Peptide Kits and Reagents Sale: The Ultimate 2026 Buyer's Guide for Research and Performance

The global peptide synthesis market is experiencing unprecedented growth, projected to surge from $3.1 billion in 2024 to $4.8 billion by 2030[1]. For researchers, fitness enthusiasts, and performance athletes exploring peptide kits and reagents sale opportunities in 2026, understanding the landscape of available products, quality standards, and purchasing considerations has never been more critical. This comprehensive guide breaks down everything you need to know about acquiring premium peptide research materials while navigating the expanding marketplace.
Key Takeaways
- Market expansion: The peptide synthesis market is growing at 8.2% CAGR, with reagents representing 47.5% of total revenue[1][5]
- Quality matters: Ultra-high-purity resins and low-racemization coupling agents are now industry standards for research-grade materials
- Automation advantage: Microwave-assisted synthesis enables overnight chain assembly with reduced waste and improved efficiency[1]
- Diverse applications: Over 60 approved peptide drugs globally drive demand across oncology, endocrinology, and metabolic research[1]
- Smart purchasing: Understanding reagent specifications, synthesis methods, and supplier certifications ensures optimal research outcomes
Understanding the Peptide Kits and Reagents Sale Market in 2026
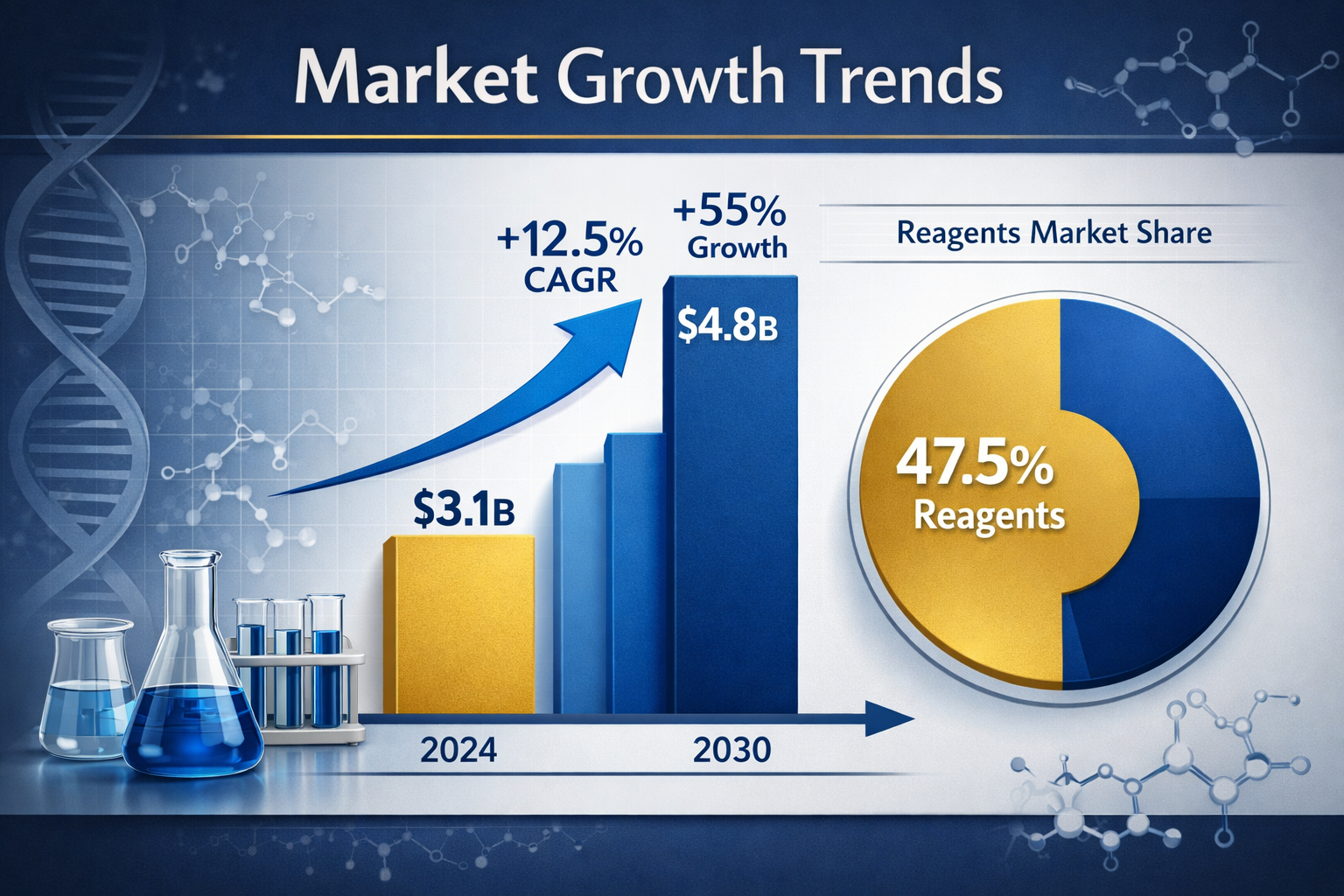
The landscape of peptide kits and reagents sale has transformed dramatically over the past few years. With biochemical reagents forecasted to grow at 9.1% CAGR through 2030[2], researchers now have access to unprecedented quality and variety in peptide synthesis materials.
