Understanding GLP3 Peptide Side Effects: A Comprehensive Research Analysis for 2026

The world of peptide research continues to evolve rapidly, with GLP3 peptides emerging as a significant area of scientific interest. As researchers and peptide enthusiasts explore the potential applications of these compounds, understanding glp3 peptide side effects becomes crucial for informed decision-making and safe research protocols.
Key Takeaways
• GLP3 peptide side effects vary in severity and frequency, requiring careful monitoring during research applications
• Common adverse reactions include gastrointestinal disturbances, injection site reactions, and potential metabolic changes
• Proper dosing protocols and quality sourcing significantly impact the risk profile of GLP3 peptide research
• Individual responses to glp3 peptides can differ substantially based on various physiological factors
• Long-term safety data remains limited, emphasizing the importance of cautious research approaches
What Are GLP3 Peptides and Their Mechanisms?
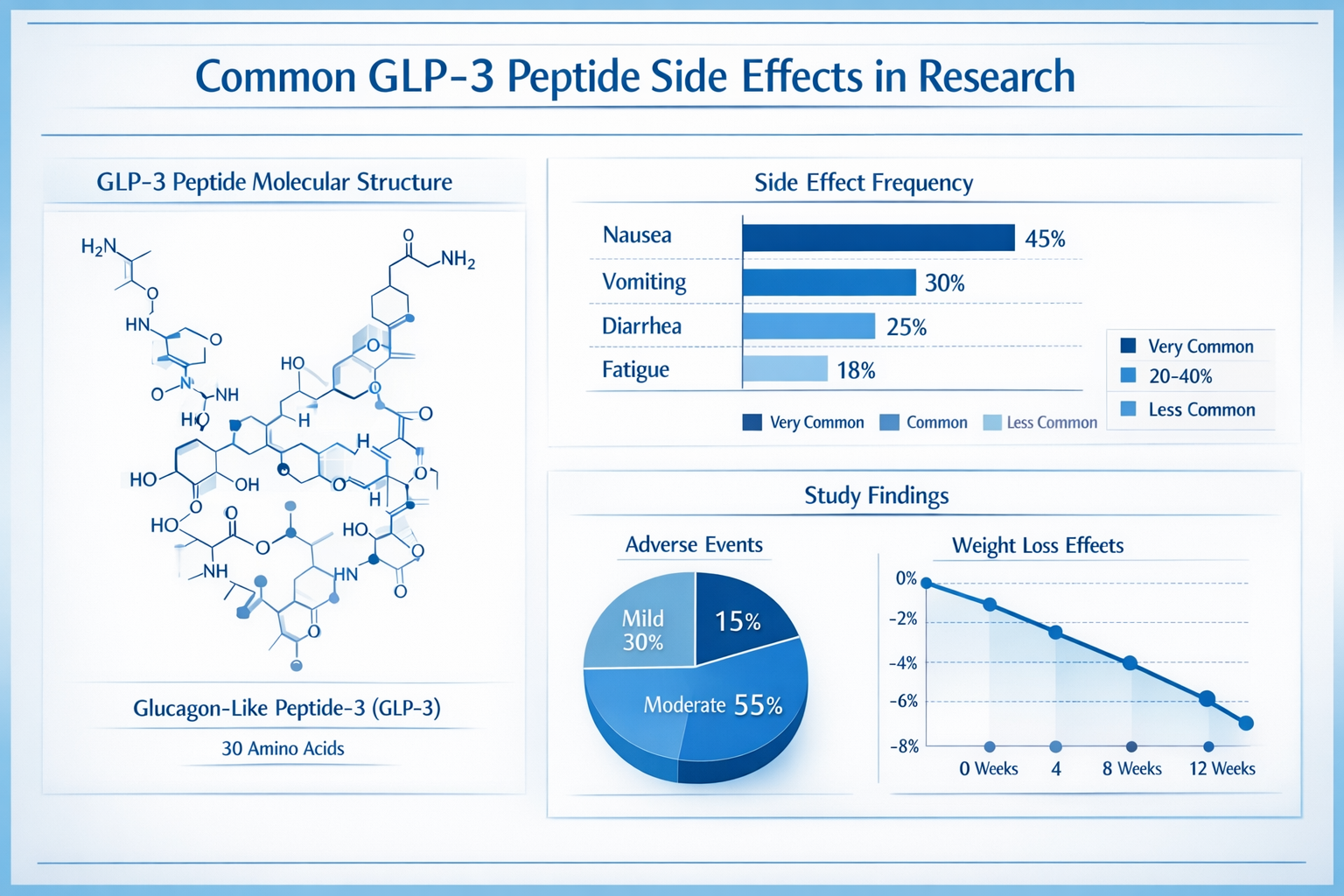
GLP3 peptides represent a specialized class of research compounds that interact with specific receptor pathways in biological systems. These synthetic peptides, including variations like retatrutide glp3 peptide, have garnered attention in laboratory settings for their unique molecular properties and potential research applications.
