The Complete Guide to Peptides for Laboratory Research in 2026

The landscape of peptides for laboratory research has transformed dramatically over the past year. What once required years of painstaking trial-and-error can now be accomplished in hours through AI-powered computational systems. Researchers in fitness, recovery science, and metabolic studies are witnessing a revolution—one where machine learning generates novel antimicrobial peptides ready for immediate validation, and automated synthesis platforms reduce production time while cutting environmental waste[1]. This comprehensive guide explores the cutting-edge developments, practical applications, and strategic considerations for anyone sourcing peptides for laboratory research in 2026.
Key Takeaways
- AI and machine learning have reduced peptide discovery timelines from years to hours, enabling rapid development of novel compounds for laboratory validation[1]
- Advanced modification techniques including stapled peptides, lipidation, and macrocyclization dramatically improve stability, bioavailability, and research outcomes[1][2]
- Automated synthesis platforms are making high-quality peptide production accessible to smaller research facilities while reducing environmental impact[1]
- Regulatory frameworks continue to evolve, requiring researchers to prioritize verified suppliers with proper documentation and quality controls[1]
- Personalized research approaches are becoming standard practice, with peptide selection increasingly tailored to specific experimental parameters and biomarker tracking[5]
Understanding Peptides for Laboratory Research: The 2026 Landscape

Peptides for laboratory research represent short chains of amino acids that serve as fundamental building blocks for studying biological processes, cellular signaling, and therapeutic mechanisms. In 2026, the field has expanded far beyond simple sequence analysis to encompass sophisticated computational design, advanced modification strategies, and precision-targeted applications.
What Makes Research Peptides Different
Research-grade peptides differ significantly from pharmaceutical preparations. These compounds are specifically manufactured for in vitro and in vivo laboratory studies, requiring rigorous purity standards, comprehensive documentation, and proper handling protocols. The distinction matters because research applications demand consistency, traceability, and verification that clinical-grade products may not always provide.
Modern peptides for laboratory research must meet increasingly stringent quality benchmarks. Researchers expect third-party testing, certificate of analysis documentation, and transparent sourcing information—standards that have become non-negotiable in 2026's regulatory environment.
The Evolution of Peptide Discovery
The traditional peptide discovery process involved extensive screening of natural sources, followed by laborious synthesis and testing cycles. This approach could consume years of research time with uncertain outcomes. The introduction of computational methods has fundamentally altered this timeline.
AI-powered peptide design has shortened discovery timelines dramatically. Systems like ProteoGPT can now move peptide development from years of trial-and-error to mere hours of in silico analysis, generating novel antimicrobial peptides ready for immediate laboratory validation[1]. This represents a paradigm shift for researchers working with peptides for laboratory research across multiple disciplines.
Top 10 Peptides for Laboratory Research Applications in 2026
1. 🔬 BPC-157: Tissue Recovery Research
BPC-157 remains one of the most extensively studied peptides for laboratory research in the recovery and tissue repair domain. This pentadecapeptide derived from body protection compound demonstrates remarkable stability and has been the subject of numerous preclinical investigations examining cellular repair mechanisms.
Laboratory studies have explored BPC-157's interaction with growth factor signaling pathways, angiogenesis promotion, and tissue regeneration processes. Researchers investigating recovery protocols frequently include this compound in their experimental designs due to its well-documented stability profile and consistent research outcomes.
For comprehensive documentation and sourcing information, researchers can review our BPC-157 research guide which details proper handling, reconstitution protocols, and experimental considerations.
2. 💪 TB-500: Muscle Recovery Research Themes
TB-500, a synthetic version of Thymosin Beta-4, represents a critical tool for studying cellular migration, differentiation, and tissue repair mechanisms. This peptide for laboratory research has garnered significant attention in studies examining muscle recovery, wound healing, and inflammatory response modulation.
Research applications focus on TB-500's ability to promote actin polymerization and cellular motility—fundamental processes in tissue repair. Laboratory investigations have documented its effects on endothelial cell differentiation and blood vessel formation, making it valuable for muscle recovery research protocols.
