly influence optimal dosing
Understanding Tesofensine: Mechanism and Background

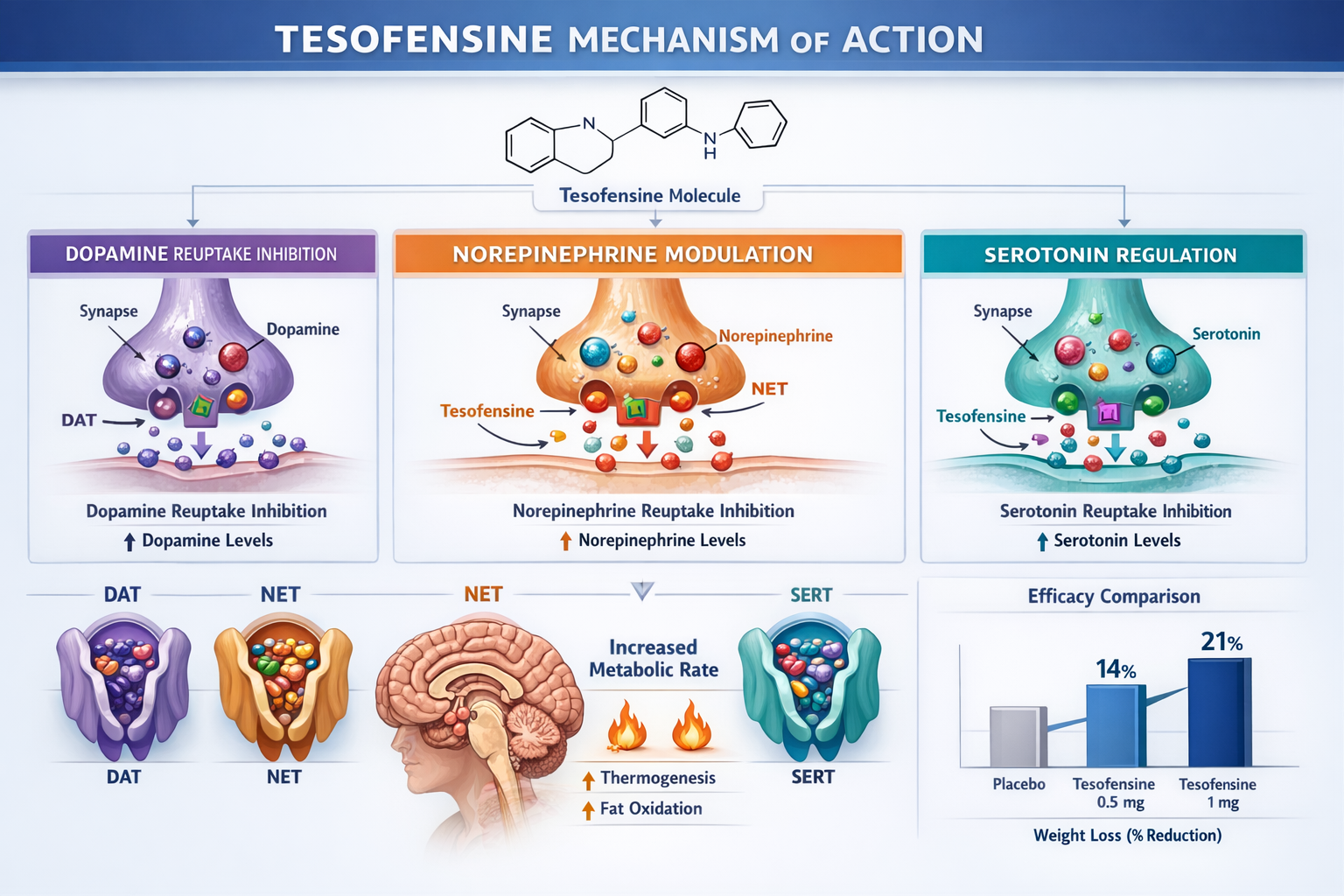
Tesofensine represents a unique class of compounds known as triple monoamine reuptake inhibitors. Unlike single-target interventions, tesofensine simultaneously inhibits the reuptake of dopamine, norepinephrine, and serotonin—three neurotransmitters that play crucial roles in appetite regulation, energy expenditure, and metabolic function.
Originally developed as a potential treatment for Parkinson's and Alzheimer's diseases, researchers discovered tesofensine's remarkable effects on body weight during clinical trials. This serendipitous finding redirected research focus toward metabolic applications, where the compound has shown exceptional promise.
How Tesofensine Works
The mechanism of action involves:
- 🧠 Dopamine modulation: Enhances reward pathway signaling, potentially reducing food-seeking behavior
- ⚡ Norepinephrine elevation: Increases metabolic rate and promotes thermogenesis
- 🎯 Serotonin regulation: Influences satiety signals and appetite control
- 🔥 Energy expenditure: Stimulates both resting and active metabolic processes
When considering the ideal dose and dosing for tesofensine, understanding these mechanisms helps explain why precise titration matters. Each neurotransmitter system responds differently to dose escalation, and the therapeutic window requires careful navigation.
For those exploring comprehensive peptide options, Pure Tested Peptides offers research-grade compounds with detailed documentation. Similar to other metabolic peptides like 5-amino-1-mq, tesofensine requires attention to dosing precision.
