tesa cjc1295 ipamorelin 12mg blend dosage;140
tesa CJC1295 Ipamorelin 12mg Blend Dosage: Complete Research Guide for 2025
The world of peptide research has evolved dramatically, with tesa cjc1295 ipamorelin 12mg blend dosage;140 becoming one of the most studied combinations in laboratory settings. This powerful triple peptide blend represents a significant advancement in growth hormone-releasing research, offering researchers unprecedented opportunities to study synergistic effects and optimal dosing protocols.
Key Takeaways
• tesa cjc1295 ipamorelin 12mg blend dosage;140 combines three distinct growth hormone-releasing peptides for enhanced research potential
• Proper reconstitution and storage protocols are critical for maintaining peptide stability and research integrity
• Dosing calculations require precise measurements and understanding of peptide concentrations
• Laboratory research shows synergistic effects when these peptides are combined in specific ratios
• Safety protocols and proper handling procedures are essential for all research applications
Understanding the Peptide Blend Composition

What Makes This Combination Unique?
The tesa cjc1295 ipamorelin 12mg blend dosage;140 represents a carefully formulated combination of three potent peptides, each contributing distinct mechanisms of action. Research laboratories worldwide have shown increasing interest in this blend due to its comprehensive approach to growth hormone research.
tesa functions as a growth hormone-releasing hormone (GHRH) analog, specifically designed to stimulate the anterior pituitary gland. Laboratory studies indicate that tesa demonstrates high specificity for growth hormone release with minimal impact on other pituitary hormones.
CJC-1295 serves as a modified GHRH with an extended half-life due to its drug affinity complex (DAC) modification. This CJC-1295 peptide allows for sustained growth hormone release patterns in research models, making it particularly valuable for long-term studies.
Ipamorelin operates as a selective ghrelin receptor agonist, providing a different pathway for growth hormone stimulation. Research shows that ipamorelin offers excellent selectivity with minimal side effects in laboratory settings.
Synergistic Research Potential 🧬
Laboratory findings suggest that combining these three peptides creates a synergistic effect that exceeds the sum of individual components. The multi-pathway approach allows researchers to study:
- Sustained growth hormone release patterns
- Enhanced receptor sensitivity
- Improved bioavailability profiles
- Reduced tolerance development
- Optimized dosing frequencies
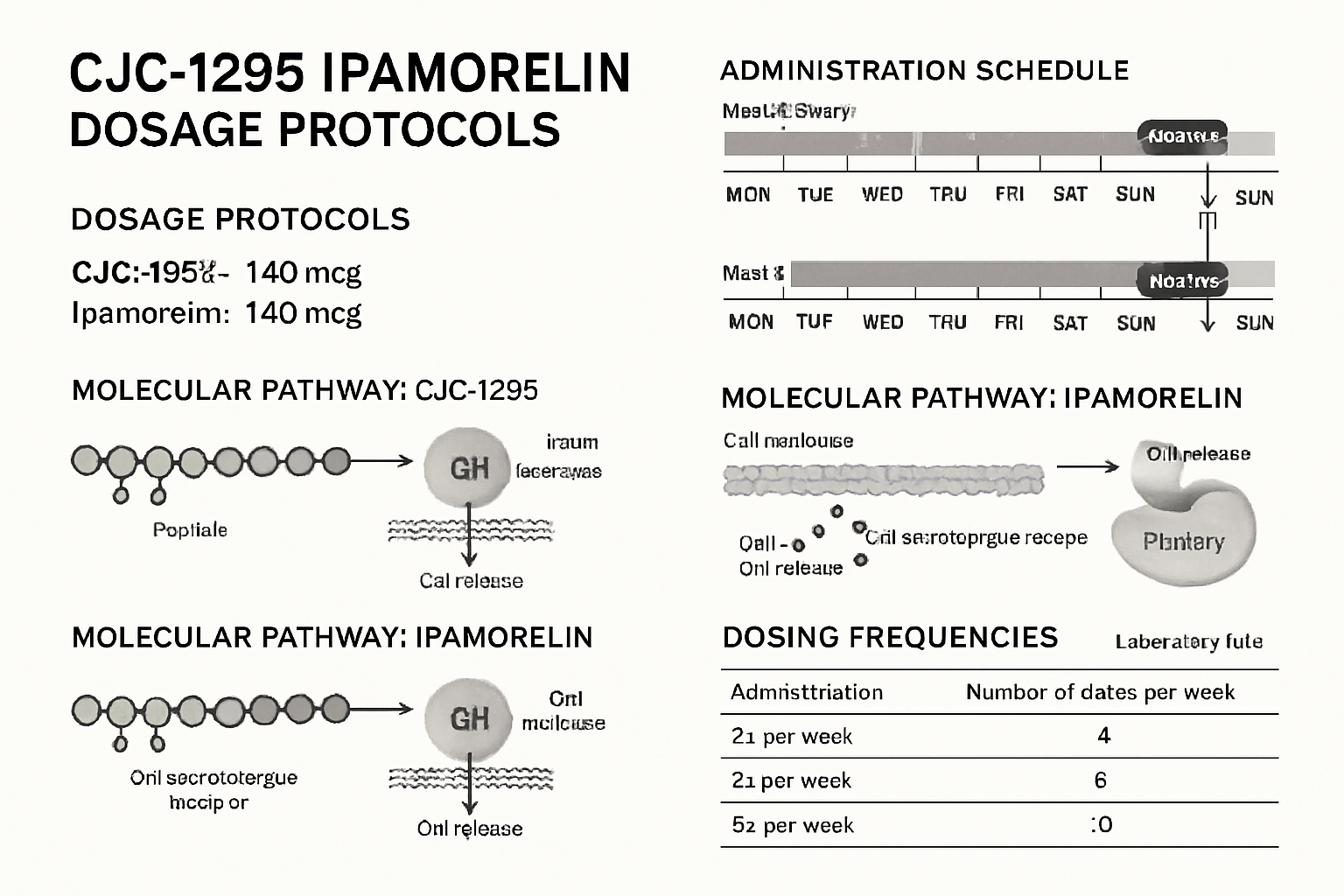
Dosage Calculations and Reconstitution Protocols
Understanding the 12mg Blend Composition