The peptide's 43-amino acid sequence provides researchers with a stable, well-characterized compound for exploring recovery mechanisms at the cellular level. When sourcing peptides for laboratory research, TB-500's documented stability profile makes it a reliable choice for extended experimental timelines.
3. 🧬 Epithalon: Longevity Research Applications
Epithalon (also known as Epitalon) has become central to longevity peptide research investigating telomerase activation and cellular aging mechanisms. This tetrapeptide (Ala-Glu-Asp-Gly) has been studied extensively for its potential effects on telomere length, circadian rhythm regulation, and age-related cellular changes.
Laboratory research with Epithalon focuses on understanding how short peptide sequences might influence fundamental aging processes. Studies have examined its effects on pineal gland function, melatonin production, and antioxidant enzyme activity in various model systems.
Researchers exploring longevity peptide research applications find Epithalon valuable for investigating the relationship between peptide signaling and cellular senescence. The compound's relatively simple structure and well-established synthesis protocols make it accessible for laboratories with varying resource levels.
4. 🧠 Selank: Cognitive Function Research
Selank represents a synthetic derivative of tuftsin with applications in neuroscience research, particularly studies examining anxiety modulation, cognitive function, and neuroprotective mechanisms. This peptide for laboratory research has been investigated for its effects on brain-derived neurotrophic factor (BDNF) expression and neurotransmitter systems.
Laboratory applications include examining Selank's influence on serotonergic and dopaminergic pathways, as well as its potential anxiolytic properties in preclinical models. Researchers studying Selank peptide benefits in controlled settings have documented its effects on learning, memory consolidation, and stress response markers.
The peptide's stability and well-characterized pharmacological profile make it suitable for extended research protocols. When working with cognitive research peptides, proper handling and storage protocols ensure consistent experimental results across multiple trial iterations.
5. 🔋 SS-31 (Elamipretide): Mitochondrial Research
SS-31, also known as Elamipretide, has emerged as a powerful tool for investigating mitochondrial function, cellular energetics, and oxidative stress responses. This peptide for laboratory research selectively targets mitochondrial membranes, making it invaluable for studies examining cellular energy production and mitochondrial dysfunction.
Research applications focus on SS-31's interaction with cardiolipin, a phospholipid critical for mitochondrial membrane integrity and electron transport chain function. Laboratory investigations have explored its effects on ATP production, reactive oxygen species generation, and mitochondrial morphology across various cell types.
Detailed information about SS-31 research peptide considerations helps researchers design appropriate experimental protocols. The compound's unique mechanism of action—concentrating specifically at inner mitochondrial membranes—provides researchers with a targeted tool for studying cellular bioenergetics.
6. 🌟 GHK-Cu: Copper Peptide Research
GHK-Cu (Glycyl-L-Histidyl-L-Lysine-Copper) represents a naturally occurring copper-binding peptide that has become central to research examining tissue remodeling, wound healing, and extracellular matrix regulation. This peptide for laboratory research demonstrates multiple biological activities related to collagen synthesis, antioxidant enzyme activation, and growth factor modulation.
Laboratory studies have investigated GHK-Cu's effects on fibroblast proliferation, metalloproteinase expression, and angiogenesis in various experimental models. The copper complex formation provides researchers with a tool for studying metal ion signaling in biological systems.
Researchers can access comprehensive sourcing information through our GHK-Cu peptide purchase guide, which details quality considerations, handling protocols, and experimental design recommendations for copper peptide research applications.
7. 📊 MOTS-c: Mitochondrial-Derived Peptide Studies
MOTS-c represents a mitochondrial-derived peptide encoded within the mitochondrial genome, making it a unique subject for peptides for laboratory research focused on metabolic regulation and cellular communication. This 16-amino acid peptide has been studied for its effects on insulin sensitivity, metabolic homeostasis, and exercise response mechanisms.
Research applications examine MOTS-c's role in AMPK activation, glucose metabolism, and mitochondrial-nuclear communication pathways. Laboratory investigations have documented its effects on skeletal muscle metabolism and age-related metabolic decline in preclinical models.