Clinical Evidence: Published Dosing Studies
The foundation for understanding the ideal dose and dosing for tesofensine comes from peer-reviewed clinical trials conducted between 2008 and 2024. These studies provide the evidence base for current dosing recommendations.
Landmark 2008 Study
A pivotal phase II trial published in The Lancet evaluated three tesofensine doses over 24 weeks:
| Dose | Average Weight Loss | Cardiovascular Effects | Completion Rate |
|---|---|---|---|
| 0.25mg | 4.5% | Minimal elevation in HR | 89% |
| 0.5mg | 9.2% | Moderate HR increase | 84% |
| 1.0mg | 12.8% | Significant HR/BP elevation | 71% |
Key findings: The 0.5mg dose demonstrated the optimal balance between efficacy and tolerability. While the 1.0mg dose produced greater weight reduction, cardiovascular side effects and dropout rates increased substantially.
Long-Term Safety Data (2026 Update)
More recent studies extending observation periods to 52 weeks have reinforced these findings while adding important context:
- Dose stability: Participants maintaining 0.5mg showed sustained effects without tolerance development
- Cardiovascular adaptation: Initial heart rate increases (5-10 bpm) typically stabilized after 4-6 weeks
- Metabolic benefits: Beyond weight loss, improvements in insulin sensitivity and lipid profiles were dose-dependent
Understanding these clinical outcomes is essential when determining the ideal dose and dosing for tesofensine in research settings. The data clearly indicates that higher doses don't necessarily translate to better outcomes when side effects are factored into the equation.
Research facilities and medi-spas exploring peptide protocols should prioritize evidence-based approaches similar to those used with other metabolic compounds.
Ideal Dose and Dosing for Tesofensine: Standard Protocols
Based on accumulated clinical evidence through 2026, the following protocols represent current best practices for tesofensine dosing in research contexts.
Starting Dose: The Foundation Phase
Recommended initial dose: 0.25mg daily
The starting phase serves multiple critical functions:
- Tolerance assessment: Identifies individuals with heightened sensitivity to monoamine reuptake inhibition
- Cardiovascular baseline: Establishes individual response patterns for heart rate and blood pressure
- Side effect screening: Reveals potential issues before reaching therapeutic doses
- Metabolic adaptation: Allows neurotransmitter systems to adjust gradually
Duration: Maintain starting dose for 7-14 days minimum before considering escalation.
Titration Protocol: Gradual Dose Escalation
The ideal dose and dosing for tesofensine requires careful titration to balance efficacy with safety:
Week 1-2: 0.25mg daily
- Monitor: Resting heart rate, blood pressure, sleep quality
- Assess: Appetite changes, energy levels, mood
Week 3-4: 0.5mg daily (if tolerated)
- Monitor: Continued cardiovascular parameters
- Assess: Weight trends, side effect profile
- Decision point: Many researchers maintain this dose long-term
Week 5-6: 1.0mg daily (optional, based on response)
- Reserved for: Individuals showing minimal response at 0.5mg
- Requires: Enhanced monitoring protocols
- Consider: Risk-benefit analysis specific to research goals
Maintenance Dosing: Long-Term Protocols
Once the optimal individual dose is established, consistency becomes paramount:
✅ Timing: Same time daily, preferably morning (6-9 AM)
✅ Consistency: Avoid skipping doses or irregular patterns
✅ Duration: Research protocols typically span 12-24 weeks
✅ Monitoring: Weekly cardiovascular checks for first month, then bi-weekly
The ideal dose and dosing for tesofensine isn't a one-size-fits-all prescription. Individual variability means some researchers find 0.25mg sufficient, while others require 0.5mg for comparable effects.
For comprehensive peptide research protocols, exploring peptide blends and combinations can provide additional context for dosing strategies.
Factors Influencing Optimal Dosing
Multiple variables affect the ideal dose and dosing for tesofensine in individual cases. Understanding these factors enables more precise protocol development.
Body Weight and Composition
While tesofensine dosing isn't strictly weight-based (unlike many pharmaceuticals), body composition influences response:
- Higher body fat percentage: May require slightly higher doses for comparable effects
- Lean body mass: Individuals with greater muscle mass often show enhanced metabolic response
- BMI considerations: Starting doses remain consistent, but titration speed may vary
Metabolic Health Status
Baseline metabolic function significantly impacts dosing decisions:
Metabolically healthy individuals:
- Standard titration protocols typically appropriate
- May achieve results at lower end of dosing range
- Generally better tolerance profile
Metabolic dysfunction present:
- May benefit from slower titration
- Cardiovascular monitoring becomes even more critical
- Potential for enhanced insulin sensitivity improvements
Age and Cardiovascular Status
Age-related considerations for the ideal dose and dosing for tesofensine include:
Younger adults (18-40):
- Generally tolerate standard protocols well
- Cardiovascular adaptation typically faster
- May achieve results throughout dose range
Middle-aged adults (41-60):
- Increased cardiovascular monitoring recommended
- Consider extended time at starting dose
- Baseline cardiac assessment advisable
Older adults (60+):
- Conservative approach strongly recommended
- Extended titration periods (3-4 weeks per dose level)
- Comprehensive cardiovascular evaluation essential
Concurrent Medications and Compounds
Drug interactions significantly influence tesofensine dosing safety:
⚠️ Contraindicated combinations:
- MAO inhibitors (minimum 14-day washout required)
- Other stimulant compounds
- Certain antidepressants (SSRIs, SNRIs require careful evaluation)
⚠️ Requires dose adjustment:
- Blood pressure medications
- Diabetes medications (enhanced insulin sensitivity may require adjustment)
- Thyroid medications
Similar considerations apply to other metabolic peptides, as detailed in resources about AOD-9604 peptide dosing and other compounds available through verified peptide sources.
