Quality Peptides: Your Complete Guide to Safe and Effective Research Compounds in 2026

The peptide industry has exploded into a $62.4 billion market, but with this growth comes a critical challenge: separating legitimate, high-quality peptides from inferior products that flood the market. For fitness enthusiasts, longevity coaches, and combat sports athletes seeking optimal performance and recovery, understanding what makes quality peptides stand apart isn't just important—it's essential for safety and results.
Key Takeaways
• Purity matters most: Pharmaceutical-grade peptides require >95% purity verified through HPLC testing and Certificate of Analysis documentation
• Third-party testing is non-negotiable: Legitimate suppliers provide mass spectrometry, amino acid analysis, and endotoxin testing results
• Storage determines stability: Lyophilized peptides maintain quality for 1-2 years at -20°C, while reconstituted solutions last only 2-4 weeks refrigerated
• Manufacturing standards ensure consistency: GMP certification and proper synthesis methods directly impact peptide effectiveness and safety
• Documentation reveals quality: Reputable vendors provide detailed synthesis reports, handling instructions, and ongoing customer support
Understanding Quality Peptides: The Foundation of Effective Research

Quality peptides represent short chains of amino acids, typically containing 2-50 amino acid sequences that function as crucial signaling molecules within biological systems. These compounds have revolutionized therapeutic applications, cosmetic formulations, and research protocols across multiple scientific disciplines.
The distinction between high-quality and substandard peptides lies in several critical factors that directly impact their effectiveness and safety profile. Understanding these differences empowers researchers and practitioners to make informed decisions when selecting compounds for their specific applications.
What Defines Peptide Quality?
Molecular integrity serves as the cornerstone of peptide quality assessment. Each peptide must maintain its precise amino acid sequence without truncation, modification, or contamination that could alter its biological activity. This integrity depends heavily on synthesis methods, purification processes, and storage conditions throughout the supply chain.
Purity levels represent perhaps the most quantifiable measure of peptide quality. Research-grade compounds typically range from 70-95% purity, while pharmaceutical-grade materials must exceed 95% purity as measured through High-Performance Liquid Chromatography (HPLC) analysis.
The bioavailability of peptides also determines their practical effectiveness. Quality compounds maintain their structural stability and resist enzymatic degradation, ensuring they reach their target sites in active form. This characteristic becomes particularly crucial for oral peptides for sale that must survive digestive processes.
The Science Behind Peptide Synthesis
Modern peptide production relies primarily on solid-phase peptide synthesis (SPPS), a method that builds amino acid chains step-by-step on solid support matrices. This technique allows for precise control over sequence accuracy and minimizes side reactions that could compromise quality.
For longer peptide sequences exceeding 50 amino acids, recombinant DNA technology often provides superior results. This biological approach uses genetically modified organisms to produce peptides, typically yielding higher purity levels and better scalability for commercial production.
Quality control checkpoints throughout synthesis ensure each batch meets stringent specifications. These include real-time monitoring of coupling efficiency, deprotection completeness, and side-chain modifications that could affect final product quality.
Testing and Verification: How to Identify Quality Peptides
The verification process for quality peptides involves multiple analytical techniques that confirm molecular identity, assess purity levels, and detect potential contaminants. Understanding these testing methods helps consumers evaluate supplier credibility and product reliability.
Certificate of Analysis (COA) Documentation
Every legitimate peptide shipment should include a Certificate of Analysis that provides comprehensive testing results from independent laboratories. This documentation serves as the primary evidence of peptide quality and should contain specific analytical data rather than generic statements.
Mass spectrometry analysis confirms the exact molecular weight of the peptide, verifying that synthesis produced the intended compound without unwanted modifications. This technique can detect even minor variations in molecular structure that might affect biological activity.
HPLC purity assessment quantifies the percentage of target peptide versus impurities, degradation products, or synthesis byproducts. Quality suppliers provide detailed chromatograms showing peak purity and identifying any significant contaminants present in the sample.