Studies exploring MOTS-c as a mitochondrial peptide have revealed its potential as a research tool for understanding how mitochondrial-encoded peptides influence whole-body metabolism. When sourcing peptides for laboratory research in this category, researchers should verify sequence accuracy and purity standards.
8. 🎯 PT-141 (Bremelanotide): Melanocortin Receptor Research
PT-141, a synthetic peptide analog of alpha-MSH, serves as a valuable research tool for investigating melanocortin receptor pathways, particularly MC3R and MC4R signaling. This peptide for laboratory research has been studied extensively in preclinical models examining central nervous system signaling and receptor activation patterns.
Laboratory applications focus on understanding melanocortin receptor pharmacology, signal transduction mechanisms, and physiological responses to receptor activation. Research protocols examining PT-141 peptide research contexts provide insights into proper experimental controls and quality assurance measures.
The peptide's cyclic structure and receptor selectivity make it useful for structure-activity relationship studies and receptor binding investigations. Researchers working with melanocortin pathways find PT-141 valuable for exploring the complex signaling networks involved in central regulation of various physiological processes.
9. 🧪 CJC-1295 with Ipamorelin: Growth Hormone Research
The combination of CJC-1295 and Ipamorelin represents a frequently studied peptide blend for laboratory research examining growth hormone releasing pathways and pulsatile hormone secretion patterns. CJC-1295, a growth hormone releasing hormone analog, and Ipamorelin, a growth hormone secretagogue, provide researchers with tools for investigating different aspects of GH axis regulation.
Laboratory studies have examined how these peptides influence pituitary signaling, receptor activation patterns, and downstream effects on IGF-1 production. Research protocols often utilize these compounds to understand the complex regulatory mechanisms governing growth hormone release and its physiological consequences.
Detailed guidance on CJC-1295 Ipamorelin assay planning helps researchers design appropriate experimental protocols, including dosing considerations, timing parameters, and quality control measures essential for reproducible results.
10. 🔄 GLP-1 Receptor Agonists: Metabolic Research
GLP-1 (Glucagon-Like Peptide-1) receptor agonists have become central to peptides for laboratory research investigating metabolic regulation, insulin secretion, and glucose homeostasis. These peptides activate incretin receptors, providing researchers with tools for studying pancreatic beta cell function, appetite regulation, and metabolic signaling pathways.
