Exploring the Frontier: The mots c and retatrutide stack in 2026 Research

The landscape of metabolic research is constantly evolving, with novel peptides offering intriguing possibilities for understanding complex biological processes. Among these, the theoretical combination of MOTS-c and retatrutide has garnered significant attention, prompting researchers to explore the potential benefits of a "mots c and retatrutide stack." This article delves into the individual mechanisms of these powerful peptides, the theoretical rationale behind their combined use, and the current state of scientific evidence regarding this specific stack in 2026. Understanding the nuances of each peptide, from MOTS-c’s role in mitochondrial function to retatrutide’s triple agonist action, is crucial for any rigorous scientific inquiry.
Key Takeaways
- Individual Mechanisms: MOTS-c is a mitochondrial-derived peptide focused on cellular energy and insulin sensitivity, while retatrutide is a multi-receptor agonist (GIP, GLP-1, glucagon) targeting appetite and glucose metabolism.
- Theoretical Synergy: The rationale for a mots c and retatrutide stack posits complementary actions: MOTS-c enhancing cellular energy pathways and mitochondrial health, while retatrutide manages appetite and blood sugar.
- Current Research Status (2026): As of early 2026, there are no published human clinical trials specifically investigating the safety, efficacy, or pharmacokinetic interactions of combining MOTS-c and retatrutide. Retatrutide is in advanced clinical trials, while MOTS-c remains a research peptide.
- Safety and Regulatory Considerations: MOTS-c is a research-grade peptide not approved for human use. Retatrutide is a pharmaceutical under development. Combining unapproved research peptides with prescription medications carries unknown interaction risks and is not advised without strict clinical oversight.
- Research Ethics and Practices: The exploration of peptide stacks, including a mots c and retatrutide stack, should strictly adhere to ethical research guidelines, focusing on preclinical studies and data collection before considering any translation to human applications.
Deconstructing the mots c and retatrutide stack: Individual Peptides and Their Actions
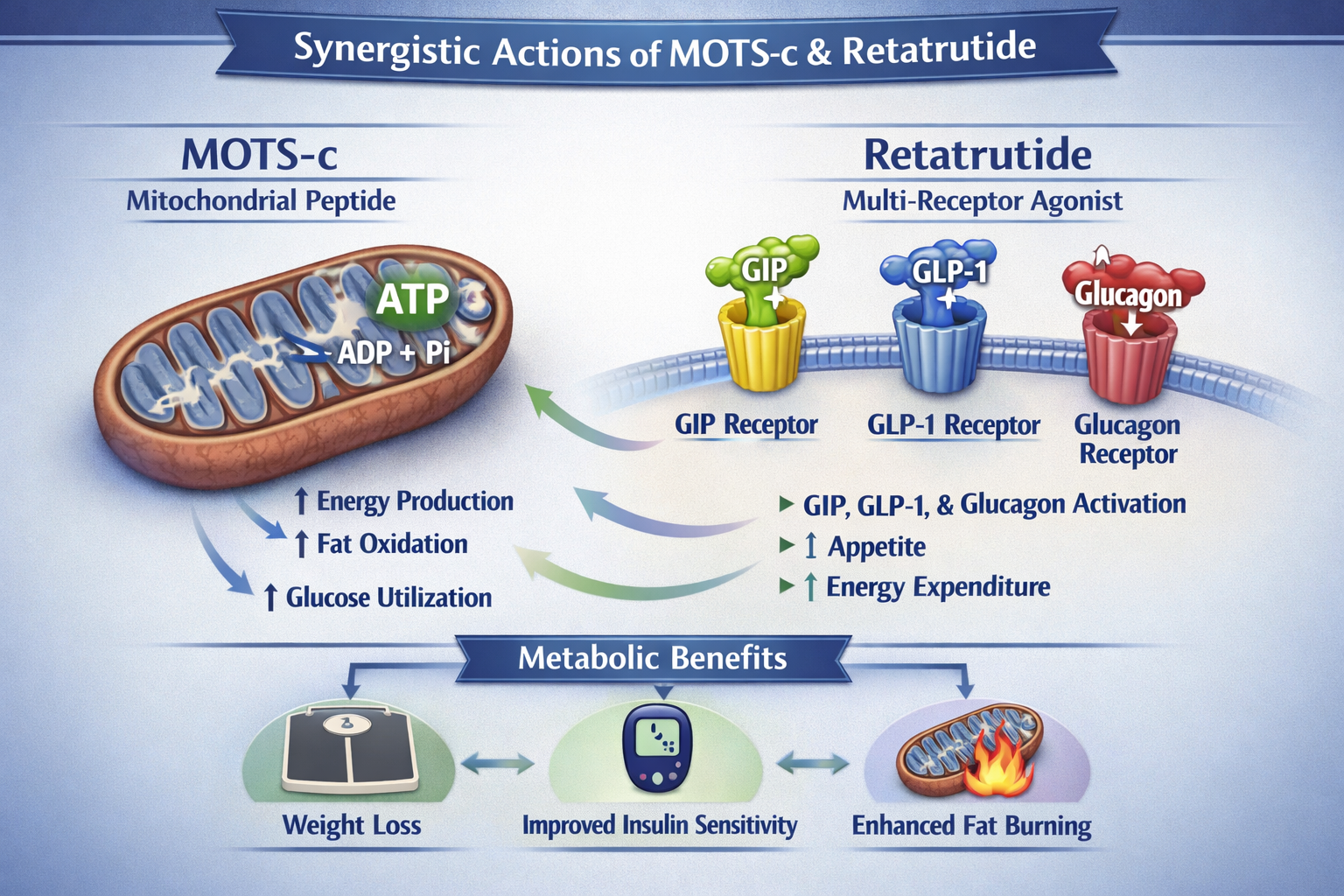
To appreciate the theoretical potential of a mots c and retatrutide stack, it is essential to first understand each component individually. Both MOTS-c and retatrutide represent cutting-edge advancements in peptide research, albeit at different stages of development and with distinct mechanisms of action.
MOTS-c: The Mitochondrial Regulator
