
MC4R Activation: Unlocking the Science Behind Metabolic Control and Weight Management
The human body's ability to regulate hunger, satiety, and energy expenditure hinges on a complex network of neural pathways and molecular signals. At the center of this intricate system lies the melanocortin-4 receptor (MC4R), a protein that has captured the attention of researchers and biohackers alike for its profound impact on metabolic health. Recent clinical breakthroughs in 2026 have revealed that MC4R activation represents one of the most promising frontiers in addressing obesity, metabolic dysfunction, and weight management challenges that affect millions worldwide.
Key Takeaways
• MC4R activation serves as a master regulator of appetite, energy expenditure, and body weight through hypothalamic signaling pathways
• Recent 2026 clinical data shows that combining MC4R agonists with existing therapies like tirzepatide produces superior weight loss results compared to single-agent treatments
• Next-generation oral MC4R agonists demonstrate approximately 50% bioavailability and extended half-life, offering convenient dosing options for patients
• FDA-approved setmelanotide represents the first successful MC4R-targeted therapy for rare genetic obesity disorders
• Emerging research reveals that MC4R activation can help prevent weight regain after discontinuing other weight loss medications
Understanding MC4R: The Body's Metabolic Master Switch
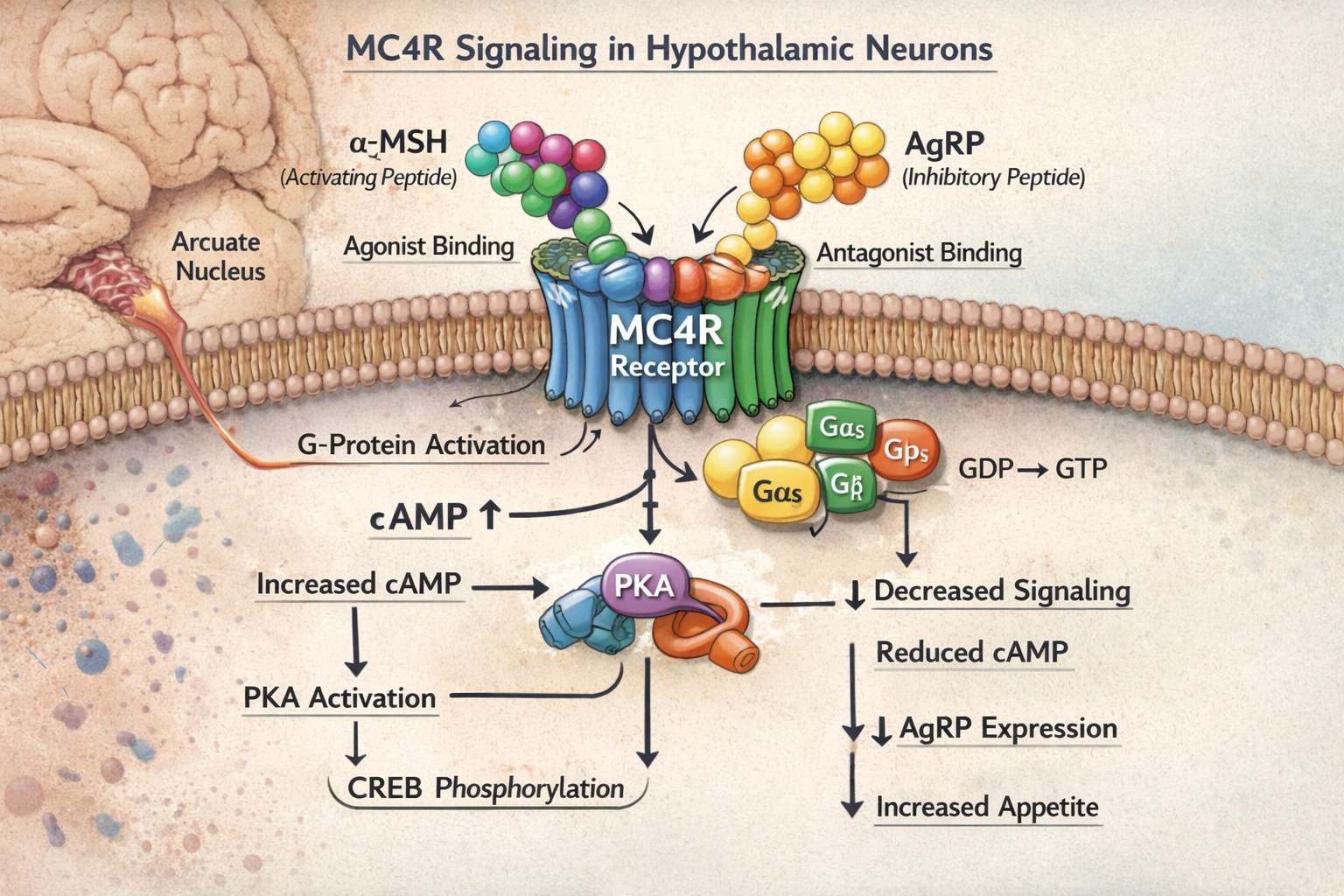
The melanocortin-4 receptor represents a critical component of the brain's appetite control center, located primarily in the hypothalamus. This G-protein coupled receptor responds to specific signaling molecules, including alpha-melanocyte stimulating hormone (α-MSH), to regulate fundamental aspects of energy balance.