Research applications examine GLP-1's effects on insulin secretion mechanisms, glucagon suppression, gastric emptying, and central nervous system appetite regulation. Laboratory investigations have documented receptor binding characteristics, signal transduction pathways, and downstream metabolic effects in various experimental models.
Comprehensive information about GLP-1 peptide research concepts helps researchers understand the evolving landscape of incretin-based research tools. When sourcing peptides for laboratory research in this category, verification of sequence accuracy and receptor selectivity becomes particularly important.
Advanced Peptide Modification Techniques Transforming Research
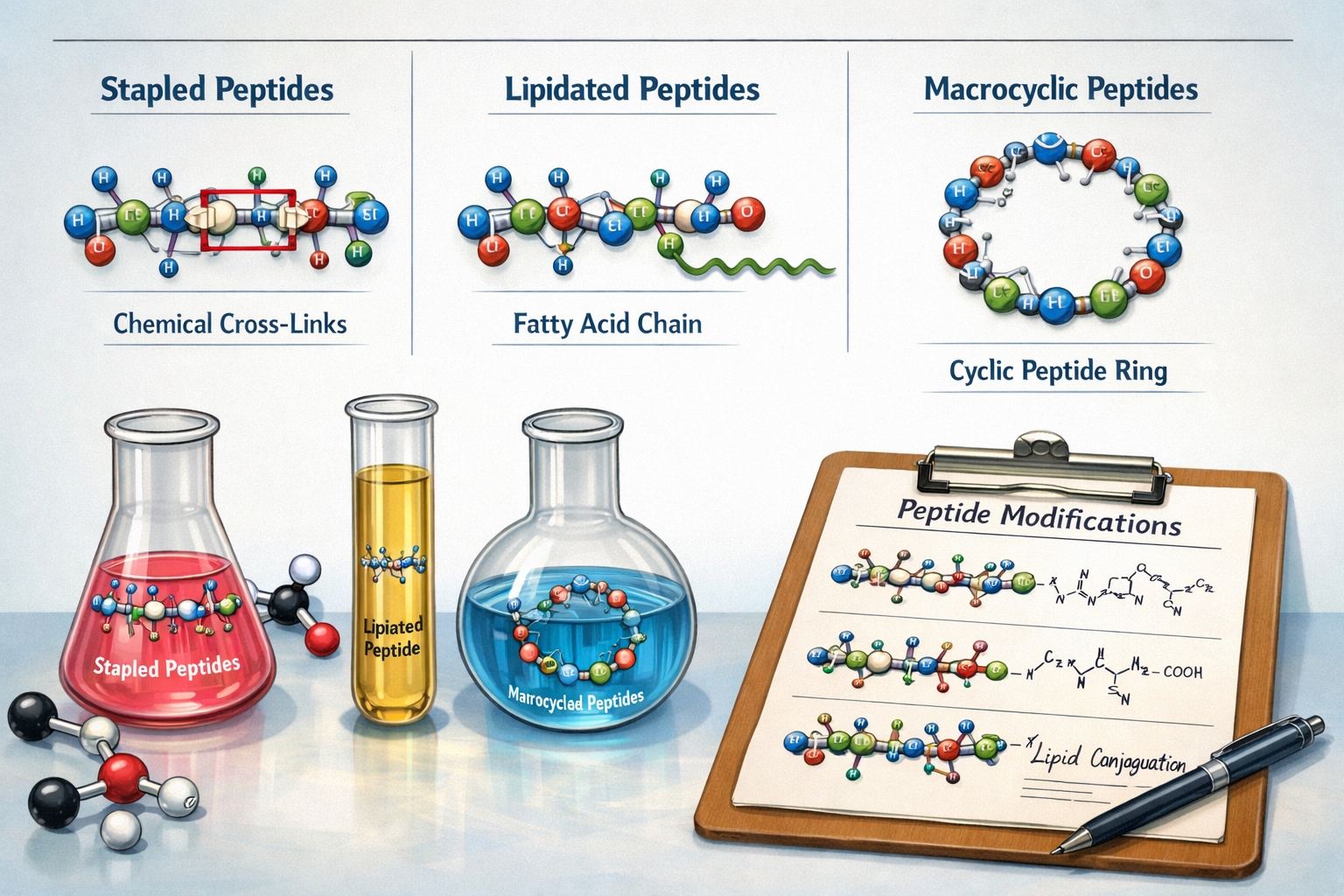
Stapled Peptides: Enhancing Stability and Permeability
Stapled peptides represent an established modification strategy where chemical cross-links stabilize peptide structure and improve cell permeability while maintaining biological activity[2]. This technique addresses traditional peptide degradation challenges that have historically limited research applications.
The stapling process involves introducing synthetic amino acids that form covalent bonds, creating a "staple" that locks the peptide into its active conformation. This modification dramatically improves proteolytic resistance, allowing peptides for laboratory research to maintain activity in biological environments where unmodified peptides would rapidly degrade.
Research applications benefit from stapled peptides' enhanced cellular uptake, enabling studies of intracellular targets previously inaccessible to peptide-based tools. The technology has proven particularly valuable for investigating protein-protein interactions and intracellular signaling pathways.
Lipidation: Extending Circulation and Bioavailability
Lipidated peptides are improving membrane permeability and circulation time through fatty acid chain modifications that extend peptide half-life and tissue distribution[2]. This modification strategy addresses fundamental limitations of unmodified peptide drugs, making them more suitable for extended research protocols.
The lipidation process involves attaching fatty acid chains to specific amino acid residues, creating amphipathic molecules with enhanced membrane interaction properties. These modifications can dramatically alter pharmacokinetic profiles, allowing researchers to design experiments with extended observation windows.
Laboratory applications of lipidated peptides for laboratory research include studies examining sustained receptor activation, prolonged signaling effects, and tissue-specific accumulation patterns. The modification technique has become standard practice for researchers requiring peptides with extended biological activity.