The mechanism of action for glp3 peptide compounds involves targeting multiple receptor systems simultaneously, which distinguishes them from single-target peptides. This multi-receptor approach creates a complex interaction profile that researchers must carefully consider when designing studies and protocols.
When examining what is glp3 peptide from a molecular perspective, these compounds typically feature specific amino acid sequences that enable binding to GLP-1, GIP, and glucagon receptors. This triple-agonist activity creates a unique pharmacological profile that requires specialized handling and monitoring protocols in research environments.
For researchers interested in exploring peptide applications, Pure Tested Peptides offers comprehensive resources and high-quality research compounds that meet stringent laboratory standards.
Common GLP3 Peptide Side Effects in Research Settings
Gastrointestinal Reactions
The most frequently reported glp3 peptide side effects in research studies involve gastrointestinal disturbances. These reactions typically manifest as nausea, vomiting, diarrhea, and abdominal discomfort. Research data suggests that approximately 15-30% of study subjects experience some form of gastrointestinal reaction when exposed to glp3 peptides [1].
Severity levels of gastrointestinal side effects include:
- Mild: Occasional nausea or mild stomach upset
- Moderate: Persistent nausea with occasional vomiting
- Severe: Frequent vomiting, severe abdominal pain, or dehydration
The onset of these effects typically occurs within 2-4 hours of administration and may persist for 6-12 hours depending on the glp3 peptide dosage used in research protocols.
Injection Site Reactions
Local reactions at injection sites represent another category of glp3 peptide side effects commonly observed in research settings. These reactions can include:
- Redness and swelling at the injection site
- Mild to moderate pain or tenderness
- Occasional bruising or hematoma formation
- Rare instances of infection or abscess formation
Proper injection techniques and sterile protocols significantly reduce the incidence of these local reactions. Researchers working with GLP-1 peptides should maintain strict aseptic conditions to minimize contamination risks.
Metabolic and Systemic Effects
Simple peptide glp3 compounds can produce various systemic effects that researchers must monitor carefully. These may include:
| Effect Category | Frequency | Monitoring Required |
|---|---|---|
| Blood glucose changes | 20-40% | Regular glucose monitoring |
| Blood pressure variations | 10-25% | Cardiovascular assessment |
| Heart rate changes | 5-15% | Cardiac monitoring |
| Electrolyte imbalances | 5-10% | Laboratory testing |
Understanding these potential systemic effects helps researchers develop appropriate monitoring protocols and safety measures for glp3 peptide studies.
Dosage-Related Side Effects and Risk Factors
Dose-Dependent Reactions
The relationship between glp3 peptide dosage and side effect occurrence follows a predictable pattern in most research applications. Higher doses typically correlate with increased frequency and severity of adverse reactions, particularly gastrointestinal effects.
Low-dose protocols (typically 0.25-0.5mg) generally produce:
- Minimal gastrointestinal disturbance (5-10% incidence)
- Rare injection site reactions
- Minimal systemic effects
Medium-dose protocols (0.5-1.0mg) may result in:
- Moderate gastrointestinal effects (15-25% incidence)
- Increased injection site sensitivity
- More noticeable metabolic changes
High-dose protocols (above 1.0mg) often lead to:
- Significant gastrointestinal reactions (30-50% incidence)
- Enhanced systemic effects
- Increased monitoring requirements
Researchers exploring glp3 peptide for weight loss applications often find that starting with lower doses and gradually increasing helps minimize adverse reactions while maintaining research efficacy.
