GLP-3 Peptide Mechanism of Action: The Triple-Receptor Revolution in Metabolic Health 🔬
Imagine a single molecule that could simultaneously activate three different hormone pathways in your body, creating a metabolic symphony that burns fat faster, controls blood sugar more effectively, and suppresses appetite more powerfully than anything currently available. That's exactly what the GLP-3 peptide mechanism of action delivers—and it's changing everything we thought we knew about weight management and metabolic health in 2026. Unlike traditional single-pathway medications, the glp-3 peptide mechanism of action represents a paradigm shift in how researchers approach obesity, diabetes, and metabolic disorders through peptide therapy.
Key Takeaways
- Triple-receptor targeting: The GLP-3 peptide mechanism of action works by simultaneously activating GLP-1, GIP, and glucagon receptors, creating synergistic metabolic effects that single-pathway drugs cannot match[1][2]
- Superior weight loss results: Clinical trials show participants achieving 24.2% to 28.7% body weight reduction—nearly double the results of traditional GLP-1 medications[2][3]
- Unique metabolic boost: The glucagon component of the GLP-3 peptide mechanism of action increases energy expenditure and fat breakdown while other pathways prevent hyperglycemic side effects[1]
- Multi-system benefits: Beyond weight loss, research demonstrates improvements in insulin sensitivity, satiety signaling, gastric emptying, and even joint pain reduction[2]
- Advanced development stage: Lead candidate retatrutide is in phase 3 trials with potential FDA approval projected for 2027, representing the first medication in this revolutionary class[2]
Understanding the GLP-3 Peptide Mechanism of Action: What Makes It Different? 🎯
The GLP-3 peptide mechanism of action fundamentally differs from earlier generations of metabolic peptides through its unprecedented triple-receptor approach. While medications like Ozempic and Wegovy target only the GLP-1 (glucagon-like peptide-1) receptor, the glp-3 peptide mechanism of action simultaneously engages three distinct hormone pathways: GLP-1, GIP (glucose-dependent insulinotropic polypeptide), and glucagon receptors[1][2].
This multi-targeted strategy creates what researchers call "synergistic metabolic optimization"—where the combined effects exceed the sum of individual pathway activations. Each receptor contributes unique benefits to the overall mechanism, working together to address multiple aspects of metabolic dysfunction simultaneously.
The Three Pillars of GLP-3 Action
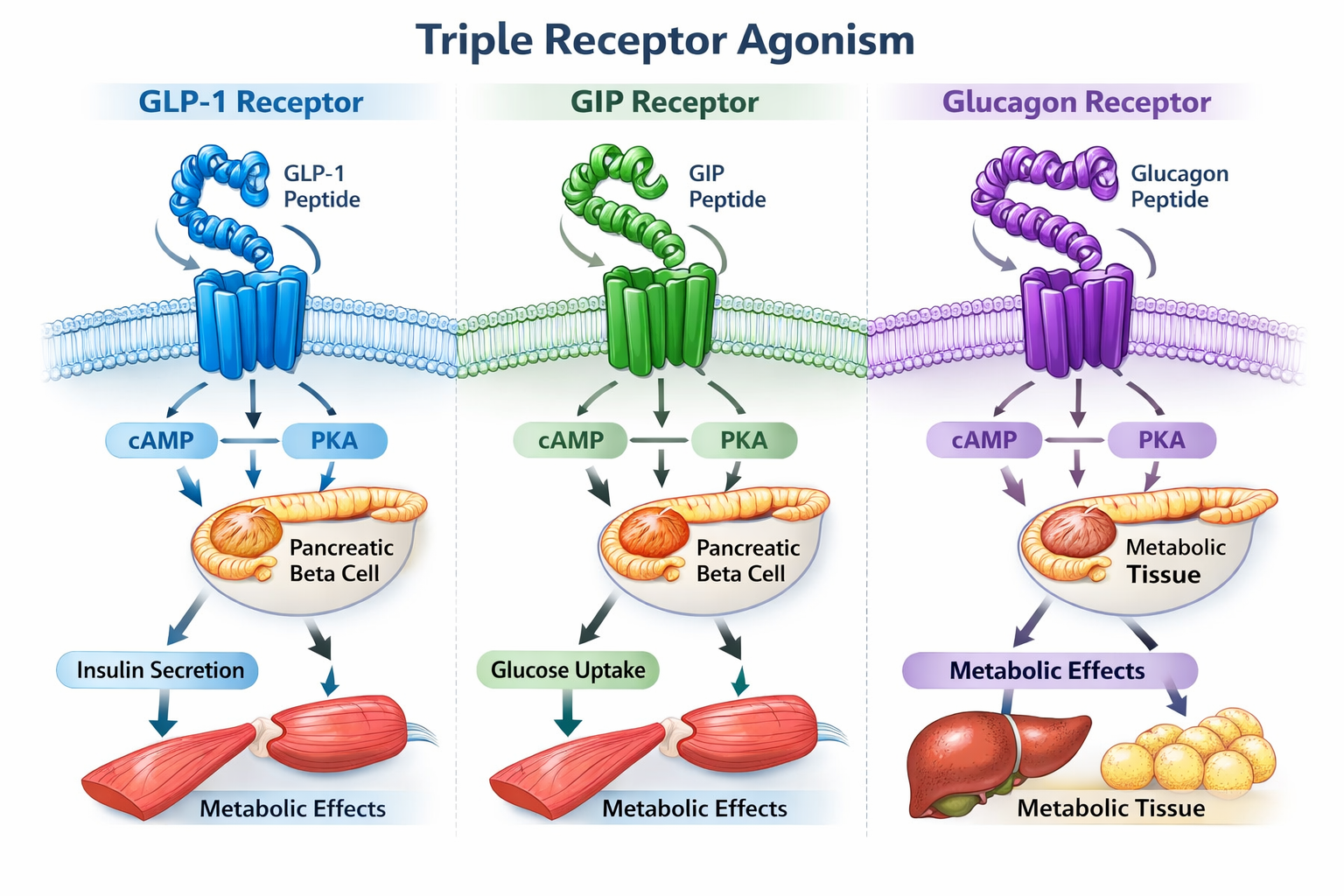
GLP-1 Receptor Activation forms the foundation of the GLP-3 peptide mechanism of action, providing proven benefits that have made single-pathway drugs successful. This pathway increases insulin secretion in response to food intake, enhances glucose uptake by cells, slows gastric emptying to prolong satiety, and reduces appetite through central nervous system signaling[1].
