
Epithalon Telomerase Activation: The Science Behind Cellular Longevity

Imagine if there was a molecular switch that could potentially slow down the aging process at the cellular level. Epithalon telomerase activation represents one of the most fascinating frontiers in longevity research, where a simple four-amino acid peptide might hold the key to extending cellular lifespan and promoting healthier aging.
In 2026, biohackers and longevity enthusiasts are increasingly turning their attention to peptides that can influence fundamental aging mechanisms. Among these compounds, Epithalon stands out for its unique ability to activate telomerase, the enzyme responsible for maintaining telomeres—the protective caps on our chromosomes that shorten with age.
Key Takeaways
• Epithalon activates telomerase enzyme in human cells, leading to measurable telomere elongation averaging 33.3% in research studies
• The peptide upregulates hTERT expression, the catalytic component of telomerase that's typically inactive in most adult cells
• Animal studies demonstrate significant lifespan extension, with mice showing 12.3% increased maximum lifespan
• Research indicates multiple pathways of action, including circadian rhythm restoration and anti-tumor effects
• Current evidence is primarily preclinical, with limited human clinical data available as of 2026
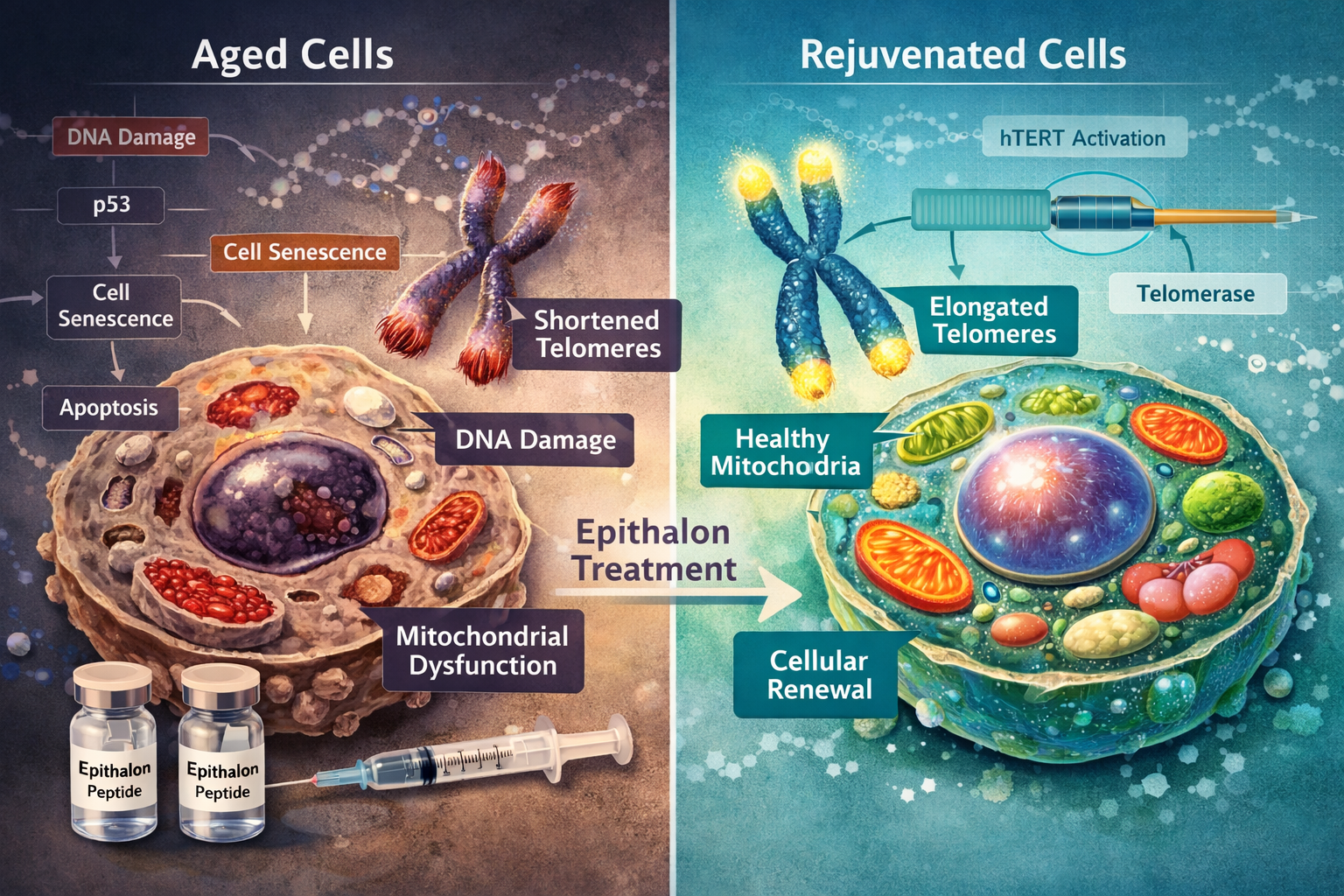
Understanding Telomeres and Cellular Aging
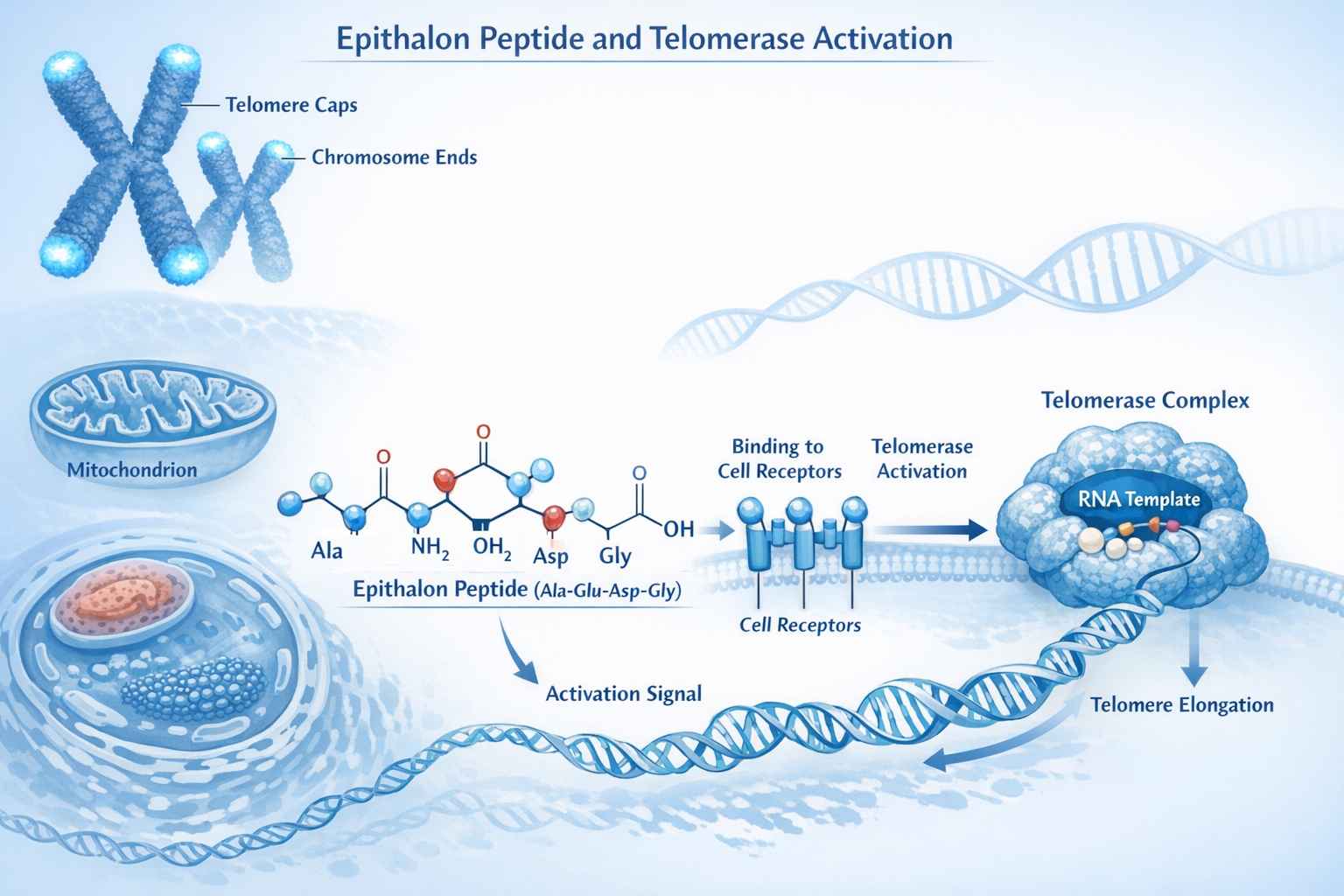
Telomeres function as molecular clocks within our cells. Every time a cell divides, these protective DNA-protein structures at chromosome ends become slightly shorter. When telomeres reach critically short lengths, cells enter senescence or die—a process directly linked to aging and age-related diseases.