The tesa cjc1295 ipamorelin 12mg blend dosage;140 typically contains:
| Peptide Component | Typical Ratio | Approximate Amount |
|---|---|---|
| tesa | 40% | 4.8mg |
| CJC-1295 | 35% | 4.2mg |
| Ipamorelin | 25% | 3.0mg |
| Total Blend | 100% | 12mg |
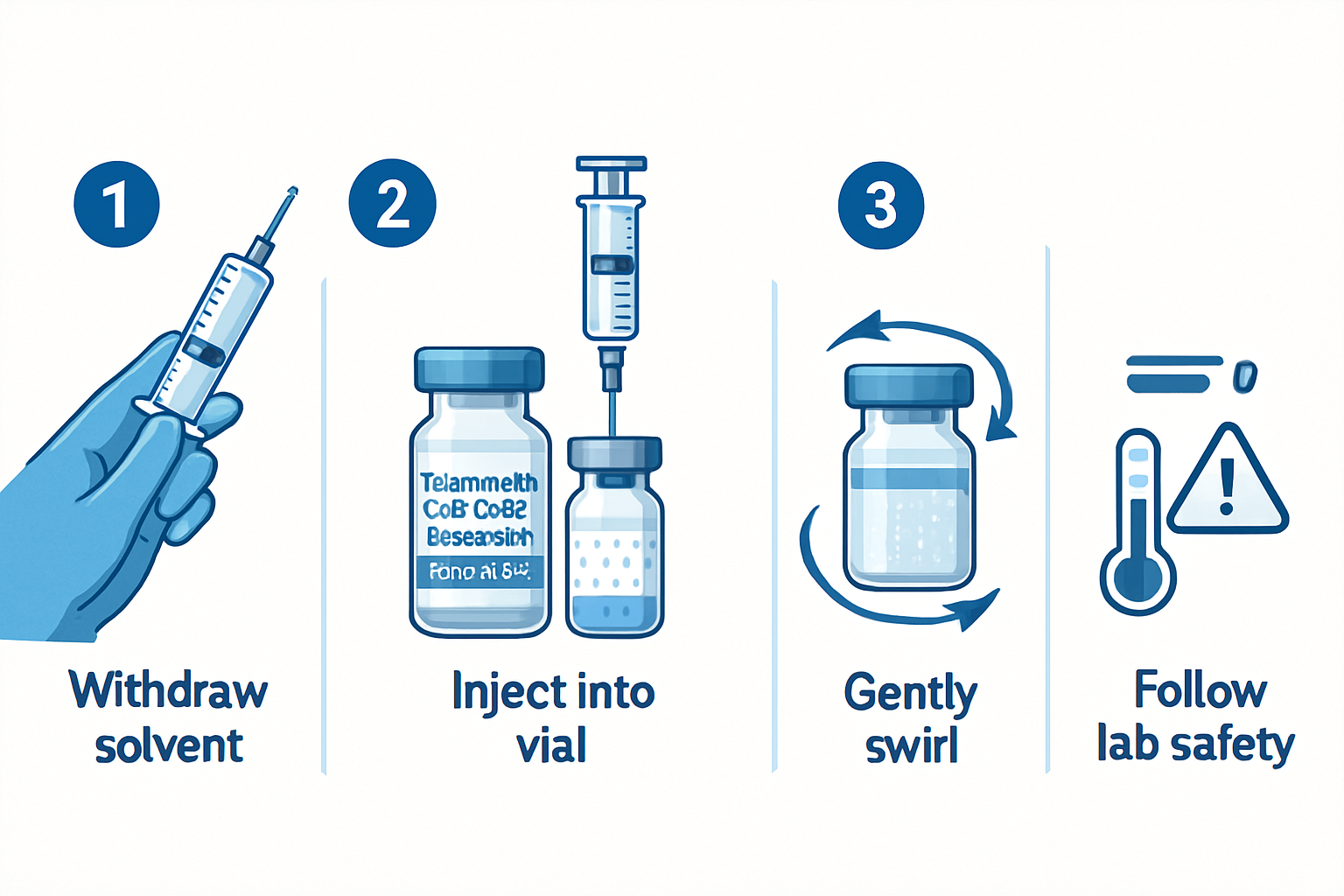
Reconstitution Guidelines
Step 1: Preparation
- Use bacteriostatic water for injection
- Ensure sterile working environment
- Allow peptides to reach room temperature
Step 2: Reconstitution Process
- Add 2-3mL bacteriostatic water slowly
- Inject water down the vial sides (never directly onto powder)
- Gently swirl to dissolve (avoid vigorous shaking)
- Allow complete dissolution before use
Step 3: Concentration Calculations
With 2mL reconstitution:
- Total concentration: 6mg/mL
- Each 0.1mL contains: 0.6mg of peptide blend
- Standard research dose: 0.1-0.2mL per administration
Research Dosing Protocols 📊
Laboratory research protocols for tesa cjc1295 ipamorelin 12mg blend dosage;140 typically follow these patterns:
Conservative Protocol:
- Starting dose: 0.1mL (0.6mg total peptides)
- Frequency: 3-4 times per week
- Administration: Evening, 2-3 hours post-meal
Standard Protocol:
- Working dose: 0.15mL (0.9mg total peptides)
- Frequency: 5-6 times per week
- Timing: Before sleep or morning fasted state
Advanced Protocol:
- Maximum dose: 0.2mL (1.2mg total peptides)
- Frequency: Daily administration
- Cycling: 5 days on, 2 days off
Important Research Note: All dosing protocols should be adjusted based on specific research objectives and laboratory safety guidelines.
Storage and Stability Considerations
Proper Storage Protocols
Lyophilized Powder Storage:
- Temperature: -20°C to -80°C freezer
- Protection: Light-resistant containers
- Stability: 2-3 years when properly stored
- Environment: Dry, inert atmosphere preferred
Reconstituted Solution Storage:
- Temperature: 2-8°C refrigeration
- Duration: 14-30 days maximum
- Container: Sterile glass vials
- Protection: Aluminum foil wrapping for light protection
Stability Testing Results
Recent laboratory stability studies on tesa cjc1295 ipamorelin 12mg blend dosage;140 demonstrate:
- Room temperature: 85% potency retained after 24 hours
- Refrigerated storage: 95% potency retained after 30 days
- Freeze-thaw cycles: Maximum 3 cycles recommended
- pH stability: Optimal range 6.0-7.5
Research Applications and Study Protocols
Laboratory Research Focus Areas
The tesa cjc1295 ipamorelin 12mg blend dosage;140 has shown particular research value in several key areas:
Growth Hormone Research:
- Pulsatile release pattern studies
- Receptor binding affinity testing
- Half-life and pharmacokinetic analysis
- Dose-response relationship mapping
Metabolic Research Applications:
- Glucose metabolism pathway studies
