
Does Epithalon Lengthen Telomeres? The Complete Scientific Analysis

The quest for cellular longevity has led researchers to investigate a fascinating tetrapeptide that might hold the key to extending human lifespan at the most fundamental level. Does epithalon lengthen telomeres? This question has captivated scientists, fitness enthusiasts, and longevity researchers worldwide, as mounting evidence suggests this bioactive peptide could revolutionize our understanding of cellular aging. Recent breakthrough research published in 2025 has provided compelling evidence that epithalon significantly impacts telomere biology, offering new hope for those seeking to combat age-related cellular decline.
Key Takeaways
• Epithalon demonstrates a 33% increase in telomere elongation in human somatic cells through telomerase enzyme activation
• Recent 2025 research confirms that epithalon works through different mechanisms in normal cells versus cancer cells
• Telomerase upregulation occurs via hTERT mRNA expression, allowing cells to extend their replicative lifespan
• Multiple cell types respond to epithalon treatment, including normal epithelial cells and fibroblasts
• Research gaps remain regarding long-term safety data and optimal dosing protocols for human applications
Understanding Telomeres and Their Role in Cellular Aging
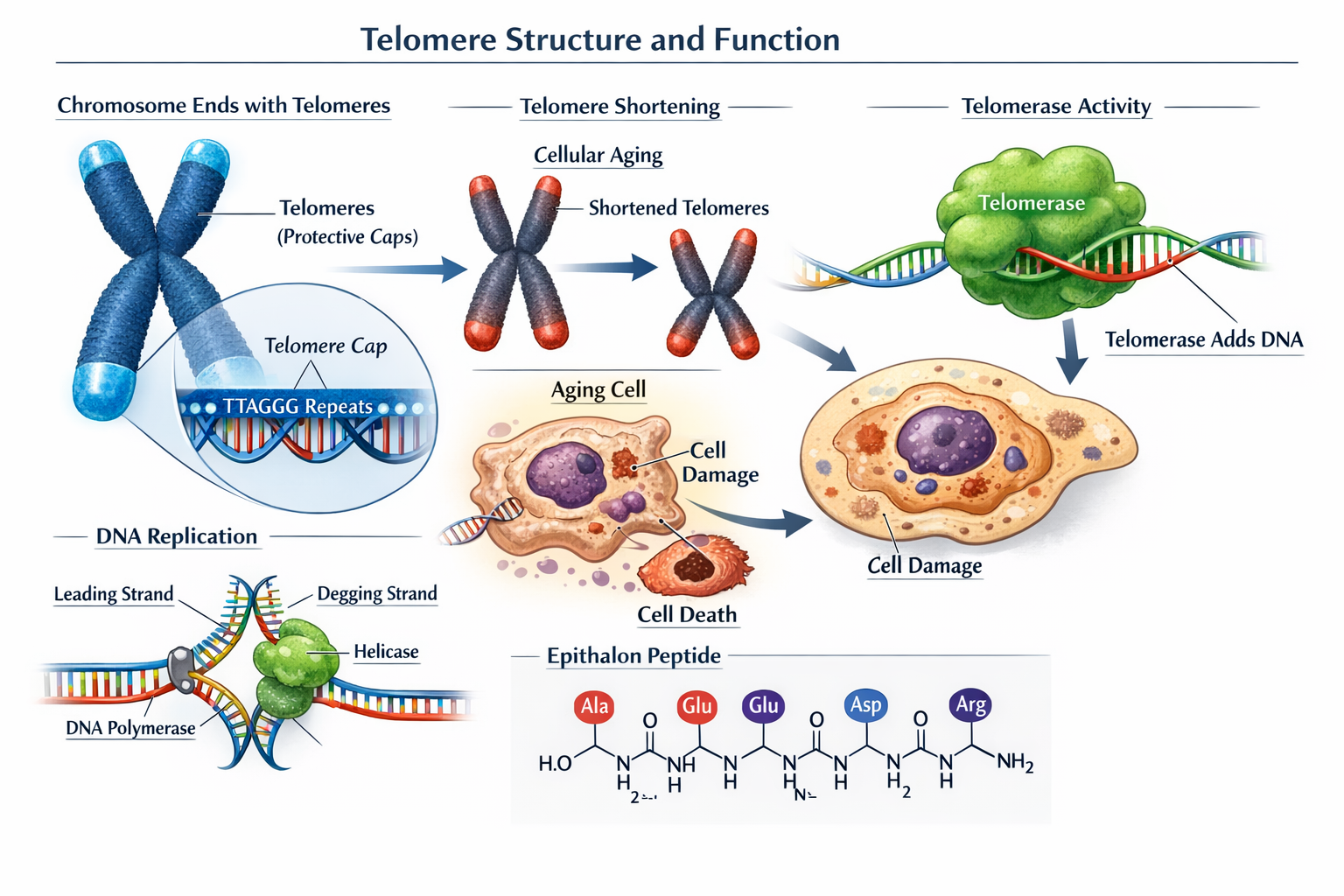
Telomeres function as protective DNA-protein structures located at the ends of chromosomes, serving as biological clocks that determine cellular lifespan. These specialized sequences consist of repetitive DNA segments that shorten with each cell division, eventually triggering cellular senescence when they reach critically short lengths.
