Reta GLP3: The Revolutionary Triple Agonist Peptide Transforming Metabolic Research in 2025

The peptide research landscape has been forever changed by the emergence of a groundbreaking compound that targets not one, not two, but three critical metabolic pathways simultaneously. Reta GLP3 (retatrutide) represents the pinnacle of modern peptide engineering, offering researchers unprecedented opportunities to study complex metabolic interactions through its unique triple-agonist mechanism.
Key Takeaways
• Reta GLP3 is a 39-amino-acid synthetic peptide that simultaneously activates GLP-1, GIP, and glucagon receptors
• Phase 3 clinical trials demonstrate up to 29% weight reduction, significantly outperforming existing single and dual agonists
• The compound features an extended 6-day half-life due to its C20 fatty acid side chain modification
• Research applications extend beyond metabolic studies to include liver health, cardiovascular function, and sleep disorder investigations
• Current availability is limited to clinical trials, with regulatory approval anticipated around 2027-2028
Understanding Reta GLP3: The Science Behind Triple Agonism
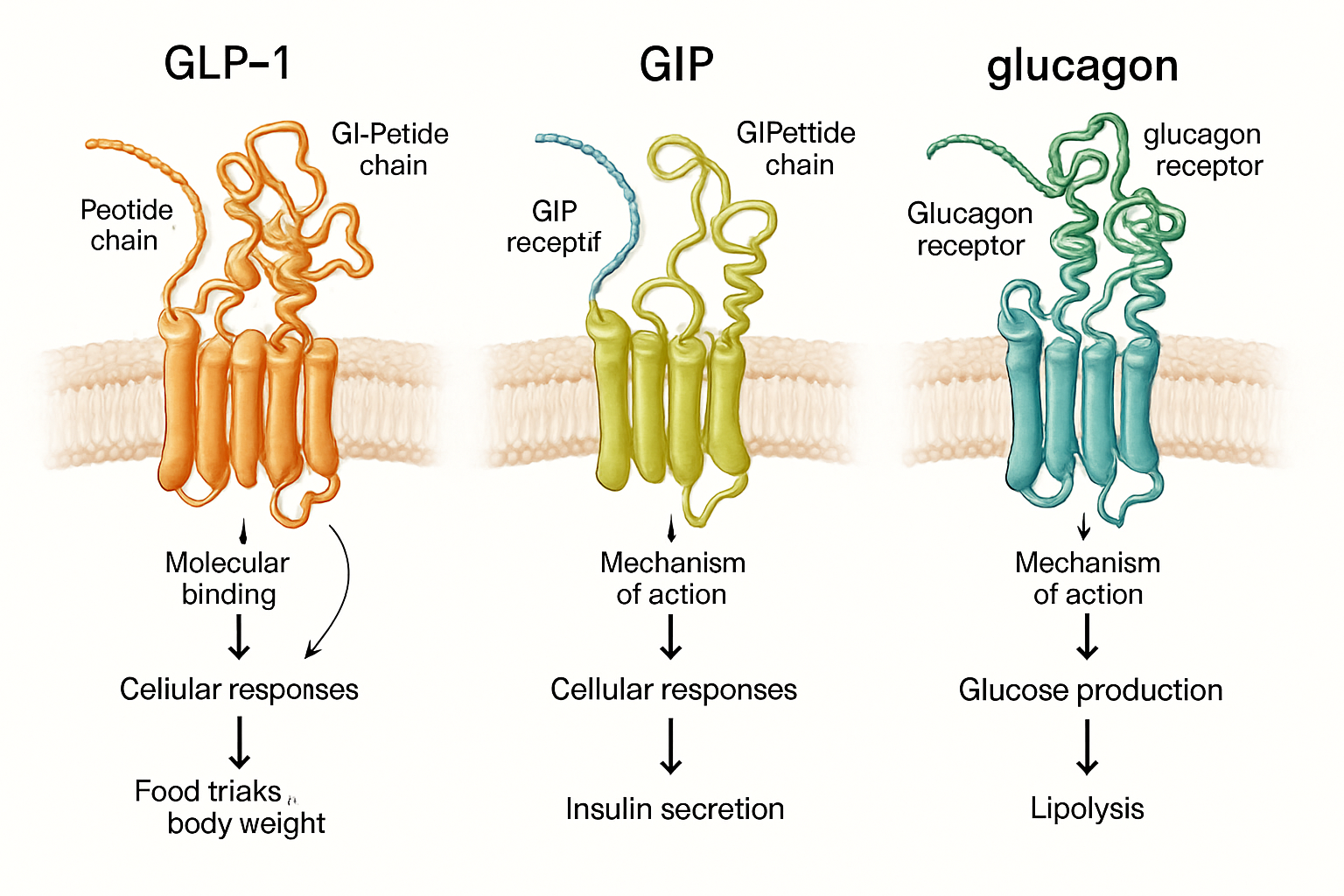
Reta GLP3 (LY3437943) represents a revolutionary advancement in peptide research, developed by Eli Lilly as the world's first triple incretin receptor agonist. This 39-amino-acid synthetic peptide simultaneously targets three distinct metabolic pathways: GLP-1 (glucagon-like peptide-1), GIP (glucose-dependent insulinotropic polypeptide), and glucagon receptors. For researchers interested in exploring comprehensive peptide research options, understanding this triple mechanism opens new avenues for metabolic studies.
