Best Peptide Suppliers for Labs: 2026 Comprehensive Guide for Research Excellence

Finding reliable sources for research-grade peptides has become increasingly critical as peptide science advances into new frontiers. In 2026, the landscape of best peptide suppliers for labs has evolved significantly, with stricter quality standards, enhanced third-party verification protocols, and more sophisticated synthesis techniques. Whether conducting cutting-edge research in athletic performance, recovery optimization, or cellular function, selecting the right peptide vendor can make the difference between breakthrough discoveries and compromised results.
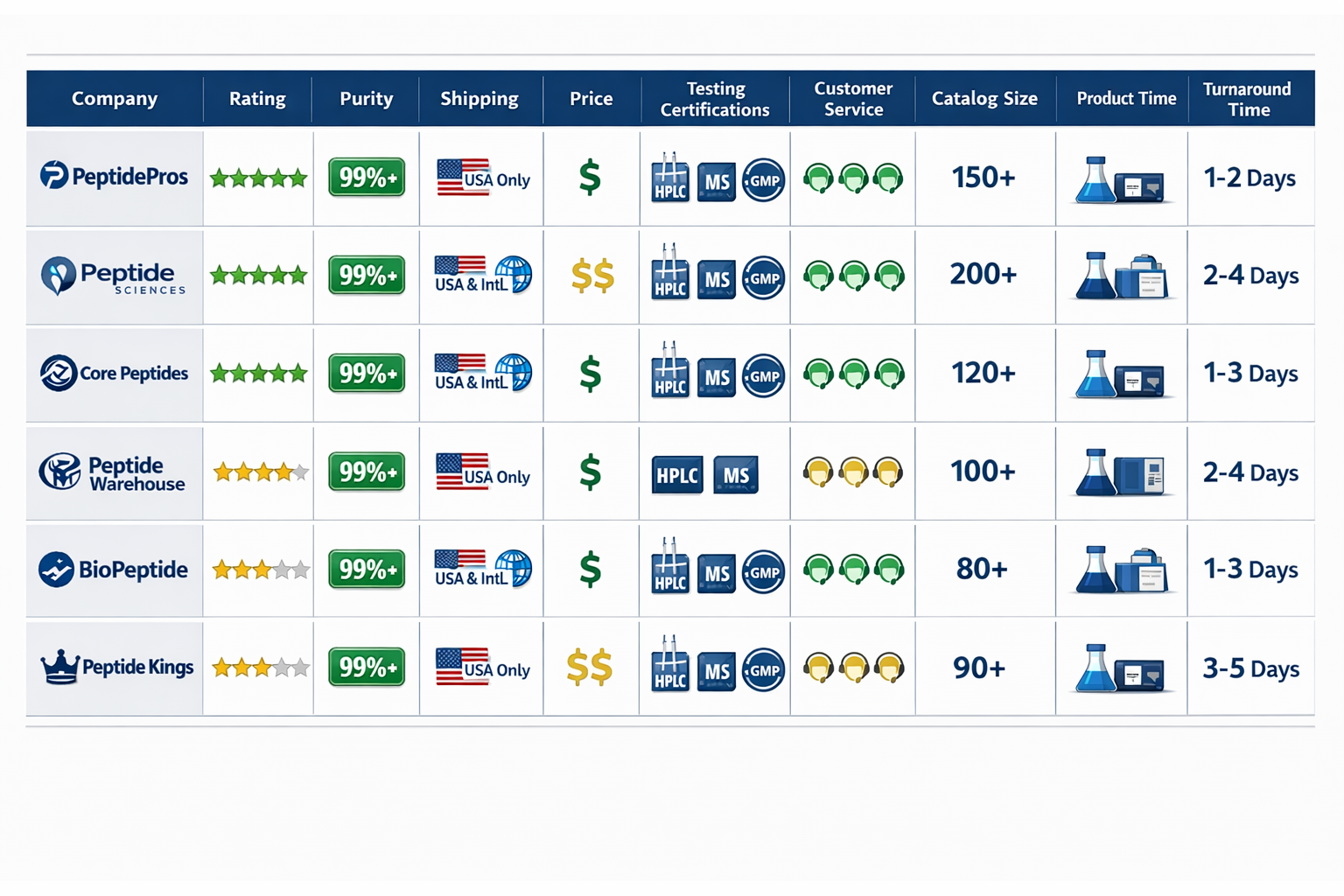
The peptide supply industry now serves thousands of research facilities worldwide, yet not all suppliers meet the rigorous standards required for legitimate scientific work. With over 171 trusted peptide suppliers tracked by independent verification services as of January 2026[6], researchers face the challenging task of identifying which vendors deliver consistent quality, proper documentation, and research-grade purity levels.