The enzyme telomerase can add DNA sequences back to telomeres, effectively "rewinding" the cellular clock. However, most adult human cells have very low or undetectable telomerase activity. This is where Epithalon telomerase activation becomes particularly intriguing for researchers studying cellular longevity.
The Telomerase Challenge
In healthy adults, telomerase remains active primarily in:
- Stem cells 🧬
- Immune system cells
- Reproductive cells
- Some highly regenerative tissues
Most other cell types—including muscle, skin, and organ cells—show minimal telomerase activity, making them susceptible to replicative aging. This selective activation pattern explains why interventions targeting Epithalon longevity pathways have captured scientific attention.
How Epithalon Telomerase Activation Works
The mechanism behind Epithalon telomerase activation involves multiple cellular pathways that researchers are still working to fully understand. Current evidence suggests this tetrapeptide (Ala-Glu-Asp-Gly) influences gene expression patterns related to cellular aging and telomere maintenance.
Primary Mechanisms of Action
hTERT Gene Upregulation: Research demonstrates that Epithalon significantly increases expression of hTERT (human telomerase reverse transcriptase) mRNA, the catalytic subunit of the telomerase enzyme complex [1][2]. This upregulation occurs through pathways that may involve:
- Transcriptional activation of telomerase genes
- Epigenetic modifications affecting gene accessibility
- Signal transduction cascades influencing cellular aging programs
Direct Telomerase Enzyme Activation: Beyond gene expression changes, Epithalon appears to directly enhance telomerase enzyme activity in cells where the enzyme is present but inactive [2].
Cellular Response Patterns
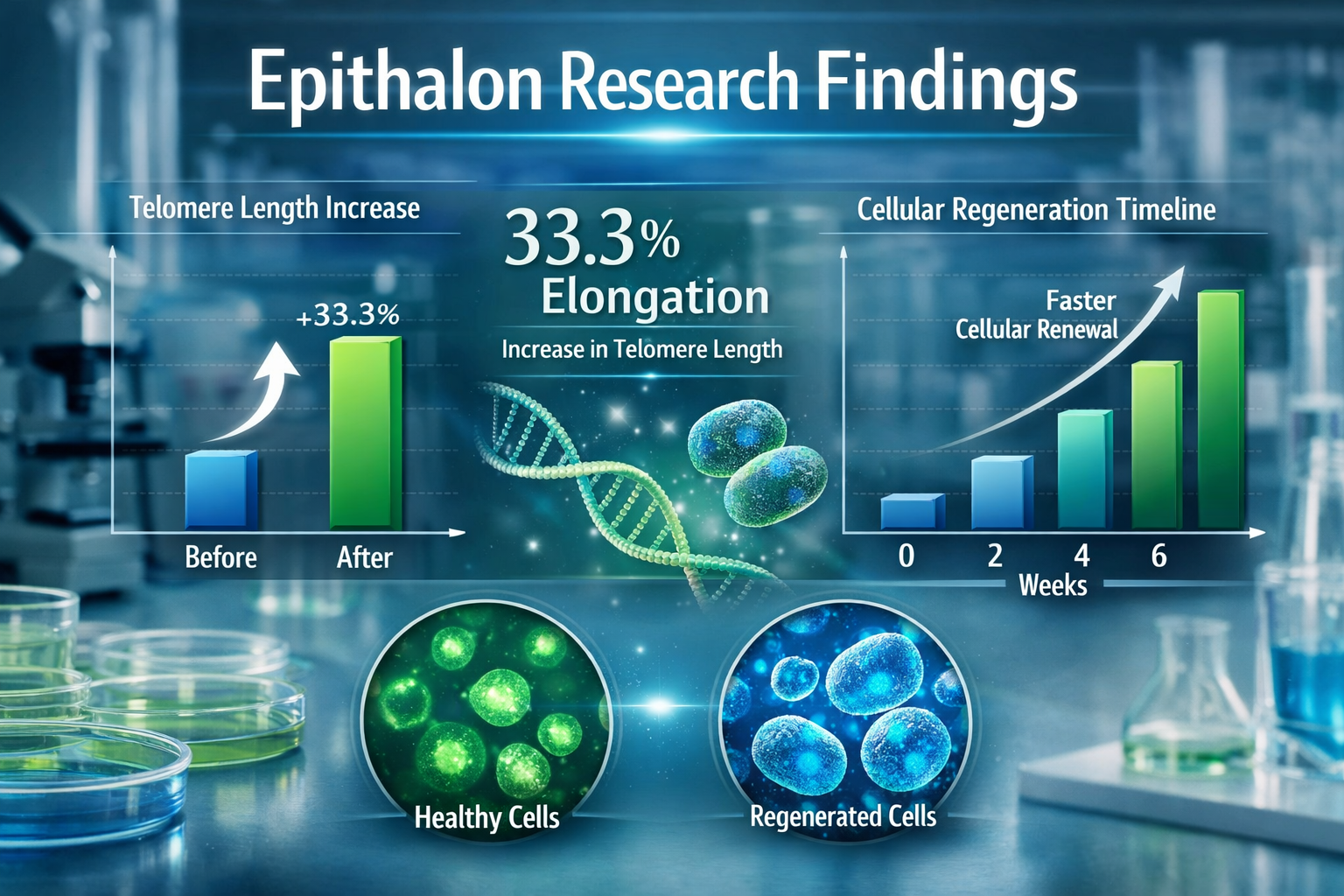
Human fetal fibroblasts treated with Epithalon showed remarkable telomere elongation averaging 33.3% in controlled laboratory studies [1][2]. This elongation occurred in a dose-dependent manner, suggesting that higher concentrations within safe ranges produced more pronounced effects.
Interestingly, the peptide's effects varied by cell type:
- Normal epithelial cells: Consistent telomere lengthening
- Fibroblast cells: Strong dose-dependent responses
- Cancer cell lines: Activation of Alternative Lengthening of Telomeres (ALT) pathways [2]