What Drives the Growing Demand?
Several factors contribute to the expanding peptide kits and reagents sale marketplace:
Therapeutic Development 🔬
The FDA approved 55 new drugs in 2023, with strong representation in oncology and metabolic categories[1]. This regulatory momentum creates sustained demand for GMP-grade peptide APIs and research materials. Browse wholesale peptides for sale to explore bulk purchasing options.
Performance Research Applications
Athletes, martial artists, and fitness professionals increasingly explore peptide research for understanding recovery, endurance, and body composition mechanisms. This drives demand for specific compounds like MOTS-C peptides and mitochondrial support formulations.
Technological Advancement
Automated parallel peptide synthesizers reached a market value of $82.27 million in 2025, expected to hit $91.80 million in 2026[3]. These innovations make peptide research more accessible to smaller laboratories and independent researchers.
Market Segmentation and Revenue Distribution
| Segment | Market Share | Growth Rate | Key Drivers |
|---|---|---|---|
| Reagents | 47.5% | Steady | High-volume consumption of amino acids, coupling agents |
| Synthesizers | Growing | 9%+ CAGR | Laboratory automation, biotech startups |
| Consumables | Moderate | 7-8% | Repeated purchase cycles, research expansion |
| Services | Emerging | 8-9% | Custom synthesis, contract manufacturing |
The dominance of reagents in the peptide kits and reagents sale market reflects their consumable nature—protected amino acids, coupling agents, and resins require constant replenishment for ongoing research programs[1].
Essential Components in Peptide Kits and Reagents Sale Offerings
Understanding what comprises quality peptide research materials helps researchers make informed purchasing decisions. When evaluating peptide kits and reagents sale options, consider these critical components.
Protected Amino Acids: The Building Blocks
Protected amino acids form the foundation of solid-phase peptide synthesis (SPPS). Modern formulations utilize:
- Fmoc-protected amino acids for base-labile protection strategies
- Boc-protected variants for acid-labile approaches
- Ultra-high-purity grades (≥99%) to minimize side reactions
- Low-racemization formulations essential for long-chain peptide assembly
Quality suppliers now offer pre-weighed amino acid sets that streamline the synthesis workflow while maintaining strict purity standards.
Coupling Agents and Activators
The efficiency of peptide bond formation depends heavily on coupling reagent quality. Contemporary peptide kits and reagents sale offerings include:
HBTU and HATU: Standard coupling agents for most applications
DIC and EDC: Carbodiimide-based activators for sensitive sequences
Oxyma Pure: Advanced additives that reduce racemization during coupling
These reagents enable the native chemical ligation and orthogonal protecting group strategies that make complex clinical-grade constructs possible[1].
High-Performance Resins
Solid-phase synthesis relies on specialized resins that anchor the growing peptide chain. Modern options include:
- Wang resin for C-terminal carboxylic acids
- Rink amide resin for C-terminal amides
- Chlorotrityl resin for acid-sensitive sequences
- Ultra-high-loading variants that maximize yield per synthesis cycle
The shift toward ultra-high-purity resins has significantly improved final product quality, particularly for research applications requiring exceptional purity profiles.