This comprehensive guide examines the best peptide suppliers for labs across multiple critical dimensions: purity verification, regulatory compliance, documentation standards, synthesis capabilities, and specialized offerings. By understanding what separates exceptional suppliers from mediocre ones, research professionals can make informed decisions that protect their work's integrity while advancing peptide science.
Key Takeaways
- Purity standards of ≥99% verified through HPLC and Mass-Spec analysis are now baseline requirements for research-grade peptides from reputable suppliers in 2026[1][4]
- Third-party testing with batch-specific Certificates of Analysis (COAs) provides essential verification that separates legitimate suppliers from questionable sources[1][4]
- GMP-certified manufacturing facilities ensure reproducibility and contamination control critical for research applications[2][5]
- Independent verification platforms like Finnrick Analytics track over 171 suppliers with quality ratings updated through January 2026[6]
- Specialized suppliers focus on different research areas, from custom synthesis to large-scale manufacturing, requiring careful matching to specific research needs[2][5]
Understanding Quality Standards for Best Peptide Suppliers for Labs

What Defines Research-Grade Peptide Quality? 🔬
The foundation of any reputable peptide supplier rests on demonstrable quality standards. In 2026, research-grade peptides require minimum purity levels of 99% or higher, verified through multiple analytical methods[1][4]. This threshold isn't arbitrary—it reflects the precision needed for reproducible research outcomes and reliable data collection.
High-Performance Liquid Chromatography (HPLC) serves as the primary verification method, separating peptide compounds to identify purity levels with exceptional accuracy. Mass spectrometry (Mass-Spec) provides secondary confirmation by analyzing molecular weight and structure. Together, these techniques create a comprehensive purity profile that legitimate suppliers willingly share with customers.
The best peptide suppliers for labs go beyond basic testing by providing batch-specific documentation. Each production run receives individual analysis, ensuring consistency across orders. This level of quality control has become standard practice among lab-tested peptides suppliers who understand research requirements.
Third-party verification adds another layer of credibility. Independent laboratories conduct unbiased testing, removing potential conflicts of interest. Suppliers confident in their products actively seek third-party validation, publishing results alongside their own internal testing data[1][4].
Regulatory Compliance and cGMP Certification
Current Good Manufacturing Practices (cGMP) certification represents the gold standard for peptide production facilities. These regulations, enforced by agencies like the FDA, establish comprehensive quality systems covering everything from raw material sourcing to final product storage[2][5].
Bachem AG, with over 50 years of experience, operates GMP-compliant facilities that demonstrate how established manufacturers maintain consistency across global markets[2][5]. Their automation-driven production processes minimize human error while maximizing reproducibility—essential factors for research applications requiring identical peptides across multiple experiments.
AmbioPharm Inc. exemplifies U.S.-based GMP compliance, providing large-scale manufacturing across preclinical, clinical, and commercial scales[2][5]. Their end-to-end support includes specialized modifications that many research projects require. For researchers exploring all peptides for sale, understanding a supplier's GMP status helps predict quality consistency.
Compliance extends beyond manufacturing to documentation practices. Proper chain-of-custody records, batch tracking systems, and stability testing protocols all fall under GMP requirements. Suppliers meeting these standards provide researchers with confidence that their peptides maintain integrity from synthesis through delivery.
Documentation and Transparency Requirements
Certificates of Analysis (COAs) serve as the primary documentation tool for verifying peptide quality. Comprehensive COAs include HPLC chromatograms, mass spectrometry results, purity percentages, peptide sequence confirmation, and storage recommendations. The best peptide suppliers for labs provide COAs automatically with each order, not just upon request[1][4].
Science.bio has built its reputation on advanced documentation practices, offering comprehensive COAs on every product in their catalog[4]. This transparency allows researchers to verify quality before conducting experiments, preventing wasted time and resources on substandard materials.
Batch-specific testing results matter more than general product specifications. A supplier might claim 99% purity for a particular peptide, but without batch-level verification, that claim remains unverifiable. Reputable vendors assign unique batch numbers and provide corresponding test results for each production run.
Storage and handling documentation also separates professional suppliers from amateur operations. Proper peptide storage requires specific temperature ranges, humidity controls, and light protection. Suppliers who provide detailed handling instructions demonstrate understanding of peptide stability and commitment to maintaining quality through the entire supply chain.
Top-Tier Best Peptide Suppliers for Labs in 2026
Cernum Biosciences: U.S.-Based Excellence
Cernum Biosciences has established itself as a trusted U.S.-based vendor specializing in research-grade peptides with verified ≥99% purity levels[1]. Their commitment to third-party testing and batch-specific COAs addresses the core concerns of serious researchers who cannot compromise on quality.
The company's U.S.-only shipping policy ensures compliance with domestic regulations while providing faster delivery times for American research facilities. This geographic focus allows Cernum to maintain tighter quality control and more responsive customer service compared to international suppliers managing complex global logistics.