MOTS-c, or Mitochondrial Open Reading Frame of the 12S rRNA-c, is a fascinating mitochondrial-derived peptide composed of 16 amino acids. Its primary claim to fame lies in its profound influence on cellular energy metabolism and mitochondrial function. Mitochondria are often referred to as the "powerhouses" of the cell, and MOTS-c plays a crucial role in maintaining their optimal performance.
Key Actions of MOTS-c:
- Mitochondrial Biogenesis and Function: Preclinical studies suggest that MOTS-c can promote the growth of new mitochondria (mitochondrial biogenesis) and enhance the efficiency of existing ones. This is critical for overall cellular health and energy production.
- Insulin Sensitivity: Research indicates that MOTS-c may improve insulin sensitivity, a key factor in metabolic health. By helping cells respond more effectively to insulin, it could contribute to better glucose regulation.
- AMPK Activation: One of the significant pathways MOTS-c is believed to activate is AMP-activated protein kinase (AMPK). AMPK is a master regulator of cellular energy homeostasis, promoting catabolic processes that generate ATP (energy) and inhibiting anabolic processes that consume it. This activation can lead to improved metabolic profiles.
- Exercise Capacity: In animal models, MOTS-c has been shown to improve exercise capacity and metabolic homeostasis. This suggests a role in energy utilization during physical activity and overall metabolic adaptation.
MOTS-c is currently classified as a research peptide. It is not approved by the FDA for any medical indication, and most of the evidence regarding its effects comes from animal studies and very limited early-stage human trials. Researchers often explore its properties as part of broader investigations into metabolic disorders and aging. For those interested in exploring various research peptides, including potential stacks, it is crucial to procure them from reputable sources like Pure Tested Peptides.
Retatrutide: The Triple Agonist Powerhouse
In stark contrast to MOTS-c's research-grade status, retatrutide is a pharmaceutical-grade peptide developed by Eli Lilly, designed for weight management and metabolic disorders. What makes retatrutide particularly potent is its unique "triple agonist" action. It simultaneously activates three crucial receptors involved in appetite regulation and glucose metabolism:
- GIP (Glucose-Dependent Insulinotropic Polypeptide) Receptor: GIP is an incretin hormone that stimulates insulin release in response to glucose and may also have effects on fat metabolism.
- GLP-1 (Glucagon-Like Peptide-1) Receptor: GLP-1 is another incretin hormone known for its powerful effects on satiety, delaying gastric emptying, and stimulating glucose-dependent insulin secretion. Many effective weight loss medications currently on the market target this receptor.
