Buy Peptide Building Blocks Bulk: The Complete 2026 Guide for Research and Performance

The global peptide building block market is experiencing explosive growth, projected to reach $282 million by 2034 from $130 million in 2025[4]. For researchers, athletes, and performance enthusiasts looking to buy peptide building blocks bulk, understanding the landscape of suppliers, quality standards, and procurement best practices has never been more critical. This comprehensive guide reveals everything needed to make informed purchasing decisions in 2026's competitive peptide marketplace.
Key Takeaways
- Market consolidation is accelerating: Top Chinese manufacturers like Kelong Chemical and GL Biochem control 24.30% of global revenue, while Western suppliers compete through technological differentiation[4]
- Quality documentation is non-negotiable: Batch-specific Certificates of Analysis (COAs), HPLC chromatograms, and mass spectrometry confirmation are essential verification tools[1]
- Bulk purchasing advantages: Established suppliers offer vertically integrated production capabilities and cost advantages for large-volume orders[4]
- Regulatory complexity is increasing: Stringent requirements and supply chain disruptions make supplier reliability paramount[5]
- Strategic sourcing matters: Understanding the difference between FMOC-protected amino acids, unnatural amino acids, and specialized building blocks optimizes research outcomes
Understanding Peptide Building Blocks: Foundation for Research
Peptide building blocks serve as the fundamental components for synthesizing complex peptide sequences used in research applications. These molecular units, primarily consisting of protected amino acids, enable scientists and researchers to construct specific peptide chains with precise sequences and properties.
What Are Peptide Building Blocks?
Peptide building blocks are chemically protected amino acids that serve as the starting materials for peptide synthesis. The most common protection strategy involves FMOC (9-fluorenylmethoxycarbonyl) protected amino acids, which allow for controlled, stepwise peptide chain assembly[2]. These building blocks include:
- Standard FMOC-protected amino acids (20 natural amino acids)
- Unnatural amino acids with modified side chains
- Specialty building blocks for specific therapeutic applications
- Resin-attached amino acids for solid-phase synthesis
The global FMOC Protected Amino Acids market demonstrates the sector's robust growth trajectory, with pharmaceutical companies increasingly demanding high-purity building blocks for peptide drug development[4].
Why Buy Peptide Building Blocks in Bulk?
Purchasing peptide building blocks in bulk quantities offers several strategic advantages:
Cost Efficiency 💰
Bulk orders typically reduce per-unit costs by 30-50% compared to small-quantity purchases. Suppliers like BACHEM and Merck KGaA offer volume discounts for research institutions and commercial buyers[4].