Macrocyclization: Creating Ring-Shaped Stability
Enzyme-enabled macrocyclization tools have expanded peptide modification capabilities through the discovery of enzymes like PapB, a radical SAM maturase that provides new enzymatic methods to stabilize peptides into ring-shaped molecules[1]. This approach makes peptides significantly more durable and bioavailable while reducing chemical waste compared to traditional synthesis.
Macrocyclic peptides combine the specificity of linear peptides with the stability of small molecules, creating research tools with unique properties. The cyclic structure restricts conformational flexibility, often enhancing receptor binding affinity and selectivity while improving resistance to enzymatic degradation.
Research applications of macrocyclic peptides for laboratory research span multiple disciplines, from studying receptor pharmacology to investigating membrane transport mechanisms. The technology enables researchers to create peptides with drug-like properties while maintaining the targeting specificity that makes peptides valuable research tools.
AI and Machine Learning: Revolutionizing Peptide Discovery
Computational Design Platforms
Machine learning is optimizing peptide sequence design through algorithms that predict peptide properties and optimize sequences for specific research applications[2]. This computational approach potentially accelerates development beyond simple modifications of natural sequences, enabling truly de novo peptide design.
Deep learning models trained on vast databases of peptide sequences and their biological activities can now predict which amino acid combinations will produce desired properties. These systems analyze structure-activity relationships at scales impossible for traditional experimental approaches, identifying promising candidates for laboratory validation.
A landmark Nature Materials study in late 2025 showcased a family of self-assembling antimicrobial peptides engineered through machine learning that demonstrated potent activity against multidrug-resistant infections in mouse models while maintaining human cell viability[1]. This breakthrough demonstrates how AI-designed peptides for laboratory research can achieve properties difficult to obtain through traditional discovery methods.
Automated Synthesis and Quality Control
Automated synthesis platforms are reducing production time and environmental waste, enabling smaller biotech firms and research laboratories to compete by translating complex syntheses into reliable protocols[1]. These systems integrate synthesis, purification, and quality control into streamlined workflows that minimize human error and maximize reproducibility.
Modern automated platforms can synthesize multiple peptide variants simultaneously, allowing researchers to rapidly test structure-activity relationships and optimize sequences for specific applications. The integration of real-time quality monitoring ensures that each synthesis batch meets predetermined purity standards before proceeding to biological testing.
For researchers sourcing peptides for laboratory research, understanding whether suppliers utilize automated synthesis platforms provides insight into product consistency and quality assurance capabilities. The technology has become increasingly important as regulatory frameworks tighten around peptide production standards.
Predictive Modeling for Stability and Activity
Computer-aided design methods are enabling rational peptide engineering through sophisticated approaches that allow researchers to create peptides with desired properties through de novo design using artificial intelligence[2]. These systems can predict not only biological activity but also stability, solubility, and potential off-target effects before synthesis.
Predictive modeling reduces the trial-and-error cycles traditionally required for peptide optimization. Researchers can now screen thousands of virtual peptide candidates computationally, selecting only the most promising designs for actual synthesis and testing. This approach dramatically reduces research costs and accelerates timeline from concept to validation.
The integration of predictive modeling with experimental validation creates a feedback loop that continuously improves algorithm accuracy. As more peptides for laboratory research are synthesized and tested, the models become increasingly precise, further accelerating the discovery process.
Quality Considerations for Sourcing Peptides for Laboratory Research
Purity Standards and Testing Protocols
Peptide purity directly impacts research reproducibility and experimental outcomes. In 2026, leading suppliers provide comprehensive testing documentation including HPLC analysis, mass spectrometry verification, and amino acid analysis for each batch of peptides for laboratory research.
High-performance liquid chromatography (HPLC) remains the gold standard for assessing peptide purity, separating the target peptide from synthesis byproducts, deletion sequences, and truncated variants. Researchers should expect minimum purity levels of 95% for most research applications, with some specialized studies requiring 98% or higher purity.
Mass spectrometry provides molecular weight confirmation, ensuring the synthesized peptide matches the intended sequence. This verification step catches synthesis errors that might not be apparent from HPLC analysis alone. Combined with amino acid analysis, these testing methods provide comprehensive quality assurance for research-grade peptides.