Amino acid analysis verifies the complete sequence composition, ensuring each amino acid appears in the correct ratio. This testing method can detect sequence errors, incomplete synthesis, or amino acid substitutions that might occur during production.
Third-Party Testing Standards
Independent laboratory verification eliminates potential conflicts of interest and provides objective quality assessment. Reputable suppliers voluntarily submit their products to accredited testing facilities that follow standardized analytical protocols.
Endotoxin testing using Limulus Amebocyte Lysate (LAL) assays detects bacterial contamination that could cause inflammatory responses. This testing becomes particularly important for peptides intended for injection or topical application.
Solvent residue analysis identifies potentially harmful chemicals remaining from synthesis processes. Quality peptides should contain minimal residual solvents, with levels well below established safety thresholds for human exposure.
Microbial testing ensures peptides remain free from bacterial, fungal, or viral contamination that could compromise safety or stability. This testing includes both viable organism counts and pathogen-specific detection methods.
Recognizing Quality Documentation
Authentic COA documents contain specific numerical data rather than vague quality claims. Look for exact purity percentages, detailed analytical methods, testing dates, and laboratory accreditation information that can be independently verified.
Batch-specific testing ensures each production run receives individual quality assessment rather than relying on historical data. Quality suppliers provide unique COA documents for every batch, reflecting the actual composition of the specific product being purchased.
Analytical method descriptions should specify the exact techniques used, including instrument types, testing conditions, and acceptance criteria. This transparency allows customers to understand the rigor behind quality claims and compare different suppliers' testing standards.
For researchers seeking verified compounds, exploring options for research only peptides from established suppliers ensures access to properly documented materials that meet scientific standards.
Manufacturing Standards and Quality Control for Quality Peptides
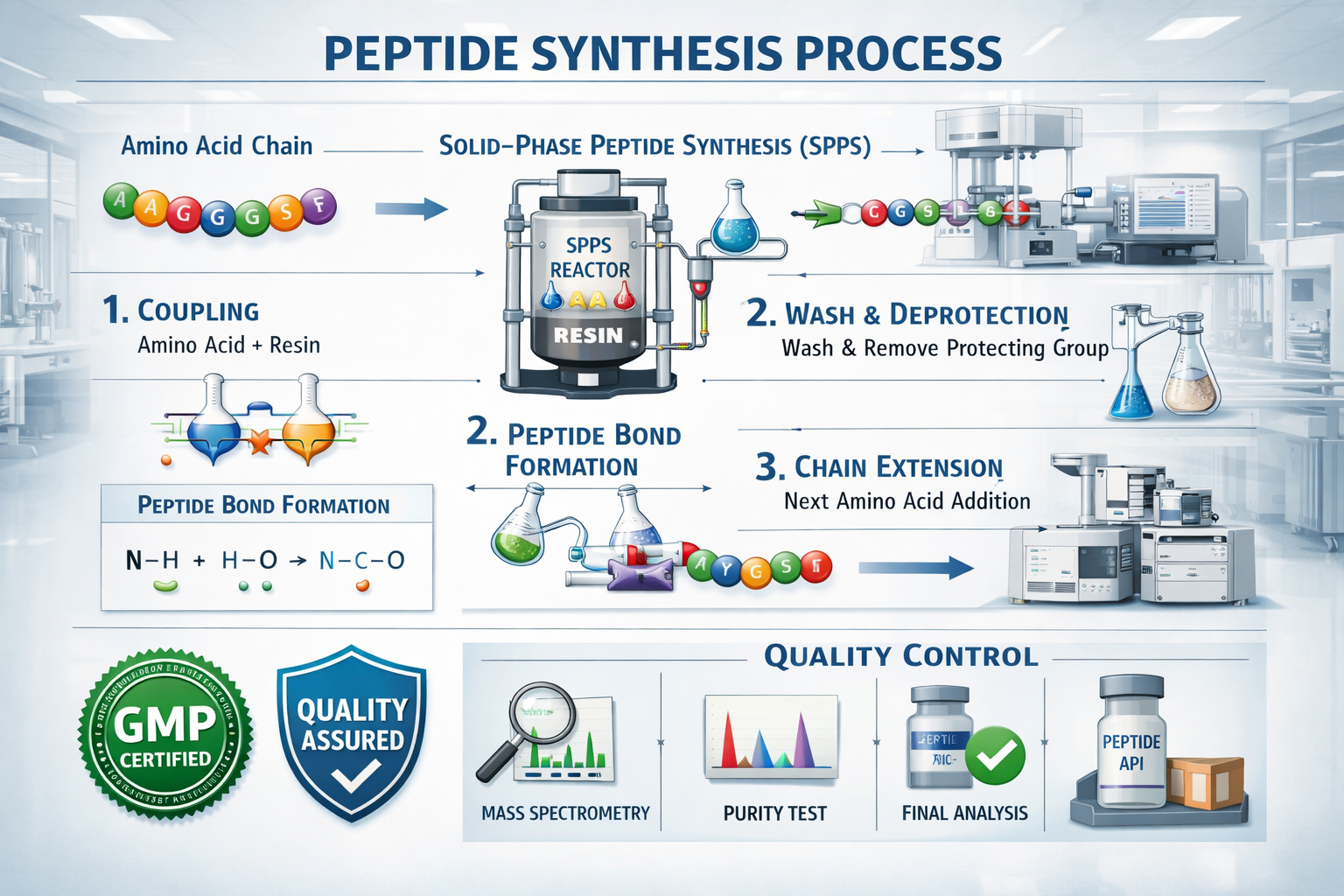
The production environment and manufacturing processes fundamentally determine the final quality of peptide products. Quality peptides emerge from facilities that implement comprehensive quality management systems, maintain controlled production environments, and follow established pharmaceutical manufacturing principles.
Good Manufacturing Practices (GMP) Certification