For those interested in exploring research-grade peptides, understanding these cellular mechanisms provides crucial context for evaluating potential applications.
Research Evidence and Clinical Findings
The scientific foundation for Epithalon telomerase activation spans multiple research domains, from basic cellular studies to animal longevity experiments. While human clinical data remains limited in 2026, the existing evidence provides compelling insights into this peptide's potential mechanisms and effects.
Animal Longevity Studies
The most striking evidence comes from controlled animal studies examining lifespan extension. In carefully designed experiments, Epithalon treatment produced measurable increases in both average and maximum lifespan:
- Maximum lifespan increase: 12.3% in treated mice compared to controls [1]
- Late-life survival: 13.3% extension in the lifespan of the last 10% of survivors [1]
- Healthspan improvements: Enhanced physical condition and reduced age-related pathology
These results suggest that Epithalon telomerase activation may influence fundamental aging processes beyond simple telomere maintenance.
Anti-Cancer Properties
Paradoxically, despite activating telomerase—an enzyme often overactive in cancer cells—Epithalon demonstrated protective effects against certain malignancies. Treatment inhibited leukemia development by 6.0-fold in animal models without increasing overall spontaneous tumor incidence [1].
This apparent contradiction highlights the complex relationship between telomerase activity and cancer development. The peptide's effects may involve:
- Immune system enhancement through improved T-cell function
- Cellular quality control mechanisms that eliminate damaged cells
- Balanced telomerase activation that supports healthy cells while not promoting malignant transformation
Circadian Rhythm Restoration
Beyond direct cellular effects, research in aged primates revealed that Epithalon administration restored normal circadian rhythm patterns [1]. Treated animals showed:
- Normalized evening melatonin levels
- Restored cortisol circadian rhythms
- Improved sleep-wake cycle regulation
These findings suggest broader physiological effects that may contribute to the peptide's longevity-promoting properties. For researchers comparing different longevity interventions, examining Epithalon vs NAD+ evidence provides valuable context for understanding complementary approaches.