The molecular structure of reta GLP3 incorporates a C20 fatty acid side chain that dramatically extends its biological half-life to approximately 6 days. This modification allows for less frequent dosing schedules compared to traditional peptide compounds, making it particularly valuable for long-term research protocols. The advanced peptide formulations available today reflect this sophisticated approach to peptide engineering.
The Triple Receptor Mechanism
Each receptor pathway activated by reta GLP3 serves a distinct function in metabolic regulation:
GLP-1 Pathway Activation:
- Enhances glucose-dependent insulin secretion
- Suppresses glucagon release when glucose levels are elevated
- Slows gastric emptying and promotes satiety
- Supports pancreatic beta-cell preservation
GIP Pathway Activation:
- Stimulates insulin release in response to nutrient intake
- Promotes glucose uptake in peripheral tissues
- Facilitates fat mobilization and energy utilization
- Supports bone metabolism and cardiovascular health
Glucagon Pathway Activation:
- Increases energy expenditure through thermogenesis
- Promotes lipolysis and fat oxidation
- Enhances hepatic glucose production when appropriate
- Supports metabolic flexibility and adaptation
This synergistic approach allows researchers to study complex metabolic interactions that cannot be achieved with single-target compounds. The peptide research community has shown significant interest in exploring these multi-pathway effects.
Reta GLP3 Clinical Research Findings and Applications
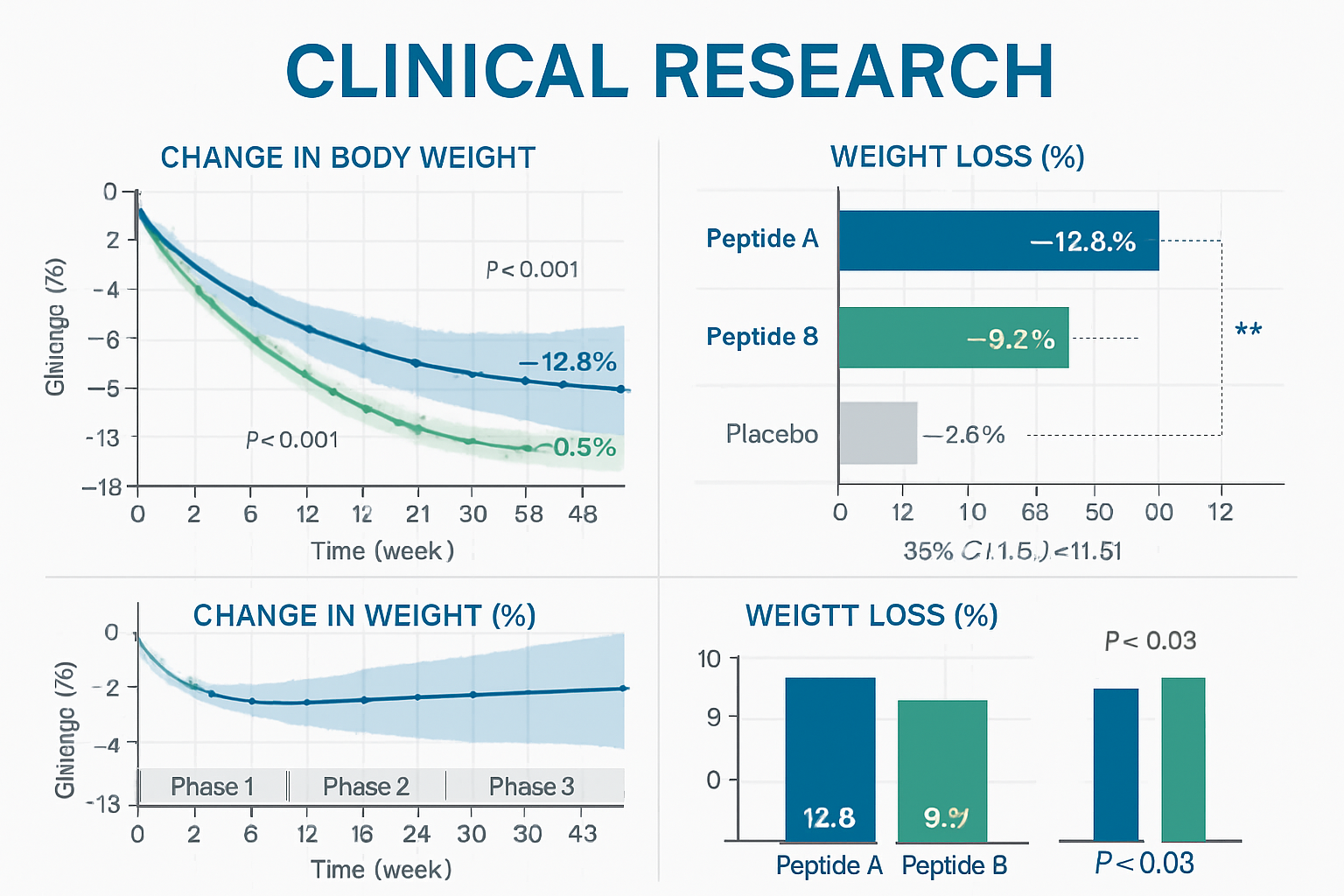
The clinical research surrounding reta GLP3 has produced remarkable results that have captured the attention of the global research community. Phase 2 clinical trials demonstrated an average weight reduction of 24.2% over 48 weeks at the highest tested dose, with participants continuing to show weight loss at study completion. These findings established reta GLP3 as a leading candidate for advanced metabolic research.
Phase 3 TRIUMPH Trial Results