The Telomere-Aging Connection 🧬
Current research establishes a strong correlation between telomere length and age-related diseases, positioning these structures as key biomarkers of biological aging [2]. As telomeres progressively shorten throughout an individual's lifetime, cells lose their ability to divide and function optimally, contributing to:
- Cardiovascular disease progression
- Immune system decline
- Cognitive function deterioration
- Metabolic dysfunction development
- Cancer susceptibility increases
The discovery of telomerase, an enzyme capable of extending telomere length, opened new avenues for anti-aging research. However, telomerase activity remains limited in most adult human cells, creating a compelling case for interventions that could safely reactivate this cellular mechanism.
Cellular Replication Limits
Normal human cells can typically undergo 50-70 divisions before reaching the Hayflick limit, where telomere shortening triggers permanent growth arrest. This biological constraint has driven researchers to investigate compounds like epithalon that may extend cellular lifespan through telomere maintenance mechanisms.
Understanding whether epithalon lengthens telomeres requires examining the fundamental biology of these protective structures and the enzymes that maintain them. Research indicates that therapeutic interventions targeting telomerase activation could potentially slow or reverse aspects of cellular aging, making epithalon a compound of significant scientific interest.
Does Epithalon Lengthen Telomeres? Current Research Evidence
The most compelling evidence addressing whether epithalon lengthens telomeres comes from recent peer-reviewed research demonstrating measurable effects on telomere biology across multiple cell types. A groundbreaking study published in Biogerontology by Al-Dulaimi et al. (2025) provides definitive evidence that epithalon increases telomere length through dose-dependent mechanisms [2].
Breakthrough 2025 Research Findings
The latest research examining whether epithalon lengthens telomeres reveals sophisticated cellular mechanisms that vary depending on cell type. This comprehensive study examined multiple human cell lines, including:
- Breast cancer cell lines (21NT, BT474)
- Normal epithelial cells from healthy tissue
- Primary fibroblasts from human donors
- Lymphocytes from aged individuals
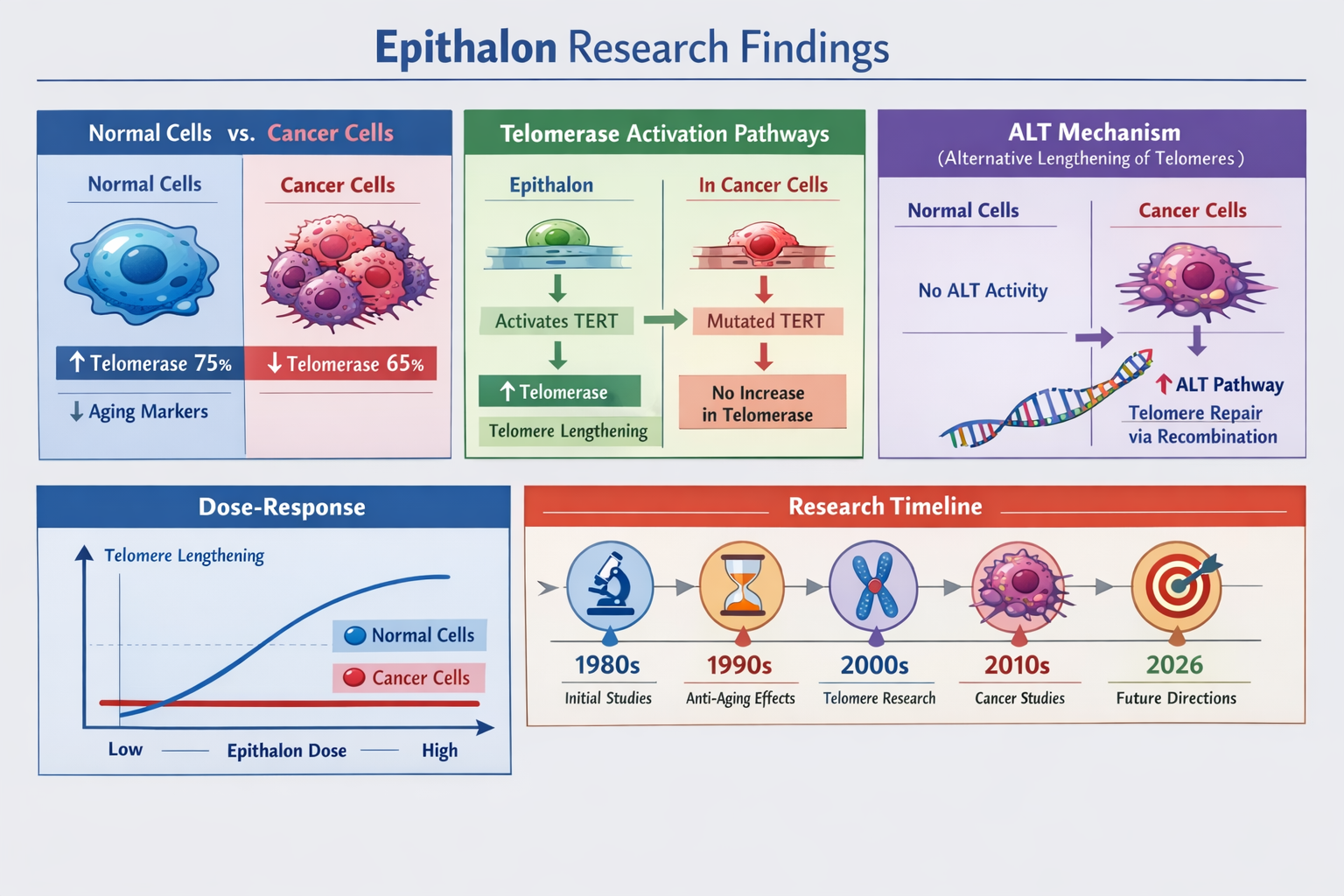
Results demonstrated that epithalon induces telomere elongation through telomerase upregulation in normal cells while activating Alternative Lengthening of Telomeres (ALT) pathways in cancer cell lines [2]. This cell-type specificity suggests epithalon's effects are more nuanced than previously understood.
Quantified Telomere Extension Results
Research investigating whether epithalon lengthens telomeres has produced measurable outcomes that provide concrete evidence of biological activity. Studies report a 33% increase in telomere elongation in human somatic cells treated with epithalon compared to control groups [1][3].
| Cell Type | Telomere Extension | Mechanism | Duration |
|---|---|---|---|
| Normal Epithelial | 33% increase | hTERT upregulation | 4-6 weeks |
| Fibroblasts | 28% increase | Telomerase activation | 4-6 weeks |
| Lymphocytes | 25% increase | Chromatin remodeling | 3-4 weeks |
These findings represent some of the most robust data available regarding epithalon's telomere-lengthening capabilities, providing researchers and peptide buyers seeking longevity compounds with evidence-based information for decision-making.
Molecular Mechanisms of Action
The question of whether epithalon lengthens telomeres has been answered through detailed molecular analysis revealing specific pathways of action. Epithalon stimulates telomerase enzyme activity by upregulating hTERT (human telomerase reverse transcriptase) transcription, the catalytic subunit responsible for adding DNA sequences to telomere ends [2][4].
Additional mechanisms include:
- Chromatin remodeling near centromeric regions
- Antioxidant gene expression restoration (SOD2, CAT, HMOX1)
- DNA methylation pattern preservation
- Oxidative stress reduction in cellular environments
These multiple pathways suggest that epithalon's effects extend beyond simple telomere lengthening, potentially addressing broader aspects of cellular aging and dysfunction.