Supply Chain Stability 🔗
Securing larger inventories protects against supply disruptions, particularly important given current raw material price volatility[5]. For those engaged in ongoing peptide research, consistent access to building blocks ensures project continuity.
Batch Consistency ✅
Ordering bulk quantities from a single production batch ensures uniformity across research protocols, eliminating variables introduced by batch-to-batch variations.
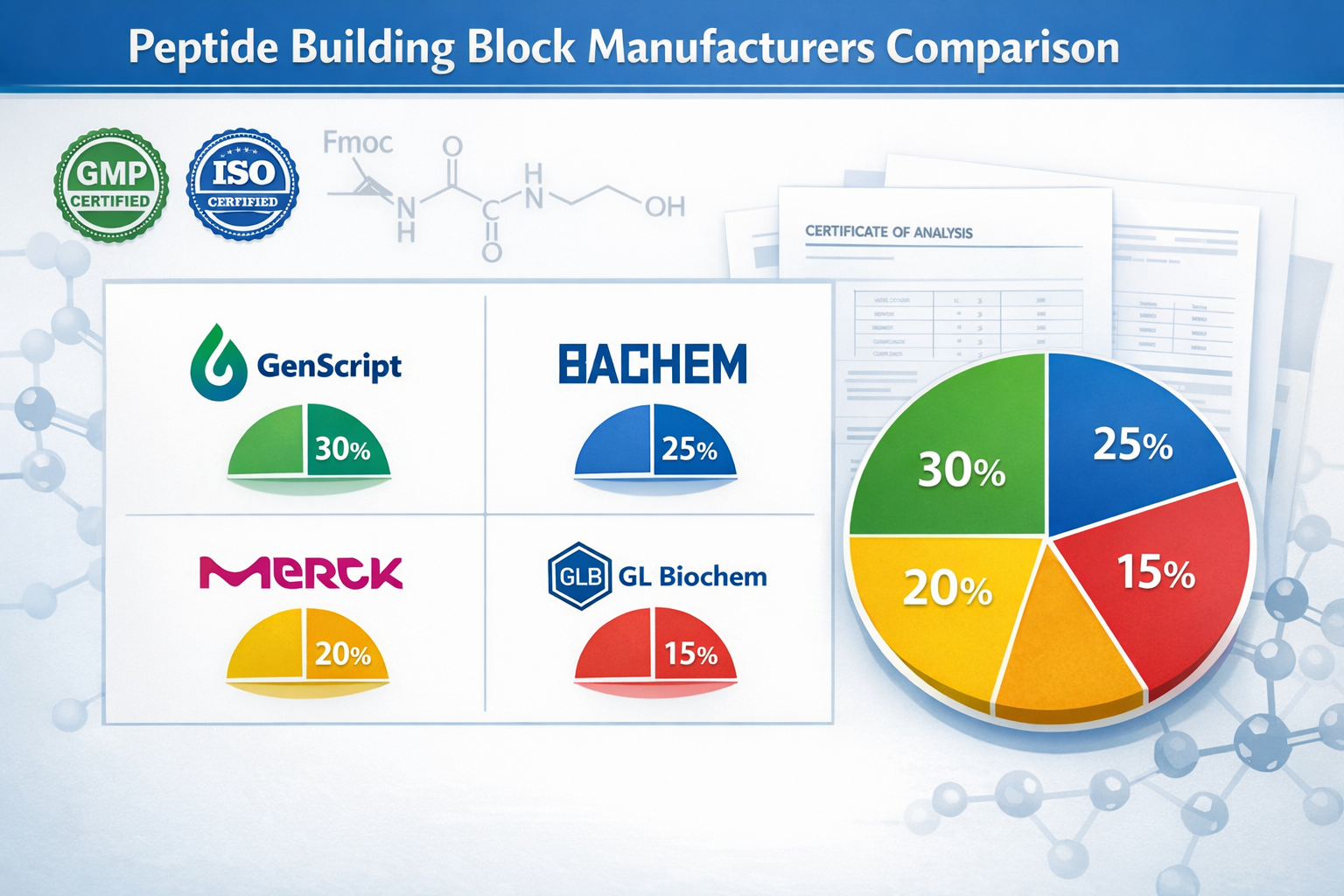
Top Manufacturers When You Buy Peptide Building Blocks Bulk
The competitive landscape for bulk peptide building blocks features both established Western manufacturers and rapidly expanding Asian suppliers. Understanding each player's strengths helps optimize procurement decisions.
Tier-One Global Suppliers
BACHEM stands as a premium supplier offering robust peptide benchmarks through advanced side-chain protection technologies and pharmaceutical-grade purity standards[4]. Their vertically integrated production capabilities ensure consistent quality for large-volume orders.
Merck KGaA competes through technological differentiation, providing high-purity FMOC amino acids with comprehensive documentation packages. Their global distribution network facilitates reliable delivery for international buyers[4].
GenScript offers extensive catalog options spanning standard and unnatural amino acids, with competitive pricing for bulk orders. Their digital ordering platform streamlines procurement for research institutions[5].
GL Biochem (Shanghai) Ltd has captured significant market share through cost-competitive bulk offerings while maintaining quality standards acceptable for pharmaceutical applications[4].
Emerging Suppliers and Market Dynamics
Second-tier suppliers including TACHEM, ZY BIOCHEM, and Sichuan Jisheng are expanding production capacity specifically to meet growing demand from GLP-1 drug development programs[4]. These manufacturers often provide attractive pricing for buyers willing to conduct thorough quality verification.
The 2 Cl Trt Cl resin segment accounts for noticeable market share and projects continued expansion, reflecting specialized applications in peptide synthesis[5]. When planning to buy peptides online, understanding these market segments helps identify optimal suppliers.
| Manufacturer | Market Position | Key Strength | Bulk Pricing |
|---|---|---|---|
| BACHEM | Premium | Quality/Documentation | High |
| Merck KGaA | Premium | Technology/Purity | High |
| GenScript | Mid-Tier | Catalog Breadth | Moderate |
| GL Biochem | Mid-Tier | Cost/Volume | Competitive |
| TACHEM | Emerging | Capacity/Price | Very Competitive |
Quality Verification: Essential Standards to Buy Peptide Building Blocks Bulk
When purchasing peptide building blocks in bulk, quality verification separates reliable suppliers from problematic vendors. Best-practice procurement demands rigorous documentation and analytical confirmation.