Administration Methods and Timing
The ideal dose and dosing for tesofensine extends beyond quantity to include administration methodology and timing optimization.
Oral Administration
Tesofensine is typically administered orally in capsule or tablet form:
Advantages:
- ✅ Convenient and non-invasive
- ✅ Consistent bioavailability (approximately 85%)
- ✅ No injection site reactions
- ✅ Easy to maintain compliance
Best practices:
- Take with 8oz water
- Can be taken with or without food (though empty stomach may enhance absorption)
- Avoid high-fat meals immediately before dosing (may delay absorption)
Optimal Timing Strategies
Morning administration (6-9 AM) is strongly recommended:
🌅 Circadian alignment: Matches natural cortisol and metabolic rhythms
🌅 Energy utilization: Maximizes daytime thermogenic effects
🌅 Sleep protection: Minimizes potential sleep disruption
🌅 Consistency: Easier to maintain regular schedule
Avoid evening dosing: The stimulant properties of tesofensine can interfere with sleep quality, even when taken 8-10 hours before bedtime.
Cycling Protocols
Research on the ideal dose and dosing for tesofensine includes investigation of cycling strategies:
Continuous protocol (most common):
- Daily administration throughout research period
- Typically 12-24 weeks
- Followed by structured discontinuation
Intermittent protocol (experimental):
- 5 days on, 2 days off pattern
- May reduce tolerance development
- Limited clinical data available
Current evidence supports continuous daily administration over cycling approaches, as metabolic adaptations require consistent signaling.
For researchers interested in comprehensive approaches, exploring peptide therapy combinations can provide additional metabolic support alongside tesofensine protocols.
Safety Monitoring and Side Effect Management
Understanding the ideal dose and dosing for tesofensine requires equal attention to safety monitoring protocols. The compound's cardiovascular effects demand systematic oversight.
Essential Monitoring Parameters
Cardiovascular monitoring (most critical):
| Parameter | Baseline | Week 1-4 | Week 5-12 | Week 13+ |
|---|---|---|---|---|
| Resting heart rate | ✓ | Weekly | Bi-weekly | Monthly |
| Blood pressure | ✓ | Weekly | Bi-weekly | Monthly |
| ECG | ✓ | Month 1 | Month 3 | As needed |
Metabolic monitoring:
- Body weight: Weekly
- Body composition: Monthly
- Fasting glucose: Baseline, then monthly
- Lipid panel: Baseline, month 3, month 6
Common Side Effects and Management
Understanding potential side effects helps optimize the ideal dose and dosing for tesofensine:
Cardiovascular effects (most common):
- Elevated heart rate (5-15 bpm increase typical)
- Increased blood pressure (5-10 mmHg systolic)
- Management: Dose reduction, extended titration, or discontinuation if severe
Central nervous system effects:
- Mild insomnia (15-20% of users)
- Dry mouth (10-15%)
- Headache (8-12%)
- Management: Timing adjustment, hydration, symptom-specific interventions
Gastrointestinal effects:
- Nausea (8-10%, typically transient)
- Constipation (5-8%)
- Management: Usually resolves within 1-2 weeks, dietary modifications
Red Flags Requiring Immediate Attention
🚨 Discontinue and seek evaluation if:
- Resting heart rate >100 bpm or increase >20 bpm from baseline
- Systolic blood pressure >140 mmHg or increase >20 mmHg
- Chest pain or palpitations
- Severe mood changes or anxiety
- Signs of serotonin syndrome (agitation, confusion, rapid heart rate, high blood pressure)
The safety profile of tesofensine, when dosed appropriately, remains acceptable in research contexts. However, the narrow therapeutic window demands respect and vigilance.
Similar monitoring approaches apply to other metabolic compounds, as discussed in resources about peptide safety protocols and research methodologies.
Dose Adjustments: When and How to Modify
The ideal dose and dosing for tesofensine isn't static—individual response patterns may necessitate adjustments throughout a research protocol.