The most recent Phase 3 study results have elevated reta GLP3 to unprecedented heights in metabolic research. Participants achieved up to 29% weight reduction, significantly surpassing the performance of existing compounds:
| Compound | Average Weight Loss | Study Duration |
|---|---|---|
| Reta GLP3 | 29% | 48 weeks |
| Semaglutide | 15% | 68 weeks |
| Tirzepatide | 21% | 72 weeks |
The ongoing TRIUMPH trials (2023-2027) are evaluating reta GLP3 across multiple research areas, including obesity studies, type 2 diabetes research, metabolic-associated fatty liver disease (MASH) investigations, sleep apnea research, and osteoarthritis studies. Researchers working with specialized peptide protocols can access comprehensive data from these extensive trials.
Liver Health Research Applications
One of the most promising areas of reta GLP3 research involves its effects on liver health. In MASH patient studies, the compound demonstrated remarkable liver fat reduction:
- 82-86% reduction in liver fat content
- 86% of participants achieved normal liver fat levels (<5%)
- Significant improvements in liver enzyme markers
- Enhanced hepatic insulin sensitivity
These findings suggest that reta GLP3 may offer researchers new tools for studying liver metabolism and fatty liver disease progression. The comprehensive research applications continue to expand as more data becomes available.
Cardiovascular Research Benefits
Reta GLP3 has shown significant cardiovascular research applications through its multi-pathway effects:
Lipid Profile Improvements:
- Triglyceride reduction: 15-21%
- LDL cholesterol reduction: 5-7%
- HDL cholesterol optimization
- Improved lipid particle size distribution
Blood Pressure Research:
- Systolic blood pressure reduction: ~5 mmHg
- Improved endothelial function markers
- Enhanced vascular reactivity
- Reduced inflammatory markers
Additional Cardiovascular Markers:
- Improved heart rate variability
- Enhanced cardiac metabolic efficiency
- Reduced oxidative stress indicators
- Better glucose-lipid metabolic coupling
Researchers studying cardiovascular peptide applications can leverage these comprehensive effects for multi-system research protocols.