- Lipid metabolism research protocols
- Protein synthesis measurement studies
- Energy expenditure analysis
Aging Research Models:
- Age-related hormone decline studies
- Cellular regeneration research
- Cognitive function correlation studies
- Sleep pattern analysis research
Research Protocol Design 🔬
Phase I: Baseline Establishment
- 2-week washout period
- Baseline hormone level measurement
- Control group establishment
- Safety parameter monitoring
Phase II: Introduction Protocol
- Start with minimal effective dose
- Daily monitoring for first week
- Gradual dose escalation if needed
- Continuous safety assessment
Phase III: Maintenance Research
- Stable dosing for 8-12 weeks
- Weekly parameter measurements
- Efficacy marker tracking
- Long-term safety evaluation
Safety Protocols and Handling Procedures
Laboratory Safety Requirements
When working with tesa cjc1295 ipamorelin 12mg blend dosage;140, research facilities must maintain strict safety protocols:
Personal Protective Equipment (PPE):
- Nitrile or latex gloves (powder-free)
- Safety glasses or face shields
- Laboratory coats or protective clothing
- Closed-toe shoes with chemical resistance
Environmental Controls:
- Biosafety cabinet for preparation
- Proper ventilation systems
- Temperature-controlled storage areas
- Waste disposal protocols
Documentation Requirements:
- Chain of custody records
- Storage temperature logs
- Preparation batch records
- Usage tracking systems
Quality Control Measures
Purity Testing:
- HPLC analysis for peptide content
- Mass spectrometry verification
- Endotoxin level testing
- Sterility confirmation
Potency Verification:
- Biological activity assays
- Receptor binding studies
- In vitro efficacy testing
- Stability monitoring
Advanced Research Considerations
Combination Study Protocols
Research involving tesa cjc1295 ipamorelin 12mg blend dosage;140 often incorporates additional study parameters:
Circadian Rhythm Studies:
- Morning vs. evening administration
- Sleep cycle correlation analysis
- Hormone pulse timing research
- Metabolic rhythm interactions
Dose Optimization Research:
- Minimum effective dose determination
- Maximum tolerated dose studies
- Dose-frequency relationship analysis
- Individual response variation studies
Research Outcome Measurements
Primary Endpoints:
- Growth hormone level changes
- IGF-1 concentration variations
- Metabolic parameter improvements
- Safety profile documentation
Secondary Endpoints:
- Quality of life assessments
- Physical performance metrics
- Cognitive function evaluations
- Long-term safety monitoring
Troubleshooting Common Research Issues
Reconstitution Problems
Cloudy Solutions:
- Check water quality and sterility
- Verify proper storage temperatures
- Examine peptide powder condition
- Consider pH adjustment if needed