When functioning properly, MC4R activation triggers a cascade of cellular events that:
- Reduces appetite and food intake
- Increases energy expenditure through thermogenesis
- Modulates glucose metabolism and insulin sensitivity
- Regulates fat storage and mobilization
The Molecular Mechanism Behind MC4R Function
The process of MC4R activation begins when specific ligands bind to the receptor, causing conformational changes that activate intracellular signaling pathways. This activation primarily works through the cyclic adenosine monophosphate (cAMP) pathway, leading to increased protein kinase A activity and subsequent phosphorylation of key metabolic enzymes.
Research has identified that disruptions in this pathway, whether through genetic mutations or acquired dysfunction, can lead to severe metabolic consequences. Individuals with MC4R pathway deficiencies often experience:
- Early-onset severe obesity
- Hyperphagia (excessive hunger)
- Impaired satiety signals
- Reduced metabolic rate
For those interested in exploring metabolic optimization through peptide therapy, understanding these mechanisms becomes crucial for making informed decisions about comprehensive peptide protocols.
Clinical Breakthroughs in MC4R Activation Research
Revolutionary Combination Therapy Results
The most significant advancement in MC4R activation research emerged from ObesityWeek 2025, where clinical evidence demonstrated that co-administration of low-dose MC4R agonists with tirzepatide produced greater weight loss compared to tirzepatide alone, with no new safety concerns observed.[2][3]
This groundbreaking finding suggests that MC4R activation can enhance the effectiveness of existing incretin-based therapies without increasing adverse effects. The combination approach addresses multiple pathways simultaneously:
| Therapy Component | Primary Mechanism | Key Benefits |
|---|---|---|
| MC4R Agonist | Hypothalamic appetite suppression | Reduced food intake, increased satiety |
| Tirzepatide | GLP-1/GIP receptor activation | Improved glucose control, gastric emptying |
| Combined Effect | Synergistic metabolic modulation | Enhanced weight loss, sustained results |
PL7737: The Next-Generation Oral MC4R Agonist
Palatin's development of PL7737 represents a significant leap forward in MC4R activation technology. This oral MC4R agonist demonstrates robust weight loss and safety in preclinical models with approximately 50% oral bioavailability and a half-life exceeding 3 hours in rats.[2][3]
The advantages of oral MC4R activation include:
- Improved patient compliance through convenient dosing
- Consistent plasma levels with extended half-life
- Reduced injection site reactions compared to subcutaneous options
- Scalable manufacturing for broader patient access
Clinical development timelines indicate that PL7737 IND-enabling toxicology studies are underway, with IND submission and Phase 1 trials planned for the first half of 2026, with clinical data expected in the second half of 2026.[2][3]
Sustained Weight Maintenance Benefits
One of the most promising aspects of MC4R activation involves its ability to prevent weight regain after discontinuing other weight loss medications. This sustained weight maintenance benefit addresses a critical challenge in obesity management, where patients often experience weight rebound after stopping treatment.[2][3]
The mechanism behind this sustained effect likely involves:
- Metabolic reprogramming at the cellular level
- Enhanced insulin sensitivity that persists beyond treatment
- Improved appetite regulation through neural pathway modifications
- Increased baseline metabolic rate through thermogenic activation
For individuals exploring advanced metabolic interventions, combining MC4R activation strategies with other proven peptide therapies may offer synergistic benefits.