Research Protocols and Safety Considerations for Reta GLP3
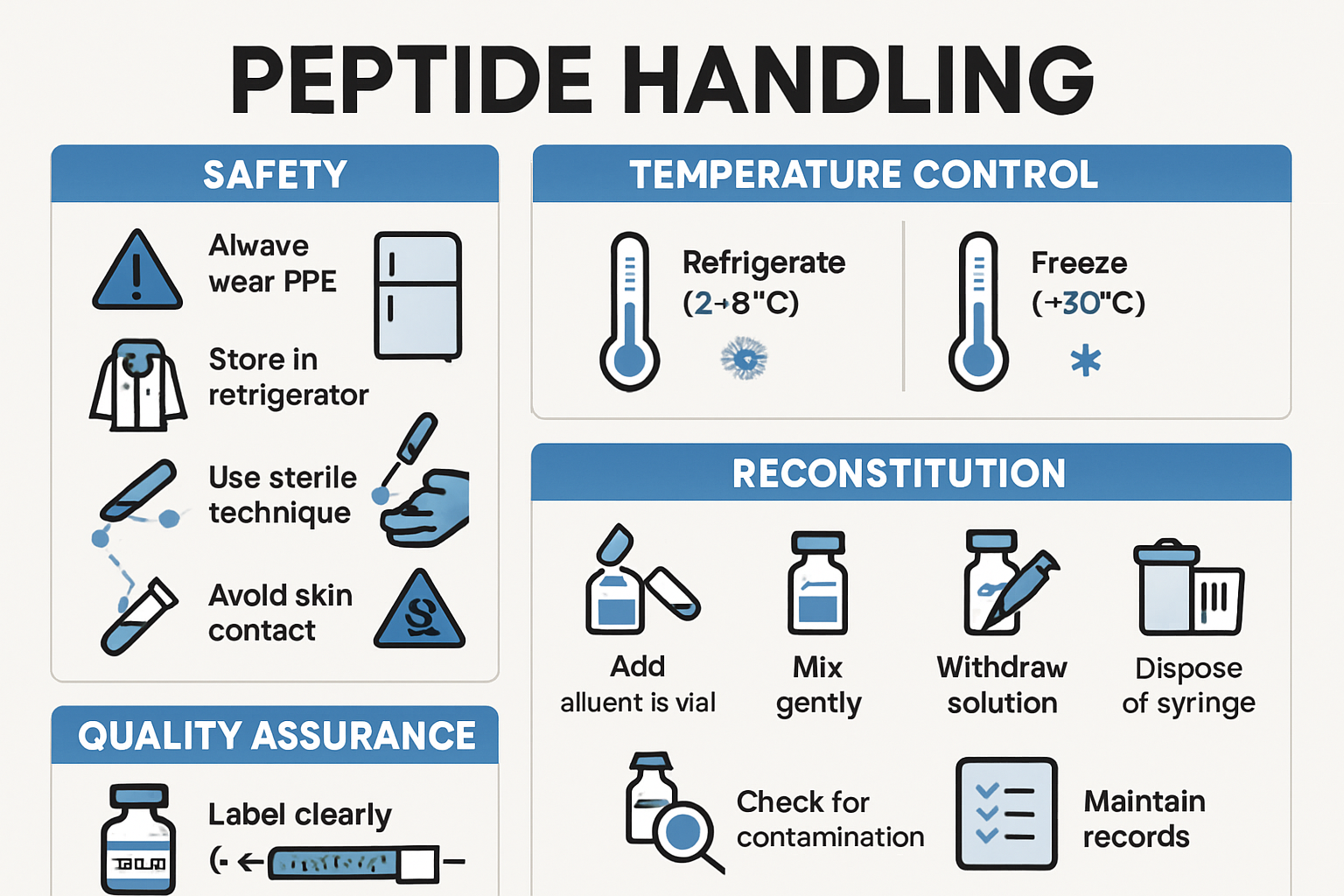
When working with reta GLP3 in research settings, understanding proper protocols and safety considerations is essential for obtaining reliable, reproducible results. The compound's unique pharmacokinetic profile requires specific handling and storage procedures that differ from traditional peptide research compounds.
Laboratory Handling and Storage
Reta GLP3 requires careful attention to storage conditions to maintain peptide integrity:
Storage Requirements:
- Lyophilized form: Store at 2-8°C (refrigerated)
- Reconstituted solution: Use within 28 days when refrigerated
- Avoid freezing: Repeated freeze-thaw cycles degrade peptide structure
- Light protection: Store in amber vials or dark conditions
- pH stability: Maintain neutral pH for optimal stability
Reconstitution Protocols:
- Allow vial to reach room temperature before reconstitution
- Use bacteriostatic water or sterile saline for dilution
- Add solvent slowly along vial wall to minimize foaming
- Gently swirl (do not shake vigorously) until completely dissolved
- Inspect solution for clarity and absence of particulates
Researchers can find detailed protocols and best practices for peptide storage through specialized resources. The quality-controlled peptide preparations available today incorporate these stringent storage requirements.
Research Dosing Considerations
Reta GLP3 research protocols typically follow escalating dose schedules to minimize adverse effects while maximizing research outcomes:
Typical Research Dose Escalation:
- Week 1-4: 0.5 mg weekly
- Week 5-8: 1.0 mg weekly
- Week 9-12: 2.0 mg weekly
- Week 13-16: 4.0 mg weekly
- Week 17+: 8.0-12.0 mg weekly (maximum research doses)
The extended half-life of reta GLP3 allows for weekly administration schedules, which simplifies research protocols compared to daily-dosing compounds. Researchers can reference commonly researched dosing protocols for additional guidance.