Current Research Limitations
While existing studies provide encouraging preliminary data, several important limitations affect interpretation of Epithalon telomerase activation research:
Limited Human Data: Most compelling evidence comes from animal studies and cell culture experiments. Long-term human clinical trials examining safety and efficacy remain sparse [3].
Dosing Protocols: Optimal dosing regimens, treatment duration, and administration routes require further investigation across different populations and health conditions.
Individual Variation: Genetic factors, baseline telomere length, age, and health status likely influence individual responses to telomerase activation interventions.
Practical Applications and Considerations
For biohackers and longevity enthusiasts considering Epithalon telomerase activation as part of their optimization protocols, several practical factors deserve careful attention. The current state of research provides guidance while highlighting areas requiring additional investigation.
Protocol Development
Research suggests that Epithalon's effects depend significantly on dosing patterns and administration methods. While specific protocols vary across studies, several principles emerge from available data:
Cycling Approaches: Many researchers recommend intermittent rather than continuous administration, potentially mimicking natural biological rhythms and avoiding cellular adaptation.
Timing Considerations: Some evidence suggests that administration timing relative to circadian rhythms may influence effectiveness, particularly given the peptide's effects on melatonin and cortisol patterns [1].
Duration Parameters: Short-term intensive cycles followed by rest periods appear in multiple research protocols, though optimal cycle lengths remain under investigation.
Quality and Sourcing Considerations
The peptide research landscape requires careful attention to compound quality and purity. When evaluating where to buy Epithalon online, researchers should prioritize:
- Third-party testing verification
- Certificate of analysis availability
- Proper storage and handling protocols
- Transparent sourcing information
Monitoring and Assessment
Tracking responses to Epithalon telomerase activation interventions presents unique challenges since direct telomere length measurement requires specialized laboratory testing. Alternative monitoring approaches might include:
Biomarker Tracking: Regular assessment of inflammatory markers, metabolic parameters, and hormonal profiles that may reflect cellular health improvements.
Subjective Measures: Sleep quality, energy levels, recovery patterns, and cognitive function changes that could indicate broader physiological effects.
Professional Oversight: Working with healthcare providers familiar with peptide research to monitor safety parameters and potential interactions.
For those exploring comprehensive approaches to longevity optimization, examining research-grade peptide options provides access to properly characterized compounds for scientific investigation.
Safety Profile and Contraindications
Current research suggests a relatively favorable safety profile for Epithalon in healthy populations, though several considerations merit attention:
Known Interactions: Limited data exists regarding interactions with medications, supplements, or other peptides commonly used in biohacking protocols.
Population-Specific Effects: Responses may vary significantly based on age, genetic background, existing health conditions, and baseline telomerase activity levels.
Long-term Considerations: While animal studies suggest safety with extended use, human long-term safety data remains limited as of 2026 [3].
The complexity of Epithalon telomerase activation mechanisms suggests that individual responses may vary considerably, emphasizing the importance of careful monitoring and gradual protocol development.
Future Directions and Research Opportunities
The field of Epithalon telomerase activation research continues evolving rapidly as scientists work to understand the full scope of this peptide's effects on cellular aging and longevity. Several key areas represent particularly promising directions for future investigation.
Mechanistic Understanding
While current research demonstrates clear effects on telomerase activity and telomere length, the complete molecular pathways remain partially understood. Future studies will likely focus on:
Gene Expression Profiling: Comprehensive analysis of how Epithalon influences cellular gene expression patterns beyond hTERT upregulation, potentially revealing additional longevity-related pathways.
Epigenetic Effects: Investigation of whether the peptide influences DNA methylation patterns, histone modifications, or other epigenetic markers associated with aging and cellular reprogramming.
Cellular Signaling Networks: Detailed mapping of the signal transduction cascades that mediate Epithalon's effects on telomerase activation and cellular function.
Clinical Translation
The transition from promising preclinical results to human clinical applications represents a critical frontier for Epithalon telomerase activation research:
Phase I Safety Studies: Comprehensive dose-escalation studies in healthy volunteers to establish safety parameters and optimal dosing ranges for human use.
Biomarker Development: Identification of reliable, accessible biomarkers that can track treatment responses without requiring direct telomere length measurement.
Population-Specific Studies: Investigation of how factors like age, genetic background, and baseline health status influence individual responses to telomerase activation.