Therapeutic Applications and FDA-Approved Treatments
Setmelanotide: The First FDA-Approved MC4R Therapy
Setmelanotide (Imcivree) represents a landmark achievement in MC4R activation therapeutics, receiving FDA approval for treating rare MC4R pathway diseases including POMC, PCSK1, LEPR, and BBS deficiencies.[4] These genetic conditions are characterized by hyperphagia, impaired satiety, and early-onset obesity.
The selectivity advantage of setmelanotide for MC4R over other melanocortin receptors results in reduced cardiovascular side effects compared to earlier agonists like LY2112688, which is crucial for obesity patients with cardiovascular comorbidities.[1]
Hypothalamic Obesity: A Specialized Application
Recent preclinical research demonstrates that setmelanotide can reactivate suppressed MC4R neurons via CaMKK2/AMPK signaling pathways in rat models of hypothalamic obesity caused by hypothalamic injury.[1] This finding opens new therapeutic avenues for patients with acquired hypothalamic dysfunction.
MC4R activation in hypothalamic obesity works by:
- Restoring neural sensitivity to satiety signals
- Reactivating dormant metabolic pathways through AMPK signaling
- Improving energy expenditure despite hypothalamic damage
- Reducing compensatory hyperphagia that typically follows brain injury
Phase 1 studies for next-generation MC4R agonists are expected to include patients with hypothalamic obesity, expanding beyond current genetic disorder indications.[2]
Expanding Clinical Applications
The success of MC4R activation in rare genetic disorders has paved the way for broader therapeutic applications. Researchers are investigating potential benefits for:
- Common obesity without genetic mutations
- Metabolic syndrome and insulin resistance
- Type 2 diabetes management
- Age-related metabolic decline
For biohackers interested in metabolic optimization, understanding these expanding applications can inform decisions about incorporating advanced peptide strategies into their protocols.
Future Directions and Next-Generation Therapies
Weekly Dosing Formulations
Palatin is advancing selective peptide MC4R agonists designed for once-weekly subcutaneous dosing, with IND filing and Phase 1 trials planned for mid-2026.[2][3] These next-generation formulations offer several advantages:
- Enhanced patient adherence through reduced dosing frequency
- Sustained therapeutic levels with extended-release technology
- Improved quality of life for long-term users
- Reduced healthcare costs through simplified administration
Combination Therapy Protocols
The well-tolerated combination of MC4R agonists with tirzepatide has opened new possibilities for multi-target therapeutic approaches. Future protocols may include:
- Triple-agonist combinations targeting GLP-1, GIP, and MC4R pathways
- Personalized dosing algorithms based on genetic profiles
- Sequential therapy protocols optimizing timing and duration
- Biomarker-guided adjustments for individual response optimization
Precision Medicine Applications
As our understanding of MC4R activation deepens, precision medicine approaches are becoming increasingly viable. Future developments may include:
- Genetic testing to identify optimal candidates for MC4R therapy
- Pharmacogenomic profiling to predict individual responses
- Real-time monitoring of metabolic parameters
- AI-driven optimization of dosing and timing protocols
For those exploring cutting-edge metabolic interventions, staying informed about these developments while working with qualified healthcare providers remains essential. The integration of MC4R activation strategies with other proven approaches like growth hormone releasing peptides may offer synergistic benefits for metabolic optimization.
Safety Considerations and Clinical Monitoring
Cardiovascular Safety Profile
One of the most significant advantages of modern MC4R activation therapies lies in their improved cardiovascular safety profile compared to earlier melanocortin receptor agonists. The selectivity of current drugs like setmelanotide for MC4R over other melanocortin receptors has substantially reduced the risk of cardiovascular side effects.[1]
Key safety considerations include:
- Blood pressure monitoring during initiation and dose adjustments
- Heart rate assessment for patients with cardiac conditions
- Electrolyte balance evaluation in long-term users
- Renal function monitoring for optimal drug clearance
Gastrointestinal Tolerance
Clinical data from combination therapy studies shows that MC4R activation does not increase gastrointestinal side effects when used with incretin-based therapies, addressing a common concern with weight loss medications.[2][3] This improved tolerance profile makes MC4R agonists attractive options for patients who cannot tolerate other weight loss interventions.
