AOD 9604 Peptide Dosage Chart: Complete Research Guidelines for 2026

The world of peptide research has exploded in recent years, with scientists and researchers seeking precise protocols for studying compounds like AOD 9604. This synthetic peptide fragment, derived from human growth hormone, has captured significant attention in laboratory settings for its unique molecular properties and research applications. Understanding the proper aod 9604 peptide dosage chart is crucial for anyone conducting serious research with this compound.
Key Takeaways
• Standard research dosages typically range from 250-500 micrograms daily, adjusted based on body weight and research objectives
• Administration timing plays a critical role, with most protocols suggesting specific intervals for optimal research outcomes
• Individual factors such as body composition, research goals, and tolerance levels significantly impact dosage requirements
• Safety monitoring remains paramount throughout any research protocol involving AOD 9604 peptide
• Quality sourcing from reputable suppliers ensures consistent research results and proper compound integrity
Understanding AOD 9604 Peptide Fundamentals
What is AOD 9604 Peptide?
AOD 9604 represents a modified fragment of human growth hormone, specifically amino acids 176-191. This aod 9604 peptide has been extensively studied in laboratory settings for its unique molecular characteristics. Unlike its parent hormone, this fragment maintains specific properties while eliminating others, making it particularly interesting for research applications.
The peptide's molecular structure allows researchers to study metabolic processes without the broader systemic effects associated with full growth hormone compounds. Laboratory studies have demonstrated that what is aod 9604 peptide can be described as a selective fragment that maintains certain biological activities while providing a more focused research tool.
Research institutions worldwide have incorporated AOD 9604 studies into their protocols, examining various aspects of cellular metabolism and energy utilization. The compound's stability and predictable behavior in laboratory conditions make it valuable for controlled research environments.
Research Applications and Scientific Interest
Current scientific literature reveals multiple research directions for aod 9604 peptides. Laboratory studies focus on cellular energy processes, metabolic pathway analysis, and molecular interaction studies. Researchers appreciate the compound's specificity, allowing for targeted investigation without interference from broader hormonal cascades.