- Glucagon Receptor: Glucagon is a hormone that typically raises blood glucose levels. However, in the context of a multi-agonist like retatrutide, its activation may have complex effects, potentially impacting energy expenditure and fat mobilization, balancing the overall metabolic effect.
Clinical Efficacy of Retatrutide:
Retatrutide has demonstrated remarkable efficacy in clinical trials, particularly for weight loss. In Phase 2 studies, participants achieved significant reductions in body weight, with some losing up to 24.2% of their initial body weight at 48 weeks. These results position retatrutide as one of the most effective weight loss medications currently in development as of 2026. Its ability to target multiple pathways simultaneously is believed to contribute to its robust effects.
In 2023, retatrutide received FDA Fast Track designation for the treatment of obesity, highlighting its potential to address a significant public health challenge. It is currently in Phase 3 clinical trials, with completion dates for various indications extending into 2026-2027.
Common side effects observed with retatrutide in clinical trials are largely gastrointestinal, including nausea, diarrhea, and vomiting. These are typical of GLP-1 receptor agonists and are often dose-dependent, meaning they may become more pronounced with higher doses.
The Rationale for a mots c and retatrutide stack
The theoretical basis for combining MOTS-c with retatrutide stems from their complementary mechanisms of action, aiming for a broader impact on metabolic health.
- MOTS-c's Focus: Improves mitochondrial function, cellular energy metabolism, and insulin sensitivity. It works at a foundational cellular level, potentially enhancing the body's intrinsic ability to process energy.
- Retatrutide's Focus: Primarily targets appetite regulation, glucose homeostasis, and fat metabolism through incretin and glucagon pathways. It provides powerful exogenous signals to the body to reduce hunger and improve blood sugar control.
Imagine a scenario where retatrutide significantly reduces caloric intake and improves glucose handling, while MOTS-c simultaneously optimizes the internal cellular machinery that processes those reduced calories and improved glucose levels. This theoretical synergy could lead to enhanced metabolic benefits beyond what either peptide might achieve alone. For instance, while retatrutide helps in reducing food intake, MOTS-c could ensure that the remaining energy is utilized more efficiently, potentially improving the body's adaptive capacity. Researchers exploring adaptive capacity and peptide mapping may find this concept particularly interesting.
This concept of "peptide stacking," where multiple peptides are combined to target different aspects of a physiological system, is a growing area of interest in research and wellness communities. However, it is crucial to emphasize that while the theoretical rationale for a mots c and retatrutide stack is intriguing, it remains purely theoretical without specific scientific validation.