Critical Documentation Requirements
Certificates of Analysis (COAs) represent the cornerstone of quality verification. Premium suppliers provide batch-specific COAs tied to unique lot numbers, ensuring traceability throughout the supply chain[1]. Each COA should include:
- HPLC chromatograms showing purity percentages (typically ≥98% for research-grade materials)
- Mass spectrometry (MS) identity confirmation displaying observed versus theoretical mass
- Optical rotation data for chiral amino acids
- Water content analysis via Karl Fischer titration
- Heavy metal screening results
Documentation access before purchase with consistent formatting serves as a key quality indicator, reducing procurement uncertainty[1]. Reputable suppliers maintain digital databases allowing buyers to review historical batch data.
Analytical Methods and Purity Standards
High-Performance Liquid Chromatography (HPLC) provides the primary purity assessment for FMOC-protected amino acids. Research-grade building blocks should demonstrate:
- Purity ≥98% by HPLC
- Single major peak in chromatogram
- Minimal degradation products
- Consistent retention times matching reference standards
Mass Spectrometry confirms molecular identity by detecting the expected mass-to-charge ratio. Discrepancies between observed and theoretical mass exceeding ±0.5 Da warrant investigation.
For researchers exploring wholesale peptides for sale, understanding these analytical standards ensures purchased building blocks meet synthesis requirements.
Red Flags and Warning Signs
Avoid suppliers who exhibit these problematic characteristics:
🚩 Refuse to provide batch-specific COAs before purchase
🚩 Offer prices significantly below market rates (>40% discount suggests quality concerns)
🚩 Cannot provide MS confirmation for identity verification
🚩 Lack GMP or ISO certifications for pharmaceutical-grade materials
🚩 Show inconsistent documentation formatting across batches
The lab tested peptides category demonstrates the importance of third-party verification for quality assurance.
Strategic Sourcing: How to Buy Peptide Building Blocks Bulk Effectively
Optimizing bulk procurement requires strategic planning beyond simply identifying low-cost suppliers. Successful buyers implement systematic evaluation frameworks and long-term supplier relationships.
Supplier Evaluation Framework
Step 1: Define Requirements
Clearly specify purity standards, quantities, delivery timelines, and budget constraints. Different research applications tolerate varying purity levels—pharmaceutical development demands higher standards than preliminary screening studies.
Step 2: Request Samples
Before committing to bulk orders, request small samples (1-5g) for in-house verification. Test samples using independent analytical methods to confirm supplier claims.
Step 3: Audit Documentation
Review multiple batch COAs spanning 6-12 months to assess consistency. Variation in purity >2% between batches suggests process control issues.
Step 4: Verify Regulatory Compliance
Confirm suppliers maintain appropriate certifications for intended applications. Pharmaceutical research requires GMP-compliant manufacturing, while basic research may accept lower standards.
Pricing Strategies and Negotiation
Bulk peptide building block pricing varies significantly based on:
- Order volume (price breaks typically occur at 100g, 500g, and 1kg thresholds)
- Amino acid complexity (standard amino acids cost less than unnatural variants)
- Protection strategy (FMOC protection costs more than BOC protection)
- Purity requirements (98% vs. 99.5% purity affects pricing)
Effective negotiation tactics include:
✅ Consolidate orders across multiple building blocks to increase total volume
✅ Commit to annual contracts for predictable pricing and supply
✅ Request custom synthesis quotes for specialized building blocks
✅ Leverage competitive quotes from multiple suppliers
For comprehensive peptide procurement, exploring all peptides for sale options provides context for building block pricing.
Supply Chain Risk Management
The peptide building block sector faces stringent regulatory requirements, supply chain disruption, and raw material price volatility[5]. Mitigate risks through:
Dual Sourcing: Qualify at least two suppliers for critical building blocks to prevent production halts during disruptions.
Inventory Buffering: Maintain 3-6 months of safety stock for essential building blocks, particularly those with long lead times.
Quality Agreements: Establish formal quality agreements defining specifications, testing requirements, and dispute resolution procedures.