Long-term Monitoring Protocols
For individuals considering MC4R activation therapies, comprehensive monitoring protocols should include:
- Baseline metabolic panels including glucose, lipids, and inflammatory markers
- Body composition analysis to track fat mass and lean muscle changes
- Hormonal assessments including thyroid function and cortisol levels
- Regular follow-up to assess efficacy and adjust protocols as needed
Those interested in incorporating these approaches should work with healthcare providers familiar with advanced peptide protocols and metabolic optimization strategies.
Conclusion
The field of MC4R activation represents one of the most promising frontiers in metabolic health and weight management. With FDA-approved therapies already available for rare genetic conditions and next-generation oral formulations entering clinical trials in 2026, the therapeutic potential of targeting this crucial metabolic pathway continues to expand.
The recent clinical evidence demonstrating enhanced efficacy when combining MC4R agonists with existing therapies like tirzepatide marks a paradigm shift toward multi-target approaches in obesity management. The sustained weight maintenance benefits observed with MC4R activation address one of the most challenging aspects of long-term weight management.
For fitness enthusiasts and biohackers seeking to optimize their metabolic health, understanding the science behind MC4R activation provides valuable insights into the body's natural weight regulation mechanisms. While these therapies show tremendous promise, working with qualified healthcare providers remains essential for safe and effective implementation.
Next Steps for Implementation
- Consult with a healthcare provider experienced in metabolic medicine and peptide therapies
- Assess your individual risk factors and metabolic profile through comprehensive testing
- Stay informed about emerging clinical trial results and FDA approvals
- Consider combination approaches that may enhance the benefits of MC4R activation
- Monitor progress through regular biomarker assessments and body composition analysis
The future of metabolic health lies in precision approaches that target multiple pathways simultaneously. As MC4R activation therapies continue to evolve, they offer new hope for individuals struggling with metabolic dysfunction and weight management challenges.
References
[1] Pmc12833056 – https://pmc.ncbi.nlm.nih.gov/articles/PMC12833056/
[2] Palatin Presents Data At Obesityweek 2025 Highlighting Promise Of Melanocortin Based Therapies For Obesity – https://palatin.com/press_releases/palatin-presents-data-at-obesityweek-2025-highlighting-promise-of-melanocortin-based-therapies-for-obesity/
[3] Palatin Presents Data At Obesityweek 2025 Highlighting Promise Of Melanocortin Based Therapies For Obesity 302606887 – https://www.prnewswire.com/news-releases/palatin-presents-data-at-obesityweek-2025-highlighting-promise-of-melanocortin-based-therapies-for-obesity-302606887.html
[4] Fda Approval Of Setmelanotide For Patients 2 Years And Older With Mc4r Pathway Diseases – https://checkrare.com/fda-approval-of-setmelanotide-for-patients-2-years-and-older-with-mc4r-pathway-diseases/
Meta Title: MC4R Activation: Science-Based Guide to Metabolic Control 2026
Meta Description: Discover how MC4R activation revolutionizes weight management. Latest 2026 clinical data, FDA-approved therapies, and next-generation treatments explained.
{“@context”:”https://schema.org”,”@type”:”Article”,”headline”:”MC4R activation”,”description”:”Discover how MC4R activation revolutionizes weight management. Latest 2026 clinical data, FDA-approved therapies, and next-generation treatments explained.”,”image”:”https://zsxkvszxbhpwnvzxdydv.supabase.co/storage/v1/object/public/generated-images/kie/50bd39d0-9fed-4605-a60d-b63290064561/slot-0-1770266886897.png”,”datePublished”:”2026-02-05T04:45:42.64441+00:00″,”dateModified”:”2026-02-05T04:48:48.237Z”,”author”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″},”publisher”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″}}