Detailed information about peptide purity testing helps researchers understand quality documentation and make informed sourcing decisions. When evaluating suppliers of peptides for laboratory research, comprehensive testing documentation should be standard, not optional.
Storage and Handling Best Practices
Proper storage and handling protocols are essential for maintaining peptide integrity throughout research protocols. Most peptides for laboratory research require storage at -20°C or -80°C in lyophilized form, protected from moisture, light, and temperature fluctuations.
Once reconstituted, peptide solutions typically require refrigeration at 2-8°C for short-term storage or freezing at -20°C for extended periods. However, freeze-thaw cycles can damage peptide structure, so researchers should aliquot reconstituted peptides into single-use portions to avoid repeated freezing and thawing.
Reconstitution protocols vary by peptide, with some requiring specific solvents or pH conditions to maintain stability and solubility. Researchers should follow supplier recommendations for reconstitution and verify that storage conditions maintain peptide activity throughout the experimental timeline.
Regulatory Compliance and Documentation
Regulatory frameworks are evolving around peptide distribution, with tightening requirements around synthetic and compounded peptides expected to shape development and clinical implementation strategies going forward[1]. Researchers must ensure their peptide sources comply with applicable regulations and provide proper documentation.
For peptides for laboratory research, proper documentation includes certificates of analysis, synthesis records, purity testing results, and clear labeling indicating "For Research Use Only" status. This documentation protects researchers from regulatory complications and ensures experimental integrity.
Understanding supplier compliance with Good Manufacturing Practices (GMP) and quality management systems provides additional assurance of product consistency. While research-grade peptides may not require the same regulatory oversight as pharmaceutical products, adherence to recognized quality standards indicates supplier commitment to product integrity.
Emerging Applications: Peptide Blends and Combination Research
Synergistic Peptide Combinations
Peptide blends represent an emerging area of laboratory research where multiple peptides are combined to investigate synergistic effects and multi-pathway targeting. These combinations allow researchers to study complex biological processes that involve multiple signaling pathways simultaneously.
Research with peptide blends examines how different peptides might interact, enhance, or modulate each other's effects. For example, combining peptides targeting different aspects of tissue repair might reveal interactions between growth factor signaling, inflammation modulation, and matrix remodeling pathways.
The development of standardized peptide blends provides researchers with consistent tools for investigating multi-factorial biological processes. When sourcing peptides for laboratory research in blend formulations, verification of individual peptide purity and accurate ratio confirmation becomes particularly important.
Peptide-Drug Conjugates
Peptide-drug conjugates are advancing in early clinical development, combining peptide targeting specificity with therapeutic agent potency[2]. These molecules represent a sophisticated approach to targeted delivery, using peptides as homing mechanisms to direct drugs to specific cell types or tissues.
Laboratory research with peptide-drug conjugates examines linker chemistry, payload release mechanisms, and targeting efficiency. Early results suggest improved efficacy and reduced toxicity compared to conventional approaches, making this an active area of investigation for peptides for laboratory research applications.
The technology requires careful consideration of peptide selection, conjugation chemistry, and payload characteristics. Researchers investigating these systems must balance peptide stability with linker cleavability, ensuring the conjugate remains intact during circulation but releases its payload at the target site.
Nanotechnology Integration
Nanotechnology approaches are protecting peptides from degradation through advanced delivery systems that enable targeted delivery and extend therapeutic windows[2]. These systems encapsulate peptides for laboratory research in protective carriers that shield them from enzymatic degradation while controlling release kinetics.
Nanoparticle formulations, liposomes, and polymeric carriers represent different strategies for peptide protection and delivery. Research applications examine how these systems affect peptide bioavailability, tissue distribution, and biological activity in various experimental models.
The integration of nanotechnology with peptide research opens new possibilities for studying peptides that would otherwise be too unstable for in vivo applications. Researchers can now investigate peptides with promising biological activities but poor pharmacokinetic profiles by incorporating them into protective delivery systems.
Practical Considerations for Research Protocol Design
Dosing and Concentration Guidelines
Designing appropriate dosing protocols for peptides for laboratory research requires consideration of peptide stability, receptor affinity, and experimental objectives. Researchers must balance achieving measurable biological effects with avoiding supraphysiological concentrations that might produce artifacts.