Regulatory Monitoring: Track regulatory changes affecting peptide building block classification and import/export requirements.
Applications and Research Contexts for Bulk Peptide Building Blocks
Understanding application contexts helps optimize building block selection and supplier choice. The peptide building block market serves diverse research domains, each with specific requirements.
Pharmaceutical Development
The pharmaceutical sector drives substantial demand for high-purity peptide building blocks, particularly for GLP-1 drug development[4]. Companies developing peptide therapeutics require:
- Pharmaceutical-grade purity (≥99%)
- GMP-compliant manufacturing
- Comprehensive regulatory documentation
- Scalable supply for clinical trials
Researchers working with GLP-1 peptides benefit from understanding pharmaceutical-grade building block requirements.
Performance and Recovery Research
The fitness, martial arts, and weightlifting communities increasingly explore peptide research for performance optimization and recovery enhancement. Common research areas include:
Tissue Repair Peptides 🏋️
Building blocks for synthesizing peptides like BPC-157 support research into tissue healing mechanisms. Understanding BPC-157 research themes provides context for building block selection.
Growth Hormone Secretagogues 💪
Amino acid sequences for peptides like Ipamorelin require specific building blocks with precise stereochemistry.
Mitochondrial Peptides ⚡
Research into MOTS-C and SS-31 peptides demands specialized building blocks for mitochondrial-targeting sequences.
Cosmetic and Longevity Applications
The cosmetic peptide sector utilizes building blocks for synthesizing:
- Copper peptides like GHK-Cu for skin health research
- Antimicrobial peptides including LL-37 for dermatological applications
- Anti-aging peptides such as Epithalon for longevity research
The longevity peptide research field demonstrates growing demand for specialized building blocks supporting telomere and cellular senescence studies.
Cost-Benefit Analysis: Bulk Purchasing vs. Small-Scale Orders

Financial planning for bulk peptide building block procurement requires comprehensive cost-benefit analysis extending beyond simple per-unit pricing.
Direct Cost Comparisons
| Order Size | Cost per Gram (FMOC-Leu) | Total Cost | Savings vs. 10g |
|---|---|---|---|
| 10g | $45 | $450 | Baseline |
| 100g | $28 | $2,800 | 38% |
| 500g | $22 | $11,000 | 51% |
| 1kg | $18 | $18,000 | 60% |
Pricing examples based on typical market rates for standard FMOC-protected amino acids[4]
Hidden Costs and Considerations
Storage Requirements 🧊
FMOC-protected amino acids require controlled storage conditions (typically -20°C, desiccated environment). Bulk orders demand adequate freezer capacity and inventory management systems.
Expiration and Degradation ⏰
Most building blocks maintain stability for 2-3 years under proper storage. Calculate realistic usage rates to prevent waste from expired materials.
Opportunity Costs 💵
Capital tied up in inventory represents opportunity costs. Balance bulk discounts against alternative uses for research funding.
Quality Verification Expenses 🔬
Large orders may justify in-house analytical verification (HPLC, MS), adding equipment and personnel costs.
Break-Even Analysis
Calculate break-even points by comparing:
- Total bulk purchase cost (including storage, verification, opportunity costs)
- Equivalent small-order costs over the same timeframe
- Risk-adjusted savings accounting for potential waste
For most research programs using >100g annually of specific building blocks, bulk purchasing provides net positive returns.
Regulatory Compliance and Import Considerations
Navigating regulatory requirements represents a critical aspect of buying peptide building blocks bulk, particularly for international transactions.
United States Regulations
The FDA regulates peptide building blocks based on intended use:
Research Use Only (RUO): Building blocks for non-clinical research face minimal restrictions but cannot be marketed for human consumption.
Pharmaceutical Development: Building blocks for drug development require GMP-compliant manufacturing and comprehensive documentation trails.
Import Requirements: Customs declarations must accurately describe materials. Peptide building blocks typically fall under HS code 2930.90 (organo-sulfur compounds) or similar classifications.
International Shipping Challenges
Documentation Requirements 📋
International shipments require:
- Commercial invoices with accurate HS codes
- Material Safety Data Sheets (MSDS)
- Certificates of Analysis
- Import permits (jurisdiction-dependent)
Temperature Control ❄️
FMOC-protected amino acids often require cold-chain shipping to prevent degradation during transit. Factor cold-pack expenses and expedited shipping into total costs.