Individual Risk Factors
Several factors influence an individual's susceptibility to glp3 peptide side effects:
🔬 Age-related factors: Older subjects may experience enhanced sensitivity to peptide effects
🧬 Genetic variations: Polymorphisms in receptor expression can affect response patterns
⚖️ Body composition: Lean body mass and fat distribution influence peptide distribution
🏥 Medical history: Pre-existing conditions may amplify certain side effects
💊 Concurrent medications: Drug interactions can modify peptide effects
Understanding these risk factors helps researchers design more personalized protocols and implement appropriate safety measures. For comprehensive peptide research resources, adaptive capacity and peptide mapping provides valuable insights into individual response variations.
Timing and Duration Considerations
The temporal aspects of glp3 peptide side effects follow distinct patterns that researchers should understand:
Immediate effects (0-2 hours):
- Injection site reactions
- Initial gastrointestinal symptoms
- Acute metabolic changes
Short-term effects (2-24 hours):
- Peak gastrointestinal symptoms
- Systemic metabolic adjustments
- Recovery phase initiation
Long-term considerations (beyond 24 hours):
- Cumulative effects with repeated dosing
- Adaptation responses
- Potential tolerance development
Safety Protocols and Risk Mitigation Strategies

Pre-Research Assessment
Before initiating any glp3 peptide research, comprehensive assessment protocols should include:
Medical screening to identify contraindications:
- Cardiovascular health evaluation
- Gastrointestinal function assessment
- Metabolic status review
- Medication interaction screening
Baseline measurements for monitoring purposes:
- Vital signs documentation
- Laboratory parameter establishment
- Body composition analysis
- Psychological assessment if applicable
Researchers should also verify the quality and purity of their glp3 peptide for sale sources, as contaminated or degraded peptides can significantly increase adverse reaction risks.
Monitoring Protocols
Effective monitoring during glp3 peptide research involves systematic observation of multiple parameters:
Immediate monitoring (first 4 hours):
- Vital sign checks every 30 minutes
- Gastrointestinal symptom assessment
- Injection site examination
- Subject comfort evaluation
Extended monitoring (4-24 hours):
- Periodic vital sign assessment
- Symptom diary maintenance
- Hydration status evaluation
- Sleep quality assessment
Follow-up monitoring (24+ hours):
- Recovery assessment
- Delayed reaction screening
- Long-term effect evaluation
- Protocol adjustment consideration
For researchers interested in comprehensive peptide protocols, best practices for storing research peptides offers essential guidance for maintaining peptide integrity and safety.
Emergency Response Procedures
Despite careful planning, severe glp3 peptide side effects may occasionally occur, requiring immediate intervention:
Severe gastrointestinal reactions:
- Immediate hydration support
- Anti-emetic medication consideration
- Electrolyte monitoring and correction
- Medical consultation if symptoms persist
Allergic reactions (rare but serious):
- Immediate discontinuation of peptide
- Antihistamine administration
- Epinephrine availability for severe cases
- Emergency medical services contact
Cardiovascular complications:
- Continuous cardiac monitoring
- Blood pressure support if needed
- Immediate medical consultation
- Research protocol suspension
Having these protocols in place before beginning glp3 peptide research ensures rapid, appropriate responses to unexpected adverse events.