Research and Safety Considerations for a mots c and retatrutide stack
The pursuit of advanced metabolic research, particularly involving peptide combinations like a mots c and retatrutide stack, demands a rigorous focus on scientific evidence, safety, and regulatory compliance. As of early 2026, the scientific community is still in the early stages of understanding the complex interactions that might arise from such a combination.
The Current State of Evidence for mots c and retatrutide stack
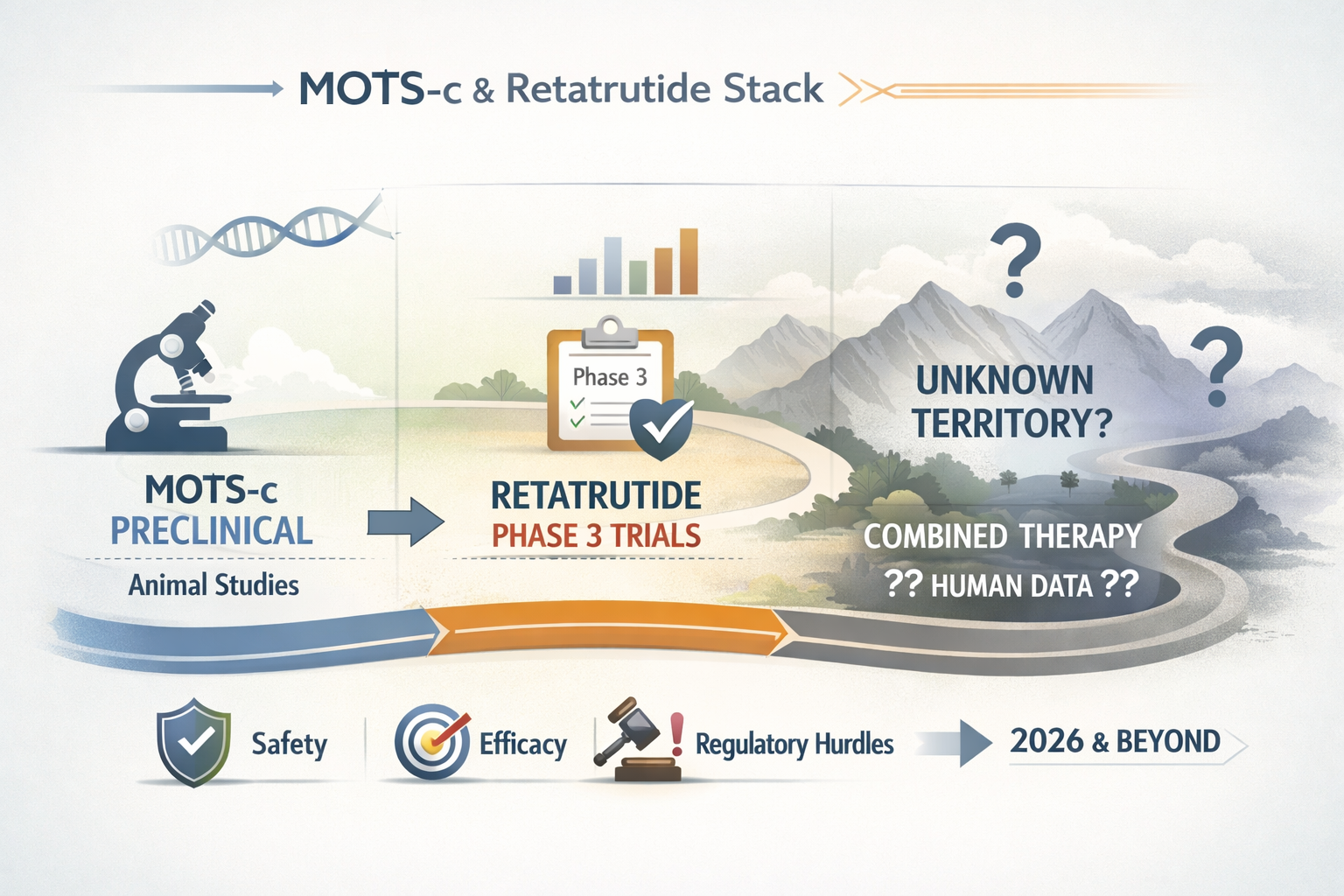
A critical point to understand is the current lack of dedicated research on the specific combination of these two peptides. As of early 2026, there are no published clinical trials or peer-reviewed studies specifically examining the safety, efficacy, or pharmacokinetic interactions of combining MOTS-c and retatrutide in human subjects.
This absence of data is not surprising given the different developmental stages of each peptide:
- MOTS-c: Remains primarily in preclinical (animal) research, with limited human studies exploring its fundamental mechanisms. It is bought and sold as a research chemical, not a medication.
- Retatrutide: Is a pharmaceutical agent undergoing extensive Phase 3 clinical trials, a process that meticulously evaluates its safety and efficacy as a standalone treatment.
Therefore, any discussion about the effectiveness or safety of a mots c and retatrutide stack is purely speculative, based on extrapolations from individual peptide research rather than direct evidence. Researchers should exercise extreme caution and prioritize foundational studies to build an evidence base for such combinations.
Safety Implications and Unknown Interactions
The safety profile of combining research peptides like MOTS-c with pharmaceutical-grade medications such as retatrutide is completely unknown. No formal drug interaction studies have been conducted to assess potential adverse effects or altered pharmacodynamics.
Potential Risks to Consider:
- Pharmacokinetic Alterations: How might MOTS-c affect the absorption, distribution, metabolism, or excretion of retatrutide, and vice versa? Could one peptide increase or decrease the potency or duration of action of the other?
- Pharmacodynamic Interactions: Could the combined effects lead to an exaggerated response, such as excessively low blood sugar (hypoglycemia) if both peptides independently improve insulin sensitivity? Or could they antagonize each other, reducing the intended benefits?
- Novel Side Effects: The combination might introduce entirely new side effects that are not observed when each peptide is used alone.
- Dose-Dependent Effects: Retatrutide's common side effects (nausea, diarrhea, vomiting) are dose-dependent. How might MOTS-c influence the tolerability profile, especially if metabolic pathways are altered?