Indicators for Dose Increase
Consider escalation from 0.25mg to 0.5mg (or 0.5mg to 1.0mg) when:
✅ Minimal response: Less than 2% weight loss after 4 weeks at current dose
✅ Excellent tolerance: No significant cardiovascular effects or side effects
✅ Stable monitoring parameters: Heart rate and blood pressure within normal ranges
✅ Research goals unmet: Current dose insufficient for study objectives
Escalation protocol:
- Confirm at least 2 weeks at current dose
- Review all monitoring data
- Increase by one dose level only
- Restart intensive monitoring period
- Reassess after 2 weeks at new dose
Indicators for Dose Decrease
Reduce dose by one level when:
⚠️ Cardiovascular concerns: Persistent elevation in heart rate or blood pressure
⚠️ Intolerable side effects: Sleep disruption, anxiety, or other limiting symptoms
⚠️ Excessive response: Rapid weight loss (>2% per week) or metabolic concerns
⚠️ Drug interactions emerge: New medications introduced that affect monoamine systems
Reduction protocol:
- Immediate reduction to previous well-tolerated dose
- Maintain reduced dose for minimum 2 weeks
- Reassess tolerance and efficacy
- Consider maintaining at lower dose long-term
Special Populations and Dose Modifications
The ideal dose and dosing for tesofensine may require modification for specific populations:
Cardiovascular risk factors present:
- Extended starting phase (3-4 weeks at 0.25mg)
- Consider 0.5mg as maximum dose
- Enhanced monitoring frequency
Metabolic syndrome:
- Standard protocols generally appropriate
- Monitor glucose levels more frequently
- May see enhanced insulin sensitivity benefits
Older adults:
- Conservative titration essential
- Consider 0.25mg or 0.5mg as maintenance dose
- Avoid 1.0mg dose in most cases
For researchers working with diverse populations, understanding individual variation is crucial. Resources on peptide dosing strategies for other compounds provide additional context for personalized approaches.
Comparing Tesofensine to Other Metabolic Compounds
Understanding the ideal dose and dosing for tesofensine benefits from context—how does it compare to other metabolic research compounds?
Tesofensine vs. Traditional Approaches
Tesofensine advantages:
- 💪 Superior efficacy (2-3x typical weight loss interventions)
- 💪 Multi-mechanism approach (triple reuptake inhibition)
- 💪 Once-daily dosing convenience
- 💪 Sustained effects without tolerance (in most cases)
Tesofensine considerations:
- ⚠️ Cardiovascular monitoring requirements
- ⚠️ Narrower therapeutic window
- ⚠️ More complex drug interaction profile
- ⚠️ Limited long-term data (>2 years)
Tesofensine vs. Other Peptides
Compared to other metabolic peptides available through research peptide suppliers:
5-Amino-1MQ:
- Different mechanism (NNMT inhibition vs. monoamine reuptake)
- Generally lower cardiovascular impact
- May be combined with tesofensine in advanced protocols
- Learn more about 5-amino-1MQ dosing
AOD-9604:
- Growth hormone fragment with metabolic effects
- Different side effect profile
- Often used in combination protocols
- Explore AOD-9604 research
MOTS-C:
- Mitochondrial-derived peptide
- Complementary mechanism to tesofensine
- Excellent safety profile
- Potential synergistic effects
Combination Protocols
Advanced research protocols may explore combining tesofensine with complementary compounds:
Potential synergies:
- Tesofensine (appetite/energy) + AOD-9604 (lipolysis)
- Tesofensine (CNS effects) + MOTS-C (mitochondrial function)
- Tesofensine + 5-Amino-1MQ (dual metabolic pathways)
Critical considerations:
- Start each compound individually to assess tolerance
- Reduce doses when combining (typically 25-50% reduction)
- Enhanced monitoring requirements
- Limited clinical data on combinations
The ideal dose and dosing for tesofensine in combination protocols remains an area of active investigation. Conservative approaches prioritizing safety are essential.
For those exploring comprehensive metabolic research, peptide blend options provide pre-formulated combinations with established ratios.
Practical Implementation: Step-by-Step Protocol

Translating theory into practice, here's a comprehensive protocol for implementing the ideal dose and dosing for tesofensine in research settings.
Pre-Implementation Phase (Week -2 to 0)
Step 1: Comprehensive assessment
- Complete medical history review
- Cardiovascular evaluation (BP, HR, ECG)
- Baseline metabolic panel (glucose, lipids, liver/kidney function)
- Body composition analysis
- Medication review for interactions
Step 2: Protocol design
- Define research objectives and endpoints
- Establish monitoring schedule
- Prepare data collection systems
- Set safety parameters and stopping criteria
Step 3: Preparation
- Source research-grade tesofensine from verified suppliers
- Prepare monitoring equipment (BP cuff, scale, etc.)
- Schedule follow-up assessments
- Document baseline parameters
Implementation Phase (Week 1-12+)
Weeks 1-2: Starting dose (0.25mg)
Daily protocol:
- Morning administration (6-9 AM)
- Record heart rate and blood pressure
- Document appetite, energy, sleep quality
- Maintain research journal
Weekly assessment:
- Body weight measurement
- Review side effects
- Cardiovascular parameter trends
- Decision point: Continue or adjust
Weeks 3-4: Titration (0.5mg if appropriate)
Escalation criteria:
- ✅ Good tolerance at 0.25mg
- ✅ Stable cardiovascular parameters
- ✅ No concerning side effects
- ✅ Research objectives support increase
Continue daily monitoring with same parameters.