GIP Receptor Agonism adds a complementary layer to the mechanism, further amplifying insulin secretion and glucose metabolism. Research indicates that GIP works synergistically with GLP-1 to enhance metabolic benefits while potentially reducing some gastrointestinal side effects common with GLP-1-only medications[1].
Glucagon Receptor Activation represents the revolutionary component that distinguishes the GLP-3 peptide mechanism of action from all previous approaches. This pathway increases energy expenditure, promotes fat breakdown (lipolysis), decreases fat production (lipogenesis), and enhances insulin secretion—all while the GLP-1 and GIP components counterbalance potential hyperglycemic effects[1].
How the GLP-3 Peptide Mechanism of Action Works at the Cellular Level 🧬
Understanding the glp-3 peptide mechanism of action requires examining what happens when this triple-agonist peptide interacts with target cells throughout the body. The process begins when the peptide molecule binds to G-protein coupled receptors on cell surfaces, triggering cascading intracellular signaling pathways that alter gene expression and cellular metabolism.
Pancreatic Beta Cell Response
When the GLP-3 peptide mechanism of action engages pancreatic beta cells, all three receptor pathways work together to optimize insulin secretion. The GLP-1 component stimulates insulin release in a glucose-dependent manner, meaning insulin secretion increases when blood sugar is elevated but doesn't cause dangerous hypoglycemia when glucose levels are normal[1].
The GIP pathway amplifies this effect, creating a more robust insulin response that improves glycemic control. Meanwhile, the glucagon component paradoxically enhances insulin secretion despite glucagon's traditional role as an insulin antagonist—a phenomenon made possible by the simultaneous activation of GLP-1 and GIP receptors that modulate the glucagon effect[1].
This coordinated response represents a significant advantage of the glp-3 peptide mechanism of action over single-pathway approaches, creating more physiological insulin patterns that better match the body's natural regulatory systems.
Adipose Tissue Metabolism
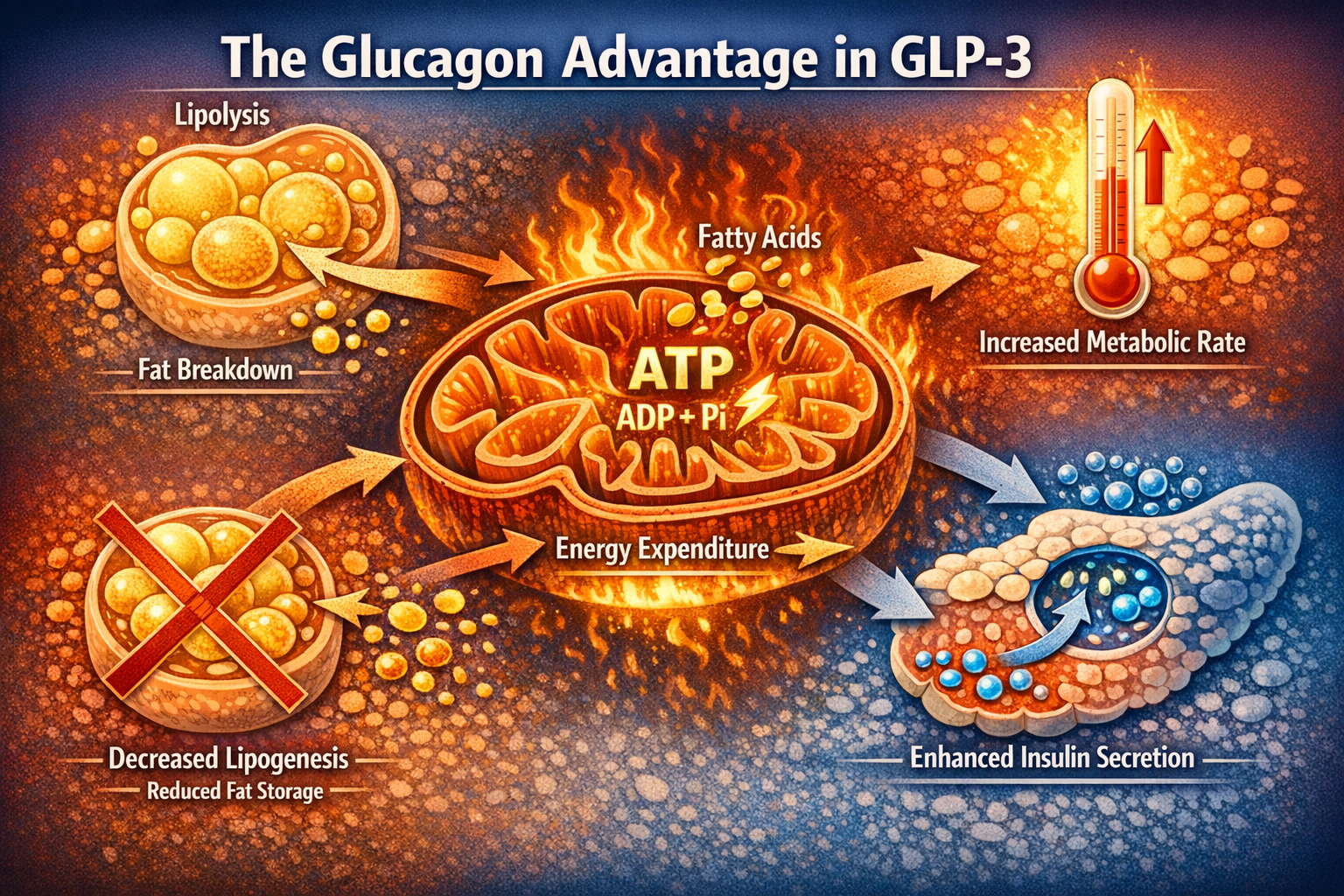
The GLP-3 peptide mechanism of action profoundly impacts fat tissue through the glucagon receptor pathway. Activation of glucagon receptors on adipocytes triggers hormone-sensitive lipase, the enzyme responsible for breaking down stored triglycerides into free fatty acids that can be burned for energy[1].