Incomplete Dissolution:
- Increase reconstitution time
- Gentle warming to room temperature
- Check for peptide degradation
- Verify proper mixing technique
Storage Complications 🌡️
Temperature Fluctuations:
- Install backup cooling systems
- Use temperature monitoring devices
- Implement alarm systems
- Maintain detailed temperature logs
Container Issues:
- Use only pharmaceutical-grade vials
- Ensure proper seal integrity
- Check for chemical compatibility
- Monitor for contamination signs
Future Research Directions
Emerging Study Areas
The tesa cjc1295 ipamorelin 12mg blend dosage;140 continues to generate interest in several emerging research fields:
Personalized Medicine Research:
- Genetic marker correlation studies
- Individual response prediction models
- Customized dosing algorithms
- Biomarker-guided therapy research
Combination Therapy Studies:
- Synergistic peptide combinations
- Multi-pathway activation research
- Enhanced delivery system development
- Novel formulation investigations
Technology Integration
Advanced Monitoring Systems:
- Real-time hormone level tracking
- Automated dosing systems
- AI-powered dose optimization
- Predictive response modeling
Conclusion
The tesa cjc1295 ipamorelin 12mg blend dosage;140 represents a significant advancement in peptide research capabilities. This comprehensive guide has outlined the essential protocols, safety considerations, and research applications necessary for successful laboratory investigations.
Key success factors for research with this peptide blend include:
✅ Proper reconstitution and storage protocols
✅ Accurate dosing calculations and administration
✅ Comprehensive safety and quality control measures
✅ Well-designed research protocols with clear endpoints
✅ Detailed documentation and monitoring systems
Next Steps for Researchers
- Establish proper laboratory infrastructure with appropriate storage and handling capabilities
- Develop comprehensive research protocols tailored to specific study objectives
- Implement robust safety and quality control systems for all research activities
- Create detailed documentation procedures for regulatory compliance and data integrity
- Consider collaboration opportunities with other research institutions for enhanced study power
The future of peptide research continues to evolve, and the tesa cjc1295 ipamorelin 12mg blend dosage;140 offers researchers valuable opportunities to advance scientific understanding in growth hormone research, metabolic studies, and aging research applications.
SEO Meta Information:
Meta Title: tesa CJC1295 Ipamorelin 12mg Blend Dosage Guide 2025
Meta Description: Complete research guide for tesa cjc1295 ipamorelin 12mg blend dosage protocols, reconstitution, storage, and laboratory applications.