Starting with published literature provides baseline dosing information, but researchers should conduct preliminary dose-response studies to optimize concentrations for their specific experimental systems. Cell culture studies typically use micromolar to nanomolar concentrations, while in vivo studies require consideration of pharmacokinetics and tissue distribution.
Documentation of dosing rationale, preparation methods, and administration routes ensures experimental reproducibility. When working with peptides for laboratory research, maintaining detailed records of reconstitution, storage conditions, and handling procedures helps troubleshoot unexpected results and ensures consistency across experimental replicates.
Control Selection and Validation
Appropriate control selection is critical for interpreting results from peptide research. Negative controls might include vehicle-only treatments, scrambled peptide sequences, or inactive peptide analogs that help distinguish specific effects from non-specific interactions.
Positive controls using established peptides with known activities provide benchmarks for assessing experimental system responsiveness. These controls verify that the experimental model can detect peptide effects and help normalize results across different experimental runs.
Validation of peptides for laboratory research effects often requires multiple complementary approaches. Combining receptor binding assays with functional readouts and downstream signaling measurements provides comprehensive evidence for peptide activity and mechanism of action.
Reproducibility and Documentation Standards
Reproducibility challenges have prompted increased emphasis on detailed documentation of peptide research protocols. Researchers should record complete information about peptide source, batch numbers, purity data, storage conditions, and preparation methods.
Standardized reporting frameworks help ensure that peptide research can be replicated by other laboratories. This includes documenting not only successful experiments but also negative results and troubleshooting steps that might inform future research efforts.
The growing emphasis on research transparency and data sharing makes comprehensive documentation increasingly important. When publishing research using peptides for laboratory research, journals increasingly require detailed methodological information and data availability statements that enable independent verification.
Future Trends: What's Next for Peptides in Laboratory Research

Personalized Research Approaches
Personalized peptide research is becoming feasible through advances in functional testing, genetics, and biomarker tracking that enable peptide selection increasingly tailored to specific experimental parameters[5]. This trend reflects broader movements toward precision medicine and individualized therapeutic approaches.
Laboratory applications of personalized approaches might involve selecting peptides based on specific receptor expression patterns, genetic backgrounds, or biomarker profiles in experimental models. This targeting improves research efficiency by focusing on peptides most likely to produce measurable effects in specific contexts.
The integration of omics technologies—genomics, proteomics, metabolomics—with peptide research enables increasingly sophisticated experimental designs. Researchers can now correlate peptide effects with comprehensive molecular profiles, revealing mechanisms of action and identifying biomarkers of response.
Expanded Therapeutic Targets
Peptide research is expanding beyond traditional targets to address previously "undruggable" proteins and protein-protein interactions. The unique properties of peptides—larger than small molecules but smaller than antibodies—position them to target binding sites inaccessible to other therapeutic modalities.
Intracellular targets, once considered beyond the reach of peptide therapeutics, are becoming accessible through advanced modification strategies and delivery technologies. Researchers are investigating peptides that disrupt transcription factor interactions, modulate enzyme activity, and interfere with intracellular signaling complexes.
The expansion of target space creates new opportunities for peptides for laboratory research across multiple disciplines. From neuroscience to immunology to metabolic research, peptides provide tools for investigating biological processes at molecular resolution.
Integration with Digital Health Technologies
Digital transformation in the peptide sector is creating new opportunities for data integration, quality control, and research optimization[7]. Laboratory information management systems (LIMS) now integrate peptide inventory management, experimental tracking, and data analysis in unified platforms.
Real-time monitoring technologies enable continuous quality assessment throughout peptide synthesis and storage. Sensors tracking temperature, humidity, and other environmental parameters ensure optimal conditions for maintaining peptide integrity.
The integration of artificial intelligence with laboratory workflows promises to accelerate peptide research further. Machine learning systems can analyze experimental results, suggest protocol optimizations, and identify patterns that might escape human observation, creating a continuous improvement cycle for peptides for laboratory research applications.
