Retatrutide and Cagrilintide: Exploring Their Potential in Metabolic Research (2025)

The landscape of metabolic research is constantly evolving, driven by the relentless pursuit of novel compounds that can address complex physiological challenges. Among the most exciting developments in recent years are the advanced peptide candidates, retatrutide and cagrilintide. For gym health enthusiasts and those keenly interested in the cutting edge of scientific inquiry, understanding these compounds, their mechanisms, and their potential applications in a research context is paramount. The year 2025 brings increased focus on these innovative peptides, particularly in areas related to weight management and metabolic health, as researchers delve deeper into their unique properties and the intriguing possibilities of a retatrutide and cagrilintide combination study.
These research peptides represent significant strides in leveraging the body's own regulatory systems to influence metabolism. While both are being explored for their roles in weight and glucose regulation, their individual mechanisms offer distinct avenues for investigation, making the potential of taking cagrilintide with retatrutide an area of intense scientific interest. This article will delve into the current understanding of each peptide, explore the intriguing concept of a cagrilintide and retatrutide stack, and discuss the broader implications for metabolic research in 2025.
Key Takeaways
- Distinct Mechanisms: Retatrutide is a triple-agonist targeting GLP-1, GIP, and glucagon receptors, while cagrilintide is an amylin analog.
- Individual Research Focus: Retatrutide is being studied for significant weight reduction and glucose control, whereas cagrilintide focuses on appetite suppression and gastric emptying modulation.
- Synergistic Potential: The combination of retatrutide and cagrilintide is an active area of research, with preclinical studies exploring additive or synergistic effects on metabolic parameters.
- Early-Stage Research: Both peptides are currently in various stages of clinical and preclinical research, primarily for the treatment of obesity and related metabolic disorders.
- No Human Use: It is crucial to remember that these are research chemicals and are not approved for human use outside of strictly controlled clinical trial settings.
Understanding Retatrutide and Cagrilintide Individually: A Scientific Overview
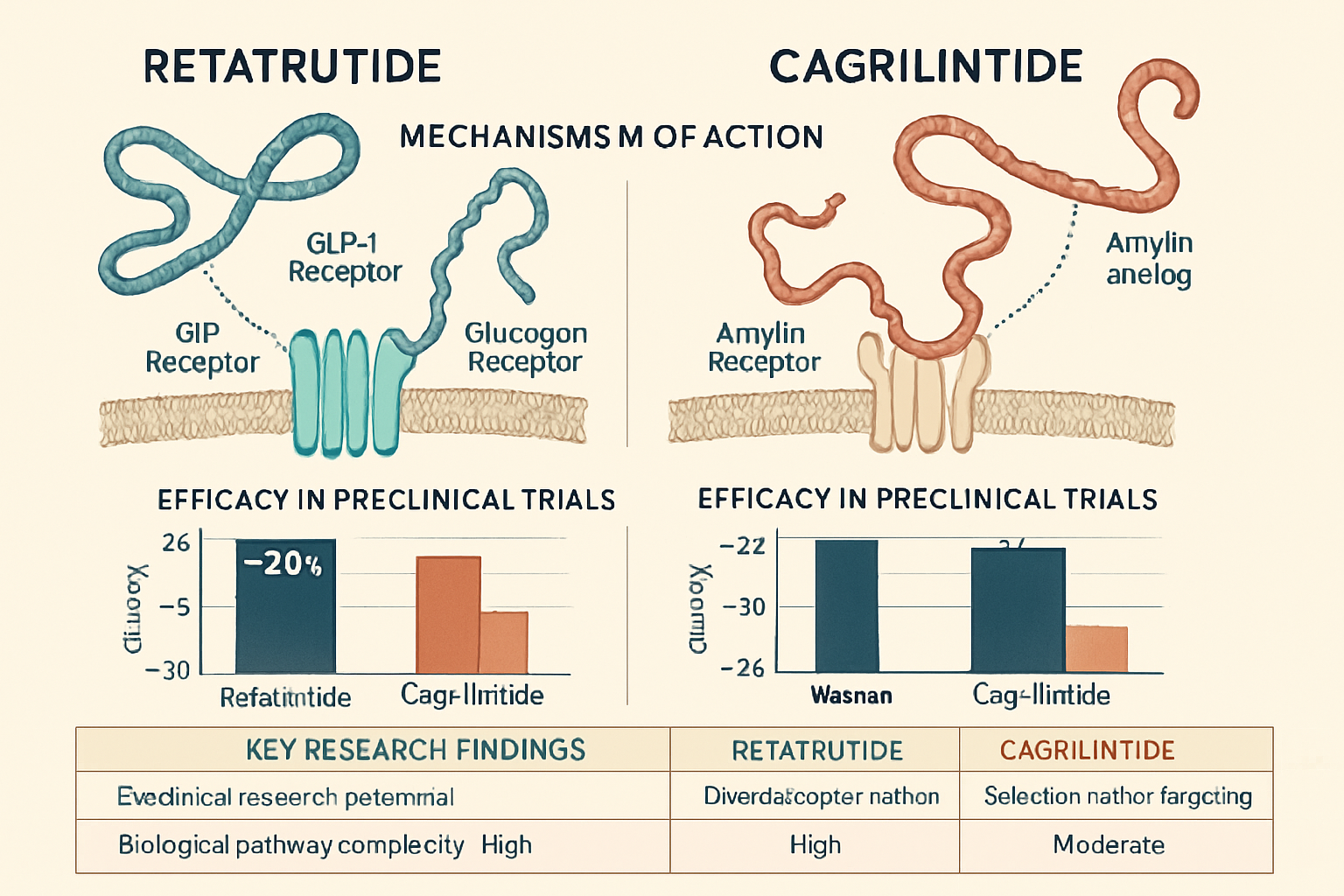
To fully appreciate the potential of combining these compounds, it is essential to first understand retatrutide and cagrilintide as standalone research peptides. Each possesses a unique biochemical profile and mechanism of action, making them distinct subjects of study in the realm of metabolic science.