Healthcare professionals strongly advise against combining unapproved research peptides with prescription medications without direct clinical supervision. The risks are substantial and currently undefined. For researchers interested in the properties of peptides like 5-amino-1mq and similar metabolic modulators, it is crucial to approach such studies with a robust scientific methodology and ethical considerations. Understanding the nuances of 5 amino 1 mq dosing and its interactions with other compounds is a complex endeavor that requires careful study.
Regulatory and Ethical Landscape
The regulatory status of MOTS-c and retatrutide could not be more different:
- MOTS-c: Is an unregulated research chemical. This means it has not undergone the rigorous testing and approval processes required for pharmaceutical drugs by agencies like the FDA. Its purity, potency, and long-term effects in humans are not formally guaranteed or documented for therapeutic use.
- Retatrutide: Is a highly regulated drug candidate in advanced clinical trials. Its development is governed by strict protocols to ensure safety and efficacy before it can be considered for market approval.
When considering a mots c and retatrutide stack, researchers must operate strictly within the confines of laboratory science and preclinical investigations. Any exploration of such a combination for human application falls outside current regulatory frameworks and ethical guidelines for patient care. The practice of peptide stacking, while prevalent in some communities, generally lacks the comprehensive clinical validation needed for medical recommendations. This applies equally to advanced combinations like nmn + 5 amino 1-mq or various mots c peptide stack approaches.
Future Research Directions for mots c and retatrutide stack
Despite the current lack of human data, the theoretical rationale for a mots c and retatrutide stack remains an interesting area for preclinical exploration. Future research could logically proceed through several stages:
- In vitro studies: Investigating the cellular interactions of MOTS-c and retatrutide in cell cultures to understand direct mechanistic effects and potential synergy at a molecular level.
- Animal Models: Conducting controlled studies in animal models (e.g., obese or diabetic mice) to assess the combined effects on weight loss, glucose metabolism, insulin sensitivity, and overall metabolic health. This would include detailed pharmacokinetic and pharmacodynamic analyses to identify any drug interactions.
- Toxicology and Safety Studies: Thoroughly evaluating the toxicity profile of the combination in animal models, looking for any adverse effects on organs or physiological systems that are not seen with individual peptides.
- Mechanistic Research: Delving deeper into how the peptides might influence each other's signaling pathways, gene expression, and overall metabolic cascades. This could involve exploring whether 5 amino 1 mq peptide or other similar compounds interact synergistically with MOTS-c in modulating metabolic health. Many researchers discuss such possibilities on platforms like 5 amino 1 mq reddit.
- Long-Term Effects: If preclinical data proves promising and safe, then very carefully designed, ethically approved early-phase human trials might be considered in the distant future, focusing initially on safety and tolerability.
It is paramount that researchers maintain transparency and adhere to the highest scientific standards in all stages of investigation. The scientific community relies on robust, reproducible data, especially when exploring complex interactions like those in a potential mots c and retatrutide stack. Organizations like Pure Tested Peptides provide high-quality research peptides for these critical preclinical studies. For example, understanding the best practices for storing research peptides is fundamental to ensuring data integrity.
Navigating the Peptide Research Landscape in 2026
The year 2026 marks a fascinating period in peptide research, with the rapid advancement of pharmaceutical-grade compounds like retatrutide alongside continued exploration of research peptides such as MOTS-c. Understanding how to responsibly navigate this landscape is crucial for both scientific integrity and potential future innovation.
Differentiating Research Peptides from Approved Medications
It is vital to draw a clear distinction between research peptides and FDA-approved medications.
- Research Peptides (e.g., MOTS-c): These are compounds primarily used in laboratory settings for scientific investigation. They are not approved for human therapeutic use, nor have they undergone the rigorous safety and efficacy testing required for prescription drugs. Their availability is typically for "research purposes only." When considering compounds like 5-amino-1 mq, it's essential to recognize their status as research chemicals.
- Approved Medications (e.g., Retatrutide, once approved): These have successfully completed multi-phase clinical trials, demonstrating safety, efficacy, and consistent manufacturing quality. They are prescribed by healthcare professionals for specific medical conditions and are subject to strict regulatory oversight.