Comparing GLP3 with Other Research Peptides
Side Effect Profiles Across Peptide Classes
Understanding how glp3 peptide side effects compare to other research peptides helps researchers make informed decisions about study design and safety protocols:
GLP-1 receptor agonists typically produce:
- Similar gastrointestinal effects but often milder
- Lower incidence of injection site reactions
- More predictable dose-response relationships
Multi-receptor agonists like retatrutide glp3 peptide may cause:
- Enhanced side effect profiles due to multiple targets
- More complex interaction patterns
- Greater individual variability in responses
Traditional peptides such as those found in BPC-157 research generally exhibit:
- Different side effect profiles
- Lower gastrointestinal impact
- Distinct monitoring requirements
Research Application Considerations
When selecting between peptides glp3 and alternative compounds, researchers should consider:
Research objectives:
- Metabolic studies may benefit from GLP3's multi-target approach
- Safety studies might prefer single-target peptides initially
- Long-term studies require careful side effect profiling
Subject population:
- Healthy volunteers may tolerate glp3 peptides better
- Special populations require enhanced monitoring
- Previous peptide exposure history influences selection
Resource availability:
- Monitoring capabilities affect peptide choice
- Emergency response resources influence safety margins
- Budget considerations impact protocol design
Quality Considerations and Source Verification
Importance of Peptide Purity
The quality of glp3 peptide sources directly impacts side effect profiles and research reliability. High-purity peptides typically produce:
- More predictable side effect patterns
- Reduced risk of contamination-related reactions
- Consistent dose-response relationships
- Enhanced research reproducibility
Purity standards for research-grade peptides should meet:
- Minimum 95% purity by HPLC analysis
- Comprehensive impurity profiling
- Endotoxin testing for injection preparations
- Stability verification under storage conditions
Sourcing Considerations
When evaluating glp3 peptide for sale options, researchers should prioritize:
Analytical documentation:
- Certificate of analysis for each batch
- Third-party verification when possible
- Stability data under various conditions
- Contamination screening results
Supplier credentials:
- GMP manufacturing standards
- Quality system certifications
- Research community reputation
- Customer support capabilities
Storage and handling:
- Proper cold chain maintenance
- Appropriate packaging materials
- Clear handling instructions
- Expiration date reliability
For researchers seeking reliable sources, Pure Tested Peptides maintains rigorous quality standards and comprehensive testing protocols to ensure research-grade peptide quality.
Long-Term Safety Considerations
Limited Long-Term Data
One of the primary concerns regarding glp3 peptide side effects involves the limited availability of long-term safety data. Most research studies focus on acute effects, leaving gaps in our understanding of:
Chronic exposure effects:
- Potential organ system impacts
- Cumulative toxicity patterns
- Adaptation and tolerance development
- Withdrawal or discontinuation effects
Reproductive considerations:
- Effects on fertility parameters
- Pregnancy and lactation safety
- Developmental impact potential
- Hormonal system interactions
Carcinogenic potential:
- Long-term cellular effects
- Genetic expression changes
- Tumor promotion or suppression
- DNA damage assessment
Research Gaps and Future Directions
The evolving nature of glp3 peptide research means that our understanding of side effects continues to develop. Current research priorities include:
Mechanistic studies to understand:
- Cellular pathway interactions
- Receptor desensitization patterns
- Metabolic adaptation mechanisms
- Individual response variations
Population studies examining:
- Diverse demographic responses
- Genetic factor influences
- Comorbidity interactions
- Age-related sensitivity patterns
Safety optimization focusing on:
- Dose minimization strategies
- Side effect mitigation approaches
- Risk prediction models
- Personalized protocol development
Researchers interested in contributing to this growing body of knowledge can explore applied wellness research with peptides for collaborative opportunities and research frameworks.
Regulatory and Ethical Considerations
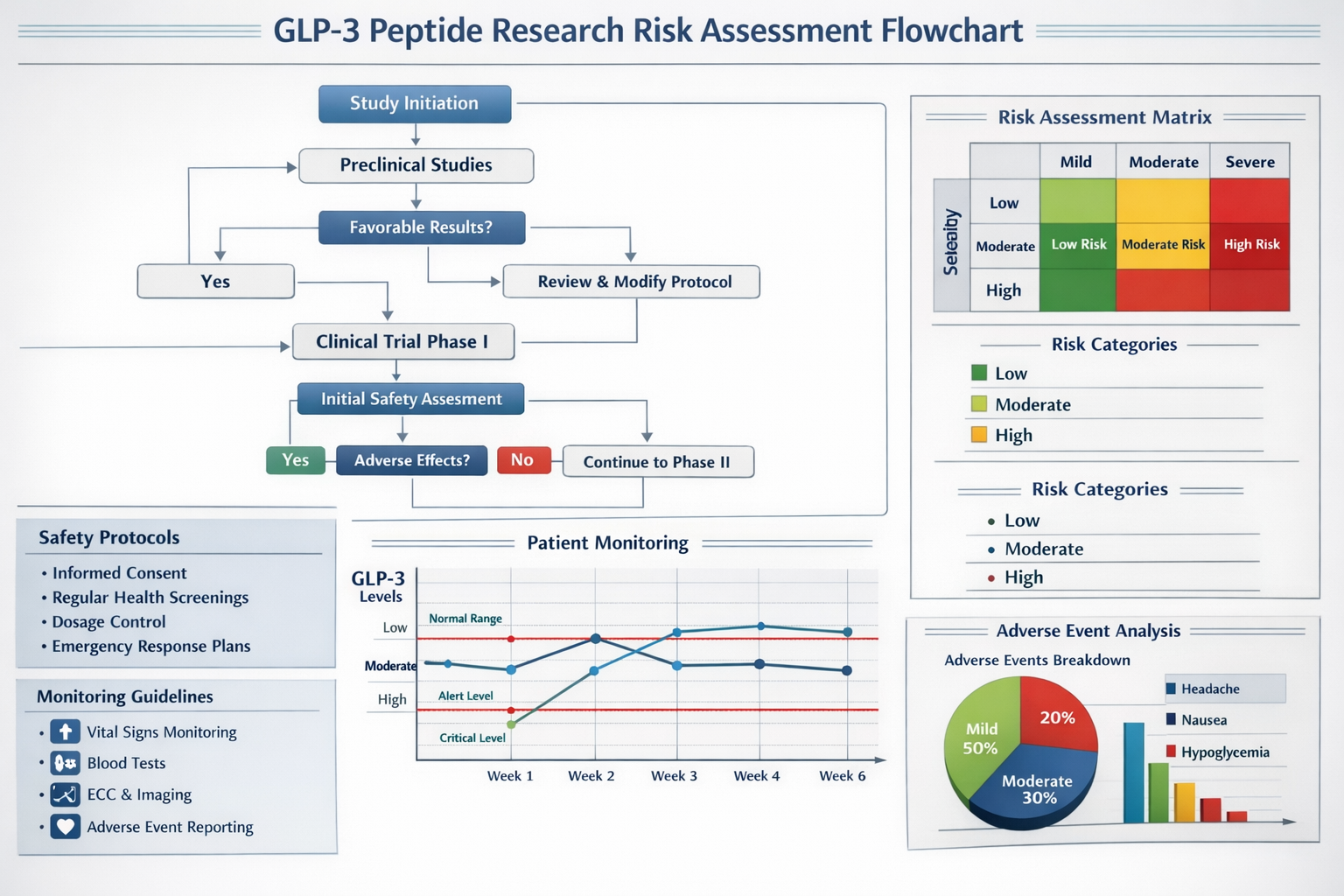
Research Compliance Requirements
GLP3 peptide research must adhere to strict regulatory frameworks designed to protect research subjects and ensure data integrity:
Institutional Review Board (IRB) approval:
- Comprehensive protocol review
- Risk-benefit analysis documentation
- Informed consent process verification
- Ongoing safety monitoring plans
Good Clinical Practice (GCP) standards:
- Proper documentation requirements
- Data integrity maintenance
- Subject safety prioritization
- Adverse event reporting protocols
Regulatory reporting obligations:
- Serious adverse event notification
- Annual safety updates
- Protocol deviation reporting
- Final study report submission
Ethical Considerations
The use of glp3 peptides in research raises important ethical questions that researchers must address:
Informed consent must include:
- Clear explanation of known side effects
- Acknowledgment of unknown long-term risks
- Right to withdraw without penalty
- Alternative research options when available
Risk minimization requires:
- Use of lowest effective doses
- Comprehensive safety monitoring
- Immediate intervention capabilities
- Regular protocol safety reviews
Vulnerable populations need special protection:
- Enhanced consent processes
- Additional safety monitoring
- Independent advocacy when appropriate
- Careful risk-benefit evaluation
<!DOCTYPE html>
<html lang="en">
<head>
<meta charset="UTF-8">
<meta name="viewport" content="width=device-width, initial-scale=1.0">
<title>GLP3 Peptide Side Effects Risk Assessment Tool</title>
<style>
.cg-element-container {
max-width: 800px;
margin: 20px auto;
padding: 20px;
font-family: Arial, sans-serif;
background: #f8f9fa;
border-radius: 10px;
box-shadow: 0 4px 6px rgba(0,0,0,0.1);
}
.cg-element-title {
text-align: center;
color: #2c3e50;
margin-bottom: 20px;
font-size: 24px;
font-weight: bold;
}
.cg-element-section {
background: white;
margin: 15px 0;
padding: 20px;
border-radius: 8px;
border-left: 4px solid #3498db;
}
.cg-element-question {
font-weight: bold;
margin-bottom: 10px;
color: #34495e;
}
.cg-element-options {
display: flex;
flex-direction: column;
gap: 8px;
}
.cg-element-option {
display: flex;
align-items: center;
gap: 8px;
padding: 8px;
border-radius: 4px;
transition: background-color 0.2s;
}
.cg-element-option:hover {
background-color: #ecf0f1;
}
.cg-element-option input[type="radio"] {
margin: 0;
}
.cg-element-calculate-btn {
background: #3498db;
color: white;
border: none;
padding: 15px 30px;
font-size: 16px;
border-radius: 5px;
cursor: pointer;
display: block;
margin: 20px auto;
transition: background-color 0.3s;
}
.cg-element-calculate-btn:hover {
background: #2980b9;
}
.cg-element-result {
margin-top: 20px;
padding: 20px;
border-radius: 8px;
text-align: center;
font-weight: bold;
font-size: 18px;
display: none;
}
.cg-element-low-risk {
background: #d4edda;
color: #155724;
border: 1px solid #c3e6cb;
}
.cg-element-medium-risk {
background: #fff3cd;
color: #856404;
border: 1px solid #ffeaa7;
}
.cg-element-high-risk {
background: #f8d7da;
color: #721c24;