Customs Delays ⏱️
Budget 2-4 weeks for international shipments clearing customs. Delays can extend to 6-8 weeks for first-time importers or complex regulatory environments.
Compliance Best Practices
✅ Maintain detailed records of all building block purchases, including supplier information, COAs, and intended uses
✅ Work with experienced freight forwarders familiar with chemical shipping regulations
✅ Verify supplier export licenses for controlled substances or dual-use materials
✅ Consult legal counsel for pharmaceutical development applications requiring regulatory submissions
Future Trends: The Evolving Landscape of Peptide Building Blocks
The peptide building block market continues evolving rapidly, driven by technological innovation and expanding therapeutic applications.
Market Growth Projections
The global FMOC Protected Amino Acids market's projected growth from $130 million (2025) to $282 million (2034) represents a compound annual growth rate (CAGR) of approximately 9%[4]. Key growth drivers include:
Expanding Peptide Therapeutics Pipeline 💊
Over 80 peptide drugs currently hold FDA approval, with hundreds more in clinical development. Each new therapeutic creates demand for specific building blocks.
GLP-1 Receptor Agonist Development 🔬
The success of semaglutide and tirzepatide has sparked intense development activity in metabolic peptides, driving demand for specialized building blocks[4].
Personalized Medicine Applications 🧬
Custom peptide synthesis for personalized therapeutics requires diverse building block inventories supporting rapid sequence optimization.
Technological Innovations
Digital Procurement Platforms 💻
Leading suppliers are adopting digital technologies to enhance product portfolios and improve plant efficiency, operations, and productivity[5]. Online platforms now offer:
- Real-time inventory visibility
- Automated quote generation
- Digital COA access
- Predictive delivery estimates
Proprietary Unnatural Amino Acids 🧪
Innovative biotech firms are developing proprietary unnatural amino acids for novel therapeutic applications[4]. These specialized building blocks enable:
- Enhanced metabolic stability
- Improved receptor selectivity
- Novel mechanisms of action
- Intellectual property differentiation
Green Chemistry Initiatives 🌱
Environmental sustainability drives development of:
- Solvent-free synthesis methods
- Recyclable protecting groups
- Reduced waste generation
- Lower energy consumption processes
Competitive Dynamics
Market consolidation continues as Kelong Chemical and GL Biochem jointly account for 24.30% of global revenue[4]. This concentration creates both opportunities and risks:
Opportunities: Larger suppliers offer enhanced reliability, broader catalogs, and competitive volume pricing.
Risks: Reduced competition may limit pricing flexibility and innovation incentives.
Strategic buyers should monitor emerging suppliers and alternative technologies to maintain negotiating leverage and supply chain resilience.
Practical Purchasing Guide: Step-by-Step Process
Implementing a systematic approach to buy peptide building blocks bulk ensures optimal outcomes and minimizes procurement risks.
Phase 1: Needs Assessment (Weeks 1-2)
Inventory Current Stock 📊
Audit existing building block inventory, noting:
- Current quantities
- Expiration dates
- Usage rates over past 12 months
- Upcoming project requirements
Forecast Future Needs 📈
Project building block consumption for next 12-24 months based on:
- Planned research projects
- Historical usage patterns
- Anticipated new initiatives
- Buffer stock requirements
Define Specifications 📝
Document required specifications:
- Purity levels (98%, 99%, 99.5%)
- Quantity ranges
- Delivery timelines
- Budget constraints
- Regulatory requirements
Phase 2: Supplier Identification (Weeks 3-4)
Research Potential Suppliers 🔍
Compile list of 5-10 potential suppliers using:
- Industry directories
- Peer recommendations
- Online research
- Trade show contacts
Consider exploring peptide supplier comparisons for detailed evaluations.
Request Quotes 💰
Submit detailed RFQs (Request for Quotations) including:
- Specific building blocks and quantities
- Required delivery dates
- Quality specifications
- Payment terms preferences
Evaluate Proposals ⚖️
Compare quotes across multiple dimensions:
- Unit pricing
- Lead times
- Quality documentation
- Minimum order quantities
- Payment terms
Phase 3: Supplier Qualification (Weeks 5-8)
Request Sample Materials 🧪
Order small samples (1-5g) from top 2-3 suppliers for verification testing.