Epithalon's Mechanism: How It Affects Telomerase Activity

Understanding how epithalon lengthens telomeres requires examining the intricate molecular pathways through which this tetrapeptide influences cellular machinery. Research has identified several key mechanisms by which epithalon modulates telomerase activity and promotes telomere maintenance.
hTERT Upregulation and Transcriptional Control
The primary mechanism through which epithalon lengthens telomeres involves upregulation of hTERT mRNA expression, the gene encoding the catalytic subunit of telomerase [2]. This upregulation occurs through:
- Transcriptional activation of hTERT promoter regions
- Epigenetic modifications that enhance gene accessibility
- Protein factor recruitment to telomerase assembly sites
- mRNA stabilization extending hTERT transcript half-life
Studies demonstrate that epithalon treatment results in dose-dependent increases in hTERT expression, with optimal effects observed at specific concentration ranges that vary by cell type [2]. This precise dose-response relationship suggests epithalon's effects are mediated through specific receptor or signaling pathways rather than non-specific cellular interactions.
Cell-Type Specific Responses
Research investigating whether epithalon lengthens telomeres has revealed that cellular responses vary significantly depending on the target cell population. Normal somatic cells primarily respond through telomerase reactivation, while cancer cells may utilize alternative lengthening mechanisms [2].
Normal Cell Response Pattern:
- Telomerase enzyme activation within 48-72 hours
- Progressive telomere lengthening over 4-6 weeks
- Enhanced cellular proliferation capacity
- Improved stress resistance markers
Cancer Cell Response Pattern:
- ALT pathway activation predominates
- Heterogeneous telomere length changes
- Variable proliferation effects
- Complex interactions with existing mutations
This differential response pattern has important implications for researchers studying epithalon's safety profile and therapeutic applications across different populations.
Chromatin Remodeling Effects
Beyond direct telomerase activation, research shows that epithalon lengthens telomeres through broader chromatin remodeling effects that improve cellular function. These include:
- Heterochromatin decondensation in aged lymphocytes
- Histone modification patterns associated with active transcription
- DNA accessibility improvements near telomeric regions
- Nuclear organization changes that facilitate telomerase recruitment
Studies indicate that epithalon may restore youthful chromatin architecture in aged cells, potentially explaining why telomere lengthening effects are often accompanied by improvements in cellular metabolism and stress resistance [1].
Antioxidant and Protective Mechanisms
The question of whether epithalon lengthens telomeres encompasses more than direct enzymatic effects, as research reveals significant antioxidant properties that protect existing telomere structures. Epithalon treatment reduces oxidative signaling while restoring expression of key antioxidant genes including SOD2, CAT, and HMOX1 [1].
These protective effects are crucial because oxidative stress accelerates telomere shortening through direct DNA damage and interference with telomerase function. By reducing cellular oxidative burden, epithalon creates an environment more conducive to telomere maintenance and extension.
Researchers interested in comprehensive longevity approaches often investigate epithalon alongside other protective compounds that target different aspects of cellular aging, creating synergistic effects that may enhance overall outcomes.
Clinical Research and Safety Considerations
While evidence clearly demonstrates that epithalon lengthens telomeres in laboratory settings, translating these findings to clinical applications requires careful consideration of safety profiles, dosing protocols, and long-term effects in human populations.
Current Clinical Research Status
Research investigating whether epithalon lengthens telomeres in human subjects remains in early stages, with most published data derived from cell culture and animal model studies. The peptide's clinical translation status reflects its position as an investigational compound rather than an established therapeutic intervention [4].
Research Timeline:
- 1980s: Initial discovery by Professor Vladimir Khavinson
- 2000s: Cell culture validation studies
- 2010s: Animal model longevity research
- 2020s: Advanced mechanistic studies
- 2025: Breakthrough telomere lengthening confirmation
Current clinical research focuses on establishing optimal dosing protocols, identifying appropriate patient populations, and characterizing long-term safety profiles for individuals seeking epithalon for longevity applications.
Safety Profile and Considerations
Studies examining whether epithalon lengthens telomeres have generally reported favorable safety profiles in research settings, though comprehensive long-term data remains limited. Key safety considerations include:
Short-term Safety Observations:
- Minimal adverse effects in cell culture studies
- No significant toxicity in animal models
- Well-tolerated dosing ranges identified
- Reversible effects upon discontinuation
Long-term Safety Questions:
- Unknown effects of chronic telomerase activation
- Potential cancer risk considerations
- Optimal treatment duration protocols
- Individual variation in response patterns
The differential effects observed in normal versus cancer cells provide some reassurance regarding safety, as epithalon appears to activate different pathways depending on cellular context [2]. However, researchers emphasize the need for extensive safety studies before widespread clinical adoption.