Their catalog emphasizes research applications, clearly distinguishing their products from consumer-grade alternatives. This positioning attracts researchers seeking wholesale peptides for ongoing projects requiring consistent supply chains.
Cernum's transparency regarding testing methodologies sets industry standards. They publish detailed HPLC results, mass spectrometry data, and purity verification for each batch. Researchers can access this information before purchase, enabling informed decision-making based on objective data rather than marketing claims.
Biotech Peptides: Ultra-High Purity Specialists
Established in 2022, Biotech Peptides quickly earned recognition for ultra-high purity standards exceeding 99% verified through both HPLC and Mass-Spec analysis[4]. Despite being relatively new, their commitment to quality has positioned them among the best peptide suppliers for labs focused on precision research.
Their cGMP certified facilities incorporate advanced synthesis technologies that minimize impurities during production. Same-day processing capabilities appeal to researchers working under tight timelines, while batch-level testing ensures each order meets documented specifications.
Biotech Peptides' focus on cutting-edge catalog offerings includes newer peptide sequences that established suppliers may not yet provide. This makes them particularly valuable for researchers exploring emerging areas like longevity peptide research or novel therapeutic applications.
The company provides full COAs with detailed analytical data, including retention times, peak purity assessments, and molecular weight confirmation. This documentation level supports researchers who must justify their material sourcing choices to institutional review boards or funding agencies.
Science.bio: Advanced Documentation and Compliance
Science.bio has built a reputation among experienced researchers requiring strict compliance protocols[4]. Their advanced documentation practices go beyond standard COAs to include comprehensive synthesis reports, stability data, and contamination testing results.
The company's cutting-edge catalog reflects current research trends, regularly updating their offerings based on emerging scientific literature. This responsiveness to the research community's evolving needs distinguishes them from suppliers offering static product lines.
Professional-grade synthesis methods ensure reproducibility across batches—a critical factor for longitudinal studies requiring identical peptides over extended periods. Their quality control processes include multiple verification steps at each production stage, from raw material inspection through final packaging.
Science.bio's customer support team includes scientific advisors who understand research applications. This expertise proves valuable when researchers need guidance on peptide selection, storage optimization, or protocol development for best peptide manufacturers products.
Specialized Peptide Suppliers and Manufacturing Excellence
Global Manufacturing Leaders
Bachem AG, headquartered in Switzerland with over 50 years of industry experience, represents the pinnacle of large-scale peptide manufacturing[2][5]. Their GMP-compliant facilities serve global research markets with customized peptides designed for specific applications.
The company's focus on reproducibility stems from automation-driven production systems that eliminate variability inherent in manual processes. This consistency proves essential for multi-site research collaborations requiring identical peptides across different geographic locations.
Bachem's expertise extends to specialized modifications including cyclization, pegylation, and complex conjugations. These capabilities support advanced research projects that standard peptide suppliers cannot accommodate. Researchers working with peptide supplier comparisons often identify Bachem as the benchmark for manufacturing excellence.
Their global distribution network ensures reliable supply chains even during international shipping disruptions. This reliability matters for ongoing research projects that cannot afford material shortages or quality inconsistencies.
AmbioPharm: End-to-End U.S. Manufacturing
AmbioPharm Inc. provides comprehensive peptide manufacturing services across preclinical, clinical, and commercial scales[2][5]. This vertical integration allows them to support research projects from initial discovery through potential commercialization.
Their U.S.-based operations appeal to researchers preferring domestic suppliers for regulatory compliance, intellectual property protection, and communication convenience. The company's end-to-end support includes formulation development, analytical method validation, and regulatory documentation assistance.
AmbioPharm specializes in complex peptides requiring sophisticated synthesis techniques. Their technical team collaborates with researchers to optimize sequences, improve stability, and enhance bioavailability. This consultative approach benefits projects exploring innovative peptide delivery systems or novel therapeutic mechanisms.
Quality assurance processes include environmental monitoring, equipment qualification, and personnel training programs that exceed basic GMP requirements. These enhanced protocols provide additional confidence for researchers conducting work that may eventually transition to clinical applications.
Thermo Fisher Scientific: Technology-Driven Solutions
Thermo Fisher Scientific Inc. brings next-generation peptide synthesis platforms to the research community[2]. Their emphasis on automation and high-throughput capabilities serves laboratories conducting large-scale screening studies or extensive peptide libraries.
The company's reagent quality directly impacts synthesis success rates. Thermo Fisher's vertically integrated supply chain ensures consistent reagent performance, reducing failed synthesis attempts and material waste.
Their technology platforms include automated synthesizers that researchers can operate in-house, providing greater control over synthesis timing and sequence confidentiality. This equipment approach appeals to well-funded laboratories seeking independence from external synthesis services.
Technical support includes synthesis protocol optimization, troubleshooting assistance, and training programs. These resources help research teams maximize their peptide synthesis capabilities while maintaining quality standards comparable to specialized manufacturers.