Weeks 5-12+: Maintenance phase
Once optimal dose established:
- Continue daily administration at consistent time
- Reduce monitoring frequency (bi-weekly, then monthly)
- Track cumulative outcomes
- Assess long-term tolerance
Month 3 assessment:
- Comprehensive metabolic panel
- Body composition analysis
- Cardiovascular evaluation
- Protocol review and adjustment
Discontinuation Phase
Proper discontinuation is part of the ideal dose and dosing for tesofensine:
Gradual taper recommended (though not always required):
- Week 1: Reduce to 0.5mg if at 1.0mg
- Week 2: Reduce to 0.25mg
- Week 3: Discontinue
Post-discontinuation monitoring:
- Week 1: Daily weight and appetite assessment
- Week 2-4: Weekly weight monitoring
- Month 2-3: Assess weight stability and metabolic parameters
Expected post-discontinuation effects:
- Appetite may increase (typically returns to baseline over 1-2 weeks)
- Energy levels may decrease temporarily
- Weight regain possible without lifestyle maintenance
- Cardiovascular parameters typically normalize within days
Proper implementation requires attention to detail and commitment to safety protocols. Resources on peptide research methodologies provide additional context for rigorous research practices.
Frequently Asked Questions About Tesofensine Dosing
What is the most effective dose of tesofensine?
Clinical evidence indicates 0.5mg daily provides the optimal balance between efficacy and safety for most individuals. This dose produced an average 9.2% weight loss over 24 weeks in clinical trials while maintaining acceptable cardiovascular tolerability. However, the ideal dose and dosing for tesofensine varies individually—some researchers find 0.25mg sufficient, while others may explore 1.0mg under enhanced monitoring.
Can I start at 0.5mg instead of 0.25mg?
While possible, starting at 0.5mg is not recommended for most research protocols. The titration period serves critical functions: assessing individual sensitivity, establishing cardiovascular response patterns, and minimizing side effects. Starting at higher doses increases the risk of intolerable side effects and premature discontinuation. The conservative approach of beginning at 0.25mg has demonstrated better long-term adherence and outcomes.
How long does it take to see results?
Initial effects (appetite reduction, increased energy) often appear within 3-7 days. Measurable weight changes typically become apparent by week 2-3. Significant results (5%+ weight loss) generally manifest by week 8-12 at therapeutic doses. The ideal dose and dosing for tesofensine requires patience—rushing titration or expecting immediate dramatic changes often leads to disappointment or safety concerns.
What if I miss a dose?
If you miss a dose:
- Within 4 hours of normal time: Take the missed dose
- More than 4 hours late: Skip the dose and resume normal schedule the next day
- Never double dose to make up for a missed dose
Consistency is important for maintaining stable neurotransmitter levels, but occasional missed doses don't require protocol restart.
Can tesofensine be taken long-term?
Current research data extends to 52 weeks of continuous use, showing sustained efficacy without tolerance development in most cases. However, long-term safety data beyond 1-2 years remains limited. Most research protocols span 12-24 weeks followed by structured discontinuation. The ideal dose and dosing for tesofensine in extended protocols (>6 months) requires enhanced monitoring and periodic reassessment of risk-benefit ratios.
Is tesofensine safe for everyone?
No. Tesofensine has specific contraindications:
❌ Absolute contraindications:
- Uncontrolled hypertension
- Significant cardiovascular disease
- History of stroke or heart attack
- Concurrent MAO inhibitor use
- Pregnancy or breastfeeding
- Severe psychiatric disorders
⚠️ Relative contraindications (require careful evaluation):
- Controlled hypertension
- Anxiety disorders
- Sleep disorders
- Concurrent stimulant use
Pre-implementation screening is essential to identify individuals for whom tesofensine research is inappropriate.
Future Directions in Tesofensine Research
As we progress through 2026, research into the ideal dose and dosing for tesofensine continues to evolve. Several promising areas of investigation may refine dosing protocols:
Personalized Dosing Algorithms
Emerging research explores genetic markers that predict tesofensine response:
- Dopamine transporter (DAT) gene polymorphisms
- Norepinephrine transporter variations
- CYP450 enzyme profiles affecting metabolism
Future protocols may incorporate genetic testing to optimize starting doses and predict optimal maintenance levels, moving beyond the current one-size-fits-all titration approach.
Extended-Release Formulations
Current research investigates modified-release tesofensine formulations that may:
- Reduce peak cardiovascular effects
- Provide more stable plasma levels
- Potentially improve tolerability
- Allow for different dosing schedules
These formulations could alter the ideal dose and dosing for tesofensine, potentially enabling lower total daily doses with comparable efficacy.
Combination Therapy Protocols
Systematic investigation of tesofensine combined with complementary compounds is expanding:
- Metabolic peptide combinations: Tesofensine + 5-Amino-1MQ or AOD-9604
- Mitochondrial support: Tesofensine + MOTS-C for enhanced energy metabolism
- Synergistic blends: Pre-formulated combinations with optimized ratios
As combination research matures, dosing protocols will need refinement to account for synergistic effects and potential interaction profiles.
Population-Specific Protocols
Ongoing research is developing specialized dosing guidelines for:
- Older adults: Conservative protocols with extended titration
- Metabolic syndrome: Enhanced monitoring with potential insulin sensitivity benefits
- Athletic populations: Performance and body composition optimization
- Post-weight loss maintenance: Lower doses for weight stability rather than active loss
These population-specific approaches will refine our understanding of the ideal dose and dosing for tesofensine across diverse research contexts.