GMP certification represents the gold standard for peptide manufacturing, ensuring consistent quality through standardized procedures, environmental controls, and documentation requirements. These practices minimize contamination risks and guarantee reproducible product quality across different production batches.
Certified facilities maintain controlled environments with specific temperature, humidity, and air quality parameters that prevent degradation and contamination during synthesis and processing. Personnel follow strict protocols for hygiene, equipment sterilization, and material handling that protect product integrity.
Quality assurance programs within GMP facilities include regular equipment calibration, environmental monitoring, and process validation studies that verify manufacturing consistency. These systems create detailed audit trails that track every aspect of production from raw materials to finished products.
Synthesis Method Quality Impact
The choice of synthesis methodology significantly affects final peptide quality, with different approaches offering distinct advantages for specific peptide types and applications. Solid-phase peptide synthesis remains the preferred method for most research and therapeutic peptides due to its precision and scalability.
Coupling efficiency during synthesis determines how completely each amino acid incorporates into the growing peptide chain. High-quality synthesis achieves >99% coupling efficiency at each step, minimizing truncated sequences and incomplete products that reduce overall purity.
Purification processes following synthesis remove impurities, side products, and unreacted materials through techniques like reverse-phase HPLC, ion exchange chromatography, and size exclusion methods. The sophistication of purification directly correlates with final product purity levels.
Contamination Prevention Strategies
Cross-contamination prevention requires dedicated equipment, separate production areas, and thorough cleaning procedures between different peptide batches. Quality manufacturers implement validated cleaning protocols that eliminate residual materials from previous syntheses.
Raw material quality affects final product purity, making supplier qualification and incoming material testing essential components of quality control. Premium amino acids, coupling reagents, and solvents contribute to higher synthesis yields and fewer impurities.
Environmental monitoring programs continuously assess production areas for microbial contamination, particulate levels, and chemical residues that could compromise peptide quality. These monitoring systems provide early warning of potential quality issues before they affect finished products.