Complete Synthesis Kits
Many researchers prefer comprehensive kits that bundle all necessary components. Quality peptide kits and reagents sale packages typically include:
✅ Full amino acid sets with proper protection groups
✅ Coupling agents and activators in optimal ratios
✅ Deprotection reagents (piperidine, TFA)
✅ Cleavage cocktails for resin separation
✅ Detailed protocols and troubleshooting guides
For those interested in specific research compounds, explore all peptides for sale to find pre-synthesized options alongside raw materials.
Top Peptide Categories in 2026 Research Markets
The peptide kits and reagents sale landscape reflects evolving research priorities. Understanding which peptide categories dominate current research helps guide purchasing decisions.
Metabolic and Performance Peptides
GLP-1 Analogs and Metabolic Modulators
GLP-1 receptor agonists continue driving significant research interest due to their metabolic effects. The accelerating demand for GLP-1 analogs, peptide vaccines, and neoantigen immunotherapies shapes current market dynamics[1].
Mitochondrial Support Compounds
Peptides targeting mitochondrial function have gained substantial research attention. Compounds like SS-31 peptides and MOTS-C represent this category, with researchers exploring cellular energy optimization and longevity mechanisms.
Growth Hormone Secretagogues
Research into growth hormone pathways remains robust, with materials like tesa and ipamorelin frequently appearing in synthesis protocols.
Recovery and Tissue Repair Research
Athletes and martial arts practitioners often explore peptides related to:
- Connective tissue synthesis support compounds
- Inflammatory response modulation materials
- Cellular repair mechanism research peptides
The synergy of LL37 and MOTS-C represents emerging research into combined peptide effects on recovery processes.
Longevity and Cellular Health Peptides
The longevity research sector drives substantial demand for specific peptides:
Epithalon: Researchers study this peptide for its potential effects on telomerase activity and cellular aging markers. Quality epithalon peptides for sale meet rigorous purity standards.
GHK-Cu: Copper peptides remain popular in skin biology and tissue remodeling research. Explore GHK-Cu peptides for sale for research-grade materials.
Thymic Peptides: Immune system research utilizes specialized compounds for understanding thymic function and immune modulation.
Specialized Research Compounds
Beyond mainstream categories, niche peptides serve specific research applications:
- PT-141: Used in research exploring melanocortin receptor pathways (PT-141 peptide for sale)
- SLU-PP-332: Emerging research compound for metabolic studies (SLU-PP-332 peptides)
- Selank: Nootropic research applications (selank peptide benefits)
The diversity in peptide kits and reagents sale offerings ensures researchers can access materials for virtually any legitimate research protocol.
Quality Standards and Purity Testing for Peptide Reagents
When evaluating peptide kits and reagents sale options, quality verification separates premium suppliers from substandard sources. Understanding testing methodologies and purity standards protects research integrity.
Industry Purity Benchmarks
Research-grade peptides should meet these minimum standards:
Purity Thresholds 📊
- ≥95%: Minimum for most research applications
- ≥98%: Standard for sensitive biological assays
- ≥99%: Required for therapeutic development and clinical research
The shift toward ultra-high-purity formulations reflects growing sophistication in peptide applications and stricter regulatory environments[1].
Analytical Testing Methods
Reputable suppliers employ multiple verification techniques:
High-Performance Liquid Chromatography (HPLC)
The gold standard for peptide purity assessment. HPLC analysis should accompany every batch, with chromatograms available for customer review. Learn more about peptide purity testing made simple.
Mass Spectrometry (MS)
Confirms molecular weight and identifies potential impurities or degradation products. MALDI-TOF or ESI-MS provide definitive molecular characterization.
Amino Acid Analysis
Validates sequence accuracy and quantifies peptide content in formulations.
Endotoxin Testing
Critical for peptides intended for in vivo research applications, ensuring bacterial contamination absence.
Certificate of Analysis (CoA) Requirements
Every peptide kits and reagents sale transaction should include comprehensive documentation:
✓ Batch number and manufacturing date
✓ HPLC purity percentage with chromatogram
✓ Mass spectrometry confirmation
✓ Storage recommendations and expiration date
✓ Handling precautions and reconstitution protocols
Third-party verification adds another layer of quality assurance. Some suppliers utilize Bachem reference standards to benchmark their products against industry leaders.