Combination Approaches
Growing evidence suggests that longevity interventions may work synergistically when combined appropriately. Research areas include:
Metabolic Optimization: Combining Epithalon with compounds targeting mitochondrial function, such as MOTS-C peptide research, to address multiple aging pathways simultaneously.
Cellular Protection: Integration with antioxidant systems and cellular repair mechanisms to maximize the benefits of telomere lengthening.
Lifestyle Synergies: Understanding how diet, exercise, sleep optimization, and stress management interact with Epithalon telomerase activation to enhance overall effectiveness.
Technology Integration
Advances in biotechnology and monitoring capabilities will likely enhance both research and practical applications:
Personalized Protocols: Development of genetic testing and biomarker panels that can guide individualized dosing and timing recommendations.
Real-time Monitoring: Wearable devices and home testing kits that allow continuous tracking of relevant physiological parameters during treatment cycles.
Delivery Optimization: Novel delivery methods that might enhance bioavailability and target specific cell populations more effectively.
The convergence of these research directions suggests that Epithalon telomerase activation may eventually become part of comprehensive, personalized longevity protocols tailored to individual genetic and physiological profiles.
For researchers interested in staying current with developments in this field, accessing high-quality research compounds remains essential for conducting meaningful investigations into peptide-based longevity interventions.
Conclusion
Epithalon telomerase activation represents a compelling intersection of cutting-edge longevity research and practical biohacking applications. The evidence demonstrating measurable telomere elongation, lifespan extension in animal models, and multi-system physiological effects positions this tetrapeptide as a significant focus for cellular aging research in 2026.
The research foundation, while promising, emphasizes the importance of approaching Epithalon telomerase activation with scientific rigor and appropriate caution. Current studies provide strong mechanistic rationale and encouraging preliminary results, but the field requires additional human clinical data to fully establish optimal protocols and long-term safety profiles.
Actionable Next Steps
For Researchers: Consider incorporating telomerase activation studies into broader longevity research programs, focusing on biomarker development and mechanistic understanding. Access to research-grade Epithalon enables controlled investigations into this promising compound.
For Biohackers: Develop comprehensive monitoring protocols that track multiple biomarkers beyond direct telomere measurement. Work with qualified healthcare providers to design safe, evidence-based approaches to peptide research.
For the Longevity Community: Stay informed about emerging research developments while maintaining realistic expectations about current evidence limitations. Support continued research through participation in appropriate studies and data sharing initiatives.
The future of Epithalon telomerase activation research will likely reveal additional mechanisms, optimal protocols, and synergistic combinations that enhance its longevity-promoting potential. As our understanding deepens, this peptide may become a cornerstone of evidence-based approaches to healthy aging and cellular optimization.
The journey toward effective longevity interventions requires patience, scientific rigor, and commitment to safety. Epithalon's unique ability to influence fundamental aging processes at the cellular level makes it a valuable tool for researchers and biohackers dedicated to advancing human healthspan and longevity in 2026 and beyond.
References
[1] Telomeres And Epithalon – https://www.peptidesciences.com/peptide-research/telomeres-and-epithalon
[2] sciety – https://sciety.org/articles/activity/10.21203/rs.3.rs-7066545/v1
[3] Epithalon The Peptide Linked To Longevity And Cellular Renewal – https://pulseandremedy.com/anti-aging/epithalon-the-peptide-linked-to-longevity-and-cellular-renewal/
SEO Meta Title: Epithalon Telomerase Activation: Science & Research Guide 2026
SEO Meta Description: Discover how Epithalon activates telomerase for cellular longevity. Research evidence, mechanisms, and practical applications for biohackers in 2026.
{“@context”:”https://schema.org”,”@type”:”Article”,”headline”:”Epithalon telomerase activation”,”description”:” Discover how Epithalon activates telomerase for cellular longevity. Research evidence, mechanisms, and practical applications for biohackers in 2026.”,”image”:”https://zsxkvszxbhpwnvzxdydv.supabase.co/storage/v1/object/public/generated-images/kie/89ea0e83-9143-4d0a-af77-a4aa50adecde/slot-0-1770266891981.png”,”datePublished”:”2026-02-05T04:45:41.283833+00:00″,”dateModified”:”2026-02-05T04:48:42.668Z”,”author”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″},”publisher”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″}}