Retatrutide: The Triple-Agonist Approach
Retatrutide, often referred to as a "triple-G agonist," is a novel unimolecular peptide designed to activate three key receptors involved in metabolic regulation: glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon [1]. This multi-faceted approach distinguishes it from earlier generations of metabolic peptides that typically target only one or two of these pathways.
- GLP-1 Receptor Agonism: Similar to established GLP-1 receptor agonists, retatrutide stimulates insulin secretion, suppresses glucagon release, slows gastric emptying, and promotes satiety, all contributing to improved glucose control and reduced food intake.
- GIP Receptor Agonism: GIP agonism further enhances the glucose-lowering effects by increasing insulin sensitivity and promoting beta-cell function. Research suggests GIP also plays a role in energy metabolism and fat deposition [2].
- Glucagon Receptor Agonism: Historically, glucagon was primarily known for its glucose-raising effects. However, strategic agonism of the glucagon receptor, as seen with retatrutide, can increase energy expenditure and potentially improve lipid metabolism by influencing hepatic fat oxidation. This nuanced interaction is a key area of ongoing research.
The hypothesis behind retatrutide's triple action is that by simultaneously engaging these three pathways, it could achieve more profound and comprehensive metabolic improvements than single or dual agonists. Preclinical research has indeed shown significant reductions in body weight and improvements in various metabolic markers, including glucose levels, insulin sensitivity, and lipid profiles. The profound effects seen in early studies have positioned retatrutide as a prominent subject in the quest for advanced metabolic therapies. Researchers interested in these types of advanced peptides can explore various peptide blends research options.
Cagrilintide: The Amylin Analog
Cagrilintide is a long-acting amylin analog, a synthetic version of the naturally occurring hormone amylin. Amylin is co-secreted with insulin by the pancreatic beta cells and plays a crucial role in glucose homeostasis and appetite regulation [3].
- Appetite Suppression: Cagrilintide primarily acts in the brain to reduce appetite and increase feelings of fullness, leading to decreased food intake. This mechanism is distinct from many other metabolic peptides that primarily influence gastrointestinal processes.
- Gastric Emptying Delay: Similar to natural amylin, cagrilintide slows the rate at which food leaves the stomach. This delay contributes to satiety and helps regulate post-meal glucose spikes.
- Glucagon Suppression: It also has a modest effect on suppressing glucagon secretion, further aiding in glucose control.