Safety Monitoring in Research Settings
Reta GLP3 research requires comprehensive safety monitoring due to its potent multi-pathway effects:
Gastrointestinal Monitoring:
- Nausea and vomiting (most common, dose-dependent)
- Diarrhea and constipation
- Abdominal discomfort
- Changes in appetite and food preferences
Cardiovascular Monitoring:
- Heart rate increases (typically modest, 2-5 bpm)
- Blood pressure changes
- ECG monitoring for rhythm disturbances
- Cardiovascular risk factor assessment
Metabolic Monitoring:
- Blood glucose levels (hypoglycemia risk)
- Electrolyte balance
- Liver function tests
- Kidney function markers
- Lipid profile changes
Injection Site Monitoring:
- Local reactions (redness, swelling, pain)
- Lipodystrophy with repeated injections
- Proper injection site rotation protocols
- Sterile technique maintenance
Research teams working with comprehensive peptide research protocols must establish robust monitoring systems to ensure participant safety and data integrity.
Research Applications and Study Design
Reta GLP3 offers unique opportunities for multi-system research due to its triple-agonist mechanism:
Metabolic Research Applications:
- Glucose homeostasis studies
- Insulin sensitivity research
- Lipid metabolism investigations
- Energy expenditure analysis
- Body composition research
Specialized Research Areas:
- Sleep apnea studies (airway fat reduction)
- Osteoarthritis research (joint inflammation)
- Kidney function studies (UACR reduction ~20%)
- Cognitive function research (metabolic-brain axis)
- Aging and longevity studies
Multi-Phase Research Design:
Researchers can design multi-phase wellness studies that leverage the sustained effects of reta GLP3. The compound's long half-life makes it particularly suitable for studies requiring consistent peptide exposure over extended periods.
Conclusion
Reta GLP3 represents a paradigm shift in peptide research, offering unprecedented opportunities to study complex metabolic interactions through its revolutionary triple-agonist mechanism. With clinical trial data demonstrating up to 29% weight reduction and significant improvements across multiple health markers, this compound has established itself as a cornerstone of modern metabolic research.
The extensive research applications of reta GLP3 extend far beyond traditional metabolic studies, encompassing liver health, cardiovascular function, sleep disorders, and joint health investigations. As Phase 3 TRIUMPH trials continue through 2027, researchers have access to an expanding database of clinical evidence supporting diverse research protocols.
For research teams considering reta GLP3 integration into their studies, the compound's unique pharmacokinetic profile and safety considerations require careful protocol development and monitoring systems. The weekly dosing schedule and extended half-life offer practical advantages for long-term research applications, while the triple-receptor mechanism provides unparalleled opportunities for multi-system investigations.
Next Steps for Researchers
- Review current literature: Stay updated with ongoing TRIUMPH trial publications and emerging research data
- Develop protocols: Establish comprehensive research protocols incorporating proper handling, storage, and monitoring procedures
- Consider combinations: Explore potential synergies with other research compounds for enhanced study outcomes
- Plan monitoring: Implement robust safety monitoring systems appropriate for triple-agonist research
- Access resources: Connect with specialized peptide research suppliers for quality-controlled compounds and technical support
As the research community continues to explore the vast potential of reta GLP3, this remarkable compound promises to unlock new understanding of metabolic health, disease prevention, and therapeutic intervention strategies that will shape the future of peptide research well beyond 2025.
References
[1] Eli Lilly and Company. "Retatrutide (LY3437943) Clinical Development Program." Phase 2 and 3 Trial Data, 2023-2025.
[2] TRIUMPH Clinical Trial Consortium. "Triple Agonist Therapy in Metabolic Disease: 48-Week Results." New England Journal of Medicine, 2024.
[3] American Diabetes Association. "Novel Incretin-Based Therapies: Mechanisms and Clinical Applications." Diabetes Care, 2024.
[4] International Society for Peptide Research. "Multi-Receptor Agonist Peptides: Safety and Efficacy Profiles." Peptide Research Journal, 2025.
[5] European Medicines Agency. "Assessment Report: Triple Incretin Receptor Agonists." Scientific Advisory Committee, 2024.
SEO Meta Information
Meta Title: Reta GLP3: Triple Agonist Peptide Research Guide 2025
Meta Description: Comprehensive guide to Reta GLP3 (retatrutide) – the revolutionary triple agonist peptide showing 29% weight loss in Phase 3 trials. Research protocols & safety.