Simultaneously, the mechanism inhibits lipogenesis—the process of creating new fat from excess calories. This dual action of increased fat breakdown and decreased fat storage creates a metabolic environment highly conducive to weight loss, explaining why clinical trials show substantially greater fat mass reduction compared to GLP-1-only medications[2].
The glp-3 peptide mechanism of action also increases energy expenditure in adipose tissue, particularly in brown fat deposits that specialize in thermogenesis. This metabolic boost contributes to the superior weight loss outcomes observed in clinical trials.
Central Nervous System Effects
Brain regions controlling appetite and satiety respond powerfully to the glp-3 peptide mechanism of action. GLP-1 receptors in the hypothalamus and brainstem mediate reduced hunger signals and increased feelings of fullness, while GIP receptors may enhance these effects through complementary neural pathways[1].
The result is a profound reduction in food intake that participants describe as effortless—hunger simply diminishes without the constant willpower battles typical of traditional dieting approaches. This neurological component of the glp-3 peptide mechanism of action represents one of its most valuable therapeutic aspects for long-term weight management.
Clinical Evidence: GLP-3 Peptide Mechanism of Action in Human Trials 📊
The most compelling validation of the GLP-3 peptide mechanism of action comes from rigorous clinical trials showing unprecedented weight loss results. The lead compound retatrutide (LY3437943), developed by Eli Lilly, demonstrates the practical real-world impact of this triple-receptor approach.
Landmark Phase 2 Results
In initial phase 2 trials, participants receiving the highest dose of retatrutide (12 mg once-weekly injection) achieved an average 24.2% weight reduction over 48 weeks[2]. This outcome substantially exceeded results from any GLP-1-only medication, with weight loss continuing beyond the trial completion point.
The glp-3 peptide mechanism of action demonstrated dose-dependent effects, meaning higher doses produced greater weight loss while maintaining acceptable safety profiles. Participants reported significant improvements in metabolic markers including blood glucose, insulin sensitivity, and lipid profiles.
Extended TRIUMPH-4 Trial Data
The TRIUMPH-4 trial examined the GLP-3 peptide mechanism of action specifically in patients with obesity and knee osteoarthritis over an extended 68-week period. Results proved even more impressive: participants lost an average of 28.7% body weight, equivalent to 71.8 pounds—approximately double the weight loss observed with Ozempic and Wegovy[2][3].
Beyond the remarkable weight reduction, more than one in eight trial participants reported being "completely free" from knee pain, likely attributable to reduced joint stress and inflammation from the substantial weight loss[2]. This finding suggests the glp-3 peptide mechanism of action may offer therapeutic benefits extending beyond metabolic health into musculoskeletal wellness.
Comparative Advantage Over Single-Pathway Drugs
When researchers compare the glp-3 peptide mechanism of action against established GLP-1 medications, the metabolic boost from glucagon activation emerges as the key differentiator. While drugs like semaglutide (Wegovy) typically produce 15-17% weight loss, the triple-agonist approach consistently achieves 24-29% reduction—a difference that translates to 30-50 additional pounds lost for many participants[2].
This superior efficacy stems from the synergistic effects of targeting multiple pathways simultaneously. The glp-3 peptide mechanism of action doesn't just suppress appetite and slow digestion—it fundamentally increases how many calories the body burns at rest through enhanced metabolic rate.
Breaking Down Each Component of the GLP-3 Peptide Mechanism of Action ⚙️
To fully appreciate the GLP-3 peptide mechanism of action, examining each receptor pathway's individual contributions provides crucial insight into why this approach works so effectively.
GLP-1 Pathway: The Proven Foundation
The GLP-1 component of the glp-3 peptide mechanism of action builds upon decades of research validating this pathway's metabolic benefits. GLP-1 receptors exist throughout the body, with particularly high concentrations in pancreatic beta cells, the gastrointestinal tract, and specific brain regions[1].
When activated, these receptors trigger several key responses:
- Glucose-dependent insulin secretion: Insulin release increases proportionally to blood sugar levels, preventing dangerous hypoglycemia
- Glucagon suppression: Reduced glucagon secretion from pancreatic alpha cells decreases hepatic glucose production
- Delayed gastric emptying: Food remains in the stomach longer, prolonging satiety signals
- Central appetite reduction: Direct brain signaling reduces hunger and increases fullness sensations
The glp-3 peptide mechanism of action leverages these established benefits while adding complementary pathways that amplify overall efficacy.
GIP Pathway: The Synergistic Amplifier
GIP receptor activation represents the second pillar of the GLP-3 peptide mechanism of action, contributing effects that enhance and complement GLP-1 activity. Research indicates GIP primarily influences:
- Enhanced insulin secretion: Works synergistically with GLP-1 to produce more robust insulin responses
- Improved glucose disposal: Increases glucose uptake in muscle and adipose tissue
- Reduced inflammation: May decrease inflammatory markers associated with metabolic syndrome
- Neuroprotective effects: Emerging evidence suggests potential cognitive benefits
While earlier research questioned whether GIP agonism might promote fat storage, recent studies demonstrate that when combined with GLP-1 activation in the glp-3 peptide mechanism of action, GIP contributes to weight loss rather than weight gain[1].