Cagrilintide's mechanism of action, focusing heavily on central appetite regulation and gastric emptying, positions it as a promising candidate for weight management. Its long-acting nature means it can potentially be administered less frequently, which is an advantage in a research setting. For those studying its effects, detailed cagrilintide 5mg peptide vial research notes can be invaluable. The distinction between cagrilintide vs retatrutide lies in their primary receptor targets and the breadth of their metabolic influence, with cagrilintide being more focused on appetite and gastric motility.
The Synergy of Retatrutide and Cagrilintide: A Combination Study Perspective
The concept of combining therapeutic agents to achieve superior outcomes is a cornerstone of pharmacological research. When considering retatrutide and cagrilintide, the question naturally arises: what could a cagrilintide retatrutide combination study reveal? The rationale for exploring retatrutide and cagrilintide together stems from their complementary mechanisms of action.
Why Combine Retatrutide and Cagrilintide?
The individual strengths of retatrutide and cagrilintide suggest that a combined approach could potentially offer more comprehensive metabolic benefits than either peptide alone.
- Multi-pronged Appetite Regulation: Retatrutide influences appetite through GLP-1 and GIP pathways, while cagrilintide primarily acts via amylin receptors in the brain. Using cagrilintide with retatrutide could create a more potent and robust appetite suppression effect by targeting multiple neural and gut-brain axis pathways simultaneously.
- Enhanced Glycemic Control: While retatrutide is a powerful modulator of glucose homeostasis through its triple-agonist action, cagrilintide's effects on gastric emptying and glucagon suppression could further stabilize blood sugar levels, especially post-prandially.
- Additive or Synergistic Weight Loss: The combination of strong appetite reduction (from both peptides) and increased energy expenditure (from retatrutide's glucagon component) could lead to more significant and sustainable weight reduction in preclinical models. Research into a cagrilintide and retatrutide stack aims to quantify these potential additive or synergistic effects.
- Lipid Metabolism Improvement: Both peptides have shown positive effects on lipid profiles individually. A combined approach might amplify these benefits, leading to further improvements in markers like triglycerides and cholesterol.
“The pursuit of advanced metabolic solutions drives the exploration of novel peptide combinations, where the distinct mechanisms of retatrutide and cagrilintide offer a compelling rationale for synergistic research.”
Research into Cagrilintide Dosage with Retatrutide
When exploring the combination of retatrutide and cagrilintide, one of the critical aspects for researchers is determining the optimal cagrilintide dosage with retatrutide. This involves meticulous preclinical studies to understand dose-response relationships for each peptide individually and then systematically testing various ratios and concentrations in a combined regimen. The goal is to maximize efficacy while minimizing potential off-target effects, a standard practice in peptide research. For instance, understanding the optimal best dosage for Klow peptide blend can provide foundational insights for similar combination studies.
A cagrilintide retatrutide blend requires careful titration and observation in laboratory settings. Researchers might start with low doses of each peptide and gradually increase them, monitoring metabolic parameters, food intake, and body weight in animal models. This iterative process helps identify concentrations that produce the desired effects without adverse reactions. The complexities of a retatrutide cagrilintide blend necessitate a thorough understanding of pharmacokinetics and pharmacodynamics when these agents are administered together.
Comparative Research: Cagrilintide vs Retatrutide vs Tirzepatide
Another significant area of research in 2025 involves comparing these cutting-edge peptides. The question of cagrilintide vs retatrutide vs tirzepatide often arises, as tirzepatide (a dual GLP-1/GIP agonist) has already shown substantial efficacy in metabolic management. This comparative research helps to:
- Identify Unique Advantages: Determine if the triple-agonist action of retatrutide or the amylin agonism of cagrilintide offers distinct benefits or superior efficacy compared to dual agonists like tirzepatide.
- Elucidate Receptor Contributions: By comparing the outcomes of different receptor engagement profiles, researchers can better understand the individual and combined contributions of GLP-1, GIP, glucagon, and amylin pathways to metabolic improvements.
- Inform Future Combination Strategies: Such comparisons could reveal whether a retatrutide and cagrilintide stack provides a metabolic advantage over existing dual agonists or even other potential future combinations. This allows for a deeper understanding of cagrilintide synergy w glp1 as well.