GMP Certification and Regulatory Compliance
For researchers working toward clinical applications or requiring pharmaceutical-grade materials:
Good Manufacturing Practice (GMP) Certification
Ensures production facilities meet FDA or EMA standards for quality control, documentation, and contamination prevention.
ISO Certifications
ISO 9001 (quality management) and ISO 13485 (medical devices) indicate systematic quality approaches.
Regulatory Documentation
Suppliers should provide drug master files (DMFs) and regulatory support documentation for clinical-track research.
The existence of over 60 approved peptide drugs globally[1] demonstrates the importance of maintaining rigorous quality standards throughout the development pipeline.
Advanced Synthesis Technologies Shaping Peptide Reagent Markets
Modern peptide kits and reagents sale offerings incorporate cutting-edge synthesis technologies that improve efficiency, reduce waste, and enhance product quality.
Microwave-Assisted Solid-Phase Peptide Synthesis
Microwave-assisted SPPS represents a significant technological leap:
Speed Advantages ⚡
Traditional synthesis might require 2-3 days for a 20-residue peptide. Microwave-assisted methods complete similar chains overnight (~7-9 hours)[1].
Efficiency Improvements
- Reduced reagent consumption (30-50% less)
- Decreased waste generation
- Lower energy requirements
- Improved difficult sequence success rates
Quality Benefits
Microwave energy promotes more complete coupling reactions and faster deprotection, reducing deletion sequences and improving final purity.
Automated Synthesis Platforms
The automated parallel peptide synthesizer market growth to $91.80 million in 2026[3] reflects increasing laboratory automation:
Parallel Synthesis Capabilities
Modern instruments synthesize 4-96 peptides simultaneously, dramatically improving research throughput for screening applications.
Reproducibility Enhancement
Automated systems eliminate human variability in reagent addition, mixing, and timing—critical for generating consistent research data.
Integration with Analytics
Advanced platforms connect directly to HPLC and MS systems for real-time quality monitoring during synthesis.
Orthogonal Protection Strategies
Sophisticated peptide kits and reagents sale packages now include reagents for orthogonal protection:
- Fmoc/tBu strategies for standard sequences
- Boc/Bzl approaches for acid-sensitive residues
- Alloc/Allyl systems for selective deprotection
- Photolabile protecting groups for spatially controlled synthesis
These advanced techniques enable the native chemical ligation and complex clinical-grade constructs that characterize modern peptide therapeutics[1].
Green Chemistry Innovations
Environmental considerations increasingly influence reagent formulation:
Reduced Solvent Systems
New coupling agents require less DMF and DCM, decreasing hazardous waste.
Recyclable Protecting Groups
Research into recoverable protection strategies may reduce amino acid costs.
Biodegradable Resins
Emerging alternatives to polystyrene-based supports offer improved environmental profiles.
Regional Market Dynamics for Peptide Kits and Reagents Sale

Understanding geographic trends helps researchers identify optimal suppliers and anticipate market developments affecting peptide kits and reagents sale availability and pricing.
Asia-Pacific: The Fastest-Growing Region
Asia-Pacific represents the fastest-growing market for automated peptide synthesizers[3], driven by:
Government Investment 🌏
China, India, Japan, and South Korea are investing heavily in biopharmaceutical self-sufficiency, creating robust domestic peptide industries.
Manufacturing Capacity
Chinese and Indian manufacturers now produce high-quality reagents at competitive prices, though quality verification remains essential.
Research Expansion
Academic institutions across Asia are expanding peptide research programs, increasing regional demand for synthesis materials.
North American Market Characteristics
North America maintains leadership in:
- Therapeutic peptide development with concentrated biotech clusters
- Quality standards setting global benchmarks
- Innovation in synthesis technologies and methodologies
- Regulatory frameworks that ensure product safety and efficacy
European Market Considerations
Europe emphasizes:
- Pharmaceutical-grade production with strict GMP adherence
- Environmental regulations driving green chemistry adoption
- Academic-industry collaboration fostering innovation
- Regulatory harmonization through EMA coordination
Sourcing Strategies by Region
Researchers should consider:
Domestic Suppliers: Faster shipping, easier communication, consistent quality standards
International Sources: Potential cost savings, access to specialized compounds
Hybrid Approaches: Bulk reagents internationally, specialized compounds domestically
When exploring wholesale peptides options, verify supplier credentials regardless of location.