The peptide's research profile includes studies on cellular metabolism, energy utilization pathways, and molecular signaling mechanisms. These investigations provide valuable insights into fundamental biological processes and contribute to our understanding of peptide-based research methodologies.
Many research facilities have established protocols for studying metabolic research applications using this compound, contributing to the growing body of scientific knowledge in peptide research.
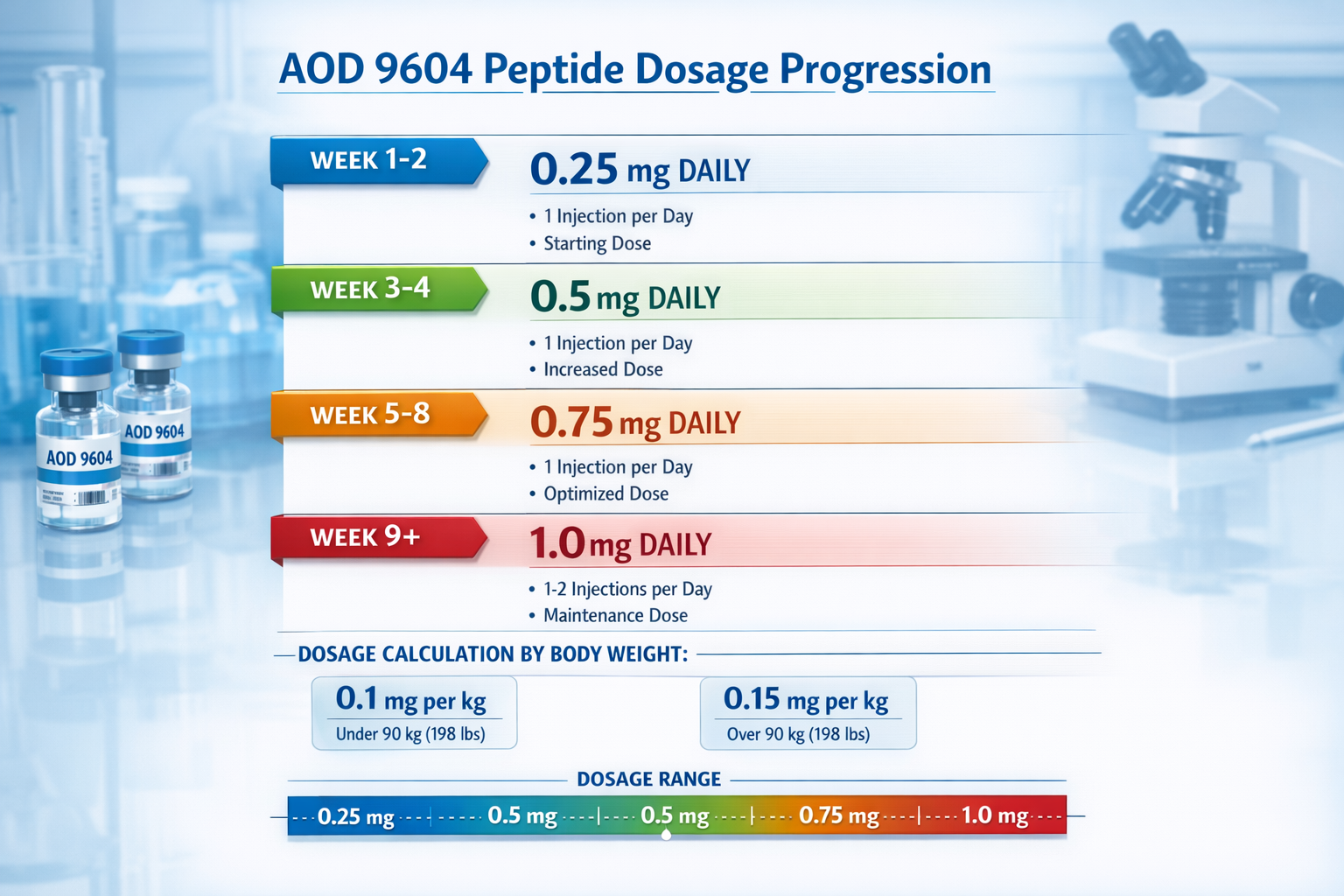
Comprehensive AOD 9604 Peptide Dosage Chart
Standard Research Dosage Protocols
The aod 9604 peptide dosage chart varies significantly based on research objectives, subject characteristics, and study duration. Most laboratory protocols establish baseline measurements before implementing any peptide administration schedule.
Basic Research Dosage Framework:
| Body Weight Range | Starting Dose | Maintenance Dose | Maximum Research Dose |
|---|---|---|---|
| 50-70 kg | 250 mcg | 300-400 mcg | 500 mcg |
| 71-90 kg | 300 mcg | 400-500 mcg | 600 mcg |
| 91-110 kg | 350 mcg | 500-600 mcg | 750 mcg |
| 110+ kg | 400 mcg | 600-750 mcg | 1000 mcg |
These measurements represent typical research parameters found in published studies. Individual research protocols may require adjustments based on specific study objectives and monitoring requirements.
Advanced Dosing Considerations
AOD 9604 peptide dosage calculations must account for multiple variables beyond simple body weight. Research experience, study duration, concurrent compounds, and monitoring capabilities all influence optimal dosing strategies.
Experienced researchers often implement graduated dosing protocols, beginning with conservative measurements and adjusting based on observed responses and research outcomes. This approach allows for comprehensive data collection while maintaining research safety standards.
The aod 9604 peptide dosing schedule typically involves daily administration, though some research protocols explore alternative timing strategies. High-quality peptide sourcing ensures consistent potency and reliable research results.
Timing and Administration Protocols
Research protocols emphasize the importance of consistent timing in aod 9604 peptide therapy studies. Most laboratory schedules implement morning administration on an empty stomach, typically 30-60 minutes before the first meal.