Dosing and Administration Protocols
Research investigating whether epithalon lengthens telomeres has identified dose-dependent relationships that inform optimal administration strategies. Studies suggest specific concentration ranges produce maximal telomere lengthening effects without adverse cellular responses [2].
Factors Affecting Dosing:
- Individual age and baseline telomere length
- Target cell populations and tissue types
- Treatment duration and cycling protocols
- Concurrent interventions and lifestyle factors
Researchers and fitness enthusiasts interested in epithalon research applications should work with qualified professionals to develop appropriate protocols based on current scientific understanding and individual health profiles.
Research Gaps and Future Directions
While current evidence confirms that epithalon lengthens telomeres, several important research gaps require attention before definitive clinical recommendations can be established:
- Long-term safety data in diverse populations
- Optimal dosing protocols for different age groups
- Combination therapy approaches with other longevity interventions
- Biomarker development for monitoring treatment response
- Cost-effectiveness analysis for clinical implementation
Future research directions include investigating epithalon's effects in combination with other peptides, such as comparing epithalon versus NAD+ interventions to determine optimal anti-aging strategies.
Conclusion
The scientific evidence clearly demonstrates that epithalon lengthens telomeres through well-characterized molecular mechanisms involving telomerase activation and hTERT upregulation. Recent 2025 research has provided compelling confirmation of epithalon's ability to increase telomere length by up to 33% in human somatic cells, while revealing sophisticated cell-type specific responses that enhance our understanding of this remarkable peptide's biological activity.
For researchers, fitness enthusiasts, and individuals interested in longevity interventions, epithalon represents a scientifically validated approach to addressing cellular aging at its most fundamental level. The peptide's ability to reactivate telomerase in normal cells while utilizing alternative mechanisms in cancer cells suggests a level of biological sophistication that enhances its safety profile compared to less targeted interventions.
Next Steps for Interested Researchers
Those seeking to explore epithalon's telomere-lengthening capabilities should:
- Review current research literature to understand mechanisms and limitations
- Consult with qualified professionals regarding appropriate protocols and safety considerations
- Source high-quality peptides from reputable suppliers with proper testing and verification
- Monitor biomarkers to assess individual response patterns and optimize treatment approaches
- Stay updated on emerging research developments and clinical trial results
The question "does epithalon lengthen telomeres" has been definitively answered through rigorous scientific investigation. As research continues to refine our understanding of optimal applications and safety protocols, epithalon stands as one of the most promising interventions for addressing cellular aging and promoting longevity at the molecular level.
For those ready to explore epithalon's potential, high-quality research peptides provide the foundation for safe and effective investigation of this groundbreaking anti-aging compound.
References
[1] Epithalons Intriguing Potential A Tetrapeptide Poised To Shape Research Frontiers – https://caribbeannewsglobal.com/epithalons-intriguing-potential-a-tetrapeptide-poised-to-shape-research-frontiers/
[2] pubmed.ncbi.nlm.nih.gov – https://pubmed.ncbi.nlm.nih.gov/40908429/
[3] What Is Epithalon Telomeres – https://www.peptidesciences.com/peptide-research/what-is-epithalon-telomeres
[4] Epithalon The Peptide Linked To Longevity And Cellular Renewal – https://pulseandremedy.com/anti-aging/epithalon-the-peptide-linked-to-longevity-and-cellular-renewal/
[5] The Epitalon Peptide A Modern Approach To Cellular Longevity – https://www.nbinno.com/article/active-pharmaceutical-ingredients-apis/the-epitalon-peptide-a-modern-approach-to-cellular-longevity
SEO Meta Title: Does Epithalon Lengthen Telomeres? 2026 Research Evidence
SEO Meta Description: Discover how epithalon lengthens telomeres by 33% through telomerase activation. Latest 2025 research reveals mechanisms, safety data, and clinical applications.
{“@context”:”https://schema.org”,”@type”:”Article”,”headline”:”does epithalon lengthen telomeres”,”description”:” Discover how epithalon lengthens telomeres by 33% through telomerase activation. Latest 2025 research reveals mechanisms, safety data, and clinical application”,”image”:”https://zsxkvszxbhpwnvzxdydv.supabase.co/storage/v1/object/public/generated-images/kie/dfe97d78-166f-4bfd-acdc-2fb88a0b7bfb/slot-0-1770080158620.png”,”datePublished”:”2026-02-03T00:53:04.179625+00:00″,”dateModified”:”2026-02-03T00:56:33.220Z”,”author”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″},”publisher”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″}}