For researchers interested in staying current with evolving peptide science, exploring comprehensive resources from established peptide research suppliers provides access to the latest protocols and formulations.
Conclusion: Implementing Evidence-Based Tesofensine Protocols
Understanding the ideal dose and dosing for tesofensine represents the cornerstone of safe, effective research with this powerful metabolic compound. As we've explored throughout this comprehensive guide, tesofensine's triple monoamine reuptake inhibition mechanism offers remarkable potential—but only when implemented with precision, respect for individual variation, and commitment to rigorous monitoring.
Key Principles for Success
Start low, go slow: The 0.25mg starting dose with gradual titration to 0.5mg (or occasionally 1.0mg) remains the evidence-based standard. This conservative approach minimizes cardiovascular side effects while allowing individual tolerance assessment.
Prioritize safety: Cardiovascular monitoring isn't optional—it's essential. Regular heart rate and blood pressure assessment, combined with periodic ECG evaluation, protects research integrity and participant wellbeing.
Individualize protocols: While standard guidelines provide the framework, the ideal dose and dosing for tesofensine varies based on body composition, metabolic health, age, concurrent medications, and individual sensitivity. Flexibility within evidence-based parameters is crucial.
Maintain consistency: Once optimal dosing is established, consistent daily administration at the same time (preferably morning) maximizes results and maintains stable neurotransmitter modulation.
Monitor comprehensively: Beyond cardiovascular parameters, tracking body composition, metabolic markers, sleep quality, and subjective experiences provides holistic assessment of protocol effectiveness and tolerance.
Actionable Next Steps
For researchers, fitness professionals, and medi-spa practitioners considering tesofensine protocols:
-
Conduct thorough pre-implementation assessment: Comprehensive cardiovascular and metabolic evaluation establishes baseline and identifies contraindications
-
Source research-grade compounds: Work with verified peptide suppliers that provide third-party testing documentation and proper handling guidelines
-
Develop detailed monitoring protocols: Establish clear schedules for cardiovascular checks, metabolic assessments, and safety evaluations
-
Start with standard titration: Begin at 0.25mg daily for 1-2 weeks, escalate to 0.5mg if appropriate, and consider 1.0mg only with excellent tolerance and specific research justification
-
Document systematically: Maintain detailed records of dosing, monitoring parameters, side effects, and outcomes to inform protocol adjustments
-
Plan discontinuation: Include structured tapering and post-discontinuation monitoring in initial protocol design
-
Stay current with research: The field of metabolic peptides evolves rapidly—ongoing education ensures protocols reflect latest evidence
The Bigger Picture
Tesofensine represents one component of the expanding toolkit available to researchers exploring metabolic optimization. When combined with other evidence-based approaches—whether complementary peptides, lifestyle interventions, or comprehensive metabolic support—tesofensine's effects can be maximized while minimizing risks.
The ideal dose and dosing for tesofensine isn't merely about numbers on a scale or milligrams in a capsule. It's about understanding mechanism, respecting individual variation, prioritizing safety, and implementing protocols with scientific rigor. As research continues to refine our understanding through 2026 and beyond, these principles will remain constant even as specific recommendations evolve.
For those embarking on tesofensine research, the journey begins with education, continues with careful implementation, and succeeds through commitment to evidence-based practices. The remarkable metabolic effects observed in clinical trials are achievable—but only when the ideal dose and dosing for tesofensine is implemented with the precision and respect this powerful compound demands.
SEO Meta Information
Meta Title: Tesofensine Dosing Guide 2026: Ideal Dose & Safety Protocol
Meta Description: Complete evidence-based guide to ideal tesofensine dosing. Clinical protocols, titration schedules, safety monitoring, and practical implementation for 2026 research.
Keywords: ideal dose and dosing for tesofensine, tesofensine dosage, tesofensine protocol, tesofensine safety, metabolic peptides, weight loss compounds, tesofensine titration, peptide research
<!DOCTYPE html>
<html lang="en">
<head>
<meta charset="UTF-8">
<meta name="viewport" content="width=device-width, initial-scale=1.0">
<title>Tesofensine Dosing Calculator</title>
<style>
* {
margin: 0;
padding: 0;
box-sizing: border-box;
}
.cg-calculator-container {
font-family: -apple-system, BlinkMacSystemFont, 'Segoe UI', Roboto, Oxygen, Ubuntu, Cantarell, sans-serif;
max-width: 800px;
margin: 0 auto;
padding: 20px;
background: linear-gradient(135deg, #667eea 0%, #764ba2 100%);
border-radius: 16px;
box-shadow: 0 10px 40px rgba(0,0,0,0.2);
}
.cg-calculator-header {
text-align: center;
color: white;
margin-bottom: 30px;
}
.cg-calculator-header h2 {
font-size: 28px;
margin-bottom: 10px;
}
.cg-calculator-header p {
font-size: 14px;
opacity: 0.9;
}
.cg-calculator-content {
background: white;
padding: 30px;
border-radius: 12px;
}
.cg-input-group {
margin-bottom: 25px;
}
.cg-input-group label {
display: block;
font-weight: 600;
color: #333;
margin-bottom: 8px;
font-size: 15px;
}
.cg-input-group select,
.cg-input-group input {
width: 100%;
padding: 12px;
border: 2px solid #e0e0e0;
border-radius: 8px;
font-size: 15px;
transition: border-color 0.3s;
}
.cg-input-group select:focus,
.cg-input-group input:focus {
outline: none;
border-color: #667eea;
}