Conduct Analytical Testing 🔬
Perform independent analysis:
- HPLC purity confirmation
- MS identity verification
- Optical rotation (if applicable)
- Visual inspection for contamination
Audit Documentation 📑
Review supplier-provided documentation:
- COA completeness and accuracy
- Regulatory certifications
- Quality management systems
- Traceability procedures
Check References 📞
Contact existing customers to assess:
- Delivery reliability
- Quality consistency
- Customer service responsiveness
- Problem resolution effectiveness
Phase 4: Purchase Execution (Weeks 9-10)
Negotiate Final Terms 🤝
Finalize pricing, delivery schedules, and contractual terms. Key negotiation points:
- Volume discounts
- Payment terms (net 30, net 60)
- Delivery schedules
- Quality guarantees
- Return policies
Execute Purchase Orders 📋
Issue formal purchase orders specifying:
- Exact building blocks and quantities
- Agreed pricing
- Delivery addresses and dates
- Quality specifications
- Special handling requirements
Arrange Logistics 🚚
Coordinate shipping details:
- Cold-chain requirements
- Customs documentation
- Insurance coverage
- Tracking procedures
Phase 5: Receipt and Verification (Weeks 11-12)
Inspect Shipments 📦
Upon receipt, immediately verify:
- Package integrity
- Temperature maintenance (if cold-shipped)
- Quantity accuracy
- Labeling correctness
Perform Incoming QC ✅
Conduct quality control testing:
- Visual inspection
- Documentation review
- Analytical verification (sample testing)
- Lot number recording
Update Inventory Systems 💾
Record received materials:
- Quantities and lot numbers
- Receipt dates
- Storage locations
- Expiration dates
- COA filing
For ongoing peptide research needs, maintaining relationships with verified suppliers ensures consistent access to all peptides for sale and related building blocks.
Common Pitfalls and How to Avoid Them
Even experienced buyers encounter challenges when purchasing peptide building blocks in bulk. Recognizing common pitfalls enables proactive risk mitigation.
Quality-Related Pitfalls
Pitfall #1: Accepting Generic COAs ❌
Some suppliers provide generic certificates not tied to specific lots. This practice prevents verification that received materials match tested batches.
Solution: Demand batch-specific COAs with unique lot numbers matching product labels[1].
Pitfall #2: Ignoring Storage Requirements ❌
Improper storage rapidly degrades FMOC-protected amino acids, wasting procurement investments.
Solution: Verify adequate cold storage capacity before ordering. Store materials at -20°C in desiccated conditions.
Pitfall #3: Overlooking Expiration Dates ❌
Bulk orders may exceed realistic usage rates, leading to expired inventory.
Solution: Calculate maximum usable quantities based on 24-month shelf life and historical consumption rates.
Financial Pitfalls
Pitfall #4: Chasing Lowest Prices ❌
Exceptionally low pricing often indicates quality compromises or hidden costs.
Solution: Evaluate total cost of ownership including quality verification, potential re-work, and project delays from substandard materials.
Pitfall #5: Inadequate Budget Planning ❌
Bulk purchases require significant upfront capital that may strain research budgets.
Solution: Negotiate payment terms (installments, net 60) to manage cash flow. Consider consortium purchasing with other research groups.
Operational Pitfalls
Pitfall #6: Single-Source Dependency ❌
Relying exclusively on one supplier creates vulnerability to disruptions.
Solution: Qualify at least two suppliers for critical building blocks. Maintain relationships through periodic orders.
Pitfall #7: Poor Documentation Management ❌
Losing COAs or failing to maintain traceability records creates regulatory and quality issues.
Solution: Implement digital documentation systems linking lot numbers to COAs, purchase orders, and usage records.
Pitfall #8: Ignoring Regulatory Changes ❌
Peptide building block regulations evolve, potentially affecting import/export permissions or usage restrictions.
Solution: Subscribe to regulatory updates from FDA, DEA, and relevant international agencies. Consult legal counsel for pharmaceutical applications.
Maximizing Value: Advanced Strategies for Bulk Buyers

Sophisticated procurement strategies extend beyond basic price negotiation, creating sustainable competitive advantages.
Consortium Purchasing
Collaborative Buying Groups 🤝
Multiple research institutions or companies can pool purchasing power to:
- Access better volume discounts
- Share quality verification costs
- Distribute inventory risk
- Negotiate favorable payment terms
Implementation: Establish formal agreements defining:
- Order quantities per participant
- Cost allocation methods
- Quality acceptance criteria
- Inventory distribution procedures
Custom Synthesis Partnerships
For specialized building blocks not available commercially, custom synthesis partnerships offer advantages:
Exclusive Access 🔐
Proprietary building blocks create intellectual property differentiation and competitive moats.