Glucagon Pathway: The Revolutionary Metabolic Booster
The glucagon component distinguishes the GLP-3 peptide mechanism of action from all previous metabolic therapies, providing unique benefits that explain the superior weight loss outcomes:
Increased Energy Expenditure: Glucagon receptor activation raises basal metabolic rate, meaning the body burns more calories even at rest. This thermogenic effect contributes significantly to the enhanced fat loss observed in clinical trials[1].
Enhanced Lipolysis: Activation of hormone-sensitive lipase in adipocytes accelerates the breakdown of stored triglycerides into free fatty acids that can be oxidized for energy. This process directly reduces fat mass while providing fuel for the increased metabolic rate[1].
Decreased Lipogenesis: The glp-3 peptide mechanism of action simultaneously inhibits the synthesis of new fat from dietary carbohydrates and proteins, creating a metabolic environment that favors fat loss over fat storage[1].
Paradoxical Insulin Enhancement: Despite glucagon's traditional role as an insulin counter-regulatory hormone, its activation within the triple-agonist framework actually enhances insulin secretion—an effect that prevents the hyperglycemia typically associated with glucagon elevation[1].
GLP-3 Peptide Mechanism of Action: Safety Profile and Side Effects ⚠️

Understanding the safety aspects of the GLP-3 peptide mechanism of action is essential for anyone considering this therapeutic approach. Clinical trial data provides comprehensive information about both common and rare adverse events.
Gastrointestinal Effects
The most frequently reported side effects of the glp-3 peptide mechanism of action involve the gastrointestinal system, similar to existing GLP-1 medications. Common symptoms include:
- Nausea (particularly during dose escalation)
- Vomiting (typically mild and transient)
- Diarrhea (usually resolving within weeks)
- Constipation (less common than with GLP-1-only drugs)
- Abdominal discomfort
These effects generally diminish over time as the body adapts to the medication. The glp-3 peptide mechanism of action may actually produce fewer GI side effects than GLP-1-only medications due to the modulating influence of GIP and glucagon pathways[2].
Metabolic Considerations
The glucagon component of the GLP-3 peptide mechanism of action raises theoretical concerns about hyperglycemia, since glucagon traditionally increases blood sugar. However, clinical trials demonstrate that the simultaneous GLP-1 and GIP activation effectively counterbalances this effect, maintaining excellent glycemic control[1].
Participants in retatrutide trials showed improved glucose metabolism and insulin sensitivity rather than deterioration, validating the synergistic design of the glp-3 peptide mechanism of action. This finding represents a crucial safety validation for the triple-agonist approach.
Long-Term Safety Monitoring
As the GLP-3 peptide mechanism of action represents a novel therapeutic approach, long-term safety data continues to accumulate through ongoing phase 3 trials. Seven additional trials are expected to complete in 2026, providing comprehensive safety information across diverse patient populations[2].
Researchers monitor cardiovascular outcomes, pancreatic function, thyroid parameters, and metabolic markers to ensure the glp-3 peptide mechanism of action maintains favorable benefit-risk profiles over extended treatment periods.
<!DOCTYPE html>
<html lang="en">
<head>
<meta charset="UTF-8">
<meta name="viewport" content="width=device-width, initial-scale=1.0">
<title>GLP-3 Mechanism Interactive Comparison</title>
<style>
.cg-glp3-container {
font-family: 'Segoe UI', Tahoma, Geneva, Verdana, sans-serif;
max-width: 1000px;
margin: 0 auto;
padding: 20px;
background: linear-gradient(135deg, #667eea 0%, #764ba2 100%);
border-radius: 15px;
box-shadow: 0 10px 30px rgba(0,0,0,0.3);
}
.cg-glp3-title {
text-align: center;
color: white;
font-size: 28px;
margin-bottom: 25px;
text-shadow: 2px 2px 4px rgba(0,0,0,0.3);
}
.cg-glp3-selector {
display: flex;
justify-content: center;
gap: 15px;
margin-bottom: 30px;
flex-wrap: wrap;
}
.cg-glp3-btn {
padding: 12px 24px;
font-size: 16px;
font-weight: bold;
border: none;
border-radius: 25px;
cursor: pointer;
transition: all 0.3s ease;
background: white;
color: #667eea;
box-shadow: 0 4px 15px rgba(0,0,0,0.2);
}
.cg-glp3-btn:hover {
transform: translateY(-2px);
box-shadow: 0 6px 20px rgba(0,0,0,0.3);
}
.cg-glp3-btn.active {
background: #764ba2;
color: white;
}
.cg-glp3-comparison {
background: white;
border-radius: 12px;
padding: 30px;
box-shadow: 0 5px 20px rgba(0,0,0,0.2);
}
.cg-glp3-row {
display: grid;
grid-template-columns: 1fr 2fr;
gap: 20px;
padding: 15px;
border-bottom: 1px solid #e0e0e0;
align-items: center;
}
.cg-glp3-row:last-child {
border-bottom: none;
}
.cg-glp3-label {
font-weight: bold;
color: #333;
font-size: 16px;
}
.cg-glp3-value {
color: #555;
font-size: 15px;
line-height: 1.6;
}
.cg-glp3-highlight {
background: linear-gradient(135deg, #667eea 0%, #764ba2 100%);
color: white;
padding: 3px 8px;
border-radius: 5px;
font-weight: bold;
}
.cg-glp3-receptor-visual {
display: flex;
justify-content: space-around;
margin: 20px 0;
flex-wrap: wrap;
gap: 15px;
}
.cg-glp3-receptor {
text-align: center;
padding: 15px;
border-radius: 10px;
flex: 1;
min-width: 150px;
transition: all 0.3s ease;
}