.cg-checkbox-group {
margin-bottom: 20px;
}
.cg-checkbox-label {
display: flex;
align-items: center;
margin-bottom: 10px;
cursor: pointer;
}
.cg-checkbox-label input[type="checkbox"] {
width: 20px;
height: 20px;
margin-right: 10px;
cursor: pointer;
}
.cg-calculate-btn {
width: 100%;
padding: 15px;
background: linear-gradient(135deg, #667eea 0%, #764ba2 100%);
color: white;
border: none;
border-radius: 8px;
font-size: 16px;
font-weight: 600;
cursor: pointer;
transition: transform 0.2s;
}
.cg-calculate-btn:hover {
transform: translateY(-2px);
}
.cg-results {
margin-top: 30px;
padding: 25px;
background: #f8f9fa;
border-radius: 12px;
display: none;
}
.cg-results.active {
display: block;
}
.cg-result-item {
margin-bottom: 20px;
padding: 15px;
background: white;
border-radius: 8px;
border-left: 4px solid #667eea;
}
.cg-result-item h3 {
color: #667eea;
font-size: 18px;
margin-bottom: 8px;
}
.cg-result-item p {
color: #555;
line-height: 1.6;
margin-bottom: 5px;
}
.cg-warning {
background: #fff3cd;
border-left-color: #ffc107;
padding: 15px;
border-radius: 8px;
margin-top: 15px;
}
.cg-warning h4 {
color: #856404;
margin-bottom: 8px;
}
.cg-warning ul {
margin-left: 20px;
color: #856404;
}
.cg-timeline {
margin-top: 20px;
}
.cg-timeline-item {
display: flex;
margin-bottom: 15px;
align-items: flex-start;
}
.cg-timeline-marker {
width: 40px;
height: 40px;
background: #667eea;
color: white;
border-radius: 50%;
display: flex;
align-items: center;
justify-content: center;
font-weight: bold;
margin-right: 15px;
flex-shrink: 0;
}
.cg-timeline-content {
flex: 1;
}
.cg-timeline-content h4 {
color: #333;
margin-bottom: 5px;
}
.cg-timeline-content p {
color: #666;
font-size: 14px;
}
@media (max-width: 600px) {
.cg-calculator-container {
padding: 15px;
}
.cg-calculator-content {
padding: 20px;
}
.cg-calculator-header h2 {
font-size: 24px;
}
}
</style>
</head>
<body>
<div class="cg-calculator-container">
<div class="cg-calculator-header">
<h2>🧬 Tesofensine Dosing Protocol Calculator</h2>
<p>Evidence-based dosing recommendations for research protocols</p>
</div>
<div class="cg-calculator-content">
<div class="cg-input-group">
<label for="cg-weight">Body Weight (kg):</label>
<input type="number" id="cg-weight" min="40" max="200" value="80" placeholder="Enter weight in kg">
</div>
<div class="cg-input-group">
<label for="cg-experience">Research Experience Level:</label>
<select id="cg-experience">
<option value="beginner">Beginner (First peptide protocol)</option>
<option value="intermediate">Intermediate (Some peptide experience)</option>
<option value="advanced">Advanced (Extensive research background)</option>
</select>
</div>
<div class="cg-input-group">
<label for="cg-age">Age Range:</label>
<select id="cg-age">
<option value="young">18-40 years</option>
<option value="middle">41-60 years</option>
<option value="older">60+ years</option>
</select>
</div>
<div class="cg-input-group">
<label for="cg-goal">Primary Research Goal:</label>
<select id="cg-goal">
<option value="weight">Weight loss research</option>
<option value="metabolic">Metabolic optimization</option>
<option value="appetite">Appetite regulation study</option>
<option value="combination">Combination protocol</option>
</select>
</div>
<div class="cg-checkbox-group">
<label style="font-weight: 600; color: #333; margin-bottom: 10px; display: block;">Risk Factors (check all that apply):</label>
<label class="cg-checkbox-label">
<input type="checkbox" id="cg-cardiovascular" value="cardiovascular">
<span>Cardiovascular concerns or history</span>
</label>
<label class="cg-checkbox-label">
<input type="checkbox" id="cg-blood-pressure" value="blood-pressure">
<span>Elevated blood pressure</span>
</label>
<label class="cg-checkbox-label">
<input type="checkbox" id="cg-anxiety" value="anxiety">
<span>Anxiety or sleep disorders</span>
</label>
<label class="cg-checkbox-label">
<input type="checkbox" id="cg-medications" value="medications">
<span>Taking other medications</span>
</label>
</div>
<button class="cg-calculate-btn" onclick="calculateDosing()">Generate Personalized Protocol</button>
<div id="cg-results" class="cg-results">
<!-- Results will be inserted here -->
</div>
</div>
</div>
<script>
function calculateDosing() {
// Get input values
const weight = parseFloat(document.getElementById('cg-weight').value);
const experience = document.getElementById('cg-experience').value;
const age = document.getElementById('cg-age').value;
const goal = document.getElementById('cg-goal').value;
// Get risk factors
const riskFactors = [];
if (document.getElementById('cg-cardiovascular').checked) riskFactors.push('cardiovascular');
if (document.getElementById('cg-blood-pressure').checked) riskFactors.push('blood-pressure');
if (document.getElementById('cg-anxiety').checked) riskFactors.push('anxiety');
if (document.getElementById('cg-medications').checked) riskFactors.push('medications');
// Calculate recommendations
let startingDose = 0.25;
let maintenanceDose = 0.5;
let titrationWeeks = 2;
let maxDose = 1.0;
let warnings = [];
// Adjust based on risk factors
if (riskFactors.length > 0) {
titrationWeeks = 3;
warnings.push('Extended titration recommended due to risk factors');
}
if (riskFactors.includes('cardiovascular') || riskFactors.includes('blood-pressure')) {
maxDose = 0.5;
warnings.push('Maximum dose limited to 0.5mg due to cardiovascular concerns');
}
// Adjust based on age
if (age === 'older') {
titrationWeeks = 4;
maxDose = 0.5;
warnings.push('Conservative protocol recommended for older adults');
}
// Adjust based on experience
if (experience === 'beginner') {
titrationWeeks = Math.max(titrationWeeks, 3);
warnings.push('Extended monitoring recommended for first-time researchers');
}
// Generate timeline
let timeline = [
{
week: '1-2',
dose: `${startingDose}mg`,
description: 'Starting dose phase. Monitor cardiovascular parameters daily. Assess tolerance and side effects.'