These comparative studies are essential for mapping the evolving landscape of metabolic pharmacology and identifying the most promising avenues for future investigation.
Practical Considerations for Research in 2025
The burgeoning interest in compounds like retatrutide and cagrilintide underscores a vibrant era in peptide research. For institutions and individual researchers, several practical considerations come into play, especially when working with advanced peptides in 2025.
Sourcing and Quality of Research Peptides
The integrity of research findings hinges entirely on the quality and purity of the peptides used. Researchers must ensure they are sourcing retatrutide and cagrilintide from reputable suppliers that provide:
- High Purity: Typically 98% or higher, verified by analytical methods such as HPLC and Mass Spectrometry.
- Detailed Documentation: Certificates of Analysis (CoAs) that confirm the identity, purity, and concentration of the peptide.
- Proper Storage and Handling: Information on how the peptides have been stored and shipped to maintain stability.
Using high-quality peptides is non-negotiable for reproducible and reliable research. Reputable vendors like Pure Tested Peptides specialize in providing research-grade peptides. Building a diverse peptide library with Pure Tested Peptides can also support various research needs.
Research Protocols for Cagrilintide and Retatrutide
Developing robust research protocols is paramount for any study involving novel peptides. When investigating can you take cagrilintide with retatrutide, researchers need to meticulously plan:
- Animal Models: Selection of appropriate animal models (e.g., diet-induced obese rodents) that mimic relevant metabolic conditions.
- Dosage and Administration: Precise determination of cagrilintide dosage with retatrutide, route of administration (e.g., subcutaneous injection), and frequency based on pharmacokinetic data and previous individual peptide studies.
- Duration of Study: Long enough to observe meaningful metabolic changes, but controlled to manage animal welfare.
- Endpoint Measurements: Comprehensive measurement of relevant metabolic parameters, including:
- Body weight and body composition (fat mass, lean mass)
- Food intake and energy expenditure
- Glucose homeostasis (fasting glucose, HbA1c, oral glucose tolerance tests)
- Insulin sensitivity
- Lipid profiles (triglycerides, cholesterol)
- Hormone levels (e.g., insulin, leptin, adiponectin)
- Histological analysis of relevant tissues (e.g., liver, pancreas, adipose tissue)
These protocols ensure that the data collected is accurate, consistent, and scientifically sound, forming the basis for understanding the efficacy and safety profile of a retatrutide and cagrilintide combination trial. Best practices for storing research peptides are also crucial for maintaining their integrity throughout the study.
Ethical Considerations and Regulatory Landscape
As with all scientific research, especially involving novel compounds with potential physiological effects, ethical considerations are paramount.
- Animal Welfare: Strict adherence to ethical guidelines for animal research, ensuring humane treatment and minimizing distress.
- Research Use Only: Emphasizing that retatrutide and cagrilintide are for "research purposes only" and are not intended for human consumption or therapeutic use outside of approved clinical trials. This distinction is critical for compliance and safety.
- Regulatory Compliance: Awareness and adherence to local and international regulations governing the acquisition, storage, and use of research chemicals.
The regulatory landscape around research peptides is dynamic, and staying informed about current guidelines in 2025 is crucial for all involved parties.
The Future of Metabolic Research with Retatrutide and Cagrilintide
The ongoing research into retatrutide and cagrilintide, both individually and as a potential cagrilintide retatrutide stack, heralds a promising future for metabolic science. The complexity of obesity and metabolic syndrome demands multi-pronged approaches, and these peptides offer precisely that.
Expanding the Understanding of Metabolic Pathways
Studies involving retatrutide and cagrilintide are not just about finding new treatments; they are also about deepening our fundamental understanding of metabolic physiology. By observing how these distinct peptide receptor interactions influence a cascade of biological events, researchers can unravel intricate connections between the gut, brain, pancreas, liver, and adipose tissue. This knowledge can lead to the identification of new therapeutic targets, even beyond the direct application of these specific peptides. For example, understanding the adaptive capacity and peptide mapping of different systems to these novel compounds is a key area of interest.