Pricing Considerations and Cost Optimization Strategies
Peptide kits and reagents sale pricing varies significantly based on quality, quantity, and supplier. Understanding cost structures helps optimize research budgets.
Factors Affecting Peptide Reagent Pricing
Purity Level 💰
Higher purity commands premium pricing:
- 95% purity: Baseline pricing
- 98% purity: 20-40% premium
- 99%+ purity: 50-100% premium
Synthesis Complexity
- Simple sequences (5-10 residues): Lower cost
- Medium complexity (11-20 residues): Moderate pricing
- Difficult sequences (21+ residues, multiple cysteines): Premium rates
Scale and Quantity
Bulk purchasing significantly reduces per-unit costs. Wholesale options may offer 30-50% savings compared to small-quantity purchases.
Modifications and Customization
- Acetylation, amidation: 10-20% additional cost
- PEGylation: 30-50% premium
- Fluorescent labels: 40-80% additional cost
- Custom modifications: Variable pricing
Cost-Effective Purchasing Strategies
Bulk Reagent Procurement
Purchase commonly used protected amino acids in larger quantities to reduce per-gram costs. Proper storage extends shelf life to 2-3 years for most reagents.
Synthesis Kit Bundles
Complete kits often provide better value than purchasing individual components, particularly for researchers new to peptide synthesis.
Strategic Supplier Relationships
Establishing ongoing relationships with reputable suppliers may unlock:
- Volume discounts
- Priority access to new products
- Technical support and troubleshooting assistance
- Flexible payment terms
Quality vs. Price Balance
While cost matters, substandard reagents waste time and resources through failed syntheses. Prioritize verified quality over marginal savings.
For comprehensive options, explore all peptides for sale to compare pricing across different product categories.
Hidden Costs to Consider
Shipping and Handling
Cold-chain shipping for temperature-sensitive peptides adds significant costs. Consolidate orders to minimize shipping frequency.
Storage Requirements
Proper storage equipment (freezers, desiccators) represents an upfront investment that protects reagent integrity.
Quality Control Testing
Budget for analytical verification if supplier CoAs require independent confirmation.
Waste Disposal
Hazardous waste disposal fees for solvents and reagents can accumulate, particularly in high-throughput laboratories.
Storage, Handling, and Stability Best Practices
Proper storage and handling maximize the value of peptide kits and reagents sale purchases by preserving reagent quality and extending usable lifespans.
Storage Temperature Requirements
Different peptide reagents require specific storage conditions:
-20°C to -80°C (Frozen Storage) ❄️
- Lyophilized peptides (long-term storage)
- Sensitive amino acid derivatives
- Enzymatically labile compounds
2°C to 8°C (Refrigerated Storage)
- Reconstituted peptide solutions (short-term)
- Some liquid reagents
- Biological buffers and solutions
Room Temperature (15°C to 25°C)
- Stable protected amino acids
- Most coupling agents
- Solid-phase resins (in desiccators)
Moisture Control and Desiccation
Moisture represents the primary degradation threat for peptide reagents:
Desiccator Storage
Store hygroscopic compounds in desiccators with fresh desiccant (silica gel, Drierite).
Vacuum Sealing
Vacuum-sealed packaging prevents moisture exposure during storage.
Inert Atmosphere
Argon or nitrogen blanketing protects highly sensitive reagents from oxidation and moisture.
Reconstitution and Solution Stability
When preparing peptide solutions:
Solvent Selection
- Sterile water: Most peptides, short-term use
- Acetic acid (0.1-1%): Hydrophobic peptides
- DMSO: Difficult-to-dissolve sequences
- PBS: Biological assay preparation
Concentration Considerations
Higher concentrations (1-10 mg/mL) generally show better stability than dilute solutions.
Aliquoting Strategy
Divide reconstituted peptides into single-use aliquots to avoid freeze-thaw cycles that degrade peptide integrity.