Evaluating Best Peptide Suppliers for Labs: Key Selection Criteria
Purity Verification and Testing Protocols
When assessing potential suppliers, purity verification protocols should top the evaluation checklist. The best peptide suppliers for labs employ multiple analytical techniques to confirm peptide identity and purity[1][4]. HPLC analysis reveals the percentage of target peptide versus impurities, with research-grade materials requiring ≥99% purity.
Mass spectrometry provides molecular weight confirmation, verifying that the synthesized peptide matches the intended sequence. Discrepancies between expected and actual molecular weights indicate synthesis errors, deletions, or unwanted modifications.
Third-party testing adds credibility by removing potential bias from internal quality control. Independent laboratories like Finnrick Analytics maintain active vendor quality ratings, with recent supplier test dates ranging from December 2024 through January 2026[6]. These ongoing verification programs help researchers identify consistently reliable suppliers.
Request sample COAs before committing to large orders. Examine chromatogram quality, peak resolution, and purity calculations. Professional COAs include detailed methodology descriptions, instrument specifications, and analyst credentials. Suppliers reluctant to share this documentation raise immediate red flags about quality claims.
Catalog Breadth and Specialization
Supplier selection should align with specific research needs. Some vendors excel at standard peptide sequences with proven synthesis protocols, while others specialize in complex modifications or novel sequences. Researchers exploring MOTS-C peptides for sale or Epithalon peptides for sale need suppliers experienced with these specific compounds.
Catalog breadth indicates manufacturing capabilities and market responsiveness. Suppliers offering hundreds of peptides demonstrate synthesis versatility and established quality control systems. However, specialized vendors focusing on particular peptide classes may provide superior expertise for niche applications.
Custom synthesis capabilities matter for research projects requiring unique sequences or modifications. Evaluate suppliers' willingness to accommodate special requests, minimum order quantities for custom work, and typical turnaround times. Some manufacturers excel at rapid custom synthesis, while others require extended timelines for non-standard requests.
Consider whether suppliers offer related products like reconstitution solutions, storage vials, or analytical standards. Comprehensive product ecosystems simplify procurement and ensure compatibility across research materials.
Pricing, Shipping, and Customer Support
Pricing transparency separates professional suppliers from questionable vendors. Reputable companies publish clear pricing structures or provide detailed quotes upon request. Be wary of suppliers with vague pricing or significant discrepancies from market averages—extremely low prices often indicate quality compromises.
Shipping policies impact peptide integrity during transit. Temperature-controlled shipping protects peptides sensitive to heat or freezing. Suppliers using insulated packaging, cold packs, or dry ice demonstrate understanding of peptide stability requirements.
Domestic versus international shipping affects delivery times, customs complications, and regulatory compliance. U.S.-based researchers often prefer domestic suppliers to avoid customs delays and ensure compliance with import regulations. However, international suppliers like Bachem offer unique capabilities that may justify additional logistical complexity[2][5].
Customer support quality reveals supplier professionalism and technical expertise. Responsive communication, knowledgeable staff, and willingness to address technical questions indicate customer-focused operations. Test support responsiveness during the evaluation phase—suppliers who ignore pre-sale inquiries will likely provide poor post-sale support.
Emerging Trends Among Best Peptide Suppliers for Labs
Advanced Quality Verification Technologies
The peptide supply industry continues evolving with enhanced verification technologies. Advanced HPLC systems with higher resolution capabilities detect impurities at lower concentrations than previous generation equipment. This improved sensitivity helps identify trace contaminants that might affect research outcomes.
Nuclear Magnetic Resonance (NMR) spectroscopy is gaining adoption as a complementary verification technique. NMR provides structural confirmation beyond molecular weight analysis, verifying peptide folding and conformation. Leading suppliers incorporate NMR data into comprehensive quality documentation.
Automated quality control systems reduce human error in testing and documentation. Robotic sample handling, integrated data management, and electronic chain-of-custody tracking enhance reproducibility and traceability. These systems appeal to researchers requiring audit trails for regulatory submissions or publication requirements.
Blockchain-based verification platforms are emerging to provide immutable quality records. These systems create permanent, tamper-proof documentation of testing results, manufacturing conditions, and supply chain custody. While still experimental, blockchain verification may become standard among premium suppliers.
Sustainability and Ethical Manufacturing
Environmental sustainability is influencing supplier selection criteria. Green chemistry approaches minimize hazardous waste, reduce solvent consumption, and optimize resource efficiency. Suppliers implementing sustainable practices appeal to institutions with environmental responsibility mandates.
Ethical sourcing of raw materials addresses concerns about supply chain transparency. Suppliers documenting their reagent sources and manufacturing conditions provide assurance that materials meet ethical standards. This transparency matters for research funded by organizations requiring responsible sourcing verification.