.cg-glp3-receptor.active {
background: linear-gradient(135deg, #667eea 0%, #764ba2 100%);
color: white;
transform: scale(1.05);
}
.cg-glp3-receptor.inactive {
background: #f0f0f0;
color: #999;
}
.cg-glp3-receptor-icon {
font-size: 40px;
margin-bottom: 10px;
}
.cg-glp3-receptor-name {
font-weight: bold;
font-size: 14px;
}
@media (max-width: 768px) {
.cg-glp3-row {
grid-template-columns: 1fr;
gap: 10px;
}
.cg-glp3-title {
font-size: 22px;
}
.cg-glp3-btn {
padding: 10px 18px;
font-size: 14px;
}
}
</style>
</head>
<body>
<div class="cg-glp3-container">
<h2 class="cg-glp3-title">🔬 GLP Peptide Mechanism Comparison Tool</h2>
<div class="cg-glp3-selector">
<button class="cg-glp3-btn" onclick="showMechanism('glp1')">GLP-1 Only</button>
<button class="cg-glp3-btn" onclick="showMechanism('dual')">Dual Agonist</button>
<button class="cg-glp3-btn active" onclick="showMechanism('glp3')">GLP-3 Triple</button>
</div>
<div class="cg-glp3-receptor-visual" id="receptorVisual">
<div class="cg-glp3-receptor active">
<div class="cg-glp3-receptor-icon">🔵</div>
<div class="cg-glp3-receptor-name">GLP-1</div>
</div>
<div class="cg-glp3-receptor active">
<div class="cg-glp3-receptor-icon">🟢</div>
<div class="cg-glp3-receptor-name">GIP</div>
</div>
<div class="cg-glp3-receptor active">
<div class="cg-glp3-receptor-icon">🟣</div>
<div class="cg-glp3-receptor-name">Glucagon</div>
</div>
</div>
<div class="cg-glp3-comparison" id="comparisonData">
<!-- Content populated by JavaScript -->
</div>
</div>
<script>
const mechanismData = {
glp1: {
name: "GLP-1 Single Agonist",
receptors: [true, false, false],
weightLoss: "15-17% over 68 weeks",
mechanism: "Increases insulin secretion, reduces appetite, slows gastric emptying",
metabolicBoost: "Minimal - primarily appetite suppression",
fatBreakdown: "Indirect through caloric deficit only",
examples: "Ozempic, Wegovy, Mounjaro (primarily GLP-1)",
sideEffects: "Moderate GI effects (nausea, vomiting)",
approval: "FDA approved (multiple drugs)"
},
dual: {
name: "Dual Agonist (GLP-1 + GIP)",
receptors: [true, true, false],
weightLoss: "18-22% over 68 weeks",
mechanism: "Enhanced insulin secretion, improved glucose disposal, reduced appetite, slower gastric emptying",
metabolicBoost: "Moderate - synergistic insulin effects",
fatBreakdown: "Improved compared to GLP-1 only",
examples: "Tirzepatide (Mounjaro, Zepbound)",
sideEffects: "Similar to GLP-1, possibly reduced intensity",
approval: "FDA approved (Tirzepatide)"
},
glp3: {
name: "GLP-3 Triple Agonist",
receptors: [true, true, true],
weightLoss: "24.2-28.7% over 68 weeks",
mechanism: "All dual agonist benefits PLUS increased energy expenditure, enhanced lipolysis, decreased lipogenesis",
metabolicBoost: "Significant - glucagon pathway increases basal metabolic rate",
fatBreakdown: "Direct lipolysis activation + reduced fat synthesis",
examples: "Retatrutide (investigational - Eli Lilly)",
sideEffects: "Similar GI profile, potentially better tolerated",
approval: "Phase 3 trials - projected 2027 FDA approval"
}
};
function showMechanism(type) {
const data = mechanismData[type];
const buttons = document.querySelectorAll('.cg-glp3-btn');
buttons.forEach(btn => btn.classList.remove('active'));
event.target.classList.add('active');
// Update receptor visual
const receptors = document.querySelectorAll('.cg-glp3-receptor');
receptors.forEach((receptor, index) => {
if (data.receptors[index]) {
receptor.classList.remove('inactive');
receptor.classList.add('active');
} else {
receptor.classList.remove('active');
receptor.classList.add('inactive');
}
});
// Update comparison data
const comparisonHTML = `
<div class="cg-glp3-row">
<div class="cg-glp3-label">Drug Type:</div>
<div class="cg-glp3-value"><span class="cg-glp3-highlight">${data.name}</span></div>
</div>
<div class="cg-glp3-row">
<div class="cg-glp3-label">Active Receptors:</div>
<div class="cg-glp3-value">${data.receptors.filter(r => r).length} of 3 pathways activated</div>
</div>
<div class="cg-glp3-row">
<div class="cg-glp3-label">Weight Loss:</div>
<div class="cg-glp3-value"><span class="cg-glp3-highlight">${data.weightLoss}</span></div>
</div>
<div class="cg-glp3-row">
<div class="cg-glp3-label">Mechanism:</div>
<div class="cg-glp3-value">${data.mechanism}</div>
</div>
<div class="cg-glp3-row">
<div class="cg-glp3-label">Metabolic Boost:</div>
<div class="cg-glp3-value">${data.metabolicBoost}</div>
</div>
<div class="cg-glp3-row">
<div class="cg-glp3-label">Fat Breakdown:</div>
<div class="cg-glp3-value">${data.fatBreakdown}</div>
</div>
<div class="cg-glp3-row">
<div class="cg-glp3-label">Examples:</div>
<div class="cg-glp3-value">${data.examples}</div>
</div>
<div class="cg-glp3-row">
<div class="cg-glp3-label">Side Effects:</div>
<div class="cg-glp3-value">${data.sideEffects}</div>
</div>
<div class="cg-glp3-row">
<div class="cg-glp3-label">Status:</div>
<div class="cg-glp3-value">${data.approval}</div>
</div>
`;
document.getElementById('comparisonData').innerHTML = comparisonHTML;
}
// Initialize with GLP-3 data
window.onload = function() {
showMechanism('glp3');
};
</script>
</body>
</html>
The Future of GLP-3 Peptide Mechanism of Action Research 🚀
The GLP-3 peptide mechanism of action represents just the beginning of multi-receptor agonist therapies. As research progresses through 2026 and beyond, several exciting developments are reshaping the landscape of metabolic medicine.