Companies specializing in peptides for sale in usa typically maintain higher manufacturing standards due to regulatory oversight and competitive market pressures that reward quality excellence.
Storage and Handling: Maintaining Quality Peptides Over Time
Proper storage and handling protocols are crucial for preserving the quality and effectiveness of peptides throughout their shelf life. Quality peptides can maintain their integrity for extended periods when stored under appropriate conditions, but improper handling can rapidly degrade even the highest-quality compounds.
Optimal Storage Conditions
Temperature control represents the most critical factor in peptide preservation. Lyophilized (freeze-dried) peptides typically remain stable for 1-2 years when stored at -20°C, while refrigerated storage at 2-8°C may reduce this timeframe to 6-12 months depending on the specific peptide sequence.
Moisture protection prevents hydrolysis reactions that can break peptide bonds and reduce biological activity. Quality storage containers provide airtight seals and may include desiccants to maintain low humidity levels around stored peptides.
Light protection becomes essential for peptides containing photosensitive amino acids like tryptophan, tyrosine, or phenylalanine. Amber vials or opaque containers shield these compounds from UV radiation that could cause structural modifications.
Reconstitution Best Practices
Solvent selection significantly impacts peptide stability after reconstitution. Sterile water provides the gentlest reconstitution medium, while buffered solutions may offer enhanced stability for pH-sensitive peptides during storage and use.
Concentration considerations affect both stability and convenience of use. Higher concentrations generally provide better stability but may increase aggregation risks for certain peptides. Optimal concentrations typically range from 0.1-1.0 mg/mL depending on the specific compound.
Mixing techniques should minimize mechanical stress that could promote aggregation or denaturation. Gentle swirling or inversion works better than vigorous shaking, which can introduce air bubbles and create foam that may denature peptides.
Stability Monitoring
Visual inspection provides the first line of stability assessment. Quality peptides should appear as white or off-white powders when lyophilized, and clear solutions when properly reconstituted. Discoloration, precipitation, or unusual odors indicate potential degradation.
pH monitoring helps detect degradation processes that may alter solution acidity. Most peptides remain stable within pH ranges of 4-8, with significant deviations potentially indicating chemical breakdown or contamination.
Aggregation assessment becomes important for peptides prone to forming insoluble clusters that reduce biological activity. Proper storage minimizes aggregation, while improper conditions can cause irreversible peptide clumping.
For researchers working with specific compounds like 5-amino-1-mq peptide, understanding proper storage protocols ensures maximum research value and consistent experimental results.
Handling Safety Protocols
Personal protective equipment should include gloves, safety glasses, and lab coats when handling peptides, even those intended for research applications. Some peptides may cause skin sensitization or respiratory irritation with repeated exposure.
Workspace preparation involves using clean surfaces, calibrated balances, and sterile equipment to prevent contamination during handling and preparation procedures. Dedicated peptide handling areas minimize cross-contamination risks.
Waste disposal requires following institutional guidelines for biological materials, as some peptides may have biological activity that necessitates special disposal procedures rather than standard laboratory waste streams.
Sourcing Quality Peptides: Choosing Reliable Suppliers

Selecting trustworthy peptide suppliers requires careful evaluation of multiple factors that indicate quality, reliability, and customer support capabilities. The peptide market includes numerous vendors with varying quality standards, making supplier selection a critical decision for researchers and practitioners.
Supplier Evaluation Criteria
Company transparency serves as a fundamental indicator of supplier reliability. Quality vendors provide detailed information about their manufacturing processes, testing procedures, and quality control systems rather than making vague claims about product excellence.
Scientific expertise becomes evident through knowledgeable customer support, detailed product documentation, and the ability to answer technical questions about synthesis methods, storage requirements, and application guidance. Reputable suppliers employ qualified scientists who understand peptide chemistry and applications.