Energy-efficient manufacturing facilities reduce environmental impact while potentially lowering production costs. Solar-powered laboratories, waste heat recovery systems, and optimized HVAC designs demonstrate commitment to sustainable operations.
Packaging innovations reduce plastic waste through recyclable materials, minimized packaging volumes, and reusable shipping containers. These improvements align with institutional sustainability goals while maintaining peptide protection during transit.
Specialized Research Applications
Supplier specialization increasingly reflects emerging research areas. Vendors focusing on longevity peptide research or healing peptides develop expertise in specific peptide classes, synthesis challenges, and application requirements.
Athletic performance research drives demand for peptides related to recovery, muscle growth, and metabolic optimization. Suppliers serving this market understand the specific purity requirements and documentation needs for sports science research.
Cosmetic peptide research has expanded supplier offerings in sequences related to skin health and anti-aging applications. Researchers exploring peptides in skincare benefit from suppliers experienced with dermatological research requirements.
Metabolic research applications, including work with GLP-1 peptides, require suppliers knowledgeable about incretin biology and related synthesis challenges. Specialized vendors provide technical support beyond basic product delivery.
Publicly Traded Peptide Companies and Market Leaders
Major Pharmaceutical Peptide Developers
Several publicly traded companies dominate peptide therapeutics development, offering insights into industry trends and quality standards. Novo Nordisk (NYSE: NVO) leads in peptide-based diabetes and obesity treatments, setting benchmarks for manufacturing quality and regulatory compliance[3].
Eli Lilly (NYSE: LLY) has invested heavily in peptide therapeutics, particularly for metabolic disorders. Their manufacturing standards influence supplier expectations across the industry, as contract manufacturers must meet pharmaceutical-grade specifications.
Pfizer (NYSE: PFE) and AstraZeneca (NYSE: AZN) maintain significant peptide development programs, driving demand for high-quality research materials[3]. Their supplier qualification processes establish quality benchmarks that research-focused vendors often adopt.
Ascendis Pharma (ASND) specializes in long-acting peptide therapeutics using proprietary delivery technologies. Their work demonstrates the importance of peptide purity and consistency for advanced formulation development.
Rhythm Pharmaceuticals (RYTM) focuses on rare genetic disorders treated with peptide therapeutics[3]. Their precision medicine approach requires exceptional peptide quality and detailed characterization—standards that benefit the broader research community.
Market Dynamics and Supplier Landscape
The peptide synthesis market includes 28 documented leading companies, each specializing in distinct service areas[2]. AnaSpec Inc., Biotage, CEM Corporation, and PolyPeptide Group AG represent diverse approaches to peptide manufacturing, from custom synthesis to large-scale production.
Market consolidation has created larger suppliers with comprehensive capabilities, while specialized boutique vendors serve niche applications. This diversity benefits researchers by providing options matching specific project requirements and budgets.
Independent verification platforms like Finnrick Analytics track 171 trusted peptide suppliers with quality ratings updated through January 2026[6]. These databases help researchers identify reliable vendors and avoid problematic suppliers.
Industry conferences and scientific meetings facilitate direct supplier evaluation. Many best peptide suppliers for labs maintain active presence at research conferences, providing opportunities for face-to-face discussions about capabilities, quality systems, and technical support.
Practical Guidelines for Selecting Best Peptide Suppliers for Labs
Initial Supplier Evaluation Process
Begin supplier evaluation by defining specific research requirements. Identify needed peptide sequences, required purity levels, quantity requirements, and timeline constraints. This clarity helps narrow the supplier pool to candidates capable of meeting project needs.
Research supplier reputations through independent verification platforms, scientific forums, and colleague recommendations. Finnrick Analytics maintains updated supplier quality ratings that provide objective performance data[6]. Cross-reference multiple information sources to build comprehensive supplier profiles.
Request detailed information packages from potential suppliers, including sample COAs, quality control procedures, manufacturing capabilities, and pricing structures. Professional suppliers provide this documentation readily, while evasive responses indicate potential quality concerns.
Conduct small test orders before committing to large purchases. Evaluate received peptides through independent verification if possible. Compare actual quality against supplier claims and documentation. This validation step prevents costly mistakes from unreliable suppliers.
Quality Verification Best Practices
Implement internal quality verification procedures regardless of supplier reputation. Independent testing through third-party laboratories provides objective quality confirmation. While adding cost, this verification protects research integrity and validates supplier claims.
Maintain detailed records of all peptide orders, including batch numbers, COAs, storage conditions, and usage dates. This documentation supports research reproducibility and helps identify quality trends across multiple orders.
Establish quality acceptance criteria before ordering. Define minimum purity levels, acceptable impurity profiles, and documentation requirements. Communicate these standards to suppliers and verify compliance upon receipt.
Monitor supplier consistency across multiple orders. Quality variation between batches suggests inadequate process controls. Consistently reliable suppliers demonstrate robust quality systems and manufacturing discipline.