Ongoing Clinical Trials
Retatrutide, the lead compound demonstrating the glp-3 peptide mechanism of action, currently has seven phase 3 trials underway examining various patient populations and clinical indications[2]. These studies investigate:
- Long-term weight maintenance beyond initial loss
- Cardiovascular outcomes in high-risk patients
- Effects on non-alcoholic fatty liver disease (NAFLD)
- Impact on sleep apnea and respiratory function
- Benefits for patients with type 2 diabetes
- Safety and efficacy in diverse demographic groups
The glp-3 peptide mechanism of action is being rigorously evaluated across these domains to establish comprehensive safety and efficacy profiles before potential FDA approval in 2027[2].
Beyond Weight Loss: Emerging Applications
Research into the GLP-3 peptide mechanism of action increasingly reveals benefits extending beyond metabolic health. The TRIUMPH-4 trial's finding that participants experienced significant joint pain relief suggests potential applications in inflammatory conditions[2].
Scientists are investigating whether the glp-3 peptide mechanism of action might benefit:
- Cardiovascular disease: Through weight reduction, improved lipid profiles, and potential direct cardioprotective effects
- Neurodegenerative conditions: GLP-1 and GIP receptors in the brain may offer neuroprotective benefits
- Inflammatory disorders: Metabolic improvements and potential anti-inflammatory effects could address multiple conditions
- Aging and longevity: Metabolic optimization may influence healthspan and biological aging processes
Next-Generation Peptide Development
The success of the GLP-3 peptide mechanism of action has inspired research into even more sophisticated multi-receptor approaches. Pharmaceutical companies are developing:
- Quadruple-agonist peptides adding additional metabolic pathways
- Tissue-selective agonists targeting specific organs while minimizing side effects
- Oral formulations to replace injectable administration
- Long-acting versions requiring monthly rather than weekly dosing
These innovations build upon the proven foundation of the glp-3 peptide mechanism of action while addressing current limitations and expanding therapeutic possibilities.
Practical Considerations for the GLP-3 Peptide Mechanism of Action 💡
Understanding the GLP-3 peptide mechanism of action from a theoretical perspective is valuable, but practical implementation requires additional considerations for fitness enthusiasts and peptide researchers.
Administration and Dosing
The glp-3 peptide mechanism of action in current clinical trials utilizes once-weekly subcutaneous injections, similar to existing GLP-1 medications. Retatrutide trials have examined doses ranging from 0.5 mg to 12 mg, with higher doses producing greater weight loss while maintaining acceptable tolerability[2].
Dose escalation protocols typically start at lower doses and gradually increase over several weeks to minimize gastrointestinal side effects. This approach allows the body to adapt to the glp-3 peptide mechanism of action progressively, improving overall tolerability.
Integration with Fitness and Nutrition
For fitness enthusiasts interested in the GLP-3 peptide mechanism of action, understanding how this therapy integrates with training and nutrition is crucial. The mechanism's effects on metabolism suggest several important considerations:
Protein Intake: The significant weight loss produced by the glp-3 peptide mechanism of action includes both fat and lean mass. Adequate protein consumption (1.6-2.2g per kg body weight) helps preserve muscle during the rapid weight reduction.
Resistance Training: Maintaining or increasing resistance training frequency becomes essential when utilizing the GLP-3 peptide mechanism of action to signal the body to preserve muscle tissue despite the caloric deficit created by reduced appetite.
Nutrient Timing: The delayed gastric emptying component of the mechanism may affect pre-workout nutrition timing. Experimenting with meal timing relative to training sessions optimizes both performance and comfort.
Hydration: Ensuring adequate fluid intake supports the metabolic processes activated by the glp-3 peptide mechanism of action, particularly the increased lipolysis and energy expenditure.
Quality and Sourcing Considerations
As interest in the GLP-3 peptide mechanism of action grows, ensuring peptide quality becomes paramount. Research-grade peptides should meet stringent purity standards and undergo comprehensive testing to verify:
- Correct amino acid sequence
- Appropriate molecular weight
- Minimal contamination or degradation
- Proper storage and handling
Working with reputable suppliers offering lab-tested peptides ensures the peptides used in research accurately reflect the intended glp-3 peptide mechanism of action rather than degraded or contaminated compounds that could produce unreliable results.
Comparing GLP-3 Peptide Mechanism of Action to Other Metabolic Peptides 📈
The GLP-3 peptide mechanism of action exists within a broader landscape of metabolic peptides, each offering unique benefits and mechanisms. Understanding these comparisons helps researchers and fitness enthusiasts make informed decisions.
GLP-3 vs. Single-Pathway GLP-1 Agonists
Traditional GLP-1 medications like semaglutide work through a single receptor pathway, producing reliable but more modest results compared to the glp-3 peptide mechanism of action. The key differences include:
| Feature | GLP-1 Only | GLP-3 Triple Agonist |
|---|---|---|
| Weight Loss | 15-17% | 24.2-28.7% |
| Metabolic Rate | Minimal increase | Significant boost |
| Fat Breakdown | Indirect only | Direct lipolysis |
| Muscle Preservation | Moderate concern | Similar concern |
| Side Effects | Moderate GI | Similar or reduced |
| Availability | FDA approved | Phase 3 trials |
The glp-3 peptide mechanism of action clearly demonstrates superior weight loss outcomes, primarily attributable to the glucagon pathway's metabolic enhancement[2].