}
];
if (maintenanceDose > startingDose) {
timeline.push({
week: `${titrationWeeks + 1}-${titrationWeeks + 2}`,
dose: `${maintenanceDose}mg`,
description: 'Titration to maintenance dose. Continue daily monitoring. Evaluate efficacy and tolerance.'
});
}
if (maxDose > maintenanceDose && experience !== 'beginner' && riskFactors.length === 0) {
timeline.push({
week: `${titrationWeeks + 3}+`,
dose: `Up to ${maxDose}mg`,
description: 'Optional escalation if needed. Enhanced monitoring required. Consider risk-benefit carefully.'
});
}
timeline.push({
week: '12+',
dose: 'Maintenance',
description: 'Long-term phase. Reduce monitoring frequency to bi-weekly, then monthly. Assess cumulative outcomes.'
});
// Display results
displayResults(startingDose, maintenanceDose, maxDose, titrationWeeks, timeline, warnings, riskFactors);
}
function displayResults(startingDose, maintenanceDose, maxDose, titrationWeeks, timeline, warnings, riskFactors) {
const resultsDiv = document.getElementById('cg-results');
let warningsHTML = '';
if (warnings.length > 0 || riskFactors.length > 0) {
warningsHTML = `
<div class="cg-warning">
<h4>⚠️ Important Considerations</h4>
<ul>
${warnings.map(w => `<li>${w}</li>`).join('')}
${riskFactors.length > 0 ? '<li>Enhanced cardiovascular monitoring required throughout protocol</li>' : ''}
<li>This calculator provides general guidance only - consult qualified professionals</li>
</ul>
</div>
`;
}
resultsDiv.innerHTML = `
<div class="cg-result-item">
<h3>📊 Recommended Dosing Protocol</h3>
<p><strong>Starting Dose:</strong> ${startingDose}mg daily (morning administration)</p>
<p><strong>Target Maintenance Dose:</strong> ${maintenanceDose}mg daily</p>
<p><strong>Maximum Dose:</strong> ${maxDose}mg daily</p>
<p><strong>Titration Period:</strong> ${titrationWeeks} weeks at starting dose before escalation</p>
</div>
<div class="cg-result-item">
<h3>📅 Implementation Timeline</h3>
<div class="cg-timeline">
${timeline.map((item, index) => `
<div class="cg-timeline-item">
<div class="cg-timeline-marker">${index + 1}</div>
<div class="cg-timeline-content">
<h4>Week ${item.week}: ${item.dose}</h4>
<p>${item.description}</p>
</div>
</div>
`).join('')}
</div>
</div>
<div class="cg-result-item">
<h3>🔬 Monitoring Requirements</h3>
<p><strong>Daily (Weeks 1-4):</strong> Resting heart rate, blood pressure, subjective assessment</p>
<p><strong>Weekly (Weeks 1-4):</strong> Body weight, side effect review, protocol adherence</p>
<p><strong>Bi-weekly (Weeks 5-12):</strong> Cardiovascular parameters, body composition</p>
<p><strong>Monthly:</strong> Comprehensive metabolic panel, ECG (if risk factors present)</p>
</div>
${warningsHTML}
<div class="cg-result-item">
<h3>✅ Next Steps</h3>
<p>1. Complete pre-implementation cardiovascular and metabolic assessment</p>
<p>2. Source research-grade tesofensine from verified suppliers</p>
<p>3. Establish monitoring schedule and data collection systems</p>
<p>4. Begin protocol at recommended starting dose</p>
<p>5. Document all parameters systematically throughout protocol</p>
</div>
`;
resultsDiv.classList.add('active');
resultsDiv.scrollIntoView({ behavior: 'smooth', block: 'nearest' });
}
</script>
</body>
</html>