Optimal Administration Timeline:
- Morning Protocol: 6:00-8:00 AM, fasted state
- Pre-workout Research: 30-45 minutes before physical activity
- Evening Protocol: 2-3 hours after last meal (alternative timing)
The timing strategy significantly impacts research outcomes and data quality. Consistent administration schedules provide more reliable data sets and improve research reproducibility across different study phases.
Administration Methods and Bioavailability
Subcutaneous Injection Protocols
Subcutaneous administration remains the gold standard for aod 9604 peptide research due to superior bioavailability and predictable absorption patterns. Research protocols typically specify injection sites, needle specifications, and rotation schedules to ensure consistent results.
Injection Site Rotation Schedule:
- Abdominal area: 2 inches from navel, alternating sides
- Thigh region: Upper outer quadrant, rotating locations
- Upper arm: Posterior aspect, suitable for assisted administration
- Hip area: Upper outer region, alternative site option
Proper injection technique ensures optimal peptide absorption and minimizes research variables. Many laboratories maintain detailed logs of injection sites, timing, and observed responses to track research progress effectively.
Research facilities often provide training on proper peptide handling and storage to maintain compound integrity throughout study periods.
Alternative Administration Routes
While subcutaneous injection provides optimal bioavailability, researchers have explored aod 9604 peptide oral administration and other delivery methods. These alternative routes offer different research opportunities and may suit specific study objectives.
Oral Administration Considerations:
- Reduced bioavailability (approximately 10-15% compared to injection)
- Longer onset time for observable effects
- Potential for digestive enzyme degradation
- Requires higher dosing to achieve equivalent research outcomes
Nasal Spray Applications:
- Moderate bioavailability (30-40% of injection efficiency)
- Rapid absorption through nasal mucosa
- Convenient for frequent dosing protocols
- Requires specialized formulation for stability
Each administration method presents unique research opportunities and challenges. Protocol selection depends on study objectives, available resources, and specific research questions being investigated.
Bioavailability and Absorption Factors
Understanding absorption kinetics helps researchers optimize their aod 9604 peptide dosage chart for specific study objectives. Factors affecting bioavailability include administration route, timing, concurrent substances, and individual physiological variables.
Research data indicates peak plasma concentrations occur 30-60 minutes post-injection, with detectable levels persisting for 4-6 hours. This timeline influences dosing frequency and measurement schedules in research protocols.
Studies examining peptide synergies provide insights into how concurrent compounds may affect absorption and research outcomes.
Safety Considerations and Monitoring
Research Safety Protocols
Implementing comprehensive safety measures ensures responsible aod 9604 peptide research while protecting study integrity. Established research facilities maintain strict protocols for peptide handling, administration, and monitoring throughout study periods.
Essential Safety Measures:
- Pre-study health screening and baseline measurements
- Regular monitoring of vital signs and research parameters
- Detailed documentation of all observations and responses
- Emergency protocols for unexpected reactions or outcomes
- Proper disposal of peptide materials and administration supplies
Research safety extends beyond immediate administration concerns to include proper storage, handling procedures, and documentation requirements. These measures ensure research quality while maintaining ethical standards.
Common Research Observations
Laboratory studies document various observations associated with aod 9604 peptide side effects research. Understanding these potential outcomes helps researchers design appropriate monitoring protocols and safety measures.
Frequently Reported Research Observations:
- Injection site reactions (mild redness, temporary discomfort)
- Transient changes in energy levels or sleep patterns
- Appetite modifications during study periods
- Minor gastrointestinal responses with oral administration
- Occasional headaches or mild fatigue
These observations typically resolve quickly and rarely interfere with research objectives. Proper documentation helps identify patterns and optimize future research protocols.
Researchers studying peptide benefits and applications contribute valuable data to the growing understanding of peptide research safety profiles.