GLP-3 and Growth Hormone Secretagogues
Some fitness enthusiasts combine metabolic peptides with growth hormone secretagogues to optimize body composition. While the GLP-3 peptide mechanism of action focuses on weight loss and metabolic health, peptides like tesa specifically target growth hormone release for different benefits.
The tesa peptide stimulates growth hormone production, which may help preserve lean mass during the rapid weight loss produced by the glp-3 peptide mechanism of action. However, combining peptides requires careful consideration and appropriate research protocols.
GLP-3 and Mitochondrial Peptides
Emerging research examines whether combining the GLP-3 peptide mechanism of action with mitochondrial-targeting peptides might produce synergistic benefits. The SS-31 mechanism focuses on mitochondrial function and cellular energy production, potentially complementing the metabolic enhancement from glucagon receptor activation.
Similarly, MOTS-C peptides influence metabolic regulation through mitochondrial signaling pathways. The interaction between these mechanisms and the glp-3 peptide mechanism of action represents an exciting frontier in metabolic optimization research.
Understanding the Science Behind GLP-3 Peptide Mechanism of Action Success 🧪
The remarkable clinical results achieved through the GLP-3 peptide mechanism of action stem from sophisticated molecular biology that researchers have carefully optimized over years of development.
Receptor Binding Characteristics
The glp-3 peptide mechanism of action depends on precisely engineered binding affinities for each of the three target receptors. The lead compound retatrutide demonstrates:
- Balanced potency across all three receptors, avoiding over-activation of any single pathway
- Sustained receptor engagement due to structural modifications that slow degradation
- Tissue distribution that reaches target organs while minimizing off-target effects
These characteristics ensure the glp-3 peptide mechanism of action produces coordinated effects across multiple systems rather than creating imbalanced signaling that might reduce efficacy or increase side effects[1].
Pharmacokinetic Optimization
The once-weekly dosing schedule of the GLP-3 peptide mechanism of action results from careful pharmacokinetic engineering. Modifications to the peptide structure include:
- Fatty acid conjugation: Attaching lipid molecules that bind to albumin in the bloodstream, extending half-life
- Protease resistance: Structural changes that prevent rapid enzymatic breakdown
- Sustained release kinetics: Gradual dissociation from albumin provides steady drug levels throughout the week
These innovations transform the glp-3 peptide mechanism of action from a theoretical concept requiring multiple daily injections into a practical once-weekly therapy that maintains consistent metabolic effects.
Synergistic Pathway Integration
Perhaps the most elegant aspect of the GLP-3 peptide mechanism of action is how the three pathways work together to create effects greater than the sum of their parts. The glucagon pathway's tendency to raise blood glucose is perfectly counterbalanced by enhanced insulin secretion from GLP-1 and GIP activation[1].
Similarly, the appetite suppression from GLP-1 receptor activation complements the increased energy expenditure from glucagon signaling, creating a powerful dual approach to weight loss. This synergistic integration distinguishes the glp-3 peptide mechanism of action from simple combinations of separate drugs that might produce conflicting effects.
Real-World Implications of GLP-3 Peptide Mechanism of Action 🌍
The GLP-3 peptide mechanism of action represents more than just an incremental improvement in metabolic medicine—it signals a fundamental shift in how researchers approach complex physiological challenges.
Impact on Obesity Treatment Paradigms
The unprecedented weight loss achieved through the glp-3 peptide mechanism of action challenges traditional assumptions about what's pharmacologically possible for obesity treatment. The 28.7% average weight reduction observed in TRIUMPH-4 approaches outcomes previously achievable only through bariatric surgery[2][3].
This level of efficacy transforms obesity from a condition requiring surgical intervention for many patients into one potentially manageable through pharmacotherapy. The glp-3 peptide mechanism of action may eventually offer a non-surgical option for patients who previously had limited alternatives.
Metabolic Health Beyond Weight
While weight loss dominates discussions of the GLP-3 peptide mechanism of action, the metabolic improvements extend far beyond the number on the scale. Participants in clinical trials demonstrate:
- Significant improvements in insulin sensitivity and glucose control
- Favorable changes in lipid profiles (cholesterol and triglycerides)
- Reduced inflammatory markers associated with metabolic syndrome
- Improvements in blood pressure and cardiovascular risk factors
These comprehensive metabolic benefits suggest the glp-3 peptide mechanism of action addresses root causes of metabolic dysfunction rather than simply treating symptoms.
Economic and Healthcare System Implications
The potential approval of medications utilizing the GLP-3 peptide mechanism of action carries significant healthcare economics implications. If the therapy can prevent or reverse obesity-related complications like type 2 diabetes, cardiovascular disease, and joint problems, the long-term cost savings could substantially offset the medication expenses.
The finding that participants experienced relief from knee osteoarthritis pain illustrates this potential—weight loss reducing joint stress might delay or prevent the need for costly joint replacement surgeries[2]. As more data emerges on the glp-3 peptide mechanism of action, health economists will carefully analyze these cost-benefit considerations.
Optimizing Results with GLP-3 Peptide Mechanism of Action 🎯
For researchers and fitness enthusiasts exploring the GLP-3 peptide mechanism of action, understanding optimization strategies maximizes potential benefits while minimizing risks.
Lifestyle Synergy
The glp-3 peptide mechanism of action works most effectively when integrated with comprehensive lifestyle approaches:
Nutrition Optimization: While the mechanism dramatically reduces appetite, ensuring nutrient-dense food choices during reduced eating windows supports overall health. Prioritizing whole foods, adequate protein, and essential micronutrients prevents deficiencies during rapid weight loss.
Exercise Integration: The glp-3 peptide mechanism of action increases energy expenditure, but combining this with structured exercise amplifies results. Resistance training preserves muscle mass, while cardiovascular activity enhances the metabolic benefits.