Building Long-Term Supplier Relationships
Develop relationships with multiple qualified suppliers to ensure supply chain resilience. Single-source dependencies create vulnerability to supplier disruptions, quality issues, or business closures. Maintain active relationships with at least two qualified vendors for critical peptides.
Provide feedback to suppliers about quality, service, and documentation. Professional vendors value customer input and use it to improve operations. This communication builds partnerships rather than transactional relationships.
Negotiate favorable terms for ongoing research projects requiring regular peptide supplies. Volume commitments often justify pricing discounts, priority processing, or enhanced technical support. However, ensure contractual flexibility for research projects with uncertain timelines.
Stay informed about supplier developments, including facility upgrades, certification achievements, or ownership changes. These events may impact quality, pricing, or service levels. Proactive monitoring helps anticipate and address potential supply chain disruptions.
Specialized Peptide Categories and Supplier Expertise
Mitochondrial and Longevity Peptides
Research into mitochondrial function and longevity has driven demand for specialized peptides like MOTS-C and SS-31. Suppliers experienced with these compounds understand unique synthesis challenges and stability requirements. Researchers exploring MOTS-C metabolic flexibility need vendors familiar with this peptide's specific characteristics.
SS-31 peptides require particular attention to synthesis precision due to their complex structure. Suppliers offering SS-31 peptides for sale should provide detailed characterization data confirming proper sequence and folding. The growing interest in SS-31 peptide benefits has expanded the supplier base, but quality varies significantly.
Epithalon represents another longevity-focused peptide with specific synthesis requirements. Researchers should verify that suppliers understand proper sequence assembly and purification protocols for this tetrapeptide. Quality Epithalon requires attention to detail that not all manufacturers provide.
Combination products pairing longevity peptides with complementary compounds require suppliers capable of handling multiple peptide synthesis projects simultaneously. Consistency across different peptides in combination formulations demands sophisticated quality control systems.
Growth Hormone Secretagogues and Performance Peptides
Ipamorelin (IPA) has become a research staple for growth hormone axis studies. The question "Is IPA the most beneficial peptide?" reflects significant research interest in this compound. Suppliers should provide high-purity IPA with comprehensive documentation supporting research applications.
CJC-1295 and related peptides require specialized synthesis expertise due to their modified structures. Research examining CJC-IPA GH axis interactions needs consistent peptide quality across both compounds. Suppliers offering combination products should demonstrate quality control for each component.
Peptide blends combining multiple growth hormone secretagogues present additional quality challenges. Each component requires individual verification, and the final blend needs stability testing. Reputable suppliers provide comprehensive documentation for blend compositions and individual component purity.
Research-grade secretagogues differ significantly from consumer products in purity, documentation, and intended use. The best peptide suppliers for labs clearly distinguish research materials from other product categories, ensuring compliance with institutional research guidelines.
Cosmetic and Dermatological Research Peptides
GHK-Cu and related copper peptides require specialized handling and storage due to metal ion sensitivity. Suppliers offering GHK-Cu peptides for sale should provide detailed stability data and storage recommendations. Research into GHK-Cu benefits from suppliers experienced with metal-peptide complexes.
Peptide blends for cosmetic research, including Glow peptide blend formulations, combine multiple sequences targeting different skin health mechanisms. Quality suppliers verify each component individually while testing the final blend for stability and compatibility.
Researchers questioning "Are peptide serums worth it?" need access to pure peptide standards for comparative studies. Suppliers supporting dermatological research provide reference materials with documented purity and stability.
The intersection of peptide science and skincare has created demand for suppliers understanding both cosmetic formulation requirements and research-grade quality standards. This dual expertise ensures materials suitable for rigorous scientific investigation.
Documentation, Compliance, and Research Integrity
Certificate of Analysis Essentials
Comprehensive COAs serve as the primary quality verification tool for research peptides. Essential components include peptide sequence confirmation, molecular weight verification through mass spectrometry, HPLC purity analysis with chromatogram, batch number and manufacturing date, storage recommendations, and expiration dating.
Chromatogram quality reveals important information beyond simple purity percentages. Sharp, well-resolved peaks indicate clean synthesis and effective purification. Broad peaks or multiple small peaks suggest impurities or degradation products that may interfere with research.
Mass spectrometry data should show the expected molecular weight with minimal deviation. Suppliers providing high-resolution mass spec data demonstrate commitment to thorough characterization. This detailed analysis helps identify unexpected modifications or synthesis errors.
Storage and handling recommendations based on stability testing prevent peptide degradation. Suppliers conducting stability studies provide evidence-based guidance rather than generic storage suggestions. This attention to detail preserves peptide integrity throughout the research timeline.
Regulatory Compliance for Research Materials
Research peptides exist in a complex regulatory environment. While not intended for human consumption, research materials must meet quality standards supporting legitimate scientific investigation. The best peptide suppliers for labs maintain clear compliance policies and provide documentation supporting institutional research requirements.