Long-term Research Considerations
Extended research protocols require additional safety considerations and monitoring strategies. Long-term aod 9604 peptide therapy studies provide valuable insights into sustained effects and optimal research methodologies.
Extended Study Protocols:
- Monthly comprehensive assessments of research parameters
- Periodic dosage adjustments based on observed responses
- Regular safety monitoring including laboratory values
- Documentation of cumulative effects and research outcomes
- Structured break periods to assess baseline return
Long-term studies contribute significantly to peptide research knowledge while requiring enhanced safety protocols and monitoring systems.
Optimizing Research Outcomes
Factors Affecting Research Results
Multiple variables influence aod 9604 peptide research outcomes beyond simple dosage considerations. Understanding these factors helps researchers design more effective protocols and achieve consistent results.
Key Research Variables:
- Individual physiological differences affecting peptide response
- Concurrent lifestyle factors that may influence outcomes
- Study duration and timing considerations
- Environmental conditions during research periods
- Measurement techniques and data collection methods
Successful research protocols account for these variables through careful study design, comprehensive monitoring, and detailed documentation procedures.
Research Protocol Optimization
Experienced researchers develop sophisticated approaches to aod 9604 peptide dosage optimization based on study objectives and available resources. These protocols evolve through careful observation and systematic adjustments.
Protocol Development Strategies:
- Baseline establishment through comprehensive pre-study measurements
- Conservative initiation with gradual dosage adjustments
- Systematic monitoring of research parameters and responses
- Data-driven modifications based on observed outcomes
- Comprehensive documentation for protocol refinement
This systematic approach ensures research quality while maximizing the potential for meaningful scientific insights.
Researchers interested in comprehensive peptide research benefit from understanding how different compounds interact and complement each other in research settings.
Advanced Research Applications
Sophisticated research facilities explore complex protocols involving aod 9604 peptides in combination with other research compounds. These advanced applications require extensive expertise and comprehensive safety protocols.
Advanced Research Considerations:
- Multi-compound protocols and interaction studies
- Specialized timing strategies for enhanced research outcomes
- Advanced monitoring techniques for comprehensive data collection
- Customized dosing algorithms based on individual responses
- Long-term follow-up studies for sustained effect analysis
These advanced applications contribute to cutting-edge peptide research while requiring exceptional attention to safety and protocol management.
Quality and Sourcing Considerations
Peptide Quality Standards
Research quality depends heavily on peptide purity, potency, and consistency. AOD 9604 peptide for sale from reputable sources undergoes rigorous testing and quality control measures to ensure research reliability.
Quality Indicators:
- Purity levels exceeding 98% through HPLC analysis
- Consistent potency across different production batches
- Proper storage conditions maintaining peptide integrity
- Third-party testing verification and documentation
- Appropriate packaging protecting against degradation
Quality sourcing directly impacts research outcomes and data reliability. Researchers prioritize suppliers with established quality control systems and comprehensive testing protocols.
Sourcing and Procurement
AOD 9604 peptide where to buy decisions significantly impact research quality and outcomes. Reputable suppliers provide detailed documentation, quality certificates, and ongoing support for research applications.
Supplier Selection Criteria:
- Established reputation in the research community
- Comprehensive quality testing and documentation
- Proper storage and shipping procedures
- Responsive customer support for research questions
- Competitive pricing without compromising quality
Researchers seeking reliable sources often turn to established peptide suppliers with proven track records in the research community.
Storage and Handling Protocols
Proper peptide storage ensures compound integrity throughout research periods. Buy AOD 9604 peptide from sources that provide detailed storage instructions and handling guidelines.
Storage Requirements:
- Refrigerated storage at 2-8°C for reconstituted peptides
- Freezer storage at -20°C for long-term powder storage
- Protection from light to prevent photodegradation
- Sterile handling techniques to prevent contamination
- Proper labeling with dates and concentration information
These storage protocols maintain peptide potency and ensure consistent research results throughout study periods.