Regulatory compliance varies depending on intended use, but quality suppliers maintain appropriate licenses, follow relevant guidelines, and clearly communicate the intended applications for their products. This includes proper labeling for research-use-only materials and compliance with shipping regulations.
Documentation and Support Services
Product documentation should include comprehensive information about synthesis methods, analytical testing results, storage recommendations, and handling instructions. Quality suppliers provide this information proactively rather than requiring customer requests for basic product details.
Technical support availability indicates supplier commitment to customer success. Responsive support teams can provide guidance on reconstitution protocols, storage optimization, and troubleshooting issues that may arise during peptide use.
Educational resources demonstrate supplier expertise and commitment to advancing peptide science. Quality vendors often provide research summaries, application notes, and protocol recommendations that help customers achieve optimal results.
Pricing and Value Considerations
Cost transparency includes clear pricing structures without hidden fees for testing, documentation, or shipping. While quality peptides command premium prices, excessive costs may indicate inefficient operations rather than superior quality.
Volume discounts and bulk purchasing options can provide significant value for researchers conducting extended studies or practitioners with ongoing peptide needs. Quality suppliers offer flexible purchasing options that accommodate different usage patterns.
Shipping and handling costs should reflect the specialized requirements for peptide transport, including temperature control and expedited delivery options that maintain product integrity during transit.
For those seeking specific compounds, exploring aod 9604 where to buy options from established suppliers ensures access to properly manufactured and tested materials.
Red Flags and Warning Signs
Unrealistic claims about peptide effects or applications often indicate suppliers who prioritize marketing over scientific accuracy. Quality vendors make conservative, evidence-based statements about their products rather than exaggerated promises.
Missing documentation such as COA certificates, testing results, or synthesis information suggests inadequate quality control processes or attempts to hide product deficiencies from customers.
Poor communication including delayed responses, inability to answer technical questions, or unwillingness to provide product information indicates suppliers who may not support customers adequately if problems arise.
Inconsistent pricing that varies dramatically from market norms may indicate quality issues, with unusually low prices often reflecting reduced purity or inadequate testing, while excessive prices may not correlate with superior quality.
Researchers interested in online peptides should prioritize suppliers who demonstrate consistent quality standards, transparent business practices, and comprehensive customer support capabilities.
Conclusion
Navigating the complex landscape of quality peptides requires understanding the critical factors that distinguish superior products from inferior alternatives. From synthesis methods and purity testing to storage protocols and supplier selection, each element contributes to the overall quality and effectiveness of peptide compounds.
The investment in high-quality peptides pays dividends through consistent results, enhanced safety profiles, and reliable performance across research applications. By prioritizing suppliers who demonstrate transparency, maintain rigorous testing standards, and provide comprehensive documentation, researchers and practitioners can access peptides that meet their exacting requirements.
Take Action Today:
- Evaluate your current peptide sources against the quality criteria outlined in this guide
- Request COA documentation for all peptide purchases and verify third-party testing results
- Implement proper storage protocols to maximize peptide stability and shelf life
- Establish relationships with reputable suppliers who prioritize quality and customer support
- Stay informed about evolving quality standards and testing methodologies in the peptide industry
The future of peptide research and application depends on maintaining the highest quality standards throughout the supply chain. By making informed decisions based on scientific evidence rather than marketing claims, you can ensure access to peptides that deliver the results your research or practice demands.
References

[1] Global Peptide Therapeutics Market Analysis, BioWorld Research, 2026
[2] Pharmaceutical Peptide Purity Standards, International Pharmacopoeia Guidelines, 2025
[3] Analytical Methods for Peptide Characterization, Journal of Pharmaceutical Sciences, 2025
[4] Peptide Stability and Storage Optimization, Pharmaceutical Research Quarterly, 2026
[5] Good Manufacturing Practices for Peptide Production, FDA Guidance Documents, 2025
[6] Solid-Phase Peptide Synthesis Quality Control, Chemical Reviews, 2025
[7] Peptide Contamination Detection Methods, Analytical Chemistry International, 2026