Institutional review boards (IRBs) and research ethics committees often require detailed supplier information for protocol approvals. Suppliers providing comprehensive quality documentation, manufacturing descriptions, and compliance statements facilitate regulatory approval processes.
Import and export regulations affect international peptide shipments. Suppliers experienced with cross-border transactions help navigate customs requirements, documentation needs, and regulatory restrictions. This expertise prevents shipment delays and regulatory complications.
Material safety data sheets (MSDS) provide essential safety information for laboratory personnel handling peptides. Comprehensive MSDS documentation demonstrates supplier professionalism and commitment to researcher safety.
Research Reproducibility and Quality Control
Batch-to-batch consistency enables research reproducibility across experiments conducted at different times. Suppliers maintaining tight quality control produce peptides with minimal variation between production runs. This consistency proves essential for longitudinal studies or multi-site collaborations.
Reference standards help verify peptide identity and purity. Some suppliers offer certified reference materials with enhanced characterization and documentation. These standards support analytical method development and quality verification protocols.
Stability testing data informs storage decisions and experimental planning. Suppliers conducting accelerated stability studies provide evidence-based shelf life estimates. This information helps researchers plan peptide procurement timing and storage strategies.
Quality control documentation should include manufacturing records, testing protocols, equipment calibration records, and analyst qualifications. While researchers rarely review these detailed records, their existence indicates robust quality systems and professional manufacturing practices.
Cost Considerations and Value Assessment
Understanding Peptide Pricing Structures
Peptide pricing reflects multiple factors including sequence complexity, synthesis scale, purity requirements, and documentation level. Simple sequences with established synthesis protocols cost less than complex peptides requiring specialized techniques or modifications.
Synthesis scale significantly impacts per-unit pricing. Large-scale production achieves economies of scale unavailable for small batches. However, researchers should order quantities matching actual needs rather than over-purchasing based solely on unit price considerations.
Purity requirements affect pricing, as higher purity levels require additional purification steps and more rigorous quality control. Research applications requiring ≥99% purity justify premium pricing compared to lower-grade materials suitable for preliminary studies.
Documentation and certification add value through enhanced confidence and regulatory support. While basic COAs may suffice for some applications, comprehensive documentation packages support publication requirements and regulatory submissions.
Balancing Cost and Quality
The best peptide suppliers for labs provide fair pricing reflecting genuine quality rather than artificially low prices indicating compromised standards. Extremely cheap peptides often contain impurities, incorrect sequences, or inadequate documentation that compromise research integrity.
Consider total cost of ownership beyond initial purchase price. Failed experiments due to poor peptide quality waste time, resources, and opportunity costs far exceeding savings from cheap suppliers. Quality peptides enable successful research and reliable data generation.
Volume discounts reward ongoing relationships and large orders. However, ensure volume commitments align with actual research needs and peptide stability timelines. Expired peptides represent wasted investment regardless of initial pricing.
Compare pricing across multiple qualified suppliers for specific peptides. Significant price variations may indicate different purity levels, documentation quality, or synthesis methods. Investigate pricing differences rather than automatically selecting the lowest-cost option.
Value-Added Services and Support
Technical support adds value beyond basic product delivery. Suppliers offering protocol guidance, troubleshooting assistance, and application expertise help researchers optimize their peptide use. This support proves particularly valuable for researchers new to peptide science.
Custom synthesis capabilities provide flexibility for unique research needs. While custom work typically costs more than catalog peptides, it enables research that standard products cannot support. Evaluate custom synthesis pricing against the value of unique research capabilities.
Expedited processing and shipping options support time-sensitive research. Premium pricing for rapid turnaround may prove worthwhile when research timelines cannot accommodate standard delivery schedules.
Consolidated ordering systems simplify procurement for laboratories using multiple peptides. Suppliers offering comprehensive catalogs reduce administrative burden and potentially qualify for volume discounts across different peptides.
Future Outlook for Best Peptide Suppliers for Labs

Technological Advances in Peptide Synthesis
Automated synthesis platforms continue advancing, enabling faster production with higher consistency. Next-generation synthesizers incorporate real-time monitoring, automated purification, and integrated quality control. These technological improvements benefit researchers through improved peptide quality and reduced costs.
Artificial intelligence and machine learning optimize synthesis protocols, predict difficult sequences, and improve purification strategies. AI-driven synthesis planning reduces trial-and-error approaches, accelerating custom peptide development and improving success rates.
Green chemistry innovations minimize environmental impact while maintaining quality standards. Solvent recycling systems, enzyme-based synthesis methods, and biodegradable reagents represent emerging approaches that may become standard practices.
Microfluidic synthesis technologies enable ultra-small scale production with exceptional efficiency. While currently experimental, these platforms may revolutionize custom peptide synthesis by reducing material waste and accelerating development timelines.