Optimized Specifications 🎯
Custom synthesis allows precise specification of:
- Protection strategies
- Purity levels
- Packaging formats
- Delivery schedules
Long-Term Cost Reduction 💰
Initial custom synthesis costs may exceed catalog pricing, but economies of scale emerge with repeated orders.
Vertical Integration Opportunities
Large-scale peptide research programs may justify backward integration into building block production:
In-House Synthesis 🏭
Organizations with sufficient scale can:
- Eliminate supplier margins
- Ensure supply security
- Optimize production schedules
- Protect proprietary sequences
Feasibility Requirements:
- Annual consumption >10kg of specific building blocks
- Access to synthesis expertise
- Capital for equipment investment
- Regulatory compliance capabilities
Strategic Inventory Management
Just-In-Time (JIT) vs. Safety Stock ⚖️
Balance competing objectives:
JIT Advantages: Reduced carrying costs, minimized expiration risk, lower capital requirements
Safety Stock Advantages: Protection against supply disruptions, price volatility hedging, project continuity assurance
Optimal Strategy: Hybrid approach maintaining:
- JIT ordering for commodity building blocks with reliable supply
- Safety stock (3-6 months) for critical or long-lead-time materials
- Strategic reserves for building blocks with price volatility
The Role of Third-Party Testing and Verification
Independent verification provides critical quality assurance when purchasing peptide building blocks bulk, particularly from new or unfamiliar suppliers.
Third-Party Testing Services
Analytical Testing Laboratories 🔬
Specialized labs offer comprehensive testing:
- HPLC purity analysis
- Mass spectrometry identity confirmation
- Optical rotation measurement
- Residual solvent analysis
- Heavy metal screening
- Endotoxin testing (for pharmaceutical applications)
Cost Considerations: Third-party testing typically costs $200-500 per building block, representing 1-5% of bulk order values.
ROI Justification: Single instance of receiving substandard materials can cost 10-100x testing expenses through:
- Failed synthesis attempts
- Wasted reagents
- Project delays
- Regulatory complications
Implementing Testing Protocols
Risk-Based Testing Strategy 📊
High-Risk Scenarios (test every batch):
- New suppliers
- Critical building blocks for pharmaceutical development
- Unnatural amino acids with complex synthesis
- Materials from suppliers with quality history issues
Medium-Risk Scenarios (periodic testing):
- Established suppliers with good track records
- Standard FMOC amino acids
- Non-critical research applications
Low-Risk Scenarios (minimal testing):
- Long-term suppliers with excellent quality history
- Materials with supplier-provided third-party COAs
- Preliminary screening studies
Certification and Accreditation
Verify testing laboratories maintain appropriate accreditations:
ISO/IEC 17025: International standard for testing laboratory competence
GLP Compliance: Required for pharmaceutical development applications
FDA Registration: Necessary for materials supporting regulatory submissions
Understanding quality testing protocols helps establish appropriate verification standards.
Building Long-Term Supplier Relationships
Strategic supplier relationships create value beyond transactional cost savings, enabling innovation, reliability, and competitive advantages.
Relationship Development Framework
Phase 1: Transactional (Years 0-1) 🤝
Initial relationship focuses on:
- Establishing reliability
- Verifying quality consistency
- Testing communication responsiveness
- Confirming regulatory compliance
Phase 2: Preferred Supplier (Years 1-3) 🌟
Deepening relationship through:
- Volume commitments
- Information sharing
- Joint problem-solving
- Priority allocation during shortages
Phase 3: Strategic Partnership (Years 3+) 💎
Collaborative relationship featuring:
- Custom synthesis development
- Exclusive building blocks
- Joint research initiatives
- Integrated planning processes
Value-Added Services
Premium suppliers offer services beyond basic product delivery:
Technical Support 🔧
- Synthesis protocol optimization
- Troubleshooting assistance
- Literature references
- Application guidance
Regulatory Assistance 📋
- DMF (Drug Master File) support
- Regulatory documentation
- Audit preparation
- Compliance consulting
Inventory Management 📦
- Consignment inventory
- Vendor-managed inventory (VMI)
- Just-in-time delivery
- Emergency supply access
Custom Packaging 📦
- Application-specific quantities
- Specialized containers
- Inert atmosphere packaging
- Custom labeling
Performance Metrics and Scorecards
Implement systematic supplier evaluation using key performance indicators:
| Metric | Target | Weight |
|---|---|---|
| On-Time Delivery | >95% | 25% |
| Quality Conformance | >98% | 30% |
| Documentation Accuracy | >99% | 20% |
| Responsiveness | <24hr | 15% |
| Competitive Pricing | Top 3 quotes | 10% |
Quarterly scorecard reviews identify improvement opportunities and inform sourcing decisions.