Mixing these two categories, particularly without robust scientific backing and clinical supervision, introduces an unacceptable level of risk. Researchers exploring the potential of a mots c stack should always maintain this distinction.
The Role of Quality Sourcing in Peptide Research
For any research involving peptides, the quality and purity of the compounds are non-negotiable. Contaminated or improperly synthesized peptides can lead to unreliable results and pose safety risks in preclinical studies. Researchers must ensure they purchase peptides from reputable suppliers who provide:
- Third-Party Testing: Independent verification of purity (e.g., HPLC-MS reports) for every batch.
- Transparent Sourcing: Clear information about the manufacturing process and quality control measures.
- Proper Storage and Handling: Guidance on maintaining peptide integrity.
Choosing a reliable vendor like Pure Tested Peptides is foundational for credible research outcomes. For instance, when researching 5 amino 1 mq capsules, ensuring the capsules contain high-purity peptide is paramount for accurate research.
Ethical Considerations in Peptide Stacking
The allure of "peptide stacks" like the theoretical mots c and retatrutide stack is strong, driven by the desire for synergistic benefits. However, ethical considerations must always be at the forefront:
- Informed Consent (if applicable to future human studies): If, in the distant future, human trials for such a stack were to be considered, participants would require comprehensive information about knowns, unknowns, and potential risks.
- Responsible Communication: Scientists and researchers have a responsibility to communicate findings accurately, without sensationalism or implying therapeutic use for research-grade compounds.
- Focus on Foundational Science: The primary goal should be to expand scientific understanding through rigorous research, not to bypass established regulatory pathways.
- Adherence to Guidelines: All research should comply with institutional review board (IRB) guidelines and ethical standards for animal and human research.
The exploration of advanced peptide combinations, including the hypothetical can you stack mots c with retatrutide question, is a testament to the dynamic nature of biomedical science. While the potential for novel metabolic interventions is exciting, the journey from theoretical concept to validated therapeutic must be paved with meticulous research, unwavering ethical principles, and a deep respect for safety. The current lack of direct evidence for a mots c and retatrutide stack in humans underscores the need for continued, diligent scientific inquiry. For further reading on best practices, consider exploring resources on best practices for storing research peptides.
Conclusion
The theoretical appeal of a mots c and retatrutide stack lies in the potential for complementary actions that address metabolic health from both a fundamental cellular level (MOTS-c) and a systemic, hormonal regulation perspective (retatrutide). While MOTS-c offers promising insights into mitochondrial function and insulin sensitivity from preclinical studies, retatrutide stands as a highly effective, pharmaceutical-grade triple agonist with compelling clinical trial data for weight management.
However, it is crucial to reiterate that as of early 2026, there is no direct scientific evidence from human clinical trials supporting the safety, efficacy, or specific interactions of combining MOTS-c and retatrutide. MOTS-c remains a research peptide, not approved for human use, while retatrutide is a medication in advanced development.
Researchers and individuals in the wellness community must approach the concept of a mots c and retatrutide stack with extreme caution, prioritizing robust scientific methodology and ethical guidelines. The responsible path forward involves dedicated preclinical research to explore potential synergies, understand pharmacokinetic and pharmacodynamic interactions, and thoroughly assess the safety profile of such a combination before any consideration for human application. The future of metabolic health interventions may well involve novel peptide combinations, but this future must be built on a solid foundation of rigorous, evidence-based science.
Actionable Next Steps for Researchers:
- Prioritize In Vitro and Animal Studies: Focus on controlled laboratory experiments and animal models to elucidate the mechanistic interactions and potential benefits of MOTS-c and retatrutide in combination.
- Conduct Interaction Studies: Specifically design experiments to assess pharmacokinetic and pharmacodynamic interactions between the two peptides.
- Evaluate Safety and Toxicology: Thoroughly investigate any potential adverse effects of the combined stack in preclinical models.
- Source High-Quality Peptides: Ensure that all research peptides, including 5 amino 1 mq or mots c stack, are sourced from reputable suppliers that provide third-party purity testing.
- Adhere to Ethical Guidelines: Always conduct research within established ethical frameworks and institutional review board protocols.
SEO Meta Title: Mots C and Retatrutide Stack: 2026 Research Insights
SEO Meta Description: Explore the theoretical mots c and retatrutide stack for metabolic research in 2026. Understand individual mechanisms, current evidence, and crucial safety considerations.