Expanding Research Applications
Peptide therapeutics development continues accelerating, driving demand for research-grade materials. As pharmaceutical companies advance peptide-based drugs through clinical development, research laboratories require corresponding materials for mechanism studies and application development.
Personalized medicine approaches incorporating peptides require flexible suppliers capable of rapid custom synthesis. This trend favors suppliers with advanced synthesis capabilities and responsive customer service over large-scale manufacturers optimized for standardized production.
Combination therapies pairing peptides with other therapeutic modalities create demand for suppliers understanding multi-component formulations. Research exploring synergistic effects requires consistent quality across different peptide sequences and complementary compounds.
Novel delivery systems for peptides, including innovative peptide delivery systems, require specialized formulation expertise. Suppliers supporting this research provide technical consultation beyond basic peptide synthesis.
Market Evolution and Supplier Landscape
Industry consolidation may reduce the number of independent suppliers while increasing average supplier capabilities. Larger organizations can invest in advanced equipment, comprehensive quality systems, and specialized expertise that smaller vendors cannot afford.
Specialized niche suppliers will likely persist by focusing on specific peptide classes, unique modifications, or particular research applications. These focused vendors provide expertise and responsiveness that diversified suppliers may not match.
Direct-to-researcher sales models enabled by e-commerce platforms democratize access to research peptides. Online ordering systems, transparent pricing, and detailed product information reduce barriers to peptide procurement for smaller research operations.
International collaboration and supply chain globalization expand researcher access to specialized suppliers worldwide. However, regulatory complexity and shipping logistics favor domestic suppliers for routine research needs while international sources serve unique requirements.
Conclusion: Making Informed Decisions About Best Peptide Suppliers for Labs
Selecting among the best peptide suppliers for labs requires careful evaluation of multiple critical factors. Quality verification through HPLC and mass spectrometry analysis, comprehensive documentation with batch-specific COAs, GMP-certified manufacturing facilities, and responsive technical support separate exceptional suppliers from mediocre alternatives.
The peptide supply landscape in 2026 offers unprecedented options, from established manufacturers like Bachem AG and AmbioPharm Inc. to specialized vendors like Cernum Biosciences and Biotech Peptides[1][2][4][5]. Independent verification platforms tracking over 171 suppliers provide objective quality data supporting informed decision-making[6].
Research success depends on peptide quality, making supplier selection a critical research decision rather than a simple procurement task. The time invested in thorough supplier evaluation pays dividends through reliable results, reproducible experiments, and research integrity.
Actionable Next Steps
Start by defining specific research requirements including needed peptide sequences, purity levels, quantities, and timelines. This clarity focuses supplier evaluation on candidates capable of meeting actual needs rather than evaluating all available options.
Request documentation packages from multiple potential suppliers, including sample COAs, quality control procedures, and pricing structures. Compare these materials objectively using consistent evaluation criteria.
Conduct small test orders with selected suppliers before committing to large purchases or long-term relationships. Verify actual quality against supplier claims through independent testing when possible.
Build relationships with multiple qualified suppliers to ensure supply chain resilience and maintain competitive pricing. Diversified sourcing protects research programs from single-supplier disruptions.
Stay informed about industry developments, emerging suppliers, and technological advances affecting peptide quality and availability. The peptide supply landscape continues evolving, creating opportunities for researchers who monitor market changes.
Prioritize quality over cost when selecting suppliers for critical research applications. The investment in premium peptides from the best peptide suppliers for labs protects research integrity and enables breakthrough discoveries that cheap, questionable materials cannot support.
By applying these principles and leveraging the comprehensive information in this guide, researchers can confidently navigate the peptide supply market, identifying vendors that support their scientific goals with exceptional quality, professional service, and unwavering commitment to research excellence. For researchers ready to explore high-quality research peptides, visit all peptides for sale to discover comprehensive options from verified suppliers.
References
[1] Top American Peptide Vendors You Can Trust – https://cernumbiosciences.com/blogs/peptide-science-guide/top-american-peptide-vendors-you-can-trust
[2] Key Companies In Peptide Synthesis – https://www.researchandmarkets.com/articles/key-companies-in-peptide-synthesis
[3] Peptide Companies – https://bullishbears.com/peptide-companies/
[4] Best Peptide Companies – https://crowncounseling.com/reviews/best-peptide-companies/
[5] Top 10 Peptide Synthesis Companies – https://www.rootsanalysis.com/key-insights/top-10-peptide-synthesis-companies.html
[6] Vendors – https://www.finnrick.com/vendors
SEO Meta Title
Best Peptide Suppliers for Labs 2026: Quality Guide
SEO Meta Description
Discover the best peptide suppliers for labs in 2026. Compare quality standards, purity testing, GMP certification, and documentation from top vendors.