Conclusion: Strategic Procurement for 2026 and Beyond
The landscape for purchasing peptide building blocks bulk in 2026 demands sophisticated procurement strategies combining quality verification, supplier relationships, and market intelligence. As the global market expands toward $282 million by 2034[4], successful buyers will differentiate through:
Quality-First Mindset ✅
Prioritizing batch-specific documentation, analytical verification, and regulatory compliance over simple cost minimization ensures research integrity and project success.
Strategic Supplier Relationships 🤝
Moving beyond transactional purchasing to collaborative partnerships unlocks value-added services, supply security, and innovation opportunities.
Risk Management 🛡️
Implementing dual sourcing, safety stock, and third-party verification protects against supply disruptions, quality failures, and regulatory complications.
Market Awareness 📊
Monitoring competitive dynamics, technological innovations, and regulatory changes enables proactive adaptation and competitive advantages.
Actionable Next Steps
Immediate Actions (This Week) 📅
- Audit current peptide building block inventory and forecast 12-month requirements
- Identify 3-5 potential suppliers for critical building blocks
- Request sample materials and quotes from qualified vendors
- Review existing supplier performance and documentation quality
Short-Term Actions (This Month) 📅
- Conduct third-party testing on samples from new suppliers
- Negotiate volume pricing and payment terms with top candidates
- Establish quality agreements defining specifications and testing requirements
- Implement digital documentation system for COA management
Long-Term Actions (This Quarter) 📅
- Develop strategic supplier relationships with 2-3 preferred vendors
- Create risk mitigation plan including dual sourcing and safety stock
- Establish supplier performance scorecard and quarterly review process
- Investigate consortium purchasing or custom synthesis opportunities
The peptide building block market's rapid evolution creates both opportunities and challenges. Organizations that invest in strategic procurement capabilities, quality verification systems, and supplier partnerships will achieve superior research outcomes while managing costs and risks effectively.
For comprehensive peptide research needs extending beyond building blocks, explore the full range of all peptides for sale to support diverse research applications from peptide therapy to specialized peptide blends.
By implementing the frameworks, strategies, and best practices outlined in this guide, researchers and procurement professionals can confidently navigate the complex peptide building block marketplace, securing high-quality materials that enable groundbreaking research and innovation.
References
[1] Best Peptide Vendors Online In 2026 Full Comparison Guide – https://cernumbiosciences.com/blogs/peptide-science-guide/best-peptide-vendors-online-in-2026-full-comparison-guide
[2] Fmocamino Acids C 60 – https://www.lifetein.com/peptide-product/fmocamino-acids-c-60.html
[3] Peptides – https://fillercloud.com/peptides/
[4] Global Fmoc Protected Amino Acids Forecast Market 27006 – https://www.intelmarketresearch.com/global-fmoc-protected-amino-acids-forecast-market-27006
[5] Peptide Building Block Market Report – https://www.cognitivemarketresearch.com/peptide-building-block-market-report
[6] Peptide Building Blocks 1232272148p – https://www.globalsources.com/Inorganic-chemical/Peptide-Building-Blocks-1232272148p.htm
[7] Peptides 101 Small Molecules Big Results – https://www.bulknaturalswholesale.com/blogs/ingredient-highlights/peptides-101-small-molecules-big-results
[8] Peptides – https://fillersupplies.com/product-category/peptides/
[9] Peptides – https://www.medicadepot.com/mesotherapy/peptides.html
SEO Meta Title and Description
Meta Title (58 characters):
Buy Peptide Building Blocks Bulk: 2026 Research Guide
Meta Description (157 characters):
Complete guide to buy peptide building blocks bulk in 2026. Compare top suppliers, quality standards, pricing strategies & procurement best practices.