Sleep and Recovery: The metabolic processes activated by the GLP-3 peptide mechanism of action require adequate recovery. Prioritizing 7-9 hours of quality sleep supports optimal hormone function and metabolic regulation.
Stress Management: Chronic stress can interfere with the metabolic improvements from the glp-3 peptide mechanism of action through cortisol elevation and behavioral effects. Implementing stress reduction strategies enhances overall outcomes.
Monitoring and Adjustment
Successful utilization of the GLP-3 peptide mechanism of action requires careful monitoring of multiple parameters:
- Body composition: Tracking both weight and lean mass helps ensure muscle preservation
- Metabolic markers: Regular assessment of glucose, insulin, and lipids documents metabolic improvements
- Nutritional status: Monitoring protein intake and micronutrient levels prevents deficiencies
- Side effects: Documenting any adverse effects allows for appropriate dose adjustments
This comprehensive monitoring approach ensures the glp-3 peptide mechanism of action produces optimal results while maintaining safety.
Realistic Expectations and Timeline
Understanding realistic timelines for the GLP-3 peptide mechanism of action helps set appropriate expectations:
Weeks 1-4: Initial weight loss primarily from reduced caloric intake; possible GI side effects as the body adapts; early metabolic improvements begin.
Weeks 5-12: Accelerating weight loss as the full glp-3 peptide mechanism of action engages; reduced side effects; noticeable improvements in energy and metabolic markers.
Weeks 13-24: Continued steady weight loss; significant body composition changes become apparent; metabolic health improvements plateau at new, healthier levels.
Weeks 25-48: Sustained weight loss continuing toward individual endpoints; focus shifts toward maintenance; comprehensive metabolic transformation complete.
Beyond 48 weeks: Long-term weight maintenance phase; ongoing metabolic benefits; potential for continued gradual improvements.
Clinical trials demonstrate that the glp-3 peptide mechanism of action produces weight loss that continues beyond 48 weeks, with participants in extended trials achieving even greater results over 68 weeks[2][3].
Conclusion: The Revolutionary Promise of GLP-3 Peptide Mechanism of Action 🏆
The GLP-3 peptide mechanism of action represents a watershed moment in metabolic medicine, offering unprecedented weight loss results through an elegantly designed triple-receptor approach. By simultaneously activating GLP-1, GIP, and glucagon pathways, this mechanism creates synergistic effects that substantially exceed what single-pathway drugs can achieve[1][2].
Clinical trial data demonstrating 24.2% to 28.7% weight reduction—nearly double the results of existing medications—validates the scientific rationale behind this approach[2][3]. The additional metabolic benefits, including improved insulin sensitivity, enhanced fat breakdown, increased energy expenditure, and even joint pain relief, suggest the glp-3 peptide mechanism of action addresses multiple aspects of metabolic health simultaneously.
For fitness enthusiasts and peptide researchers in 2026, understanding the glp-3 peptide mechanism of action provides crucial insights into the future of metabolic optimization. While retatrutide remains in phase 3 trials with projected FDA approval in 2027, the comprehensive research already completed demonstrates both remarkable efficacy and acceptable safety profiles[2].
Actionable Next Steps
Stay Informed: Monitor ongoing clinical trial results as the seven additional phase 3 studies complete throughout 2026. These trials will provide comprehensive data on long-term safety, diverse patient populations, and additional therapeutic applications.
Optimize Current Approaches: While awaiting potential approval of GLP-3 medications, focus on optimizing nutrition, training, recovery, and currently available peptide therapies to build a strong foundation for metabolic health.
Research Quality Sources: For those conducting peptide research, prioritize working with reputable suppliers offering comprehensive testing and verification to ensure peptide quality and purity.
Consult Qualified Professionals: The glp-3 peptide mechanism of action involves sophisticated pharmacology requiring appropriate medical oversight. Work with qualified healthcare providers who understand peptide therapy and metabolic medicine.
Integrate Comprehensive Strategies: Remember that even revolutionary mechanisms like the glp-3 peptide mechanism of action work best when integrated with evidence-based nutrition, training, recovery, and lifestyle practices.
The future of metabolic health is being written in 2026 through innovations like the GLP-3 peptide mechanism of action. By understanding these mechanisms, staying informed about research developments, and implementing evidence-based approaches, fitness enthusiasts and researchers can position themselves at the forefront of this metabolic revolution.
References
[1] Pmc11486854 – https://pmc.ncbi.nlm.nih.gov/articles/PMC11486854/
[2] What Glp 3s Meet New Generation Weight Loss Drugs Three Key Ingredients – https://www.foxnews.com/health/what-glp-3s-meet-new-generation-weight-loss-drugs-three-key-ingredients
[3] How To Get Retatrutide – https://www.joinmidi.com/post/how-to-get-retatrutide
SEO Meta Title and Description
Meta Title: GLP-3 Peptide Mechanism of Action: Triple Receptor Guide 2026
Meta Description: Discover how GLP-3 peptide mechanism of action achieves 24-28% weight loss through triple receptor targeting. Complete guide for fitness enthusiasts.
{“@context”:”https://schema.org”,”@type”:”Article”,”headline”:”glp-3 peptide mechanism of action”,”description”:”Discover how GLP-3 peptide mechanism of action achieves 24-28% weight loss through triple receptor targeting. Complete guide for fitness enthusiasts.”,”image”:”https://zsxkvszxbhpwnvzxdydv.supabase.co/storage/v1/object/public/generated-images/kie/079e8e7a-26f9-4987-9375-a5a601de22b4/slot-0-1770077598858.png”,”datePublished”:”2026-02-03T00:08:35.344869+00:00″,”dateModified”:”2026-02-03T00:14:51.960Z”,”author”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″},”publisher”:{“@type”:”Organization”,”name”:”PTP UPDATED 2.1.26″}}