Potential for Precision Medicine Approaches
As our understanding of individual responses to various metabolic interventions grows, peptides like retatrutide and cagrilintide could eventually contribute to more personalized, precision medicine approaches. Some individuals might respond better to a triple-agonist, while others might benefit more from the central appetite suppression of an amylin analog, or a specific retatrutide cagrilintide blend. Research into genetic and phenotypic markers that predict responsiveness to different peptide mechanisms is an exciting frontier.
Beyond Weight Loss: Broader Metabolic Benefits
While weight reduction is a primary focus, the metabolic benefits of retatrutide and cagrilintide extend to other critical areas. Improvements in fatty liver disease (NAFLD/NASH), cardiovascular risk factors, and even certain inflammatory markers are being investigated. The holistic impact of these peptides on overall metabolic health could be substantial, offering hope for mitigating the long-term complications associated with obesity and type 2 diabetes.
The development pipeline for these types of peptides continues to expand, with many researchers seeking to buy peptides online USA to advance their studies. The meticulous work of scientists exploring can you take retatrutide and cagrilintide together and optimizing the cagrilintide retatrutide combination trial is laying the groundwork for potentially transformative insights in metabolic health.
Conclusion
The scientific exploration of retatrutide and cagrilintide represents a significant leap forward in the quest to understand and address complex metabolic disorders. Individually, these research peptides exhibit compelling mechanisms of action: retatrutide with its innovative triple-agonist approach to GLP-1, GIP, and glucagon receptors, and cagrilintide as a potent amylin analog primarily focused on appetite suppression and gastric emptying.
The prospect of a retatrutide and cagrilintide combination, whether as a cagrilintide retatrutide stack or a carefully formulated blend, is particularly exciting for the research community. The complementary nature of their actions suggests a potential for synergistic effects, leading to enhanced weight reduction, improved glycemic control, and broader metabolic benefits in preclinical models. In 2025, ongoing research is diligently exploring optimal dosages, comparing their efficacy against other leading compounds like tirzepatide, and meticulously charting the complex interactions within metabolic pathways.
For gym health enthusiasts and individuals invested in understanding the forefront of health science, it is crucial to remember that retatrutide and cagrilintide are cutting-edge research compounds. They are not approved for human use outside of controlled clinical trials and should only be handled by qualified researchers in appropriate laboratory settings. The rigorous scientific process, from careful sourcing to detailed protocol execution, is essential to unlock the full potential of these promising peptides.
Actionable Next Steps for Researchers:
- Stay Informed: Continuously monitor published research and clinical trial updates on both retatrutide and cagrilintide from reputable scientific journals.
- Prioritize Quality Sourcing: Always acquire research peptides from trusted suppliers that provide comprehensive Certificates of Analysis to ensure purity and authenticity.
- Design Rigorous Protocols: Develop and implement detailed, ethical research protocols for any studies involving these compounds, especially for combination investigations.
- Collaborate and Share: Engage with the broader scientific community to share findings and insights, accelerating the pace of discovery in metabolic research.
- Explore Relevant Resources: Investigate other advanced peptide research, such as AOD9604 metabolic research for additional insights into metabolic management.
The journey of discovery with retatrutide and cagrilintide is still unfolding, and the insights gained in 2025 will undoubtedly shape the future of metabolic health science for years to come.
References
[1] Jastreboff AM, et al. Triple-Hormone-Receptor Agonist Retatrutide for Obesity — A Phase 2 Trial. The New England Journal of Medicine. 2023;389(6):513-526. DOI: 10.1056/NEJMoa2301992.
[2] Finan B, et al. A rationally designed GLP-1/GIP co-agonist for the treatment of type 2 diabetes. Science Translational Medicine. 2013;5(178):178ra43. DOI: 10.1126/scitranslmed.3005011.
[3] Christensen M, et al. Cagrilintide, a Novel Long-Acting Amylin Analogue, in People With Overweight or Obesity: A Randomised, Double-Blind, Placebo-Controlled, Dose-Escalation Trial. Diabetes, Obesity and Metabolism. 2021;23(4):1047-1056. DOI: 10.1111/dom.14323.
SEO Meta Title: Retatrutide & Cagrilintide: Metabolic Research Insights for 2025
SEO Meta Description: Explore retatrutide and cagrilintide in 2025: understanding their mechanisms, combination potential, and impact on metabolic research for gym enthusiasts.