Conclusion: Navigating the Future of Peptides for Laboratory Research
The field of peptides for laboratory research has entered an unprecedented era of innovation and accessibility. AI-powered design tools, advanced modification strategies, and automated synthesis platforms have democratized peptide research, making sophisticated tools available to laboratories of all sizes. The transformation from years-long discovery timelines to hours of computational design represents a fundamental shift in how researchers approach peptide-based investigations[1].
For researchers in fitness science, recovery studies, and metabolic research, understanding the landscape of available peptides for laboratory research and their applications is essential for designing robust experimental protocols. The peptides highlighted in this guide—from BPC-157 and TB-500 to emerging compounds like MOTS-c and advanced GLP-1 analogs—represent tools with well-documented research applications and established quality standards.
Actionable Next Steps
For researchers beginning peptide studies:
- Identify specific research questions and select peptides with documented relevance to your experimental objectives
- Prioritize suppliers offering comprehensive quality documentation, including HPLC analysis, mass spectrometry verification, and certificates of analysis
- Review current peptide research developments to stay informed about emerging tools and methodologies
For established research programs:
- Explore advanced modification strategies like stapling, lipidation, or macrocyclization to overcome limitations of unmodified peptides
- Consider peptide blends and combination approaches for investigating multi-pathway biological processes
- Implement comprehensive documentation protocols to ensure reproducibility and facilitate data sharing
For quality assurance:
- Establish relationships with suppliers demonstrating commitment to quality standards and regulatory compliance
- Verify batch-to-batch consistency through regular testing and documentation review
- Maintain detailed records of peptide storage, handling, and preparation to troubleshoot unexpected results
The regulatory landscape continues to evolve, making supplier selection increasingly important. Researchers should prioritize sources that provide transparent documentation, comprehensive testing, and clear communication about product specifications and limitations[1].
As we advance through 2026, the convergence of artificial intelligence, advanced chemistry, and digital technologies promises to accelerate peptide research further. The tools available today—from computationally designed novel sequences to precisely modified analogs with enhanced properties—provide researchers with unprecedented capabilities for investigating biological processes at molecular resolution.
The future of peptides for laboratory research lies in personalized approaches, expanded target engagement, and integration with comprehensive data systems that enable increasingly sophisticated experimental designs[5]. By staying informed about emerging technologies, maintaining rigorous quality standards, and implementing robust experimental protocols, researchers can leverage these powerful tools to advance understanding across multiple scientific disciplines.
Whether investigating recovery mechanisms, metabolic regulation, or cellular signaling pathways, the expanding toolkit of peptides for laboratory research offers solutions for addressing complex biological questions. The key to success lies in careful peptide selection, rigorous quality verification, and thoughtful experimental design that leverages the unique properties of these versatile research tools.
References
[1] Ten Peptides 2025 Medical Trends 2026 – https://www.intelligentliving.co/ten-peptides-2025-medical-trends-2026/
[2] Emerging And Approved Therapeutic Peptides Mechanisms Clinical Uses – https://globalrph.com/2025/12/emerging-and-approved-therapeutic-peptides-mechanisms-clinical-uses/
[3] Peptides In Practice What It Takes To Advance These Therapies To Clinic – https://www.drugtargetreview.com/article/191795/peptides-in-practice-what-it-takes-to-advance-these-therapies-to-clinic/
[4] Peptides In 2026 – https://veltrigen.com/blogs/peptide-research-insights/peptides-in-2026
[5] Peptide Therapy Trends To Watch In 2025 What Clinics Need To Know – https://www.optimantra.com/blog/peptide-therapy-trends-to-watch-in-2025-what-clinics-need-to-know
[6] Peptide Targets – https://www.chi-peptalk.com/peptide-targets
[7] The Critical Need For Digital Transformation In The Peptide And Api Sectors – https://www.pharmtech.com/view/the-critical-need-for-digital-transformation-in-the-peptide-and-api-sectors
SEO Meta Title and Description
Meta Title: Peptides for Laboratory Research: 2026 Complete Guide
Meta Description: Discover the top peptides for laboratory research in 2026. Expert guide covering AI-designed compounds, quality standards, and research applications.