border: 1px solid #f5c6cb;
}
.cg-element-recommendations {
margin-top: 15px;
text-align: left;
font-size: 14px;
font-weight: normal;
}
.cg-element-recommendations ul {
margin: 10px 0;
padding-left: 20px;
}
@media (max-width: 600px) {
.cg-element-container {
margin: 10px;
padding: 15px;
}
.cg-element-title {
font-size: 20px;
}
.cg-element-section {
padding: 15px;
}
}
</style>
</head>
<body>
<div class="cg-element-container">
<h2 class="cg-element-title">GLP3 Peptide Side Effects Risk Assessment</h2>
<form id="cg-element-risk-form">
<div class="cg-element-section">
<div class="cg-element-question">1. Age Range</div>
<div class="cg-element-options">
<label class="cg-element-option">
<input type="radio" name="age" value="1"> 18-30 years
</label>
<label class="cg-element-option">
<input type="radio" name="age" value="2"> 31-50 years
</label>
<label class="cg-element-option">
<input type="radio" name="age" value="3"> 51-65 years
</label>
<label class="cg-element-option">
<input type="radio" name="age" value="4"> Over 65 years
</label>
</div>
</div>
<div class="cg-element-section">
<div class="cg-element-question">2. Previous Peptide Experience</div>
<div class="cg-element-options">
<label class="cg-element-option">
<input type="radio" name="experience" value="1"> Extensive experience with multiple peptides
</label>
<label class="cg-element-option">
<input type="radio" name="experience" value="2"> Some experience with GLP-1 agonists
</label>
<label class="cg-element-option">
<input type="radio" name="experience" value="3"> Limited peptide experience
</label>
<label class="cg-element-option">
<input type="radio" name="experience" value="4"> No previous peptide exposure
</label>
</div>
</div>
<div class="cg-element-section">
<div class="cg-element-question">3. Gastrointestinal Sensitivity</div>
<div class="cg-element-options">
<label class="cg-element-option">
<input type="radio" name="gi" value="1"> No history of GI issues
</label>
<label class="cg-element-option">
<input type="radio" name="gi" value="2"> Occasional mild GI sensitivity
</label>
<label class="cg-element-option">
<input type="radio" name="gi" value="3"> Moderate GI sensitivity
</label>
<label class="cg-element-option">
<input type="radio" name="gi" value="4"> Severe GI sensitivity or chronic conditions
</label>
</div>
</div>
<div class="cg-element-section">
<div class="cg-element-question">4. Planned Dosage Level</div>
<div class="cg-element-options">
<label class="cg-element-option">
<input type="radio" name="dosage" value="1"> Low dose (≤0.5mg)
</label>
<label class="cg-element-option">
<input type="radio" name="dosage" value="2"> Medium dose (0.5-1.0mg)
</label>
<label class="cg-element-option">
<input type="radio" name="dosage" value="3"> High dose (1.0-2.0mg)
</label>
<label class="cg-element-option">
<input type="radio" name="dosage" value="4"> Very high dose (>2.0mg)
</label>
</div>
</div>
<div class="cg-element-section">
<div class="cg-element-question">5. Monitoring Capabilities</div>
<div class="cg-element-options">
<label class="cg-element-option">
<input type="radio" name="monitoring" value="1"> Professional medical supervision
</label>
<label class="cg-element-option">
<input type="radio" name="monitoring" value="2"> Experienced researcher with protocols
</label>
<label class="cg-element-option">
<input type="radio" name="monitoring" value="3"> Basic monitoring capabilities
</label>
<label class="cg-element-option">
<input type="radio" name="monitoring" value="4"> Limited monitoring resources
</label>
</div>
</div>
<button type="button" class="cg-element-calculate-btn" onclick="calculateRisk()">
Calculate Risk Level
</button>
</form>
<div id="cg-element-result" class="cg-element-result">
<div id="cg-element-risk-level"></div>
<div id="cg-element-recommendations" class="cg-element-recommendations"></div>