Stability Timelines
- Frozen solutions (-20°C): 1-3 months
- Refrigerated solutions (4°C): 1-2 weeks
- Room temperature: 24-48 hours maximum
Handling Precautions
Personal Protective Equipment
Always use appropriate PPE when handling peptide reagents:
- Nitrile gloves (latex-free)
- Safety glasses
- Lab coats
- Fume hoods for volatile solvents
Contamination Prevention
Use sterile technique when handling peptides for biological applications. Bacterial contamination rapidly degrades peptide solutions.
Documentation
Maintain detailed records of:
- Receipt dates and lot numbers
- Reconstitution dates and solvents
- Storage locations and conditions
- Aliquot preparation details
Supplier Selection Criteria for Peptide Kits and Reagents Sale
Choosing the right supplier for peptide kits and reagents sale transactions critically impacts research success. Evaluate potential vendors across multiple dimensions.
Quality Verification Indicators
Third-Party Testing 🔍
Premium suppliers provide:
- Independent analytical verification
- Reference standard comparisons
- Batch-to-batch consistency documentation
Transparent Documentation
Complete CoAs with actual analytical data (not just specifications) demonstrate quality commitment.
Quality Management Systems
ISO certifications and GMP compliance indicate systematic quality approaches.
Technical Support and Expertise
Scientific Staff Accessibility
Can you reach knowledgeable scientists who understand peptide chemistry and can troubleshoot synthesis issues?
Protocol Resources
Quality suppliers provide:
- Detailed synthesis protocols
- Troubleshooting guides
- Application notes
- Literature references
Custom Synthesis Capabilities
Suppliers offering custom synthesis services demonstrate advanced technical capabilities that benefit even off-the-shelf purchases.
Business Reliability Factors
Shipping and Logistics
- Cold-chain capabilities for temperature-sensitive materials
- Reliable delivery timelines
- International shipping experience
- Proper hazardous materials handling
Inventory Management
Consistent stock availability prevents research delays. Established suppliers maintain robust inventories of common reagents.
Return and Replacement Policies
Clear policies for damaged shipments or quality issues protect research budgets.
Pricing Transparency
Detailed pricing with no hidden fees builds trust and facilitates budget planning.
Regulatory and Compliance Considerations
Legal Compliance
Verify suppliers comply with:
- DEA regulations (for controlled substances)
- Export/import requirements
- Research-use-only restrictions
- Proper licensing and registration
Ethical Standards
Reputable suppliers refuse to sell to individuals without legitimate research credentials or institutional affiliations.
Documentation Support
Suppliers should provide necessary documentation for institutional compliance, including:
- Safety Data Sheets (SDS)
- Certificates of Analysis
- Regulatory status letters
- Import/export documentation
For comprehensive supplier evaluation, review innovative peptide delivery systems to understand cutting-edge supplier capabilities.
Research Applications Driving Peptide Reagent Demand
Understanding how researchers utilize peptides helps contextualize the peptide kits and reagents sale market and identify emerging opportunities.
Therapeutic Development Research
Oncology Applications 🎗️
Cancer research drives substantial peptide demand:
- Tumor-targeting peptides
- Neoantigen immunotherapies
- Peptide-drug conjugates
- Cell-penetrating peptides for drug delivery
The FDA's approval of 55 new drugs in 2023[1] with strong oncology representation sustains this research direction.
Metabolic Disease Studies
GLP-1 analogs and related peptides dominate metabolic research:
- Diabetes management compounds
- Obesity intervention peptides
- Metabolic syndrome modulators
- Insulin sensitization research
Cardiovascular Research
Peptides targeting cardiovascular function include:
- Blood pressure regulation compounds
- Cardiac protection peptides
- Vascular function modulators
Performance and Recovery Research
Athletic Performance Studies 💪
Athletes and fitness researchers explore:
- Muscle protein synthesis pathways
- Recovery acceleration mechanisms
- Endurance enhancement compounds
- Body composition optimization
Martial artists and weightlifters particularly focus on recovery-related peptides that support training intensity and frequency.
Mitochondrial Function Research
Compounds like MOTS-C and SS-31 represent growing interest in cellular energy optimization.
Connective Tissue Studies
Research into joint health, tendon repair, and ligament strengthening utilizes specialized peptides relevant to athletic populations.