Research Documentation and Record Keeping
Comprehensive Documentation Systems
Successful aod 9604 peptide research requires meticulous documentation of all procedures, observations, and outcomes. Proper record keeping ensures research integrity and enables meaningful data analysis.
Documentation Requirements:
- Detailed dosing logs with times, amounts, and administration methods
- Comprehensive observation records including subjective and objective measures
- Safety monitoring data with regular health assessments
- Protocol modifications and rationale for changes
- Outcome measurements using standardized assessment tools
This documentation provides the foundation for meaningful research conclusions and contributes to the broader scientific understanding of peptide applications.
Data Analysis and Interpretation
Research data requires systematic analysis to extract meaningful insights about aod 9604 peptide dosage effectiveness and optimal protocols. Statistical analysis helps identify trends and validate research hypotheses.
Analysis Considerations:
- Baseline comparisons to measure change over time
- Dose-response relationships to optimize protocols
- Individual variation analysis to understand response patterns
- Safety profile assessment throughout study periods
- Long-term outcome evaluation for sustained effects
Proper data analysis transforms raw research observations into actionable insights for future protocol development and scientific advancement.
Researchers contributing to peptide research databases help advance the collective understanding of peptide applications and optimal research methodologies.
Future Directions in AOD 9604 Research
Emerging Research Trends
The field of aod 9604 peptide research continues evolving with new applications, delivery methods, and combination protocols. Current trends focus on precision dosing, personalized protocols, and enhanced delivery systems.
Research Innovation Areas:
- Personalized dosing algorithms based on individual characteristics
- Advanced delivery systems for improved bioavailability
- Combination protocols with complementary research compounds
- Extended-release formulations for sustained research applications
- Novel administration routes expanding research possibilities
These innovations promise to enhance research capabilities while maintaining safety standards and scientific rigor.
Technology Integration
Modern research facilities integrate advanced technology to optimize aod 9604 peptide dosage chart development and implementation. Digital tools enhance precision, monitoring capabilities, and data collection efficiency.
Technological Advances:
- Digital dosing calculators for precise protocol development
- Automated monitoring systems for continuous data collection
- Advanced analytics platforms for comprehensive data analysis
- Mobile applications for real-time documentation and tracking
- Cloud-based databases for collaborative research initiatives
Technology integration streamlines research processes while improving accuracy and data quality.
Conclusion
Understanding the aod 9604 peptide dosage chart represents just the beginning of successful peptide research. Comprehensive protocols require attention to quality sourcing, proper administration techniques, safety monitoring, and detailed documentation. The research community continues advancing our understanding of optimal dosing strategies while maintaining the highest standards of scientific rigor.
Successful AOD 9604 research depends on multiple factors working in harmony: precise dosing based on individual characteristics, consistent administration protocols, comprehensive safety monitoring, and meticulous documentation. Researchers who master these elements contribute valuable insights to the growing body of peptide research knowledge.
Next Steps for Researchers:
- Establish comprehensive protocols incorporating all safety and quality considerations
- Source high-quality peptides from reputable suppliers with proven track records
- Implement systematic monitoring and documentation procedures
- Connect with research communities to share insights and best practices
- Stay current with emerging research and protocol developments
The future of peptide research depends on responsible, well-designed studies that prioritize safety while advancing scientific understanding. By following established guidelines and maintaining rigorous standards, researchers contribute to the expanding knowledge base that benefits the entire scientific community.
Whether beginning initial research or optimizing existing protocols, the principles outlined in this comprehensive guide provide the foundation for successful AOD 9604 peptide research. The combination of proper dosing, quality sourcing, safety protocols, and detailed documentation ensures research integrity while maximizing the potential for meaningful scientific discoveries.
SEO Meta Title: AOD 9604 Peptide Dosage Chart: Complete Research Guide 2026
Meta Description: Comprehensive AOD 9604 peptide dosage chart with research protocols, safety guidelines, and administration methods for laboratory applications in 2026.
Images