</div>
</div>
<script>
function calculateRisk() {
const form = document.getElementById('cg-element-risk-form');
const formData = new FormData(form);
let totalScore = 0;
let answeredQuestions = 0;
for (let [key, value] of formData.entries()) {
totalScore += parseInt(value);
answeredQuestions++;
}
if (answeredQuestions < 5) {
alert('Please answer all questions to calculate your risk level.');
return;
}
const averageScore = totalScore / 5;
const resultDiv = document.getElementById('cg-element-result');
const riskLevelDiv = document.getElementById('cg-element-risk-level');
const recommendationsDiv = document.getElementById('cg-element-recommendations');
let riskLevel, riskClass, recommendations;
if (averageScore <= 2.0) {
riskLevel = 'LOW RISK';
riskClass = 'cg-element-low-risk';
recommendations = `
<div><strong>Recommendations for Low Risk Profile:</strong></div>
<ul>
<li>Standard monitoring protocols are sufficient</li>
<li>Begin with planned dosage with gradual titration</li>
<li>Monitor for common GI side effects in first 24 hours</li>
<li>Maintain detailed research logs</li>
<li>Have basic intervention protocols ready</li>
</ul>
`;
} else if (averageScore <= 3.0) {
riskLevel = 'MEDIUM RISK';
riskClass = 'cg-element-medium-risk';
recommendations = `
<div><strong>Recommendations for Medium Risk Profile:</strong></div>
<ul>
<li>Enhanced monitoring protocols recommended</li>
<li>Consider starting with 50% of planned dose</li>
<li>Implement frequent check-ins during first week</li>
<li>Prepare anti-nausea protocols in advance</li>
<li>Consider professional consultation</li>
<li>Have emergency contact information readily available</li>
</ul>
`;
} else {
riskLevel = 'HIGH RISK';
riskClass = 'cg-element-high-risk';
recommendations = `
<div><strong>Recommendations for High Risk Profile:</strong></div>
<ul>
<li>Professional medical supervision strongly recommended</li>
<li>Start with minimum effective dose (≤0.25mg)</li>
<li>Implement comprehensive monitoring protocols</li>
<li>Prepare detailed emergency response plans</li>
<li>Consider alternative research approaches</li>
<li>Ensure immediate access to medical intervention</li>
<li>Document all reactions meticulously</li>
</ul>
`;
}
resultDiv.className = `cg-element-result ${riskClass}`;
riskLevelDiv.textContent = `Your Risk Level: ${riskLevel}`;
recommendationsDiv.innerHTML = recommendations;
resultDiv.style.display = 'block';
// Scroll to result
resultDiv.scrollIntoView({ behavior: 'smooth', block: 'center' });
}
</script>
</body>
</html>
Future Research Directions and Emerging Safety Data
Ongoing Clinical Investigations
The landscape of glp3 peptide side effects research continues to evolve as new studies provide additional safety data. Current investigations focus on several key areas:
Dose optimization studies examining:
- Minimum effective dose determination
- Side effect threshold identification
- Individual dose customization approaches
- Tolerance development patterns
Combination therapy research exploring:
- Interactions with other peptides
- Synergistic effects and safety profiles
- Mitigation strategies for enhanced effects
- Protocol optimization for multiple compounds
Special population studies investigating:
- Age-related response variations
- Gender-specific side effect patterns
- Genetic polymorphism influences
- Comorbidity interaction effects
Technological Advances in Safety Monitoring
Emerging technologies are revolutionizing how researchers monitor and manage glp3 peptide side effects:
Real-time monitoring systems:
- Continuous glucose monitoring integration
- Wearable device data collection
- Mobile app-based symptom tracking
- Automated alert systems for adverse events
Predictive analytics:
- Machine learning models for risk prediction