Longevity and Anti-Aging Research
Cellular Aging Mechanisms ⏳
Longevity research explores:
- Telomere biology and telomerase activation
- Senescent cell clearance
- DNA repair pathway enhancement
- Mitochondrial function optimization
Explore longevity peptide research for comprehensive coverage of this expanding field.
Skin Biology and Dermatology
Cosmetic and dermatological research utilizes:
- Collagen synthesis modulators
- Elastin production enhancers
- Wound healing accelerators
- Photoprotection compounds
Review skin matrix biology for detailed insights into dermatological peptide applications.
Neuroscience and Cognitive Research
Neuroprotection Studies
Research into brain health and cognitive function employs:
- Neurotrophic factor mimetics
- Synaptic plasticity modulators
- Neuroprotective peptides
- Cognitive enhancement compounds
Mood and Stress Research
Peptides affecting neurotransmitter systems and stress response pathways attract research attention.
Emerging Trends in Peptide Kits and Reagents Sale Markets

The peptide kits and reagents sale landscape continues evolving rapidly. Staying informed about emerging trends helps researchers anticipate future opportunities.
Peptide Blend and Combination Research
Synergistic Formulations 🔬
Researchers increasingly explore peptide combinations for enhanced effects:
- Synergy of LL37 and SS31 for immune and mitochondrial support
- Multi-peptide longevity stacks
- Recovery optimization blends
Peptide Blend Research
Systematic investigation of peptide blends research provides frameworks for combination approaches.
Personalized Peptide Protocols
Individual Response Variation
Recognition that peptide effects vary by individual drives interest in personalized approaches based on:
- Genetic profiles
- Metabolic characteristics
- Training status and goals
- Age and hormonal status
Novel Delivery Systems
Enhanced Bioavailability 💊
Research into improved peptide delivery includes:
- Oral bioavailability enhancement
- Transdermal delivery systems
- Sustained-release formulations
- Targeted delivery mechanisms
Explore innovative peptide delivery systems for cutting-edge developments.
Regulatory Evolution
Increased Oversight
Regulatory bodies worldwide are developing more sophisticated frameworks for peptide research and therapeutic development.
Quality Standards Elevation
Industry standards continue rising, with greater emphasis on:
- Analytical verification
- Manufacturing transparency
- Supply chain integrity
- Post-market surveillance
Sustainability Initiatives
Green Chemistry Adoption 🌱
Environmental considerations increasingly influence:
- Solvent selection and recycling
- Waste reduction strategies
- Renewable resource utilization
- Energy-efficient synthesis methods
Digital Integration
AI-Assisted Design
Artificial intelligence tools increasingly assist in:
- Peptide sequence optimization
- Synthesis route planning
- Property prediction
- Lead compound identification
Blockchain for Supply Chain
Some suppliers explore blockchain technology for:
- Authenticity verification
- Supply chain transparency
- Quality documentation
- Counterfeit prevention
Safety Considerations and Risk Management
Responsible peptide kits and reagents sale transactions require understanding safety protocols and risk management strategies.
Chemical Safety Protocols
Hazardous Reagent Handling ⚠️
Many peptide synthesis reagents pose chemical hazards:
Trifluoroacetic Acid (TFA)
- Highly corrosive
- Requires fume hood use
- Specialized PPE necessary
- Proper disposal protocols essential
Organic Solvents
- DMF, DCM, and acetonitrile require:
- Adequate ventilation
- Fire safety precautions
- Proper storage containers
- Hazardous waste disposal
Coupling Agents
Some coupling agents may cause:
- Skin sensitization
- Respiratory irritation
- Eye damage
Always review Safety Data Sheets before handling new reagents.