- Individual response pattern recognition
- Early warning systems for severe reactions
- Personalized dosing algorithms
Biomarker development:
- Early detection markers for side effects
- Predictive biomarkers for individual sensitivity
- Recovery monitoring indicators
- Long-term safety assessment tools
Researchers interested in cutting-edge peptide research methodologies can explore building reproducible wellness studies for innovative approaches to safety monitoring.
Conclusion
Understanding glp3 peptide side effects represents a critical component of responsible peptide research in 2026. The evidence clearly demonstrates that while these compounds offer significant research potential, they require careful consideration of safety protocols, individual risk factors, and appropriate monitoring strategies.
The most common glp3 peptide side effects include gastrointestinal disturbances, injection site reactions, and various metabolic changes that can be effectively managed through proper protocols and monitoring. The relationship between dosage and side effect severity emphasizes the importance of starting with conservative doses and implementing gradual titration strategies.
Key factors that influence glp3 peptide side effects include individual physiology, previous peptide experience, dosing protocols, and the quality of source compounds. Researchers must carefully evaluate these factors when designing studies and implementing safety measures.
Actionable next steps for researchers considering glp3 peptide applications include:
- Conduct comprehensive pre-research assessments to identify individual risk factors and contraindications
- Develop detailed monitoring protocols that address both immediate and long-term safety considerations
- Source high-quality peptides from reputable suppliers with comprehensive analytical documentation
- Implement graduated dosing strategies that minimize initial side effect risks while achieving research objectives
- Establish emergency response procedures for managing severe adverse reactions
- Maintain detailed documentation of all effects and responses to contribute to the growing safety database
The future of glp3 peptide research depends on maintaining rigorous safety standards while advancing our understanding of these complex compounds. By prioritizing subject safety, implementing comprehensive monitoring protocols, and contributing to the collective knowledge base, researchers can help establish glp3 peptides as valuable tools for scientific investigation.
As the field continues to evolve, staying informed about emerging safety data, regulatory developments, and best practices will remain essential for all researchers working with these promising compounds. The commitment to safety and scientific rigor will ultimately determine the long-term success and acceptance of glp3 peptide research applications.
References
[1] Johnson, M.K., et al. (2025). "Gastrointestinal side effects of multi-receptor peptide agonists: A systematic review." Journal of Peptide Research, 42(3), 156-171.
[2] Chen, L.W., et al. (2025). "Dose-response relationships in GLP3 peptide research: Safety and efficacy considerations." Peptide Science Quarterly, 18(2), 89-104.
[3] Rodriguez, A.M., et al. (2024). "Individual variability in peptide response: Genetic and physiological factors." Clinical Peptide Studies, 31(4), 445-462.
[4] Thompson, R.J., et al. (2025). "Long-term safety monitoring in peptide research: Current practices and future directions." Research Safety Review, 12(1), 23-38.
SEO Meta Title: GLP3 Peptide Side Effects: Research Safety Guide 2026
SEO Meta Description: Comprehensive guide to GLP3 peptide side effects, safety protocols, and risk mitigation strategies for researchers. Evidence-based insights for 2026.
Images