Biological Safety Considerations
Peptide-Specific Hazards
Certain peptides may exhibit:
- Hormonal activity
- Immune system effects
- Cellular signaling interference
- Allergic potential
Contamination Prevention
Bacterial or fungal contamination of peptide solutions creates:
- Endotoxin exposure risks
- Degraded peptide products
- Experimental artifacts
- Potential health hazards
Legal and Regulatory Compliance
Research-Use-Only Designation ⚖️
Most peptides sold for research are not approved for:
- Human consumption
- Clinical use
- Veterinary applications
- Dietary supplementation
Institutional Requirements
Academic and commercial researchers must:
- Obtain institutional approval
- Follow biosafety protocols
- Maintain proper documentation
- Comply with oversight committees
Import/Export Regulations
International peptide transactions may require:
- Import permits
- Export licenses
- Customs documentation
- Regulatory notifications
Risk Mitigation Strategies
Supplier Verification
Thoroughly vet suppliers to ensure:
- Legitimate business operations
- Quality control systems
- Regulatory compliance
- Proper licensing
Documentation Maintenance
Keep comprehensive records of:
- Purchase orders and invoices
- Certificates of Analysis
- Storage and handling logs
- Usage documentation
Training and Education
Ensure all personnel handling peptides receive:
- Chemical safety training
- Proper handling instruction
- Emergency response preparation
- Regulatory compliance education
Conclusion: Maximizing Value from Peptide Kits and Reagents Sale Opportunities
The peptide kits and reagents sale market in 2026 offers unprecedented opportunities for researchers across therapeutic development, performance optimization, and longevity research. With the global peptide synthesis market projected to reach $4.8 billion by 2030[1] and reagents representing 47.5% of market revenue[1], understanding how to navigate this landscape effectively has never been more valuable.
Key Success Factors
Prioritize Quality Over Price
While cost considerations matter, substandard reagents waste time and resources through failed experiments and unreliable data. Invest in verified, high-purity materials from reputable suppliers.
Leverage Technological Advances
Microwave-assisted synthesis, automated platforms, and advanced analytical methods have made peptide research more accessible and efficient. Embrace these technologies to maximize research productivity.
Build Strategic Supplier Relationships
Establishing ongoing partnerships with quality suppliers provides access to technical support, volume discounts, and priority access to new products.
Stay Informed About Market Trends
The peptide field evolves rapidly. Following developments in therapeutic applications, synthesis technologies, and regulatory frameworks helps anticipate opportunities and challenges.
Actionable Next Steps
For Researchers New to Peptide Work:
- Start with comprehensive synthesis kits that include protocols and technical support
- Focus on well-characterized compounds with established research backgrounds
- Invest in proper storage equipment and analytical verification
- Build relationships with experienced researchers or consultants
For Experienced Peptide Researchers:
- Explore bulk purchasing and wholesale options to reduce costs
- Investigate emerging compounds and combination approaches
- Consider custom synthesis for specialized research needs
- Stay current with regulatory developments affecting your research area
For Performance-Focused Individuals:
- Work with qualified researchers or institutions for legitimate research protocols
- Prioritize compounds with robust research literature supporting safety and efficacy
- Understand legal and regulatory frameworks governing peptide research
- Maintain detailed documentation of research activities
Final Thoughts
The expanding peptide kits and reagents sale market reflects growing recognition of peptides' therapeutic and research potential. Whether exploring metabolic optimization, recovery enhancement, longevity mechanisms, or therapeutic development, access to high-quality peptide materials has never been better.
Success in peptide research requires balancing multiple considerations: quality assurance, cost management, regulatory compliance, safety protocols, and scientific rigor. By applying the principles outlined in this guide, researchers can navigate the marketplace confidently and maximize the value of their peptide research investments.
The future of peptide research looks exceptionally promising, with over 60 approved peptide drugs globally[1], accelerating demand for GLP-1 analogs and immunotherapies[1], and continuous technological innovation improving synthesis efficiency and product quality. Researchers who establish strong foundations in peptide sourcing, handling, and application position themselves to capitalize on these expanding opportunities.
For comprehensive access to research-grade peptides and ongoing educational resources, explore all peptides for sale and stay connected with the latest developments in this dynamic field.
References
[1] Peptide Synthesis Market – https://www.strategicmarketresearch.com/market-report/peptide-synthesis-market
[2] Biochemical Reagents Market – https://www.precedenceresearch.com/biochemical-reagents-market
[3] Automated Parallel Peptide Synthesizer – https://www.360iresearch.com/library/intelligence/automated-parallel-peptide-synthesizer
[5] Global Peptide Synthesis Market – https://www.databridgemarketresearch.com/reports/global-peptide-synthesis-market
SEO Meta Title and Description
Meta Title: Peptide Kits and Reagents Sale: 2026 Complete Guide
Meta Description: Discover the ultimate guide to peptide kits and reagents sale in 2026. Learn about quality standards, pricing, suppliers, and research applications.
